Abstract
Objective
Enteral immunonutrition is a nutritional intervention that has been studied in postoperative patients with gastric cancer, but its effectiveness is controversial. This study aimed to investigate the effects of enteral immunonutrition and enteral nutrition on immune function in patients who undergo gastric cancer surgery.
Methods
We performed a systematic review and meta-analysis. A comprehensive search was conducted in PubMed, Embase, Cochrane, Web of Knowledge, and ClinicalTrials.gov from the inception of the review until 10 March 2023. Twelve studies were included for qualitative and quantitative analyses.
Results
We studied 1124 patients, including 565 patients in the enteral immunonutrition group and 559 in the enteral nutrition (controls) group. All included randomized, controlled trials were high quality. CD4+ levels, lymphocytes, transferrin concentrations, and systemic inflammatory response syndrome were not significantly different between the enteral immunonutrition and enteral nutrition groups. However, CD8+, immunoglobulins G and M, and proalbumin concentrations, CD4+/CD8+, and infectious complications were significantly higher in the enteral immunonutrition group than in the enteral nutrition group. A sensitivity analysis showed consistent results after excluding each study. Begg’s test showed no publication bias.
Conclusions
Enteral immunonutrition is an effective nutritional intervention that improves immune function in patients who have undergone gastric cancer surgery.
Keywords
Introduction
Gastric cancer is a frequent malignant gastrointestinal (GI) tumor that seriously threatens human health and life. The incidence and mortality rates of gastric cancer are ranked fifth and fourth, respectively, among all the cancers globally. 1 Adenocarcinoma is the most common histological type of gastric cancer (89.7%), followed by GI stromal tumors (4.6%). 2 Currently, surgery is the principal treatment plan for gastric cancer. 3 Patients with gastric cancer often suffer from malnutrition due to poor nutrient absorption and active cancer cell metabolism, and surgical treatment also aggravates the degree of malnutrition.4,5 Therefore, malnourished patients tend to have an elevated risk of delayed postoperative recovery, infectious complications, and a prolonged hospital stay.6,7 Malnutrition is also associated with substandard overall, cancer-specific, and recurrence-free survival. 8
The prognosis of patients who require gastrectomy can be improved by perioperative enteral nutrition (EN) or parenteral nutrition (PN). 9 Supplemental nutrition via enteral or parenteral feeding is believed to be an essential adjunctive therapy for individuals undergoing surgery. The selection of EN or PN is based on the individual’s gut function and nutrient supply pattern tolerance. If the patient’s intestinal status allows, EN is more efficient, comparable to the patient’s physiological characteristics, and has lower complications and costs than PN. Although EN provides essential protein, energy, carbohydrates, minerals, fat, and vitamins, its effect is not as good as expected.10,11 Recently, enteral immunonutrition (EIN), including ω-3 fatty acids, arginine, glutamine, nucleotides, and other nutritional formulations, has attracted great attention from nutritionists and surgeons. EIN is considered a nutritional preparation that can not only provide required energy, but also regulate immune function. 12 Arachidonic acid is replaced by ω-3 fatty acids in the cell membrane and regulates immune and anti-inflammatory processes.13,14 The safety and efficacy of ω-3 fatty acids have been observed in liver resection, critically ill patients, and patients with colorectal surgery.15–17 Although the effects of EIN on clinical outcomes and the immune and nutritional status are convincing, they are still controversial. 18 Giger et al. 19 showed a substantial decrease in serum C-reactive protein and tumor necrosis factor-α concentrations postoperatively in patients with malignancy in the GI tract in the EIN group. Evidence suggests that n-3 polyunsaturated fatty acid-enriched nutritional regimens efficiently and safely reduce the length of the hospital stay and postoperative complications,20,21 but this is controversial. 22 A systematic review on immunonutrition vs a standard diet in patients with cancer of the GI tract only discussed the duration of the hospital stay and morbidity due to post-surgical infectious complications. 23 Another meta-analysis reported the pooled results of EIN use in patients with gastric cancer, 24 but the search term for EIN only used the medical subject term “enteral immunonutrition,” and several articles were not included. The meta-analyses by Cheng et al. 25 and Fu et al. 26 only focused on patients with gastric cancer who underwent total gastrectomy, and some recent high-quality, randomized, controlled trials were excluded.
Therefore, this meta-analysis and systematic review of relevant, randomized, controlled trials was conducted to determine the effect of EIN and EN on immune-inflammatory factors, serum proteins, infectious complications, and cellular immunity in patients who have gastric cancer surgery. We also aimed to discuss the relevant literature on clinical strategies.
