Abstract
Objective. Feeding intolerance during the early postnatal period can be distressing for families. To assess this, infants (n = 150) of mothers who had previously decided to exclusively or partially formula feed were enrolled ≤24 hours after birth. Methods. Infants were fed with a single ready-to-feed, 100% partially hydrolyzed whey protein-based formula until discharge, in accordance with standard hospital practice. Parents recorded daily the presence/severity of gastrointestinal (GI) symptoms/behaviors, rated from 0 (never) to 5 (always). A validated questionnaire was completed at discharge to evaluate symptoms of GI discomfort; scores can range from 0 to ~140 (lower scores indicate fewer symptoms). Results. Mean ± SD daily scores ranged from 0.12 ± 0.40 (fussiness/irritability) to 1.26 ± 0.90 (spitting up), indicating that GI symptoms/behaviors occurred, on average, “never” or “almost never.” Mean GI discomfort scores were also very low (9.9 ± 7.4). Conclusion. These results indicate that the ready-to-feed formula was very well accepted and well tolerated during this period among healthy newborns in Saudi Arabia.
Introduction
Transient neonatal feeding intolerance is common in preterm infants but has also been described in full-term infants.1,2 Some symptoms of feeding intolerance (e.g., recurrent vomiting and refusal to feed) during the early postnatal period can be distressing for families and may delay discharge from the postnatal ward if symptoms are severe. Infant formulas made using hydrolyzed proteins have been shown to promote more favorable markers of gastrointestinal (GI) tolerance as they are more easily digested than formulas made with intact proteins.3-5
Although trypsin preparations derived from animal sources have historically been used to hydrolyze whey proteins in these formulas, infant formulas hydrolyzed by these enzymes may not be suitable for families with dietary restrictions due to religious or cultural concerns, including those following strict vegetarian or halal diets. 6 A partially hydrolyzed formula prepared without animal-derived enzymes may be particularly appropriate in regions where the majority of families are Muslim, likely to follow a halal diet, and rely on infant formulas as a primary source of infant nutrition. For example, in Saudi Arabia, more than 90% of the population is Muslim 7 and less than 10% of Saudi infants are exclusively breastfed during the first 6 months of life. 8
A whey-based, partially hydrolyzed ready-to-feed (RTF) infant formula prepared with microbially-derived enzymes has been recently developed to address the needs of families following halal diets. The microbially-derived enzymes have trypsin and chymotrypsin activity, mimicking the activity and the specificity of the animal-derived enzymes that are traditionally used for hydrolysis. More specifically, the microbial enzymes are endoproteases that cleave peptide bonds on the carboxyl side of the amino acids lysine and arginine (similar to trypsin) or phenylalanine, tryptophan, tyrosine, leucine, methionine, and histidine (similar to chymotrypsin). In vitro and in vivo data indicate that the protein hydrolysate profile obtained from this microbial enzymatic process is generally comparable to that observed with the use of an animal-derived trypsin preparation. 9 However, clinical data on the tolerability and acceptability of this formula are lacking.
The purpose of this real-world observational study was to evaluate the GI tolerance of healthy, term infants fed a partially hydrolyzed RTF infant formula prepared with microbial enzymes during the immediate postnatal (birth hospitalization) period. We hypothesized that this formula would be well-tolerated during this period.
Methods
Study Design
This open label (unblinded), single-arm, post-market, real-world effectiveness study was conducted at Dr. Sulaiman Al Habib Hospital in Riyadh, Saudi Arabia (Clinicaltrials.gov registration: NCT05097924) between February and March 2022. Real-world effectiveness studies are intended to provide information about the usage and potential benefits and risks of a medical product when it is administered to a diverse population in a real-life clinical setting. 10 Infants enrolled in the study were fed a single formula (halal, RTF, 100% partially hydrolyzed whey protein-derived in line with the protein requirements set in Annex I of Delegated Regulation [EU] 2016/127 [ 11 ]) during the birth hospitalization period (until approximately 2 to 4 days after birth).
