Abstract
Biliary atresia is a common cause of cholestasis in infants and is a time-sensitive diagnosis. A survey was distributed to pediatric primary care providers in order to assess variations in diagnosis and management of cholestasis. Participants were identified from physician parent groups on social media and regional pediatric residency programs. Information on knowledge and interpretation of screening tests, past experience/behavior, confidence, and comfort level managing cholestasis, as well as demographic information was collected. Out of 116 eligible respondents, 94.8% were confident in diagnosing hyperbilirubinemia but only 10.3% knew the biochemical definition of direct hyperbilirubinemia. Of the 56% of providers who had some knowledge of the guidelines, 18.5% stated the guidelines changed the way they evaluate cholestasis. These results demonstrate a gap in knowledge of diagnosing and evaluating cholestasis, which could provide the framework for standardized screening, leading to earlier identification of biliary atresia.
Introduction
Jaundice is a common clinical finding in the neonatal period (infants <28 days old) and is a normal finding in most newborns within the first 2 to 3 days of life. However, jaundice present at <24 hours or >14 days old, especially in a formula-fed infant, is concerning for pathologic causes and warrants further evaluation. Jaundice can be due to conjugated (direct) or unconjugated (indirect) hyperbilirubinemia. Conjugated hyperbilirubinemia is uncommon and usually caused by impaired bile acid flow and conjugated bilirubin excretion into bile, also known as cholestasis. Presence of cholestasis indicates hepatobiliary dysfunction and is diagnosed by elevated serum direct bilirubin levels. Hyperbilirubinemia in newborns >35 weeks’ gestation is defined as total bilirubin level >95th percentile,1,2 whereas direct hyperbilirubinemia (a surrogate marker of cholestasis) is defined as a serum level >1.0 mg/dL (17 mmol/L) in the setting of an elevated serum total bilirubin level.3,4
In a newborn with concern for cholestatic jaundice, urgent evaluation and/or referral is imperative as appropriate intervention can have an impact on clinical outcome. Biliary atresia (BA) is the most common identifiable cause of cholestatic jaundice and indication for liver transplant in children. 5 The treatment for BA is Kasai portoenterostomy, and in the United States, most infants undergo this procedure after 60 days of life.5,6 This surgery performed in infants <60 days old results in better outcomes, specifically improved survival with native liver.5,7
In the continental United States, BA has a prevalence of 1/12,000 to 1/19,500 live births and an incidence of 6.5/100,000 to 7.5/100,000 live births.8,9 Diagnosing BA relies on persistently elevated serum direct bilirubin levels and a history of acholic stools. Although direct bilirubin levels are also elevated shortly after birth and remain elevated on subsequent laboratory draws,10,11 clinical presentation varies. Screening for BA is challenging as there are no screening guidelines that currently exist in the United States and results of previous scoring systems have been suboptimal.12 -14 Stool cards have shown promising results in Taiwan and British Columbia15,16; however, lack of standardized screening in the United States provides a challenge to pediatric primary care providers as to the best way to manage and diagnose cholestasis.
The North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition (NASPGHAN) and the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) recently published updated guidelines on the evaluation of cholestatic jaundice in infants. Historically, clinical practice guidelines have not had a significant effect on changing how providers practice.17,18 It is important that community pediatric primary care providers are aware and familiar with these guidelines as a systematic approach when evaluating for cholestasis can help shed light on the cause of delay in the diagnosis of BA.
Methods
The study was designed as a cross-sectional electronic survey, approved by the Institutional Review Board at Rhode Island Hospital. Questions were developed based on the cholestatic jaundice guidelines published by members of NASPGHAN/ESPGHAN and reviewed by pediatric gastroenterologists at our institution for accuracy and functionality prior to distribution. Study participants were identified from regional pediatric residency programs in Rhode Island, Connecticut, and Massachusetts, as well as through the physician parent Facebook groups, Physician Moms’ Group, and Physician Dads’ Group (Figure 1). Requirements to join either group involves verification by a current member and he or she must hold a medical degree (MD/DO/MBBS). Further verification is also performed through other professional social media accounts (Doximity, LinkedIn). Physician Moms’ Group also requires applicants to submit credentials (photo badge, medical school diploma, and residency training certificate). The electronic survey was designed using REDCap software. 19 Ethics approval was not required and consent for participation was implied by completion of the survey. After the initial email inviting providers to participate in the survey, there was 1 reminder email sent 2 weeks later. Participants were excluded if they were not a pediatric primary care provider taking care of infants <28 days old.