Methods
Search strategy
This investigation followed the statement of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 27 The procedure used was registered (INPLASY202340030) and is available at inplasy.com (https://doi.org/10.37766/inplasy2023.4.0030). The Embase, ClinicalTrials.gov Cochrane, PubMed, and Web of Knowledge databases were surveyed from inception to 10 March 2023. All languages were selected for the article search. PubMed was searched using the following terms: (“gastrointestinal stromal tumors”[MeSH Terms] OR (“gastrointestinal”[All Fields] AND “stromal”[All Fields] AND “tumors”[All Fields]) OR “gastrointestinal stromal tumors”[All Fields] OR (“gastrointestinal”[All Fields] AND “stromal”[All Fields] AND “tumor”[All Fields]) OR “gastrointestinal stromal tumor”[All Fields] OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“neoplasm”[All Fields] AND “stomach”[All Fields]) OR “neoplasm stomach”[All Fields]) OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“stomach”[All Fields] AND “neoplasm”[All Fields]) OR “stomach neoplasm”[All Fields]) OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“neoplasms”[All Fields] AND “stomach”[All Fields]) OR “neoplasms stomach”[All Fields]) OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“gastric”[All Fields] AND “neoplasms”[All Fields]) OR “gastric neoplasms”[All Fields]) OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“gastric”[All Fields] AND “neoplasm”[All Fields]) OR “gastric neoplasm”[All Fields]) OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“neoplasm”[All Fields] AND “gastric”[All Fields]) OR “neoplasm gastric”[All Fields]) OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“neoplasms”[All Fields] AND “gastric”[All Fields]) OR “neoplasms gastric”[All Fields]) OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“cancer”[All Fields] AND “stomach”[All Fields]) OR “cancer of stomach”[All Fields]) OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“stomach”[All Fields] AND “cancers”[All Fields]) OR “stomach cancers”[All Fields]) OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“gastric”[All Fields] AND “cancer”[All Fields]) OR “gastric cancer”[All Fields]) OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“cancer”[All Fields] AND “gastric”[All Fields]) OR “cancer gastric”[All Fields]) OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“cancers”[All Fields] AND “gastric”[All Fields]) OR “cancers gastric”[All Fields]) OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“gastric”[All Fields] AND “cancers”[All Fields]) OR “gastric cancers”[All Fields]) OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“stomach”[All Fields] AND “cancer”[All Fields]) OR “stomach cancer”[All Fields]) OR (“stomach neoplasms”[MeSH Terms] OR (“stomach”[All Fields] AND “neoplasms”[All Fields]) OR “stomach neoplasms”[All Fields] OR (“cancer”[All Fields] AND “stomach”[All Fields]) OR “cancer of the stomach”[All Fields]))) AND (((“enteral”[All Fields] OR “enterally”[All Fields]) AND (“immunonutrition diet”[MeSH Terms] OR (“immunonutrition”[All Fields] AND “diet”[All Fields]) OR “immunonutrition diet”[All Fields] OR “immunonutrition”[All Fields] OR “immunonutritional”[All Fields])) OR (“enteral nutrition”[MeSH Terms] OR (“enteral”[All Fields] AND “nutrition”[All Fields]) OR “enteral nutrition”[All Fields]) OR (“arginin”[All Fields] OR “arginine”[MeSH Terms] OR “arginine”[All Fields] OR “arginine s”[All Fields] OR “arginines”[All Fields] OR “argininic”[All Fields]) OR (“glutamin”[All Fields] OR “glutamine”[MeSH Terms] OR “glutamine”[All Fields] OR “glutamine s”[All Fields] OR “glutamines”[All Fields]) OR (“fatty acids, omega 3”[MeSH Terms] OR (“fatty”[All Fields] AND “acids”[All Fields] AND “omega 3”[All Fields]) OR “omega-3 fatty acids”[All Fields] OR “omega 3 fatty acid”[All Fields])). All of the potential and eligible articles were examined, regardless of their primary outcomes and language.
The inclusion criteria were the PICOS framework applied as eligibility criteria as follows: population (P), patients undergoing gastric cancer surgery; intervention (I), EIN; comparison (C), EN; outcomes (O), serum proteins, infectious complications, immune and inflammatory factors, and cellular immunity; and study design (S), randomized, controlled trials. The exclusion criteria were as follows: (1) articles lacking the effects of EIN and EN on serum proteins, immune and inflammatory factors, infectious complications, and cellular immunity in surgical patients with gastric adenocarcinoma or gastrointestinal stromal tumors; (2) articles on EIN and management beyond EN; (3) articles that compared outcomes and effects; (4) articles lacking experimental data; (5) articles comparing EIN with EN in primary tumors other than gastric cancer; and (6) composite articles on individuals with gastric cancer or other malignancies that lacked isolated results.
