Abstract
Introduction
Children with developmental delays often experience oral-motor dysfunction, which can lead to choking during feedings and aspiration. Long-term, these feeding issues often lead to poor nutritional and health status; thus, interventions to improve nutritional intake are needed.1-3 Nutritional interventions often include the use of enteral feedings via tube to increase intake and improve growth.4,5 While initiation of enteral feedings often leads to improved nutritional status, intolerance of feedings in this population is common.4,5 These feeding intolerances are often a result of gastrointestinal dysmotility, such as esophageal dysmotility, delayed gastric emptying, and altered intestinal motility, and often present by gagging, retching, reflux, vomiting, and abdominal distention.2,6 In addition, vomiting, constipation, and diarrhea are also commonly identified. As a result of intolerance, nutritional goals are often compromised leading to inappropriate growth and development.4,5 Identifying appropriate interventions to treat feeding intolerances is imperative to support the child’s growth and development. Nutritional management of feeding intolerance in developmentally delayed children is by necessity highly individualized and may include the use of different enteral formulas and formula modulars. Whey-based formulas have been shown to empty from the stomach more rapidly than casein-based formulas, which may be helpful for patients presenting with delayed gastric emptying.3,7 Furthermore, improved absorption of hydrolyzed over intact proteins may be observed in certain patient populations.
Use of peptide-based, 100% whey protein formulas are associated with improved feeding tolerance, increased consistency in meeting nutritional needs, and a reduction in gastrointestinal issues associated with vomiting and aspiration of feeds. 7 Peptide-based diets are often prescribed to patients with gastrointestinal illnesses such as pancreatic insufficiency or malabsorption. These diets may also be used in children with developmental delays who do not consistently have definitive diagnoses of gastrointestinal impairment. Lack of evidence of benefit in this population may limit their application. Only one small prospective trial assessed the effects of switching from an intact casein-predominant formula to different whey-predominant formulas on frequency of emesis in developmentally delayed children and reported significant reductions in emesis after children were switched to whey-based formulas. 7
Crossover trials to determine improved tolerance to a change in diet are challenging and particularly difficult in children that are almost exclusively enterally fed. In fact, only one other abstract explored multiple tolerance outcomes related to switching the patient from one formula to another. 8 The primary objective of this retrospective chart review was to evaluate changes in feeding tolerance in children with developmental delays who were switched from an intact protein-based enteral formula to a 100% whey, peptide-based formula.
Materials and Methods
Medical records of children were selected for review if they met the following criteria: (1) exhibited significant developmental delays, (2) were receiving at least 90% of energy requirements through a gastrostomy tube, (3) were considered by the primary practitioner to have significant signs of intolerance, and (4) were switched from an intact protein formula to a 100% whey, peptide-based formula (Peptamen products, Nestlé Health Science, Florham Park, NJ). Additional criteria included the following: (1) age 1 to 18 years at time of formula change, (2) received an intact protein formula for a minimum of 2 weeks, and (3) did not have any extraneous factors influencing feeding tolerance (change of tube location or abdominal surgery, acute illness or infection, or documented cow’s milk protein allergy) within 30 days of feeding change.
Demographic data including age, diagnoses, and gender were collected. Data on feeding intolerances, such as vomiting, constipation, diarrhea, poor weight gain, and feeding volume, were also gathered pre- and post-formula change. Medications utilized to facilitate feeding tolerance and for bowel and use of nutritional modular products (such as protein) were also captured. Change in tolerance as assessed by the health care provider was categorized as “Improved,” “No change,” or “Worsened.”
The study protocol was approved by Copernicus Group Independent Review Board (CGIRB), Durham, North Carolina. As this was a retrospective chart review, prior to study initiation, the investigator requested and CGIRB approved a request for waiver/alteration of authorization for use and disclosure of protected health information.
Statistical Analyses
Demographic data were summarized using means ± standard deviations and percentages for continuous and categorical data, respectively. All data were analyzed using SAS version 9.4.
