Abstract
Advancing age is accompanied by decreased immunity, poor health, and physiological changes, which render the elderly population highly susceptible to infectious diseases. We aim to identify the guidelines for pneumococcal vaccines in old-age facilities in India. We performed an extensive review of Indian literature (indexed and non-indexed publications) from 2010 to 2020 using search strings “Pneumococcal vaccine AND Recommendations AND India,” “Pneumococcal vaccine AND Guidelines AND India,” followed by a hand search to identify the most updated versions of recommendations. We reviewed immunization guidelines recommended by nine medical associations and societies in India—Association of Physicians of India (API), Geriatric Society of India (GSI), Indian Society of Nephrology (ISN), Mass Gathering Advisory Board Consensus Recommendation, Indian Medical Association (IMA), Indian Chest Society and National College of Chest Physicians (ICS-NCCP), Research Society for Study of Diabetes in India (RSSDI), Indian Association of Occupational Health Guidelines for Working Adults (IAOH), and API guidelines for immunization during COVID19 pandemic. All bodies recommend pneumococcal vaccines, sequence and preference of which depend on factors such as age, underlying conditions, and immune status. Integration of society recommendations and their implementation into public and private vaccination programs are required to promote adult immunization.
Introduction
Pneumococci (Streptococcus pneumoniae) cause severe infections such as pneumonia, septicemia, and meningitis (Ramasubramanian, 2015). Pneumococcal disease can be classified as invasive and non-invasive. Invasive pneumococcal diseases (IPD) continue to be a global cause of morbidity and mortality, particularly in children <2 years and adults >65 years (Tan, 2012). Incidence rates of IPD in Europe ranged from 11 to 27/100,000, versus North America ranged from 15 to 49/100,000. Even higher incidence rates of IPD have been reported from Asia (Drijkoningen & Rohde, 2014).
The 2011 census revealed 8.6% of the total Indian population as elderly (Singh et al., 2017). Moreover, the population aged over 60 years has been continuously increasing and is projected to increase from 5.6% in 1961 to 22.5% in 2051 (Singh et al., 2017). Despite a huge population size of the elderly in India, their preventive care remains a challenge owing to no established immunization schedule. Furthermore, decreased immunity associated with advancing age, accompanied by poor health and physiological changes, increases their susceptibility to both communicable and non-communicable diseases, with pneumococcal, influenza, tetanus, and herpes zoster, being more common (Verma et al., 2014).
In India, S. pneumoniae accounts for 30% to 55% of CAP in adults (Ramasubramanian, 2015). The highest case-fatality rate in invasive disease (30.2%) has been reported in adults 50 to 60 years (Thomas et al., 2013). The Invasive Bacterial Infections Surveillance (IBIS) study indicated that 46.3% of IPD cases showed clinical worsening (Thomas et al., 2013). In addition, IPD was associated with an overall case fatality rate of 26.4% in adults even in the hospital settings (Ramasubramanian, 2015). Meningitis, sepsis, and invasive pneumonia were associated with case-fatality rates of 36.9%, 36.6%, and 20.8%, respectively (Ramasubramanian, 2015). In a laboratory-based surveillance study of IPD cases in adults >18 years at a tertiary care hospital in South India, pneumonia accounted for 146/408 cases (39%), and the case fatality rate was 19.3 (Jayaraman et al., 2019).
Multidrug resistance has emerged as a serious concern in the treatment of invasive pneumococcal diseases, especially in Asian countries (WHO, 2008). Although pneumococcal and Haemophilus influenzae type b conjugate vaccines have reduced the burden of antibiotic-resistant bacterial diseases globally, the continued use of antibiotics favors selection of antibiotic-resistant strains among the remaining non-vaccine serotypes. Serotype replacement among asymptomatic carriers has negatively affected the vaccination drive (Ginsburg & Klugmann, 2017). In India, IPD is associated with a high mortality rate despite the availability of treatment (Koul et al., 2019).
In March 2018, the government in the Indian state of Tamil Nadu launched a vaccination drive in old-age homes in collaboration with state and central governments. The State Social Welfare Department administered PCV13 vaccine to 1,853 and 2,280 elderly people residing in non-government organizations (NGOs; 50 old-age homes, 3 multi-care centers for widows, 4 day care centers, and 2 respite care homes) with state and central grants, respectively (“Grants—Immunization Programme,” 2018; The Times of India, 2018).