Data extraction and quality assessment
Two researchers (QLX and ARW) independently screened the articles by initially identifying the subject and standard subject selection, and then the abstract and the full text were read. The quality of randomized, controlled investigations was cross-estimated by two researchers using the Jadad scale, including random allocation, double-blind method setting, randomized hiding, exit, and loss to follow-up (7 points score: 1–3 = inferior quality and 4–7 = good quality) The quality of the method was assessed using the Cochrane Review handbook recommendations. Two researchers separately collected the necessary published data, such as the specificities of the first author, number of subjects, publication date, experimental and control groups’ mode of feeding, initiation and total time of nutritional support, and EIN formula. Any conflict of decision between the two researchers was resolved by a third researcher (JL).
Statistical analysis
The data were analyzed using Review Manager (5.3) (Cochrane Collaboration), and P < 0.05 indicated statistical significance. The odds ratio (OR) or mean difference (MD) and 95% confidence interval (CI) were computed with a fixed or random-effect model. Inter-study differences were assessed using the I2 statistic and Cochran’s Q test, where cut-off values of 25%, 50%, and 75% indicated low, moderate, and high, respectively. 28 In case of <50% for I2, the OR values were combined using the fixed-effects (Mantel–Haenszel) model; otherwise, the random-effect (DerSimonian and Laird) model was used. A sensitivity analysis was performed in relation to the assessed effect sizes and heterogeneity of the studies. The risk of publication bias was assessed using funnel plots, and asymmetry of the plot indicated potential bias. Asymmetry was analyzed by Begg’s test. Intercept significance was assessed using t-tests (P < 0.05).
Results
Characteristics of the included studies