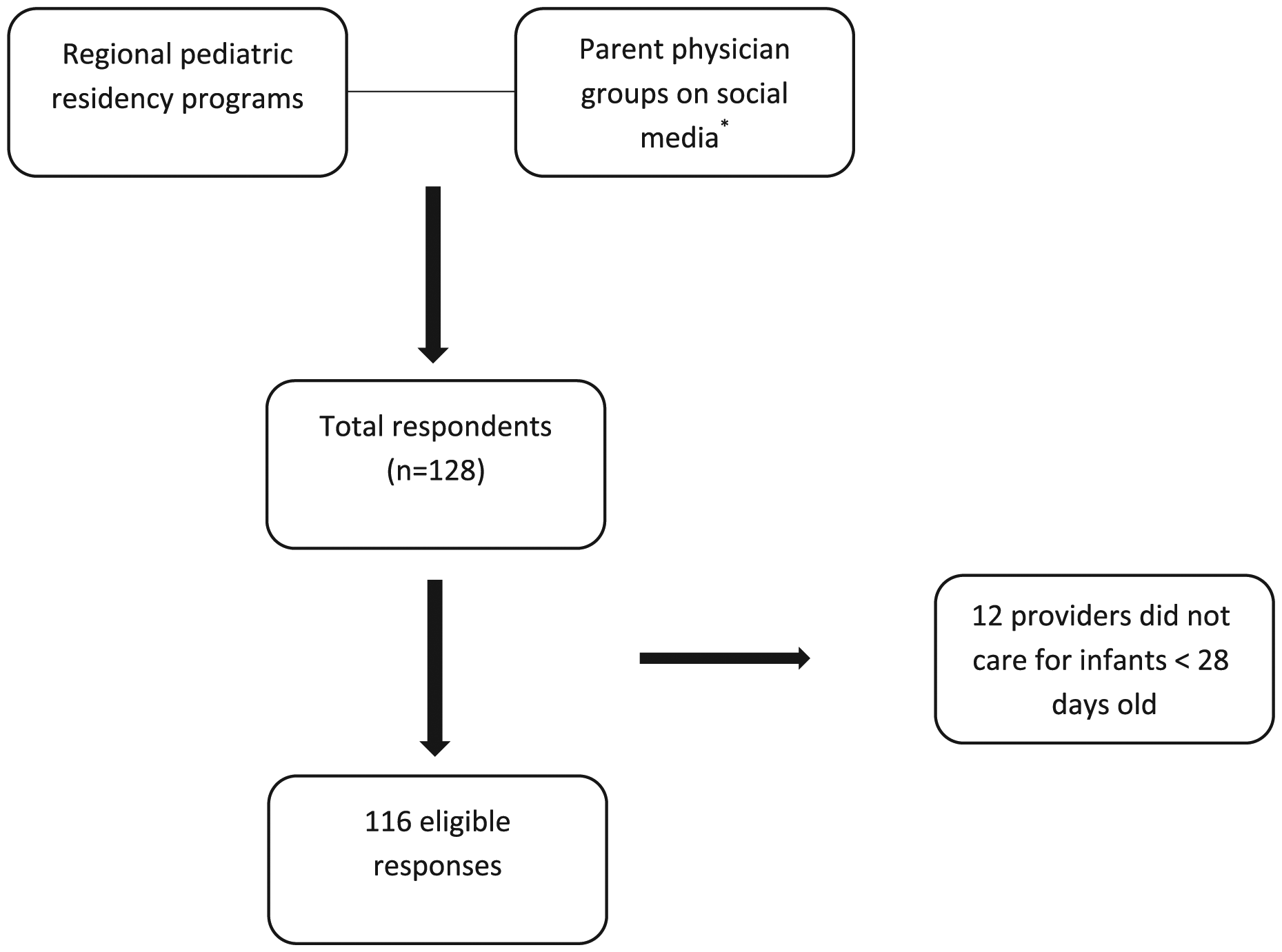
Survey participants.