In April 2018, the Geriatric Medicine Department of the All India Institute of Medical Sciences (AIIMS), in collaboration with an NGO “Healthy Aging India,” announced a free vaccination program for the elderly staying in old-age homes. Under this initiative, free pneumococcal vaccine was administered to 300 old people in 20 old-age homes in New Delhi. In addition, the old-age home residents were screened for cancer to facilitate their early diagnostic treatment at AIIMS (The Better India, 2018; India Today, 2018).
However, India still lacks a large-scale implementation of adult vaccination programs. We hereby provide a review of different guidelines and recommendations released by esteemed medical societies over the last few years for the use of pneumococcal vaccines in the country. The attempt is to help integrate the pneumococcal vaccination into the access programs in old-age homes where the elderly population have the right to age well.
Methods
We performed an extensive review of Indian literature which included both indexed and non-indexed publications from 2010 to 2020. The following search strings were used: “Pneumococcal vaccine AND Recommendations AND India,” “Pneumococcal vaccine AND Guidelines AND India”
Resources used for our search included PubMed-Medline, Scopus, Google Scholar, CINAHL (Cumulative Index to Nursing and Allied Health Literature), Embase, Science Direct databases of National Institute of Science Communication and Information Resources (NISCAIR), Annotated Bibliography of Indian Medicine (ABIM), and Google Scholar. Furthermore, a hand search was performed to identify the most updated versions of recommendations from published articles/reports and bibliographies of published works. Reference lists of eligible studies were searched for additional publications. For any association, only the most updated version of the guideline was considered for the review. The inclusion and exclusion criteria are listed below:
Inclusion Criteria
• Publications in the period 2010 to 2020 for Indian adults
• Review articles published in peer review journals or medical textbooks for guidelines/consensus recommendations for pneumococcal vaccine use in Indian adults
• Literature published by accredited medical associations and societies for use of pneumococcal vaccine Indian adults as special supplements of journals or conference proceedings
Exclusion Criteria
• Publications before 2010
• Publications by pediatric asssociations and age group <18 years
• Publications for use of pneumococcal vaccines non-Indian adult population
Results
We reviewed immunization guidelines recommended by nine medical associations and societies in India. This review discusses various guidelines by Indian associations such as the Association of Physicians of India (API), Geriatric Society of India (GSI), Indian Society of Nephrology (ISN), Mass Gathering Advisory Board Consensus Recommendation, Indian Medical Association (IMA), Indian Chest Society and National College of Chest Physicians (ICS-NCCP), Research Society for Study of Diabetes in India (RSSDI), Indian Association of Occupational Health Guidelines for Working Adults (IAOH), and API guidelines for immunization during COVID19 pandemic. All guidelines recommend pneumococcal vaccines, their sequence and preference of which depend on factors such as age, underlying conditions, and immune status.
The details of the guidelines are as follows.
Association of Physicians of India
API recommends PCV13 may be offered to all eligible adult patients following a discussion with currently available evidence, scientific rationale, and limitations. The need for re-vaccination with a subsequent dose of PCV13 has not been established. At present the available evidence to recommend widespread use of PPCV23 especially for adults those with underlying chronic diseases is less convincing (Muruganathan et al., 2015).
Geriatric Society of India
The Geriatric Society of India (GSI, 2015) recommends both PCV13 and PPSV23 for all individuals older than 50 years and for those with the following conditions:
Individuals aged 50 years and older.
Individuals with certain underlying medical conditions such as coronary artery disease, congestive heart failure, cardiomyopathy, diabetes mellitus, bronchial asthma, chronic obstructive pulmonary disease, cirrhosis of liver, and chronic renal failure (CRF).
Immunocompromised individuals with high risk for pneumococcal infections, for example, those with immunoglobulin deficiency, HIV infection, leukemia, lymphoma, multiple myeloma, Hodgkin’s disease, non-Hodgkin’s lymphoma, disseminated malignancy, those with either a reduced response to vaccination or a decline in serum antibody concentrations.
Individuals with organ or bone marrow transplantation.
Individuals receiving long-term therapy with corticosteroids or immunosuppressive agents.