The study formula, which provided 47 kcal, 0.9 g of protein, 5.5 g of carbohydrates, and 2.4 g of lipids per 70 mL, was fed orally per usual hospital practice and in accordance with recommended feeding guidelines. During the first 1 to 2 days of life, newborn infants generally consume about 15 mL of formula every 2 to 3 hours (or 8 to 12 times per day), with the amount increasing to 30 to 60 mL per feed over the next few days. Mixed feeding (i.e., both breastmilk and formula) in any proportion was acceptable. Daily formula intake was adjusted as needed by healthcare personnel depending on the amount of breastfeeding (if the infant was mixed fed) and the investigator’s clinical judgement. At discharge, infants were transitioned to a different standard formula (as the study formula is available exclusively in hospitals) and/or breastfed.
Ethical Approval and Informed Consent
The study was approved by the Institutional Review Board of the Dr. Sulaiman Al Habib Research Center (study number RC21.09.26) and written informed consent was obtained from at least one parent/legally acceptable representative before any study-related procedures were conducted.
Participants
Study subjects included healthy infants of mothers who had previously and independently decided to formula feed. Infants were eligible if they were born full term (≥37 completed weeks of gestation) and weighed ≥2500 and ≤4500 g at birth. Infants who were exclusively breastfed, who exhibited any evidence of significant cardiac, respiratory, endocrine, hematologic, or gastrointestinal diseases/conditions, or who had conditions that required specialized feeding, were excluded. All infants were enrolled ≤24 hours after birth.
Objectives and Outcome Measures
The main objective of the study was to document parent- and clinician-reported symptoms of GI tolerance using the Newborn Infant GI Tolerance eDiary and the Gastrointestinal Tract Function subscale of the NeoEAT questionnaire. 12
The Newborn Infant GI Tolerance eDiary consists of 9 questions documenting the frequency and severity of GI symptoms (diarrhea, gassiness, spitting-up, vomiting, reflux) and GI-related behaviors (fussiness, crying, crying in conjunction with feeding, and sleep patterns) using a 6-point scale ranging from “0 (Never)” to “5 (Always).” The GI Total Scale was calculated as the averaged sum of all 9 items, while the GI Symptoms subscale was calculated as the averaged sum of questions 1 to 5 and the GI Behaviors subscale was calculated as the averaged sum of questions 6 to 9. Lower scores indicate fewer symptoms.
The eDiary was completed by parents every evening during the study period. Depending on the timing of study enrollment in relation to the time of birth of the baby, the eDiary may have been skipped on Visit 1. For example, if the baby was born before 12 noon, the eDiary was completed on the same day, but if the baby was born after 12 noon, the eDiary was completed on the next day.
Two versions of the Gastrointestinal Tract Function (GI Function) subscale of the NeoEAT questionnaire were used in the study, depending on the mother’s chosen feeding mode: the Bottle-Feeding version 13 or the Mixed-Feeding version. 14 The subscale consists of 28 or 27 questions, respectively, which are used to evaluate symptoms related to gastroesophageal reflux, swallowing coordination, GI discomfort, and gagging, with total scores ranging from 0 to ~140. Parents completed this questionnaire once at hospital discharge, reflecting on any symptoms that had occurred since birth. Similar to the eDiary, lower scores indicate fewer symptoms.
The NeoEAT questionnaire was translated into Arabic per the guidelines issued by the International Society for Pharmacoeconomics and Outcomes Research. 15 The reliability of the translations was assessed using the Cronbach’s alpha method, providing estimates of 0.64 for the Bottle-Feeding version (floor, 0%; ceiling, 100%) and 0.79 for the Mixed-Feeding version (floor, 0%; ceiling, 98.4%).