Initially, 1389 articles were identified. After the removal of duplicates, 1186 articles were screened on the basis of their titles and abstracts, and only 47 articles met the eligibility guidelines. Of these, 35 articles were excluded for the following reasons after examination of the full text: 1) they lacked focus on postoperative indicators; 2) they lacked the outcomes of interest; 3) the full text was unable to be located; and 4) they had a quality rating of a score <2 on the Jadad scale. Finally, 12 studies9,13,29–38 were selected (Figure 1) and these included 1124 patients, with 565 in the EIN group and 559 in the EN (controls) group. Table 1 shows the details of the included studies.

Flow chart of the screening strategy for the included studies.
Main characteristics of all studies included in the meta-analysis.

EIN, enteral immunonutrition; Arg, arginine; Gln, glutamine; ω-3-FAs, ω-3 fatty acids; EPA, eicosapentaenoic acid; DHA, docosahexaenoic acid.
Quality assessment of the selected studies
The quality of the randomized, controlled trials, as assessed using the Jadad scale, was high (Table 2).
Quality assessment of included trials using the Jadad scale.
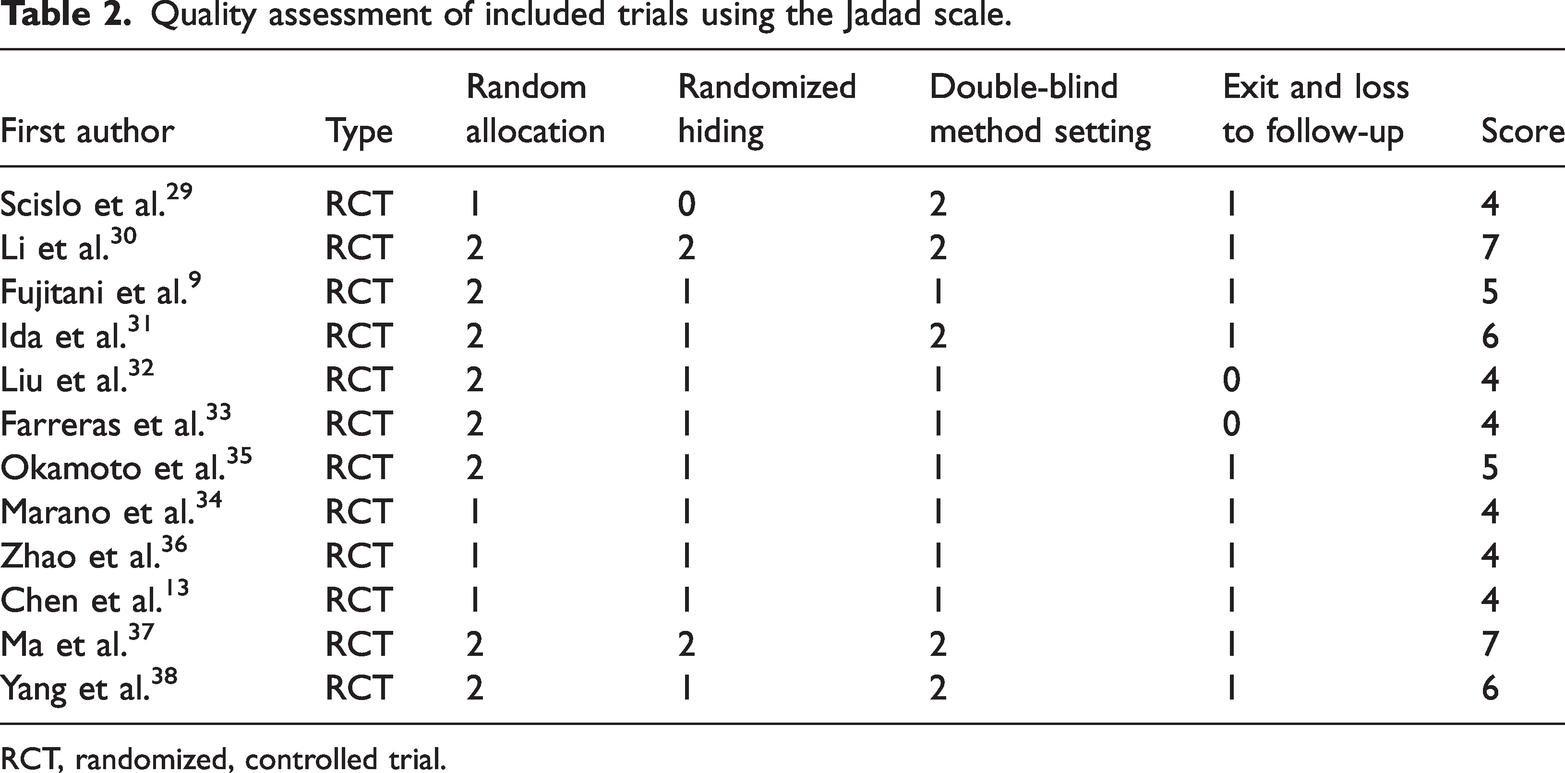
RCT, randomized, controlled trial.
Meta-analysis
CD4+ levels (MD: 2.92, 95% CI: −3.84 to 9.69, I2 = 99%, Figure 2a) were not significantly different between the EIN and EN groups. CD8+ levels (MD: 1.16, 95% CI: 0.06 to 2.26, P = 0.04, I2 = 0%, Figure 2b) and CD4+/CD8+ (MD: 0.48, 95% CI: 0.08 to 0.88, P = 0.02, I2 = 92%, Figure 2c) were significantly higher in the EIN group than in the EN group. Immunoglobulin (Ig) G (MD: 2.46, 95% CI: 1.47 to 3.46, P < 0.001, I2 = 86%, Figure 3a) and IgM (MD: 0.47, 95% CI: 0.44 to 0.50; P < 0. 001, I2 = 0%, Figure 3b) concentrations were significantly higher in the EIN group than in the EN group. Lymphocytes (MD: 0.01, 95% CI: −0.88 to 0.89, I2 = 100%, Figure 4a) and transferrin concentrations (MD: 0.03, 95% CI: −0.01 to 0.07, P = 0.14, I2 = 52%, Figure 4c) were not significantly different between the EIN and EN groups. However, proalbumin concentrations (MD: 24.98, 95% CI: 3.21 to 46.74, P = 0.02, I2 = 92%, Figure 4b) were significantly higher in the EIN group than in the EN group. Systemic inflammatory response syndrome (MD: −0.50, 95% CI: −1.4 to 0.89, I2 = 96%, Figure 5a) was not significantly different between the EIN and EN groups. However, the rate of infectious complications (OR: 0.61, 95% CI: 0.43 to 0.86; P = 0.005, I2 = 35%, Figure 5b) was significantly lesser in the EIN group than in the EN group.
