Abstract
The efficacy and therapeutic mechanisms of continuous renal replacement therapy (CRRT) for improvement of oxygenation in acute respiratory distress syndrome (ARDS) remain controversial. These questions were addressed by retrospective analysis of severe ARDS patients admitted to the pediatric intensive care unit of our hospital from 2009 to 2015 who received high-volume continuous veno-venous hemofiltration during mechanical ventilation. There was a significant improvement in partial oxygen pressure/fraction of inspired oxygen (PaO2/FiO2) 24 hours after CRRT onset compared with baseline (median change = 51.5; range = −19 to 450.5; P < .001) as well as decreases in FiO2, peak inspiratory pressure, positive end-expiratory pressure, and mean airway pressure (P < .05). The majority of patients had a negative fluid balance after 24 hours of CRRT. White blood cell (WBC) count decreased in the subgroup with high baseline WBC count (P < .05). PaO2/FiO2 was higher in ARDS patients with extrapulmonary etiology than in those with pulmonary etiology (P < .05). Improvement in oxygenation is likely related to both restoration of fluid balance and clearance of inflammatory mediators.
Introduction
Acute respiratory distress syndrome (ARDS) is an inflammatory lung disorder characterized by increased permeability of the alveolar-capillary barrier, leading to alveolar leukocyte infiltration and protein-rich pulmonary edema. Clinically, it presents as refractory hypoxemia and bilateral pulmonary infiltrates without evidence of left-heart failure or volume overload. ARDS is driven primarily by inflammatory chemokines and cytokines produced endogenously in response to a variety of inciting agents and events. In children, one study reported that the mortality from mild and moderate ARDS was about 10%, whereas that from severe ARDS was as high as 25%. 1 The severity of the oxygenation deficit as measured by the partial oxygen pressure/fraction of inspired oxygen (PaO2/FiO2) correlates well with mortality.
Hemofiltration has been proposed as one possible therapeutic option for improving oxygenation by promoting fluid removal, but its efficacy is still contentious. Some studies have concluded that continuous renal replacement therapy (CRRT), which is indicated for acute fluid overload (FO) as well as acute renal failure, improves oxygenation in patients with respiratory failure.2-4 However, others have found no improvement in oxygenation. 5 Even if CRRT can improve oxygenation in children with ARDS, there is still debate as to whether the beneficial effects on pulmonary gas exchange are mediated by mechanisms other than fluid removal, such as through continuous removal of inflammatory mediators.4,6
To examine the efficacy of CRRT for improvement of lung function in pediatric ARDS and the contribution of inflammatory mediators, we retrospectively compared gas exchange and inflammatory parameters before and after continuous veno-venous hemofiltration (CVVH) in pediatric patients with severe ARDS. We selected only patients with severe ARDS because patients in this high-mortality group may derive the greatest benefit from CRRT.7,8
Materials and Methods
Patients
This was a retrospective cohort study in which clinical and laboratory data were gathered for 30 consecutive children with severe ARDS treated at GuangZhou Women and Children’s Medical Center. Inclusion criteria were the following: (1) age 1 month to 18 years, (2) admission to the pediatric intensive care unit (PICU) between January 2009 and December 2015, (3) diagnosis of severe ARDS according to the Berlin definition criteria, 9 (4) need for mechanical ventilation, and (5) initiation of CRRT. The Berlin criteria define severe ARDS as the following: PaO2/FiO2 ≤ 100 mm Hg with PEEP ≥ 5 cm H2O occurring within 1 week of a known clinical insult or new or worsening respiratory symptoms; bilateral opacities not fully explained by effusions, lobar/lung collapse, or nodules on chest radiography without evidence of cardiac failure or FO; and objective assessment to exclude hydrostatic edema. 9
The present study was approved by the ethics committee of Guangzhou Women and Children’s Medical Center. Written informed consent was obtained from the parents or legal guardians of all participants.
Treatment
Routine Treatment
Respiratory support consisted of mechanical ventilation with relatively small tidal volumes (VTs; 6-8 mL/kg) in the prone position with allowable PaCO2 within 45 to 70 mm Hg. However, respiratory support was not regulated by protocol.