Secondary endpoints evaluated during the study included specific GI characteristics, including GI symptoms and GI-related behaviors, as documented by health care providers in the infants’ hospital medical charts: the number of bowel movements per day, the incidence of abdominal distention, and the incidence of vomiting. Study formula administration was also evaluated using information recorded from the medical chart. Data evaluated regarding formula administration included the number of formula feedings per day, the number of breast feedings per day (if infants were mixed fed), and the total volume of formula consumed per feeding.
Infant discharge characteristics were also recorded based on information provided in medical charts. These included the infant’s weight at discharge, the name and dosage of any medication prescribed at discharge, and the date and time of discharge.
Formula safety was evaluated by monitoring the incidence, severity, and possible relation to study formula of reported adverse events (AEs). Adverse events (AEs) and serious AEs (SAEs) were collected from the time the informed consent form was signed until discharge from the maternity ward (end of study participation).
Finally, parents were asked to provide their overall opinion regarding the study formula using a 7-point scale, ranging from 1 (extremely poor) to 7 (excellent).
Statistical Analysis
This study was descriptive in nature and the sample size was not based on any statistical inference. Instead, the sample size was based on the birth rate at the study hospital (300-400 births per month) and the estimated proportion of mothers who exclusively breastfeed (5%). Given the estimate that approximately 50% of mothers will decline to participate, a sample size of approximately 150 infants was calculated, with the objective to complete enrollment within about 1 month.
Three study populations were intended to be analyzed: the full analysis set (FAS), which would include all enrolled infants; the per-protocol set (PPS), which would include subjects from the FAS without any protocol deviations that could impact the assessment of GI tolerance; and the Safety Analysis Set (SAF) which would include all subjects with at least one documented feeding of the study formula. There were no major protocol deviations, and all enrolled infants completed the study, therefore the FAS, PPS, and SAF populations were identical.