The survey contained 20 questions and required less than 5 minutes to complete. There was no incentive to participate (survey available online as Supplementary Material). The survey included multiple-choice and Likert-type scale questions. Information on knowledge and interpretation of appropriate screening tests, past experience/behavior, and confidence/comfort level managing cholestasis, as well as demographic information was collected. Providers were also asked their level of knowledge regarding the recently published guidelines on cholestatic jaundice by NASPGHAN and ESPGHAN. At the completion of the survey, providers were directed to a 1-page summary of the guidelines on cholestatic jaundice and a link to the full article (available online as Supplementary Material).
Data were compiled and analyzed through the REDCap software program to characterize the overall study population. No identifying information was connected to the surveys.
Ethics Approval and Informed Consent
This study was approved by the Institutional Review Board at Rhode Island Hospital. Ethics approval was not required and written consent was waived by the institutional review board as the research presents no more than minimal risk of harm to subjects and involves no procedures for which written consent is normally required outside of the research context. All subject information was deidentified and subjects had the option to stop participation at any point if they found any questions offensive or disturbing. Knowledge, attitudes, and practice were not disclosed to departmental chairs or clinical supervisors. Completion of the survey served as consent to participate in the study.
Results
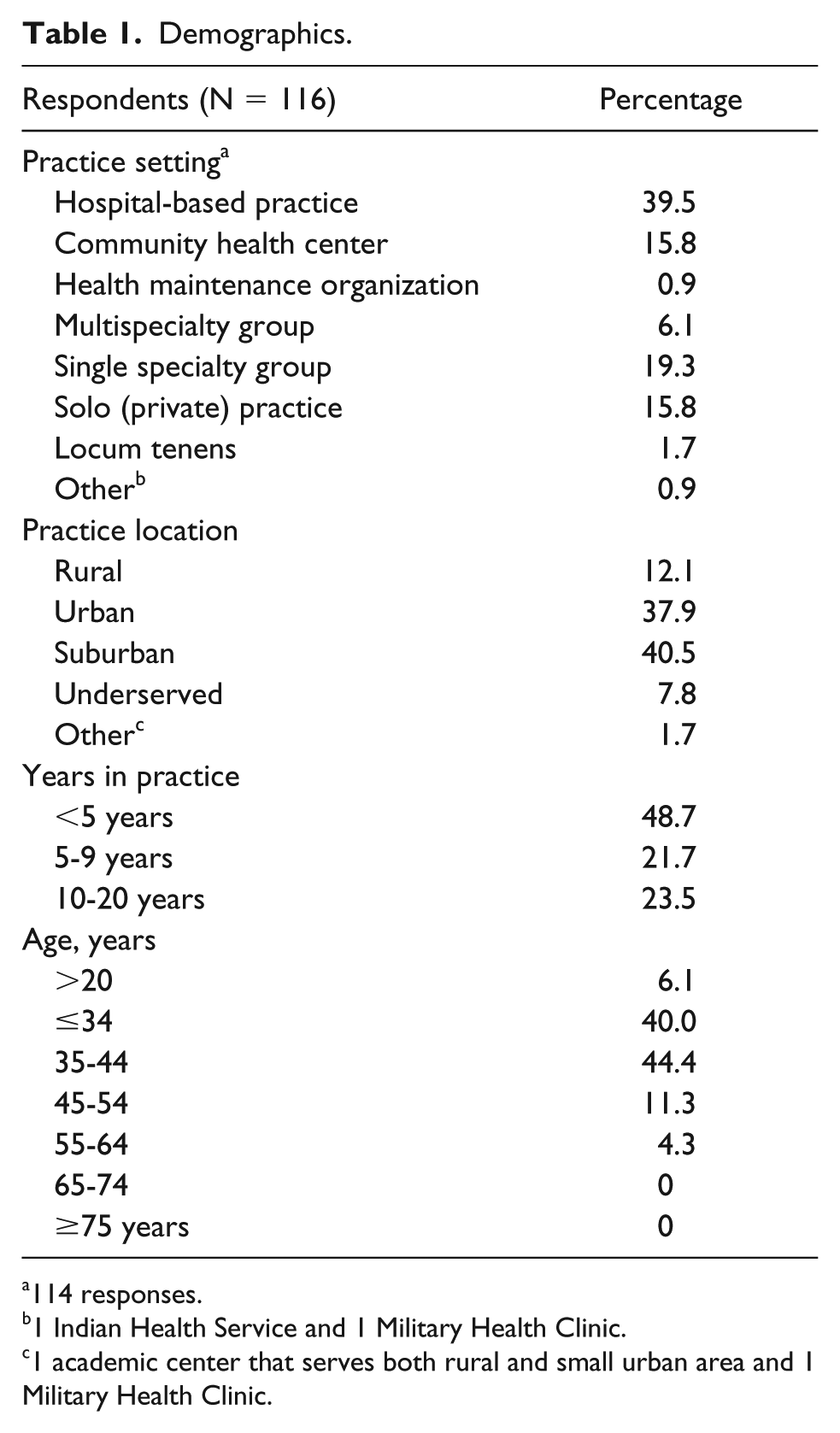
There were 128 total respondents, 12 who were ineligible to participate based on the initial survey question asking if the participating provider cared for infants younger than 28 days old, resulting in 116 eligible responses. Demographics of the 116 eligible participants is illustrated in Table 1. Providers participating in the survey were from varying practice settings with <2% from health maintenance organizations or representing locum tenens. Most providers were from urban or suburban settings (78.4%). A majority of providers had been in practice for less than 5 years (48.7%) and were less than 45 years of age (84.4%).