Individuals who have undergone splenectomy, or have anatomic asplenia, or sickle cell disease as they have reduced clearance of encapsulated bacteria from the blood stream.
Chronic smokers.
Individuals suffering from sleep disorders.
Individuals who are prone to nocturnal aspiration.
The recommended vaccination schedule is as follows:
• PCV13 for elderly individuals who have previously received PPSV23 (to maintain immune protection).
• PCV13 may be preferred in vaccine-native or previously vaccinated adults.
• In case both the vaccines are considered, PCV13 should be administered first, followed by PPSV23.
• For certain risk groups such as immunosuppressed or immunocompromised populations, if previously unvaccinated, a dose of PCV13 is recommended, followed by a dose of PPSV23 8 weeks later.
PPSV23 is administered only once as a single dose to elderly individuals.
The GSI guidelines recommend revaccination with PPSV23 for individuals with an increased risk of pneumococcal infection and those likely to have a rapid decline in pneumococcal antibody levels, given 5 years have elapsed since the administration of the first dose of pneumococcal vaccine.
Mass Gatherings Consensus Document
The consensus guidelines for India and Bangladesh for the use of pneumococcal vaccine in mass gatherings and for Hajj pilgrims recommend (“Grants—Immunization Programme,” 2018; Mathai et al., 2016)
• PCV13 may be recommended 4 weeks before starting Hajj pilgrimage, depending on the economic status of the individuals.
• PPSV23 may be administered to these individuals upon their return from the pilgrimage based on their risk status.
The Indian Society of Nephrology
The Indian Society of Nephrology (2016) guidelines state that pneumococcal vaccination is cost-effective compared with the global health costs in patients with chronic kidney disease (CKD), and there are no data indicating its potential disadvantages. Pneumococcal vaccination should be administered to all patients with CKD as early as possible (The Indian Society of Nephrology, 2016). The vaccination recommendations to be followed in all patients with CKD is shown in Table 1.
Recommendations for Administering PCV13 and PPSV23 Vaccines in Patients With CKD, by the Indian Society of Nephrology (2016).

Note . PCV = pneumococcal conjugate vaccine; PPSV = pneumococcal polysaccharide vaccine.
Recommendations for transplant patients
In case of patients with kidney transplant, the guidelines advise that the immunization schedule should be same as that for general adults. Due to intense immunosuppression following kidney transplant, vaccination should be avoided within 3 to 6 months of transplantation. For preventing infections in patients with kidney transplant, pneumococcal vaccines should be administered to all household contacts, including pets of the transplant recipients. The guidelines suggest the following comprehensive approach for optimal vaccine efficacy among immunocompromised adults, including kidney transplant recipients:
• The first dose of PCV13 should be followed by PPSV23 at about 8 weeks later.
• If the patient had received PPSV23 in the past, a PCV13 dose should be administered only after at least a year.
• If a patient who has received PPSV23 requires further doses of PPSV23, it should be administered at least 5 years after the last dose of PPSV23.
Indian Medical Association
The Indian Medical Association (IMA, 2018) guidelines recommend the following dosage, sequence, and route of administration (in at-risk cases) of pneumococcal vaccines (Tandon et al., 2018):
• PCV13 injection (0.5 mL) administered intramuscularly, followed by 0.5 mL PPSV after 8 weeks.
• PPSV23 may be repeated once after 5 years, if required.
The recommended vaccine schedules for adolescents, adults, and solid organ transplant recipients in the IMA guidelines are shown in Tables 2 and 3.
Vaccines for Adolescents and Adults Recommended per IMA Guidelines.

Source. Tandon et al. (2018).
Note. PPSV = pneumococcal polysaccharide vaccine; PCV = pneumococcal conjugate vaccine; HSCT = hematopoietic stem cell transplant; HIV = human immunodeficiency virus.
Vaccines for Solid Organ Transplant Recipients Recommended per IMA Guidelines.

Source. Tandon et al. (2018).
Furthermore, the IMA guidelines mention that both primary and secondary immunocompromised patients, pregnant women, diabetics, and individuals traveling who are at increased risk of pneumococcal infections should receive pneumococcal vaccines.