For the eDiary and the GI Function subscale of the NeoEAT questionnaire, subscale scores were derived by adding the individual average scores for all items within the given subscale. For the eDiary, a total score was also calculated by summing the individual average scores for all items for all subjects. These scores as well as secondary outcomes were analyzed using standard summary statistics for continuous or categorical variables, as appropriate. The average number of bowel movements per day, feedings per day, and average daily formula intake were calculated excluding the day of enrollment and day of discharge, since these days were usually partial days and therefore the values would be unrepresentatively low on these days.
All statistical analyses were conducted using Statistical Analysis System software (SAS Institute, Cary, NC, United States), version 9.4.
Results
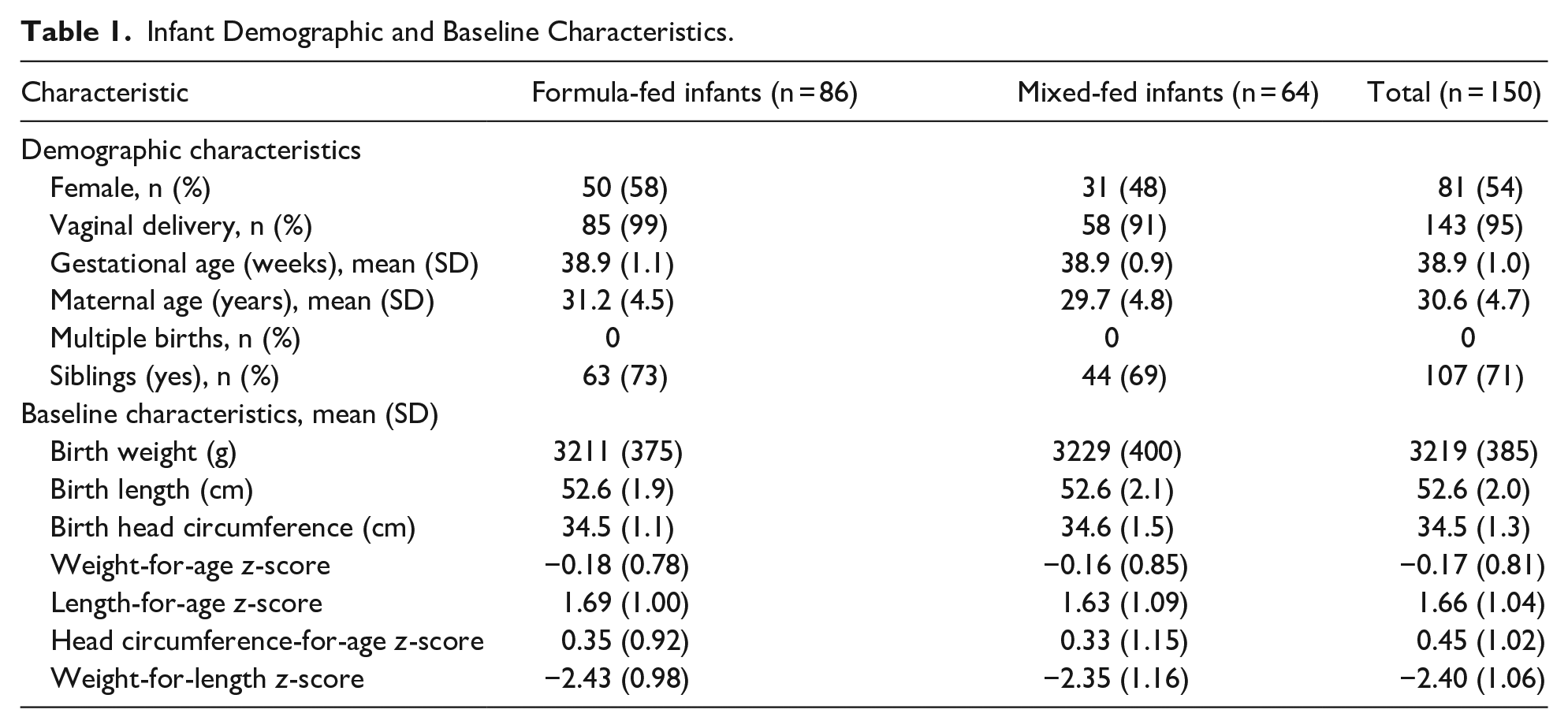
A total of 150 infants, including 86 formula-fed infants and 64 mixed-fed infants, enrolled and completed the study. No screen failures, protocol deviations, or dropouts occurred in the study. All infants were included in all analysis populations. Infant demographics and baseline characteristics, which were similar between the formula-fed and mixed-fed groups, are shown in Table 1.
Infant Demographic and Baseline Characteristics.
For the eDiary, mean scores for the total population (formula-fed infants and mixed-fed infants) are shown in Table 2. Mean GI total scores were 4.66 (SD, 3.55) in the total population, 4.65 (SD, 3.85) among formula-fed infants, and 4.67 (SD, 3.13) among mixed-fed infants. In the overall population, eDiary subscale scores were 3.69 (SD, 2.29) for the GI symptom subscale and 0.97 (SD, 1.67) for the GI behavior subscale. Subscale scores were similar between formula-fed and mixed-fed subgroups.
Parent-Reported GI Symptoms as Assessed by the Newborn Infant GI Tolerance eDiary and the GI Function Subscale of the NeoEAT Questionnaire (Mean ± SD). The Range Is Presented for Each Scale and Subscale; Lower Scores Indicate Fewer Symptoms.

Mean scores for individual items on the eDiary, as shown in Table 2, ranged from a minimum score of 0.12 (SD, 0.40) for fussiness/irritability to a maximum score of 1.26 (SD, 0.90) for spitting-up for formula-fed infants and 0.13 (SD, 0.35) for fussiness/irritability to 1.09 (SD, 0.64) for gassiness for mixed-fed infants. These ranges indicate that each GI symptom/behavior occurred, on average, “almost never” or “never.” Mean scores on the GI Function subscale of the NeoEAT questionnaire were 8.6 (SD, 5.3) for formula-fed infants and 9.9 (SD, 7.4) for mixed-fed infants (Table 2).