Renal Replacement Therapy
CVVH was performed with a flow-controlled blood roller pump machine to treat FO unresponsive to diuretics. The parameters were replacement solution infusion at 50 to 60 mL/kg/h and blood pump flow rate at 5 to 10 mL/kg/min. An automated system (PRISMA, Gambro AB, Stockholm, Sweden) was used to deliver CVVH continuously for at least 24 hours. The initial replacement solution was composed of 2000 mL normal saline, 500 mL of 5% glucose, 1 mL of 25% magnesium sulfate, 10 mL of 10% calcium gluconate, 10 mL of 10% potassium chloride, and 250 mL of 5% sodium bicarbonate. The formula was adjusted according to individual blood glucose and electrolyte levels. Conventional heparin was used as the anticoagulant. Fluid removal rate was determined by the PICU physician according to clinical condition and fluid status of the patient.
Data Collection
Data were extracted from the ICU medical records. Fluid balance/weight was defined as [Fluid in] − [Fluid out] (in mL)/[Admission weight] (in kg). The degree of FO from PICU admission to CRRT initiation (%FO) was calculated using the following formula: %FO = {[Fluid in] − [Fluid out] (in L)/[Admission weight] (in kg)} × 100. 10 The rationale for using FO from PICU admission to CRRT initiation was based on many previous pediatric studies.10-12 The following variables were collected just before CVVH (0 hours) and 24 hours after initiation: arterial blood gas levels; ventilator settings, including FiO2; peak inspiratory pressure (PIP); positive end-expiratory pressure (PEEP); VT; mean airway pressure (Paw); PaO2/FiO2; and laboratory values for white blood cell (WBC) count and creatinine. The hemodynamic variables heart rate and mean arterial blood pressure (MAP) were also recorded before and after CRRT. PaO2/FiO2 at 6 hours before CRRT was also collected to assess any spontaneous change prior to intervention. Patients were also classified according to etiology as having ARDS of pulmonary or extrapulmonary origin.