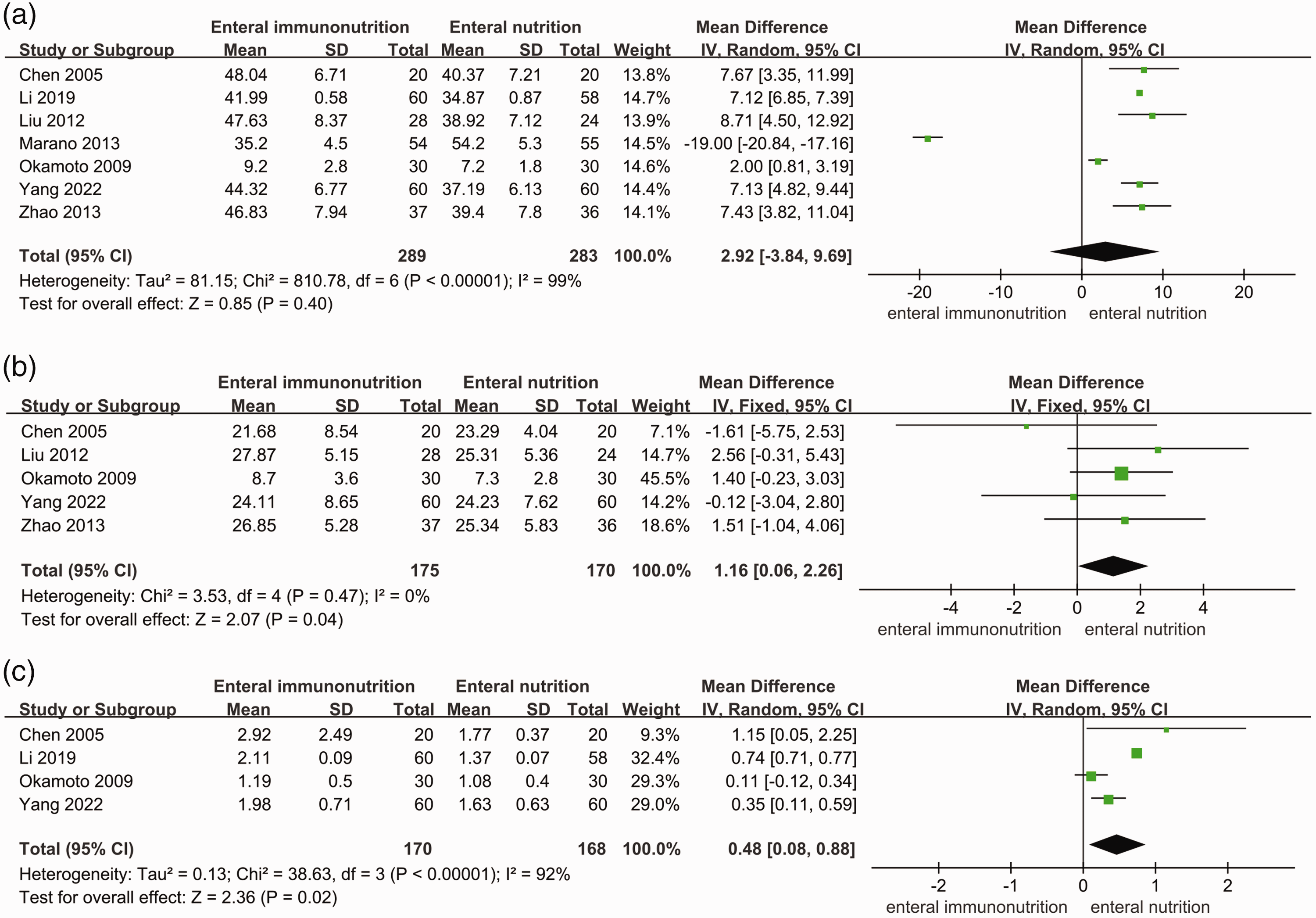
Forest plots of comparison of CD4+ levels (a), CD8+ levels (b), and CD4+/CD8+ (c) between patients with enteral immunonutrition and those with enteral nutrition who had gastric cancer surgery. SD, standard deviation; IV, inverse variance; CI, confidence interval.

Forest plots of comparison of immunoglobulin G (a) and immunoglobulin M (b) concentrations between patients with enteral immunonutrition and those with enteral nutrition who had gastric cancer surgery. SD, standard deviation; IV, inverse variance; CI, confidence interval.

Forest plots of comparison of lymphocytes (a), proalbumin concentrations (b), and transferrin concentrations (c) between patients with enteral immunonutrition and those with enteral nutrition who had gastric cancer surgery. SD, standard deviation; IV, inverse variance; CI, confidence interval.