The average number of bowel movements per day over the study duration was similar between the formula-fed and mixed-fed groups (4.0 [SD, 1.3] vs 3.7 [SD, 1.5]). Vomiting was rarely reported during the study (≤ 2.0%), occurring in only 3 infants on day 0 and 2 infants on day 1. Abdominal distension was not reported for any infant in the study.
The mean number of feedings over all days was 7.0 (SD, 0.9) in the formula-fed group and 5.6 (SD, 1.3) in the mixed-fed group. On average, infants consumed 221.8 mL/day (SD, 40.5) in the formula-fed group and 175.9 mL/day (SD, 52.7) in the mixed-fed group. In the mixed-fed group, infants breastfed, on average, 1.4 (SD, 1.3) times per day during the study period.
Overall, the mean duration of hospitalization was 1.8 days (SD, 0.6) and similar between the formula-fed and mixed-fed groups (1.7 [SD, 0.6] and 1.9 [SD, 0.6] days, respectively). As expected, the majority (about 95%) of vaginally born infants were discharged by study day 2, whereas 71.5% of C-section born infants were discharged on study day 3 or 4. At discharge, mean weight in the overall population was 3189 g (SD, 381). Mean weight at discharge was similar between the groups (formula fed, 3177 g [SD, 372]; mixed fed, 3205 g [SD, 395]). Mean weight-for-age z-score at discharge was −0.20 (SD, 0.77) in the formula-fed group and −0.18 (SD, 0.83) in the mixed-fed group. No medications were prescribed during the study or at discharge.
Among the total population, 15% of parents rated the study milk as excellent, 37% as very good, and 35% as quite good. Approximately 11% rated the study milk as neither good nor poor and 1% as quite poor, with no parents submitting ratings of very poor or extremely poor.
Only 5 infants (3.3%) experienced an AE (2 in the formula-fed group and 3 in the mixed-fed group), all of which were reported to be mild in severity and resolved prior to discharge. There were 2 reports of mild vomiting, one in each group, and 3 reports of hematemesis due to swallowed maternal blood during delivery (1 in the formula-fed group; 2 in the mixed-fed group). No SAEs were reported. All AEs were deemed unrelated or unlikely to be related to the study formula by the investigator.
Discussion
This brief report describes the GI tolerance of healthy, formula-fed newborn infants in Saudi Arabia during the immediate postnatal period. Results of the eDiary and GI Function subscale of the NeoEAT questionnaire indicate that gastrointestinal symptoms and behaviors associated with GI distress were very infrequent and mild in these infants during this period. In fact, mean scores for the GI Function subscale of the NeoEAT questionnaire were substantially lower than the 25th percentile values observed in reference populations of young infants (9 vs 25 and 10 vs 28 in formula-fed and mixed-fed infants, respectively).13,14
This study is the first to assess the tolerability of this partially hydrolyzed, whey-based formula produced using microbially-derived enzymes among healthy term infants in Saudi Arabia. The results of our study align with those from a recently published, prospective, randomized trial that reported the safety and tolerability of an extensively hydrolyzed, whey-protein infant formula derived from non-porcine enzymes in infants with CMA 16 and other studies of partially hydrolyzed whey-based formulas in older infants.17-20
Limitations of the study include the lack of a comparator group and its potential lack of generalizability given that it only evaluated infants at a single hospital in Saudi Arabia. Moreover, although our intention was to evaluate the discriminative validity of the translated NeoEAT questionnaire, the number of infants with reported AEs was too low to fully assess this aspect of validity using our study results. Finally, another limitation is the very short duration of formula feeding and assessment of GI symptoms, which was limited to the time between birth and hospital discharge. Additional studies with longer periods of formula consumption and GI symptom assessment are needed to confirm and extend these findings.