Indian Chest Society and National College of Chest Physicians of India
The ICS-NCCP (2019) guidelines state (Dhar et al., 2020):
• Vaccination with PPSV23 in all adults older than 65 years is recommended because of the overall higher incidence of invasive pneumococcal disease in this age group.
• Vaccination with PCV13 first, followed by PPSV23, is recommended for individuals having immunocompromising conditions, functional or anatomic asplenia, cochlear implant, CSF leak, or history of invasive pneumococcal disease. However, in this group of patients, the decision regarding administering PCV13 before PPSV23 can be discussed between the physician and the patient on a case‑to‑case basis.
• For adults with chronic conditions such as chronic heart disease, chronic liver disease, poorly controlled diabetes mellitus, chronic lung disease, and in current smokers and those with alcohol abuse, the decision to administer PCV13 preceding PPSV23 should be taken jointly by the physician and the patient, on a case‑to‑case basis.
• Because re-vaccination with PPSV23 may cause hypo responsiveness, it must solely be based on clinical judgment.
Research Society for the Study of Diabetes in India
The Research Society for the Study of Diabetes in India (RSSDI, 2020) 2020 recommends vaccination in diabetic patients to prevent pneumococcal diseases (Chawla et al., 2020).
• PCV13 is recommended for adults ≥50 years followed by a dose of PPSV23 at least 1 year later (and at least 5 years after their previous PPSV23 dose) depending on the clinical judgment of the physician.
• PCV13 must be considered for older diabetic patients aged >50 years.
• PPSV23 may be offered to immunocompromised patients with diabetes for additional coverage after PCV13.
• Re-vaccination with PPSV23 must be avoided to prevent hypo responsiveness.
Indian Association of Occupational Health (IAOH) Guidelines for Working Adults
The Indian Association of Occupational Health (IAOH) guidelines highlight that ensuring both health and safety from vaccine-preventable diseases (VPDs) in the workplace significantly reduces absenteeism. A healthy workplace ensures greater productivity in terms of work output, thereby generating profits and savings higher than medical expenditure (Koul et al., 2020; Table 4).
IAOH Recommendations for Administering PCV13 and PPSV23 Vaccines for Working Adults.

Source. Koul et al. (2020)
Note. PPSV = pneumococcal polysaccharide vaccine; PCV = pneumococcal conjugate vaccine. aAdminister one dose of PCV13 followed by one dose of PPSV23 at least 1 year after PCV13. PCV13 is recommended based on the shared clinical decision-making for adults who do not have an immunocompromising condition, CSF leak, or cochlear implant, and who have not previously received PCV13. Immunocompromising conditions include chronic renal failure, nephrotic syndrome, immunodeficiency, iatrogenic immunosuppression, generalized malignancy, HIV infection, Hodgkin’s disease, leukemia, lymphoma, multiple myeloma, solid organ transplants, congenital or acquired asplenia, sickle cell disease, or other hemoglobinopathies.
API Guidelines on Immunizations During COVID 19 Pandemic
During the COVID19 pandemic, API guidelines stated that as per the World Health Organization (WHO), “pneumococcal vaccines are highly recommended to protect one’s health during the pandemic” (Arulrhaj et al., 2020; Tables 5 and 6).
Recommended Immunization Schedule for Pneumococcal Vaccine in Adult Individuals per API Guidelines.

Source. Arulrhaj et al. (2020).
Note. PCV = pneumococcal conjugate vaccine; PPSV = pneumococcal polysaccharide vaccine.
Differences Between Plain Polysaccharide and Protein-Polysaccharide Conjugate Vaccines.

Source. Clutterbuck et al. (2012), Pollard et al. (2009), and Siegrist (2008).
Note. PPSV = pneumococcal polysaccharide vaccine; PCV = pneumococcal conjugate vaccine.
Discussion
Elderly people are at a significantly higher risk of getting infected and experiencing worse outcomes of an episode of pneumococcal disease, with higher chances of developing complications. The lack of data on CAP epidemiology is attributed to the unavailability of effective surveys in India and unreliability of available hospital statistics from certain centers (“Epidemiology of community-acquired pneumonia,” 2013). A study conducted on CAP-related hospitalized patients in New Delhi reported a mortality rate of nearly 2.5-fold higher in patients >50 years than in those aged <50 years. S. pneumoniae was the commonest identified bacteria (Dey et al., 1997).