Results
Subject Characteristics
A total of 375 charts were reviewed. Medical records of 13 children met eligibility criteria and were included in analyses. Subjects were aged 8.4 ± 4.6 years, and 53.8% were female. The majority of subjects were fed by gastrostomy tube (77%). Of these, 85% had a Nissen fundoplication (Table 1). All subjects had a primary diagnosis of developmental delay (Table 2). Intolerance experienced by the subjects included vomiting (53.8%), gagging and retching (30.7%), high gastric residual volumes (61.5%), constipation (53.8%), diarrhea (23.1%), and poor weight gain (38.5%). The children were switched from an intact polymeric formula to one of the following peptide-based, 100% whey formulas: Peptamen Junior (n = 6), Peptamen Junior 1.5 (n = 6), and Peptamen Junior Prebio (n = 1).
Demographics.
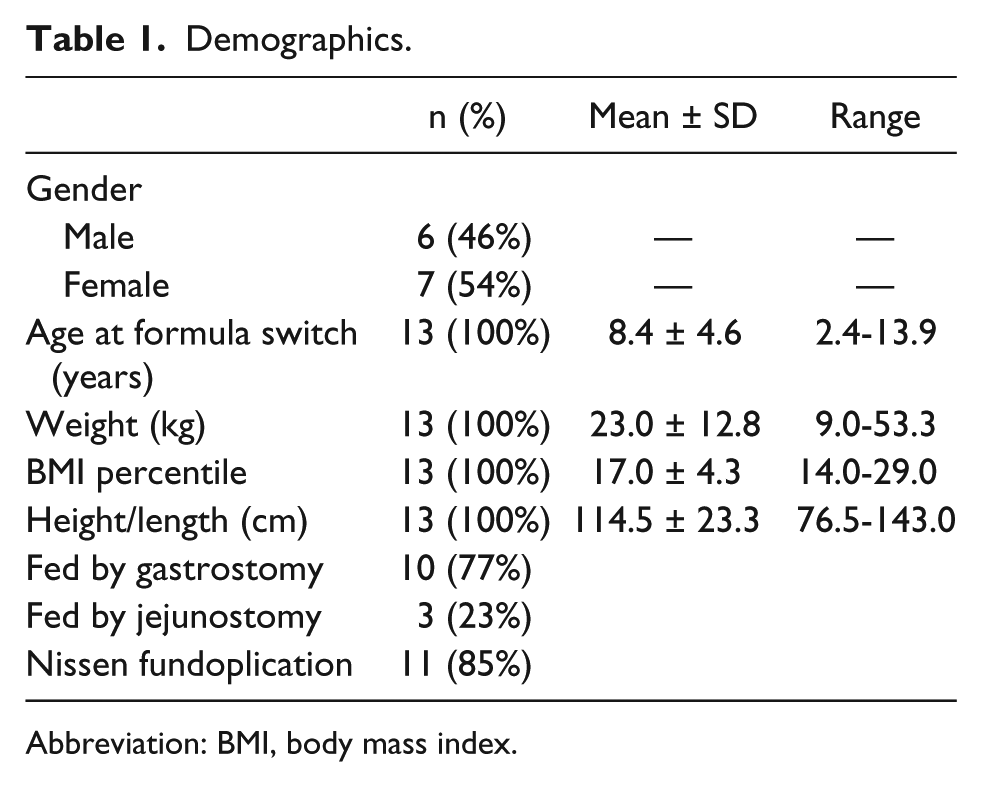
Abbreviation: BMI, body mass index.
Patient Diagnoses.