Demographics.
114 responses.
1 Indian Health Service and 1 Military Health Clinic.
1 academic center that serves both rural and small urban area and 1 Military Health Clinic.
Providers were asked 6 questions regarding past behaviors and patterns on diagnostic testing and medical knowledge regarding cholestasis (Table 2). A majority of providers felt that the most important condition to be ruled out in a newborn (<28 day old) with jaundice for >2 weeks duration is BA (80.2%). Most providers (73.3%) relied on a serum-fractionated bilirubin level when evaluating for possible cholestasis in a newborn, but not all agreed on the biochemical definition of direct hyperbilirubinemia (a surrogate marker of cholestasis). A total of 89.7% of providers were familiar with the biochemical definition of direct hyperbilirubinemia; however, 63.8% believed it to be defined as >20% of the serum total bilirubin and 25.9% defined it as a direct bilirubin >1.0 mg/dL. Most providers were appropriately concerned about jaundice if present at 1 to 2 weeks of age regardless of diet (62.9%) and 44.3% of providers always saw infants for a 4-week follow-up while only 54.7% always or often saw infants for a 4-week follow-up. A majority of providers (63.8%) believed that surgery for BA improved outcomes if performed before 60 days of life; however, 30.2% of providers did not know at what age surgery should be performed once BA is diagnosed.
Provider Patterns Working up Cholestasis.

Abbreviation: HIDA, hepatobiliary iminodiacetic acid.
Did not have enough clinical information to answer question.
2 respondents did not know.
1 respondent wrote >2 mg/dL and 1 respondent left the field blank.
Providers were asked if they were confident/comfortable evaluating, diagnosing, and managing cholestasis using a Likert-type scale from 1 (“strongly disagree”) to 5 (“strongly agree”; Table 3). A total of 94.8% of providers who participated in the survey were confident in diagnosing hyperbilirubinemia but only 70.7% were confident on what tests to order to assess for cholestasis. Of the survey respondents, 87.1% were comfortable interpreting the serum-fractionated bilirubin level but 28.5% and 17.8% of providers were not comfortable determining if stool is acholic or knowing when to refer to pediatric gastroenterology, respectively.
Confidence Regarding Workup for Cholestasis.

Knowledge of the NASPGHAN/ESPGHAN guidelines on cholestatic jaundice was assessed using a Likert-type scale ranging from 1 (no knowledge at all) to 5 (very knowledgeable). Fifty-six percent of providers had some knowledge of the guidelines (Likert “2” or greater) and 44% had no knowledge at all (Figure 2). Of the providers who had some knowledge of the guidelines, only 18.5% stated the guidelines changed the way they workup cholestasis.

Knowledge of NASPGHAN/ESPGHAN guidelines on cholestatic jaundice.