Infections acquired in older adults are more severe than those in children. Immunization with available pneumococcal vaccines is an effective preventive measure to combat pneumococcal infection. In the United States, CDC recommends the elderly to receive pneumococcal vaccination. In India, PCV13 has been approved for the elderly population to prevent pneumonia and invasive pneumococcal disease caused by 13 pneumococcal serotypes in adults >50 years (Verma et al., 2014).
A 14-valent polysaccharide vaccine was first licensed in 1977. In 1983, a 23-valent polysaccharide vaccine was licensed (Immunization Action Coalition, 2022). PPSV23 contains serotypes 1, 2, 3, 4, 5, 6B, 7F, 8, 9N, 9V, 10A, 11A, 12F, 14, 15B, 17F, 18C, 19F, 19A, 20, 22F, 23F, and 33F (Solanki et al., 2017). A 7-valent PCV was first licensed in 2000. In 2010, PCV13, the 13-valent pneumococcal conjugate vaccine was launched, (The Better India, 2018) which includes serotypes 1, 3, 4, 5, 6A, 6B, 7F, 9V, 14, 18C, 19A, 19F, and 23F (Solanki et al., 2017).
Among older adults, PCV13 was effective in preventing vaccine-type pneumococcal, bacteremic, and non-bacteremic CAP and vaccine-type invasive pneumococcal disease (Bonten et al., 2015). The Louisville study demonstrated real-world effectiveness of PCV13 against vaccine-type CAP in adults aged ≥65 years following its introduction into a national immunization program (McLaughlin et al., 2018). A study reported that bacteremic pneumococcal infections caused high mortality, especially in patients aged ≥65 years living in old-age homes. The wider use of pneumococcal vaccine can substantially benefit the elderly population (Ho et al., 2007).
There is only one published multicenter study (Solanki et al., 2017) that assessed the safety and immunogenicity of PCV13 in 1,000 Indian adults 50 to 65 years of age. In this study, apart from being safe, PCV13 elicited robust immune responses against all 13 pneumococcal serotypes as reflected by the magnitude of geometric mean fold rises in functional antibody levels from before to 1 month after vaccination. This study Madhu, S. V., Makkar, B. M., indicated that a single dose of PCV13 has the potential to protect against vaccine-type pneumococcal disease in adults aged 50 to 65 years.
In the absence of empirical studies, each medical society in India formulates its own recommendations and guidelines based on the consensus of the members of the society; available literature from peer-reviewed journals, and international guidelines. Thus we attempted a narrative literature review to discuss in detail the Pneumococcal vaccine recommendations in India from all published guidelines/consensus recommendations from diverse medical societies in the country.
On June 20, 2012, the Advisory Committee on Immunization Practices (ACIP) recommended the use of PCV13 for adults (high risk) aged ≥19 years with immunocompromising conditions. However, the ACIP decision to recommend PCV13 use (routine) among older adults was deferred until data became available on the efficacy of PCV13 against non-invasive pneumococcal pneumonia among adults. In June 2014, the results of CAPiTA trial became available, following which the ACIP recommended routine use of PCV13 and PPSV23 among adults aged ≥65 years on August 13, 2014 (Centers for Disease Control and Prevention [CDC], 2012; Ramasubramanian, 2015).
The ACIP recommendations were amended in 2015 to simplify the spacing between PCV13 and PPSV23 in adults more than 65 years (Tomczyk et al., 2014). The guidelines state that the recommended interval for adults receiving PCV13 and PPSV23 should be at least 1 year, irrespective of the sequence. In summary, PCV13 is administered first followed by PPSV23 with a gap of at least 1 year. If an adult above 65 years received PPSV23, he/she will receive PCV13 after 1 year as per the older recommendation (CDC, 2022).
The ACIP 2019 guidelines recommend PCV13 for all adults aged ≥65 years based on shared clinical decision-making. This group includes those aged ≥65 years, who do not have CSF leak, immunocompromising condition, or cochlear implant. This clinical decision-making may also consider the risk for pneumococcal disease from underlying medical conditions or an individual patient’s risk for exposure to PCV13 serotypes. The 2019 ACIP recommendations for the use of PCV13 and PPSV23 vaccines in adults ≥65 years are depicted in Table 7 (Matanock et al., 2019).