Conclusions
In conclusion, our results show that symptoms and behaviors indicative of GI distress were very infrequent and mild in formula-fed infants during the immediate postnatal period. There was no evidence of transient neonatal feeding intolerance in this population of healthy infants. The partially hydrolyzed RTF infant formula was well tolerated by infants, well accepted by parents, and may represent a suitable option for families seeking a halal formula to provide to their infants.
Supplemental Material
sj-docx-1-gph-10.1177_2333794X241260261 – Supplemental material for Gastrointestinal Tolerance of Formula-Fed Infants During the Immediate Postnatal Period in Saudi Arabia: An Observational Study
Supplemental material, sj-docx-1-gph-10.1177_2333794X241260261 for Gastrointestinal Tolerance of Formula-Fed Infants During the Immediate Postnatal Period in Saudi Arabia: An Observational Study by Nicholas P. Hays, Rasha Alenazi, Reham Al-Zaatreh, Luca Lavalle, Moustafa Sameer, Inez Sroda, Yvonne Vissers and Mohammad Mizyed in Global Pediatric Health
Supplemental Material
sj-docx-2-gph-10.1177_2333794X241260261 – Supplemental material for Gastrointestinal Tolerance of Formula-Fed Infants During the Immediate Postnatal Period in Saudi Arabia: An Observational Study
Supplemental material, sj-docx-2-gph-10.1177_2333794X241260261 for Gastrointestinal Tolerance of Formula-Fed Infants During the Immediate Postnatal Period in Saudi Arabia: An Observational Study by Nicholas P. Hays, Rasha Alenazi, Reham Al-Zaatreh, Luca Lavalle, Moustafa Sameer, Inez Sroda, Yvonne Vissers and Mohammad Mizyed in Global Pediatric Health
Supplemental Material
sj-docx-3-gph-10.1177_2333794X241260261 – Supplemental material for Gastrointestinal Tolerance of Formula-Fed Infants During the Immediate Postnatal Period in Saudi Arabia: An Observational Study
Supplemental material, sj-docx-3-gph-10.1177_2333794X241260261 for Gastrointestinal Tolerance of Formula-Fed Infants During the Immediate Postnatal Period in Saudi Arabia: An Observational Study by Nicholas P. Hays, Rasha Alenazi, Reham Al-Zaatreh, Luca Lavalle, Moustafa Sameer, Inez Sroda, Yvonne Vissers and Mohammad Mizyed in Global Pediatric Health
Footnotes
Acknowledgements
We sincerely thank the parents and caregivers who consented to their infant’s participation in the study, as well as the study team at Dr Sulaiman Al Habib Hospital in Riyadh, Saudi Arabia. We especially wish to thank Audrey Ballarin, Madeleine Blankenspoor, Sonia El Hamdi, Peter Erdmann, Ludivine Ferraille-Naze, Mazen Hemdan, and Ahmed Zamel for assistance with study planning and/or conduct. We also thank Britt Pados, PhD, NNP, CLC, founder of Infant Feeding Care and developer of the NeoEAT Questionnaires for assistance with questionnaire translation, and Nicole Cooper of Cooper Johnson Communications, LLC, for providing medical writing support (which was funded by Nestlé). Portions of these results were presented in abstract form at the Excellence in Pediatrics conference in Amsterdam, the Netherlands, in December 2022.
Author Contributions
NPH, LL, MS, YV, and MM designed the study; R. Alenazi, R. Alzaatreh, IS, and MM conducted the research, LL analyzed the data; NPH wrote the first draft of the manuscript; all authors revised it critically for important intellectual content; and all authors provided approval of the final manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors NH, LL, MS, IS, and YV are employed by Société des Produits Nestlé S.A, the sponsor of the study. MM received funding from the sponsor to conduct this research. The remaining authors have no conflicting interests to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Société des Produits Nestlé S.A. Scientific representatives of the funding body were involved in the design of the study and in the analysis and interpretation of data.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