Discussion
Based on the responses, results demonstrate an overall gap in knowledge regarding the guidelines on cholestatic jaundice. Before completing the survey, only 56% of providers were familiar with the NASPGHAN/ESPGHAN guidelines on cholestatic jaundice. These results are suboptimal as pediatric primary care providers are responsible for the timeliness of workup and referral. For those providers who had some knowledge of the guidelines, a majority (>80%) did not report that the guidelines changed the way they evaluate cholestasis. This is problematic but is not an uncommon phenomenon. Petrova et al demonstrated that although the majority of a cohort of pediatricians in New Jersey were adherent to the 1994 guidelines on the management of neonatal hyperbilirubinemia developed by the American Academy of Pediatrics (AAP), there was variability in screening, follow-up, and treatment strategies. 20 Profit et al also discovered a wide variety in adherence to the 2004 AAP guidelines on management of severe hyperbilirubinemia in healthy full-term infants, resulting in delayed follow-up. 21
The variability of medical knowledge, diagnostic testing, and management of cholestasis, as illustrated in this study, can have a profound effect on the timing of diagnosing BA. Although the majority of providers felt that BA was the most important condition that needed to be ruled out after 2 weeks of prolonged jaundice, almost one third of providers did not know at what age surgery should be performed once BA is diagnosed. This lack of knowledge minimizes the sense of urgency, which can lead to a delay in BA diagnosis. A total of 87.1% of providers were comfortable interpreting the serum-fractionated bilirubin level but only 70.7% of providers were confident on what tests to order, highlighting another gap in knowledge as serum-fractionated bilirubin level is the first screening test for a newborn with prolonged jaundice based on the NASPGHAN/ESPGHAN guidelines. Even in the cases with confident pediatricians as the survey results demonstrate, evaluation for cholestatic jaundice remains variable with areas of deficits that need improvement. The fact that 28.5% of providers were not comfortable determining if stool was acholic again reemphasizes the importance of increasing awareness of the guidelines as this is a key finding when investigating for cholestasis.
This study did have its limitations. First, the lack of monetary incentive or continuing medical education offered could have limited the respondents. Similarly, there could have been a selection bias favoring providers who are more, or less, interested and knowledgeable about hyperbilirubinemia and cholestasis. A majority of respondents were younger than 45 years of age suggesting an increased comfort level with technology, web-based surveys, and/or social media in this younger age group. Additionally, there is always potential for various interpretations regarding the language and terms used in each survey question, affecting providers’ responses by not accurately reflecting their true thoughts or beliefs.
Screening for cholestasis is challenging but these results could potentially help identify areas of improvement to direct resources (distributing guidelines, identifying appropriate screening tools) in order to identify cholestasis earlier. Although our study did not evaluate patients with cholestasis or those diagnosed with BA, additional studies would be helpful comparing how knowledge of the guidelines on cholestatic jaundice affect the timing of testing, follow-up, and referrals. With increased awareness of the guidelines on cholestatic jaundice and appropriate timing of testing and referral, BA can be identified in a timely fashion, which can lead to improved outcomes including decreased morbidity, increased survival, and decrease in health care cost.
Supplemental Material
Checklist_Internet_E-Surveys_(CHERRIES)_MENZ_05OCT18 – Supplemental material for Knowledge, Attitudes, and Behaviors of Pediatric Primary Care Providers on Management of Cholestasis
Supplemental material, Checklist_Internet_E-Surveys_(CHERRIES)_MENZ_05OCT18 for Knowledge, Attitudes, and Behaviors of Pediatric Primary Care Providers on Management of Cholestasis by Timothy Joseph Menz, Michael Herzlinger, Albert Ross and Mark R. Zonfrillo in Global Pediatric Health
Supplemental Material
Cholestasis_survey_MENZ_06OCT18 – Supplemental material for Knowledge, Attitudes, and Behaviors of Pediatric Primary Care Providers on Management of Cholestasis
Supplemental material, Cholestasis_survey_MENZ_06OCT18 for Knowledge, Attitudes, and Behaviors of Pediatric Primary Care Providers on Management of Cholestasis by Timothy Joseph Menz, Michael Herzlinger, Albert Ross and Mark R. Zonfrillo in Global Pediatric Health
Footnotes
Author Contributions
TJM: Contributed to conception or design; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy
MH: Contributed to conception or design; contributed to acquisition, analysis, and interpretation of data; critically revised the manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
AR: Contributed to conception or design; contributed to acquisition of data; critically revised the manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
MRZ: Contributed to conception or design; contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