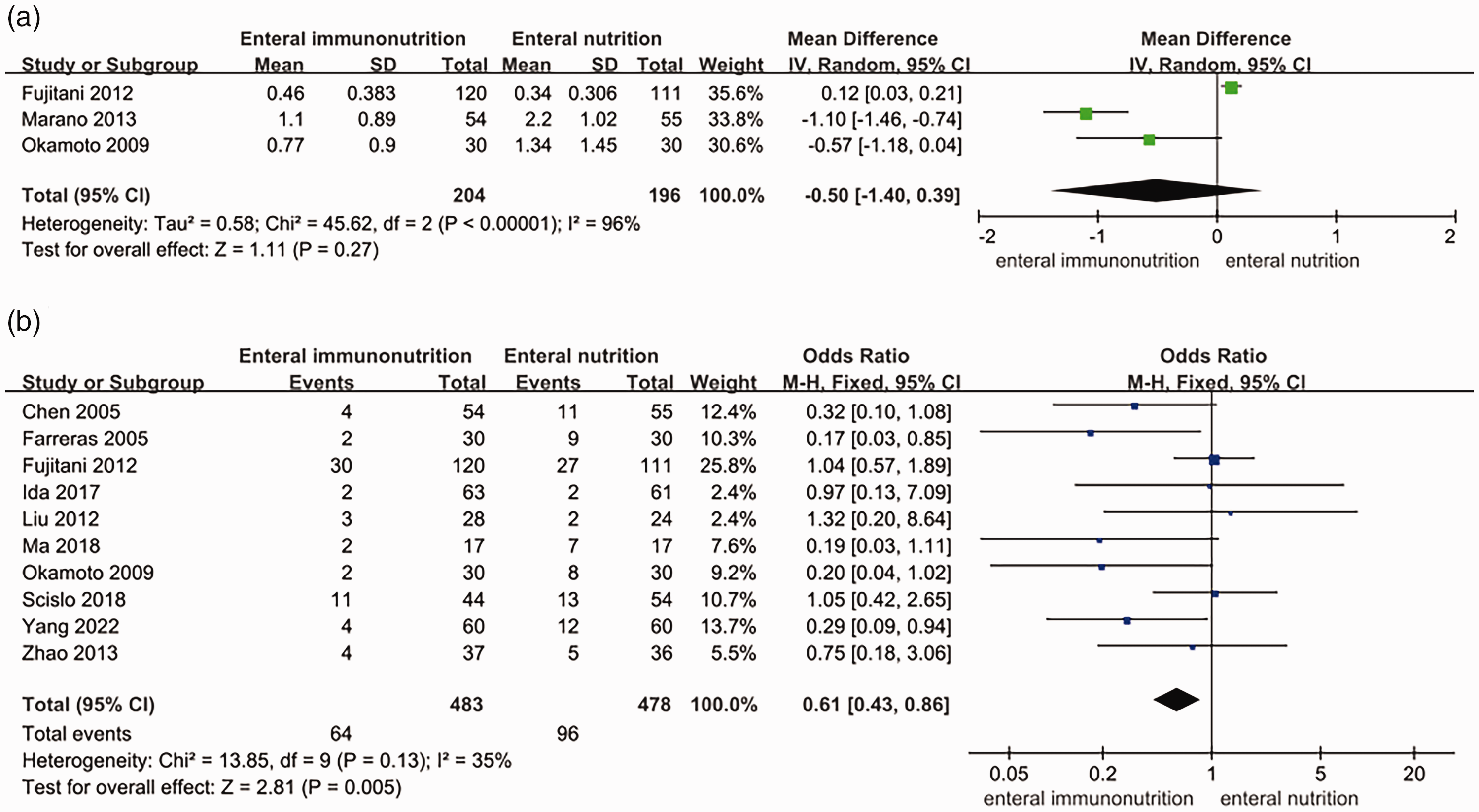
Forest plots of comparison of systemic inflammatory response syndrome (a) and infectious complications (b) between patients with enteral immunonutrition and those with enteral nutrition who had gastric cancer surgery. SD, standard deviation; IV, inverse variance; CI, confidence interval.
Publication bias analysis
A sensitivity analysis was performed for all of the results. No significant difference was found in the combined analysis results after excluding individual studies, which indicated the reliability of the results. With regard to publication bias, Begg test P values of CD4+ levels, CD8+ levels, and CD4+/CD8+ were 0.329, 0.364, and 0.324, respectively. The Begg test P values of IgG and IgM were 0.659 and 0.292, respectively. The Begg test P values of lymphocytes, proalbumin concentrations, and transferrin concentrations were 0.487, 0.126, and 0.845, respectively. The Begg test P values of systemic inflammatory response syndrome and infectious complications were 0.349 and 0.074, respectively. No obvious asymmetry was found in Begg’s funnel plots (Figures 6–9). The P values of Begg’s test for all studies were >0.05, which indicated no publication bias.

Begg’s funnel plots of CD4+ levels (a), CD8+ levels (b), and CD4+/CD8+ (c) after enteral immunonutrition versus enteral nutrition in patients with gastric cancer surgery.

Begg’s funnel plots of immunoglobulin G (a) and immunoglobulin M (b) concentrations after enteral immunonutrition versus enteral nutrition in patients with gastric cancer surgery.

Begg’s funnel plots of lymphocytes (a), proalbumin concentrations (b), and transferrin concentrations (c) after enteral immunonutrition versus enteral nutrition in patients with gastric cancer surgery.