Statistical Analysis
Changes of each variable were calculated for all patients from baseline (0 hours) to 24 hours after onset of CRRT. The Wilcoxon signed-rank test was applied to test whether the median change was 0. PaO2/FiO2 at 6 and 0 hours before CRRT were also compared. The Mann-Whitney U test was used for comparison of patients with pulmonary versus extrapulmonary ARDS. Significance was set at P < .05. All statistical calculations were performed using SPSS 11.5 for Windows.
Results
Patient Characteristics
Between 2009 and 2015, 30 patients meet the inclusion criteria. Patient characteristics are summarized in Tables 1 and 2. Before the start of CRRT, all children had positive fluid balance, and most had pre-CRRT %FO <10%. There was no significant difference between PaO2/FiO2 at 6 and at 0 hours before CRRT.
Baseline Clinicodemographic Characteristics (n = 30).
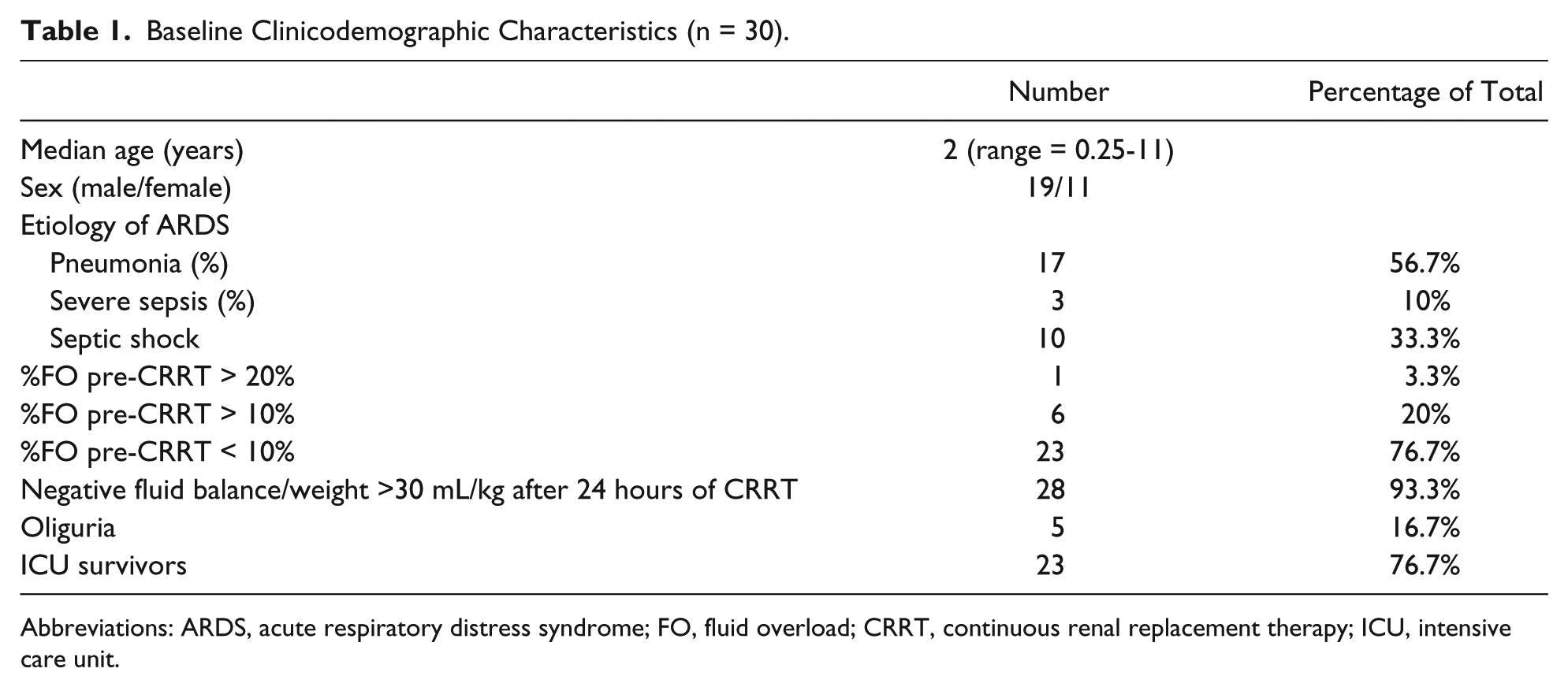
Abbreviations: ARDS, acute respiratory distress syndrome; FO, fluid overload; CRRT, continuous renal replacement therapy; ICU, intensive care unit.
Baseline Respiratory, Ventilator, Hemodynamic, and Laboratory Values.
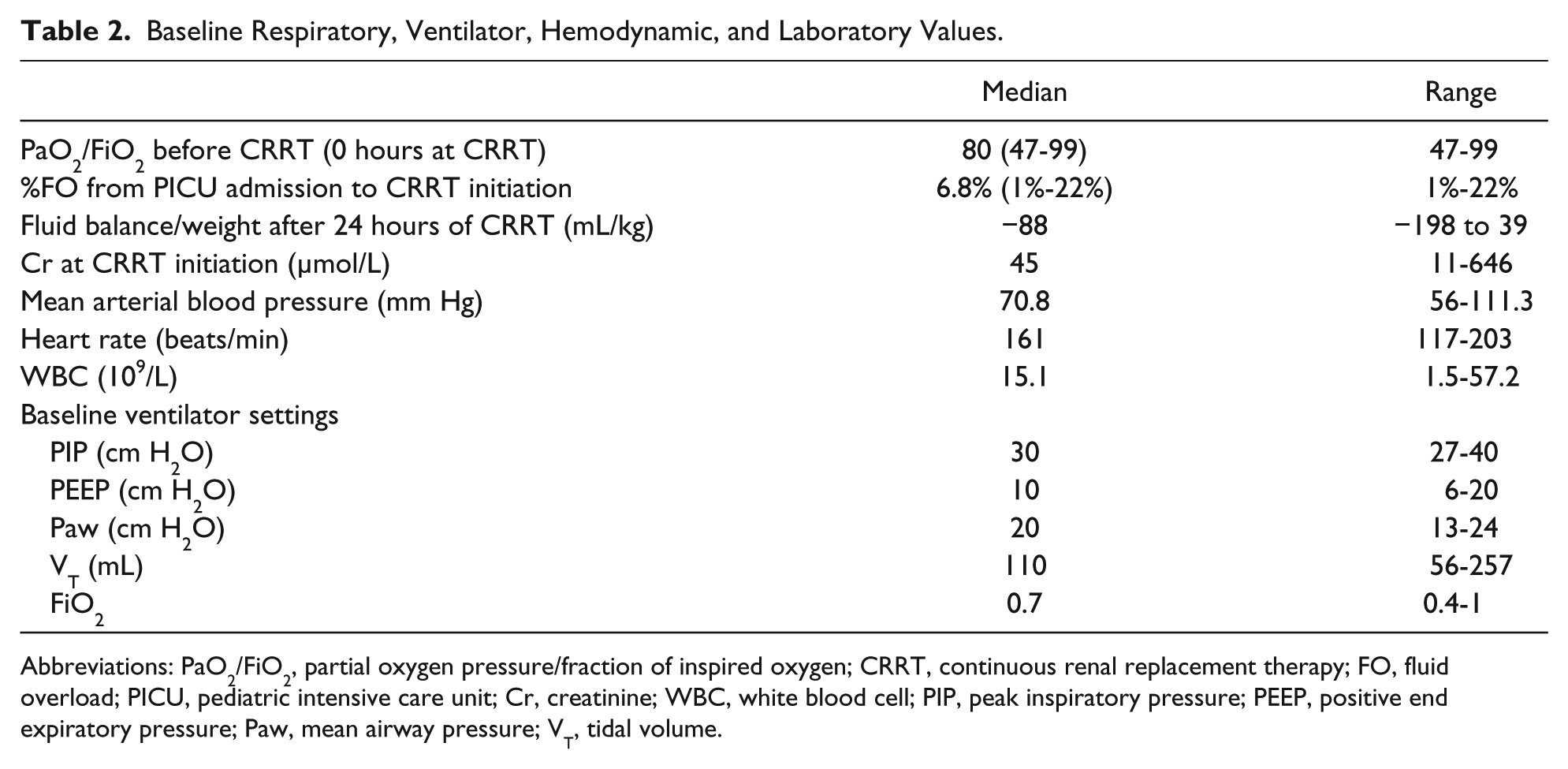
Abbreviations: PaO2/FiO2, partial oxygen pressure/fraction of inspired oxygen; CRRT, continuous renal replacement therapy; FO, fluid overload; PICU, pediatric intensive care unit; Cr, creatinine; WBC, white blood cell; PIP, peak inspiratory pressure; PEEP, positive end expiratory pressure; Paw, mean airway pressure; VT, tidal volume.
Effect of CRRT on Oxygenation, Ventilator Settings, Hemodynamics, and WBC Count
Median changes in respiratory, ventilator, hemodynamic, and laboratory parameters from baseline (0 hours) to 24 hours after initiation of CRRT are shown in Table 3. The main gas exchange parameter PaO2/FiO2 improved significantly 24 hours after the start of CRRT compared with the 0-hour baseline value (increase in median = 51.5; range = −19 to 450.5; P < .001), whereas the ventilatory parameters, FiO2, PIP, PEEP, and Paw, all decreased significantly (P < .05). MAP increased and heart rate decreased after CRRT (P < .05).