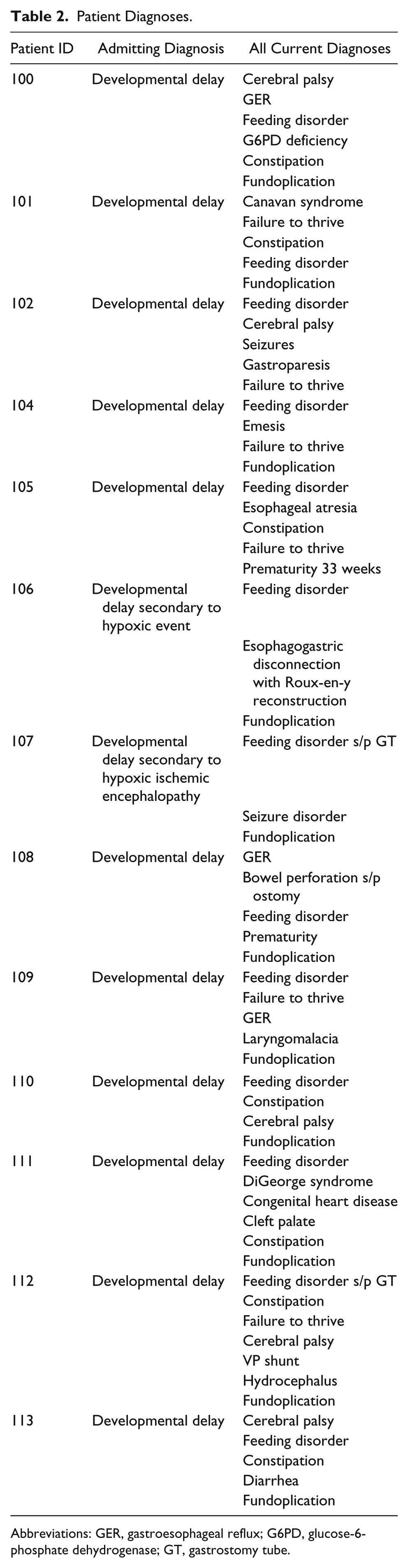
Abbreviations: GER, gastroesophageal reflux; G6PD, glucose-6-phosphate dehydrogenase; GT, gastrostomy tube.
Tolerance Parameters
Of the included subjects, 92% (12 of 13 subjects) demonstrated improved feeding tolerance that was attributed to the change to 100% whey peptide formula. Furthermore, 75% (9 of 12 subjects) of these reported improvements occurred within 1 week after the formula change. The remaining subject also had an improvement in feeding tolerance; however, the improvement was attributed to the venting of the stomach via gastrostomy, which was implemented at the same time as the formula change. In subjects with specific feeding intolerance issues, feeding tolerance improvements were also observed in vomiting (86%, n = 6), gagging and retching (75%, n = 4), high residual volumes (63%, n = 8), constipation (43%, n = 7), and diarrhea (100%, n = 3; Figure 1).

Tolerance parameters changes.
Medication Use
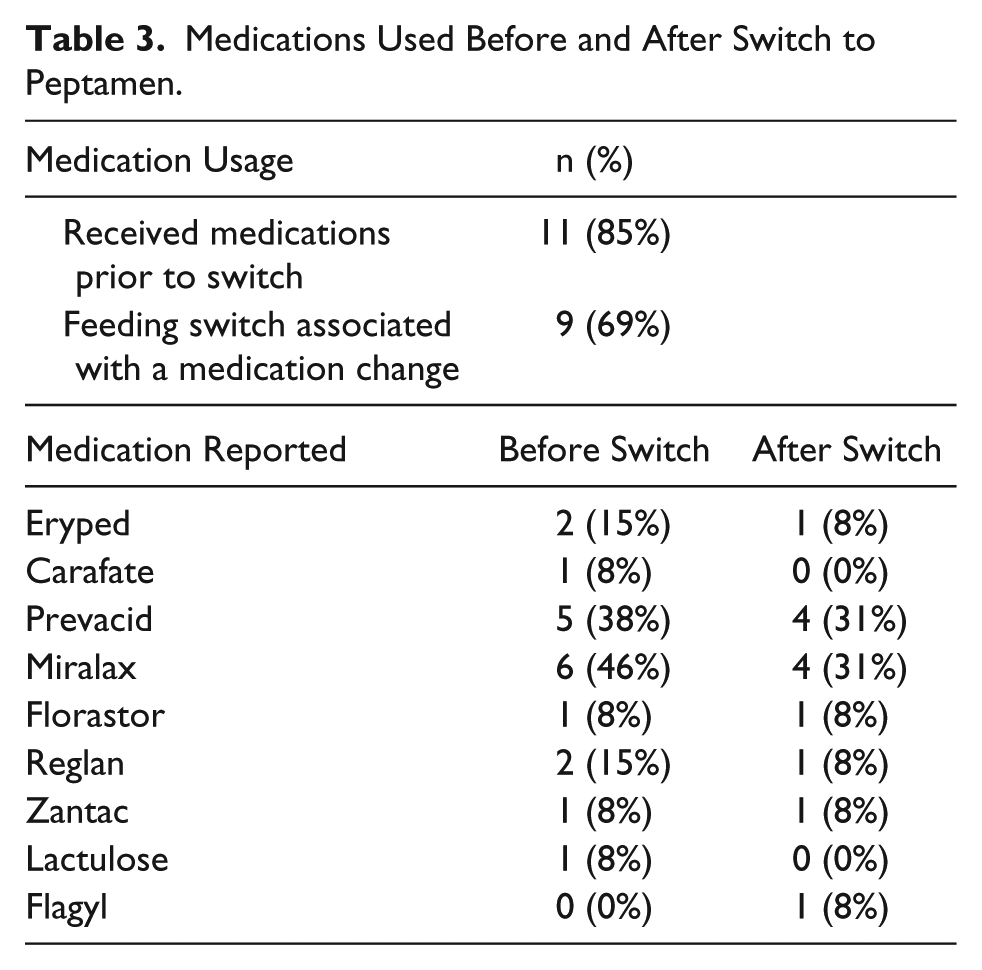
Medical record reviews identified 8 medications used to manage feeding intolerance. Prior to formula change, 11 (85%) subjects received at least one of these medications (1.54 ± 0.27). After the switch to a 100% whey, peptide-based formula, medication dosage was either stopped or decreased in 81.8% of subjects. Of the 8 medications used prior to formula change, 6 (66.6%) were used in fewer subjects (Table 3). In no cases was there a need to increase the use of medications prescribed for intolerance once the change to a 100% whey, peptide-based diet was made.
Medications Used Before and After Switch to Peptamen.
A consistent improvement in meeting nutritional goals was also observed; 71% were able to tolerate increased feeding volumes and all subjects who had experienced poor weight gain on an intact protein formula achieved an increase in weight after formula change (see Table 4 and Figure 1).
Intolerance Signs and Assessment of Status After Formula Switch.

Discussion
The purpose of this retrospective chart review was to evaluate changes in tolerance parameters in children with developmental delays who were switched from an intact protein formula to a 100% whey, peptide-based formula. Results from this review found that children experiencing significant gastrointestinal signs and symptoms of intolerance benefited from this change in formula. Whey-based, semi-elemental formulas are used sporadically in children with developmental delays and gastrointestinal issues hindering feeding tolerance. This retrospective review highlights the gastrointestinal and nutritional improvements that can be achieved with these diets.
Gastrointestinal intolerance is frequent in patients with developmental delays and can negatively affect these patients in many different ways.1,2 Intolerance can cause physical discomfort, growth failure, and can necessitate the use of medications with potentially significant side effects. Feeding intolerance also imposes burdens to caregivers, requiring additional time, financial resources, and affecting quality of life.