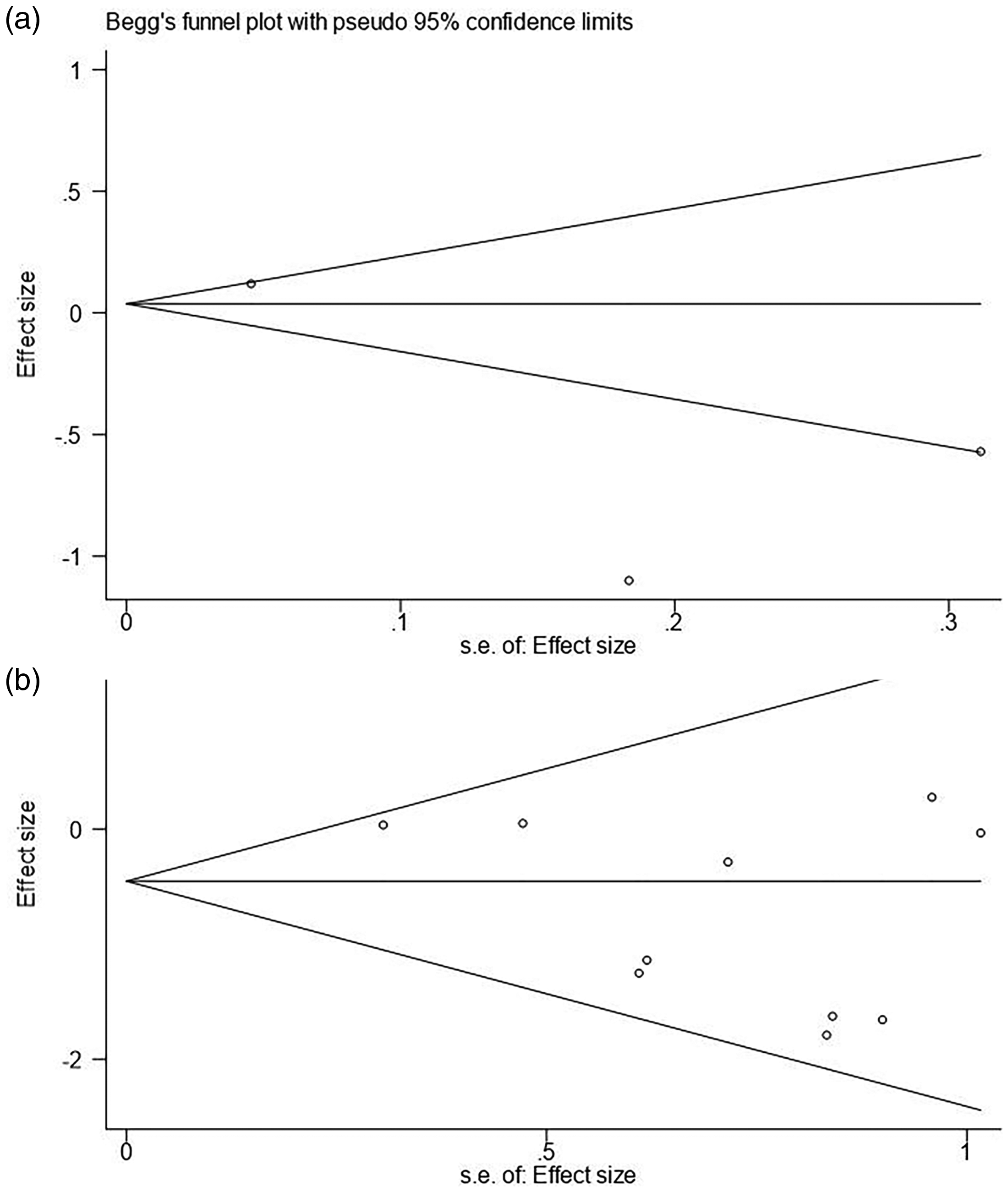
Begg’s funnel plots of systemic inflammatory response syndrome (a) and infectious complications (b) after enteral immunonutrition versus enteral nutrition in patients with gastric cancer surgery.
Discussion
The objective of the current meta-analysis and systematic review was to compare the effect of EN and EIN on patients who have gastric cancer surgery. The outcomes of infectious complications, serum proteins, immune-inflammatory factors, and cellular immunity in the EIN and EN groups were assessed. This meta-analysis and systematic review included more randomized, controlled trials and showed different conclusions compared with Fu et al.’s study. 26 Fu et al.’s study showed that IgG, IgM, and protein concentrations in the EIN group were significantly higher from higher in the EIN group than in the EN group. Our study showed that patients in the EIN group had higher CD8+, proalbumin, and immunoglobulin concentrations, higher CD4+/CD8, and a lower incidence of postoperative infectious complications than those in the EN group. These findings provide evidence for the effectiveness of EIN in patients undergoing gastric cancer surgery and set the stage for future prospective studies in such patients.