Median Changes in Respiratory, Ventilator, Hemodynamic, and Laboratory Values. a

Abbreviations: CRRT, continuous renal replacement therapy; PaCO2, carbon dioxide pressure; PaO2, partial oxygen pressure; FiO2, fraction of inspired oxygen; PIP, peak inspiratory pressure; PEEP, positive end expiratory pressure; Paw, mean airway pressure; VT, tidal volume; MAP, mean arterial blood pressure; WBC, white blood cell.
From baseline (0 hours) to 24 hours after CRRT.
Number of patients with available data.
P value from Wilcoxon signed-rank test.
WBC count elevated for age.
WBC count depressed for age.
WBC count decreased significantly after 24 hours of CRRT in the 18 patients with elevated baseline WBC count (P = .001), whereas no significant change was observed in patients with normal or below normal baseline WBC count. However, of those with baseline WBC count in the high-normal range, there was a trend toward a drop after treatment.
Effect of CRRT on Fluid Balance
It was found that 28 patients had negative fluid balance >30 mL/kg following 24 hours of CRRT, whereas only one had a zero fluid balance and one a positive fluid balance of +39 mL/kg.
Comparison of ARDS Patients With Pulmonary Versus Extrapulmonary Etiology
In a second analysis, respiratory and ventilatory changes were evaluated separately (Table 4) in those patients with pulmonary etiology (n = 17) and those with extrapulmonary etiology (n = 13). PaO2/FiO2 after 24 hours of CRRT was higher in ARDS patients with extrapulmonary etiology than in those with pulmonary etiology (P = .024). FiO2 did not differ between the 2 groups after CRRT, but there was a trend for lower post-CRRT FiO2 in the extrapulmonary group. There were no group differences in the other respiratory and ventilatory parameters.
Comparison of Respiratory and Ventilatory Variables for ARDS Patients With Pulmonary (n = 17) and Extrapulmonary Etiology (n = 13).