Most of the intolerance reported in these children, however, has traditionally focused on signs and symptoms of upper gastrointestinal dysmotility including impaired gastric emptying and gastroesophageal reflux. This study demonstrates that in addition to upper gastrointestinal problems, intolerance phenomena distal to the upper gastrointestinal tract are common and may be improved by changes in forms of feeding. The results of this exploration corroborate with previous reports that the use of 100% whey, peptide-based diets can improve signs and symptoms of upper gastrointestinal intolerance, specifically decreases in vomiting and gastric residuals.7,9 In addition, this review provides evidence of further benefit documenting improvements in other parameters of gastrointestinal tolerance, namely, constipation and diarrhea.
Several studies have evaluated diet modification to improve gastric dysmotility and reflux.1-7 For example, 2 trials demonstrated improvements in gastric emptying using whey-based versus casein-predominant formulas.3,7 One of these trials also reported reduced vomiting 7 with the use of whey-predominant or 100% whey, peptide-based formulas versus casein-predominant formulas.
Medications to improve feeding tolerance are often prescribed for patients with developmental delays. 10 These medications (like many medications) may have adverse effects, increase the complexity of care, and add to treatment cost. In this study, medications were used to promote motility, manage gastric acidity, and improve stool consistency. Use of 100% whey, peptide-based diets reduced the need for these medications, yielding an additional benefit of their use in enterally fed children with developmental delay and feeding intolerance.
In the presence of significant intolerance, feeding volumes and concentrations cannot be easily advanced. Perhaps the most prominent observation in this exploration was the improvement in achievement of nutritional goals. Meeting nutritional goals is critical for growth and for preventing nutrition deficiencies that affect overall health.
At present, little data exist to support clinicians in their selection of enteral formula for use in children with developmental delays and feeding intolerance. Despite the limited published evidence, clinicians anecdotally use 100% whey, peptide-based formulas when intolerance occurs. To our knowledge, this is the first report to objectively document improvements in tolerance parameters following a change from intact protein feedings to a 100% whey, peptide-based formula, thus providing substantiation for use of such a formula in this population.
This study has a number of strengths. All included subjects were switched from an intact protein formula to a 100% whey, peptide-based formula; thus, each subject served as his or her own control. Second, detailed medical records on tolerance and growth parameters existed for all included subjects. Finally, as children with developmental delays are a somewhat small segment of the population, the use of a retrospective study design allowed for subjects to be pooled and analyzed over a longer period of time, allowing for a larger sample size than may have been possible using a prospective study design.
Study limitations include the retrospective chart review design; thus, by nature, the results cannot prove causality. Nevertheless, it is improbable that the clinical benefits observed by switching patients to a 100% whey, peptide-based formula occurred randomly. The population captured in this sample was small and homogenous, which limits the ability to generalize the results to a larger population. However, this is a population with significant needs that are clearly differentiated from healthy children. Finally, while qualitative data on feeding volumes were present, consistent quantitative data on feeding volumes and energy intake were not available; thus, it was not possible to compare exact volume and energy intakes prior to and after formula changes.
In conclusion, changing from an intact protein enteral feeding to a 100% whey, peptide-based formula improved multiple symptoms of feeding intolerance in a population of children with developmental delays. Most improvements occurred within a week of formula change. Change in the formula was associated with a reduction in medications and improved growth. Clinical practice in switching patients with developmental delays and intolerance to a 100% whey, peptide-based formula may generate significant clinical benefits in these patients. Future research using a more rigorous study design and larger sample would be beneficial, but significantly difficult to perform.
Author Contributions
GM: Contributed to conception and design; contributed to acquisition; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
SP: Contributed to analysis and interpretation; drafted manuscript; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
JBO: Contributed to analysis and interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
HS: Contributed to conception and design; contributed to acquisition, analysis, and interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Footnotes
Author Note
Current affiliation for Heidi Storm is Nestle Nutrition, Florham Park, NJ
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Ochoa and Ms Periman are employees of Nestle Health Science in Florham Park, NJ. Ms Storm was employed at Nestle Health Science in Florham Park, NJ, at time of study completion and is now employed at Nestle Nutrition in Florham Park, NJ.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Nestlé Healthcare Nutrition, Florham Park, NJ.