Patients with gastric cancer often have malnutrition and immunodeficiency due to the hypermetabolic state of the tumor, loss of appetite, and digestion and absorption dysfunction. 39 Additionally, surgery impairs the structural and functional integrity of the GI tract, causing a systemic inflammatory stress response that includes various endocrine, immune, and hematological effects. 40 In perioperative patients, a nutritional support strategy, such as PN and EN, is a popular and essential strategy.41,42 EN is more frequently preferred than PN because of its safety, and physiological and economic benefits. 43 EN is also widely used in critically ill patients. Evidence suggests that the intake of certain essential nutrients over the normal daily requirements can modulate various inflammatory, metabolic, and immunological processes. However, because of the complexity of the tumor, the clinical effect of EN is not ideal. 25 Recently, immunonutritional substrates were shown to modulate the immune system. Zhao et al. 44 showed that adding ω-3 fatty acid emulsion to a PN formula improved postoperative immune function indices and efficacy and reduced the inflammatory response.
Zheng et al. found that EIN reduced postoperative complications and shortened the hospital stay. 45 The mechanisms associated with EIN have also been studied. Other studies showed that EIN aided the regulation of inflammation and enhanced systemic immunity by markedly reducing tumor necrosis factor-α, interleukin-6, and C-reactive protein concentrations, and increasing CD4+ and IgA levels.46,47 The ω-3 fatty acids comprise eicosapentaenoic acid and docosahexaenoic acid, which are primary effective fish oil components that can modulate the immune inflammatory response. This modulation occurs by T 48 and B cell activation, 49 alterations in membrane fluidity, lipid peroxide formation, eicosanoid generation, gene expression regulation, and the inhibition of subsequent inflammatory processes. These inflammatory processes include leukocyte chemotaxis, leukocyte–endothelial adhesive interactions, adhesion molecule expression, eicosanoid production (prostaglandins and leukotrienes) from n-6 fatty acid arachidonic acid, and stimulation of proinflammatory cytokines. 50 Glutamine is consumed by immune, intestinal mucosal, and tumor cells, and is markedly associated with their functions, such as proliferation, superoxide production, antigen presentation cytokine synthesis, and phagocytosis.51,52 Arginine, which is a semi-essential amino acid, stimulates the multiplication of T-cells in response to mitogens or cytokines. 53 These findings provide theoretical support that EIN can improve the immune level of patients who have gastric cancer surgery. However, some studies have suggested that EIN does not reduce the occurrence of complications, 54 while it lacks some cost-effectiveness. 55 Cheng et al. 25 suggested that EIN support requires more than 7 days before the immunonutrition formula can increase immune factors. Furthermore, Song et al. 24 suggested that EIN elevates the levels of some immunogenic factors, except for CD8+, and does not reduce postoperative infectious complications. These inconsistent results between studies were mainly due to the preoperative nutritional status of the experimental cohort and different immunonutritional components, administration times, and routes. Song et al 24 suggested that the formula of arginine, ω-3 fatty acids, glutamine, and RNA was the optimal regime. However, only arginine and RNA were not recommended by De Luis et al 56 who considered that an increased dose of arginine-enriched enteral formula was more clinically beneficial. Seven to 14 days of perioperative immunonutritional intervention in moderately or severely malnourished patients who have gastric surgery is recommended. 57 Additionally, pre- and post-EIN were not found to provide double benefits. 58 However, Song et al. 24 showed that perioperative EIN was the optimum timing for this intervention.
This study has a few limitations as follows. 1) All included studies focused on short-term outcomes, and therefore, assessing the long-term outcomes of EIN was difficult. Evaluating the clinical long-term results can provide more reliable evidence. 2) The included studies were heterogeneous, which may have been related to the situation of the patients, the nutritional formula, and the administration time of EIN. Unpublished articles and excluded data may have caused bias in the pooled result. 3) Relevant subgroup analyses to further investigate the optimal EIN nutritional formulation and the timing of administration were not conducted.
Conclusion
The high-quality studies in this meta-analysis and systematic review suggest that EIN improves immune activity and post-surgical infectious complications compared with EN in patients with gastric cancer. More high-quality, randomized, controlled trials are required for in-depth evaluation of the most effective formulation, administration time, and duration for optimal intervention using EIN.
Footnotes
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author contributions
JL and QLX contributed to the conception, design, and modification of the study. QLX, SQL, YXZ, and JXZ extracted the data and organized the database search. JL and QLX performed the statistical analysis. JL, SQL, YXZ, and JXZ drafted the manuscript. JL and SQL confirm the authenticity of all of the raw data. All authors contributed to revision of the submitted manuscript. All authors agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