Abbreviations: ARDS, acute respiratory distress syndrome; PaCO2, carbon dioxide pressure; PaO2, partial oxygen pressure; FiO2, fraction of inspired oxygen; CRRT, continuous renal replacement therapy; PIP, peak inspiratory pressure; PEEP, positive end expiratory pressure; Paw, mean airway pressure; VT, tidal volume.
P value from Mann-Whitney U test.
Discussion
Our study demonstrates that CRRT can substantially improve oxygenation (as measured by PaO2/FiO2) and decrease ventilatory parameters (FiO2, PIP, PEEP, and Paw) in pediatric patients with severe ARDS. Moreover, these results also strongly suggest that suppression of inflammation is a major contributor to this therapeutic effect, in addition to restoration of fluid balance.
There was no change in PaO2/FiO2 from 6 hours before to immediately before CRRT, indicating no spontaneous improvement, whereas a significant increase was observed after 24 hours of CRRT, indicating that improved oxygenation is the result of CRRT. The effect was at least partially a result of reduction of FO. All patients had positive fluid balance prior to CRRT, and the majority had a negative fluid balance 24 hours following CRRT. Many clinical studies have shown that reducing or eliminating positive fluid balance in patients with ARDS can reduce mechanical ventilation times 13 and mortality, 14 presumably by preventing fluid accumulation in the lungs, which aggravates respiratory insufficiency in ARDS.
However, most patients with severe ARDS in our study did not have severe FO before CRRT (23 of 30 or 76.7% had baseline %FO <10% and only one had %FO >20%). Moreover, many studies have shown that <10% FO is advantageous for both PaO2/FiO2 and survival.11,13-15 Nonetheless, all patients had very low baseline PaO2/FiO2. Considering the lack of severe FO, we suggest that mitigating severe FO was not the only mechanism by which CRRT improved PaO2/FiO2. Patients with high baseline WBC counts showed significant WBC count reductions after CRRT, suggesting that suppression of inflammation may be an additional contributing mechanism for improved oxygenation. Indeed, several studies have shown that CRRT can remove serum systemic inflammatory mediators that contribute to ARDS pathogenesis by convection and absorption.6,16-19
To further elucidate whether modulation of inflammation is clinically relevant, we compared patients with ARDS having an extrapulmonary cause with those having a pulmonary etiology because these 2 ARDS subgroups have distinct pathogenic characteristics. Edema and alveolar collapse caused by capillary leak and systemic inflammation play a central role in extrapulmonary ARDS, whereas consolidation secondary to pneumonia and lung inflammation is essential for progression of pulmonary ARDS. 20 In our study, PaO2/FiO2 after CRRT was higher in ARDS of extrapulmonary origin, again suggesting that suppression of the systemic inflammatory response could be one of the mechanisms for improving oxygenation.
We used high-volume hemofiltration rates of 50 to 60 mL/kg/h to facilitate the removal of inflammatory mediators associated with ARDS. Much experimental and some clinical evidence also suggests that high-volume hemofiltration efficiently removes cytokines by a convective mechanism and can be beneficial for critically ill patients with severe inflammatory states such as septic shock.21-23 Indications for CRRT in our institution are broader than in many other institutions, where CRRT is indicated primarily for renal failure and FO. In our study, early CRRT was implemented for severe ARDS patients with renal laboratory parameters and urine output within normal limits. CRRT use was based on past successes treating severe inflammatory states, and some of our severe ARDS patients were still deteriorating at the time of treatment, even in the absence of FO.
Limitations of this study include those inherent to the retrospective design and single-center case population, including the potential for selection bias. Thus, large multi-institutional studies are needed to verify if CRRT can improve oxygenation by reducing inflammation.
We demonstrate that 24 hours of CRRT can improve oxygenation in mechanically ventilated pediatric patients with severe ARDS, likely because of clearance of inflammatory mediators as well as mitigation of FO. However, determining the relative contributions of these 2 mechanisms requires further study.
Author Contributions
WY contributed to the design and drafted the manuscript. JH contributed to the conception. QZ critically revised the manuscript for important intellectual content. JT critically revised the manuscript for important intellectual content. FC drafted the manuscript. RD gave final approval. YL gave final approval. ZW agreed to be accountable for all aspects of the work, in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. YY agreed to be accountable for all aspects of the work, in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Footnotes
Authors’ Note
Wenmin Yang and Jie Hong made equal contributions to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Guangzhou Science and Technology Project (No. 2014Y2-00181).