ACIP Recommendations for the Use of PCV13 and PPSV23 Vaccines in Adults Aged 19 to 64 and ≥65 Years.
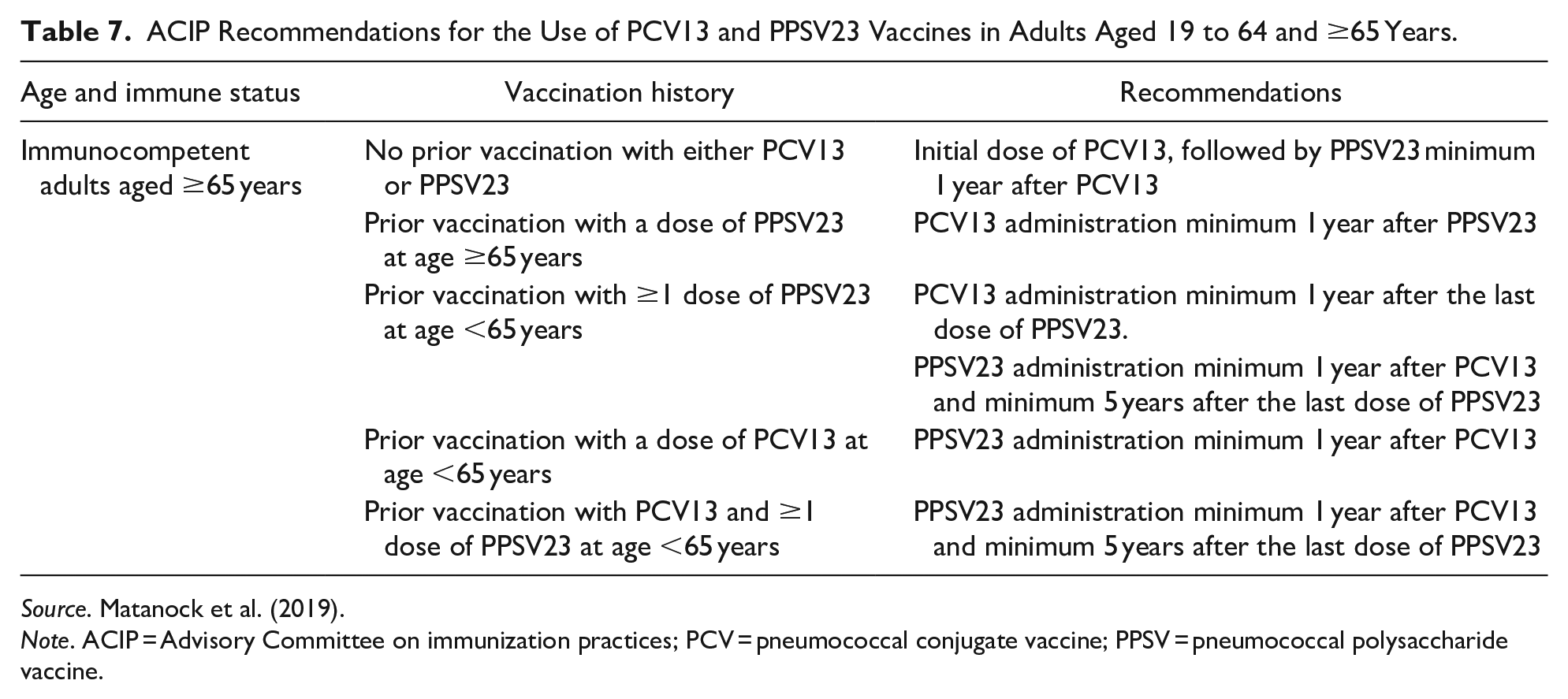
Source. Matanock et al. (2019).
Note. ACIP = Advisory Committee on immunization practices; PCV = pneumococcal conjugate vaccine; PPSV = pneumococcal polysaccharide vaccine.
In summary, the ACIP recommends administration of PCV13 to:
• Patients aged ≥65 years who have not previously received PCV13.
• Persons residing in nursing homes or other long-term care facilities.
• Persons residing in settings with low pediatric PCV13 uptake.
• Persons traveling to settings with no pediatric PCV13 program.
• Patients aged ≥65 years with chronic heart, lung, or liver disease, diabetes, alcoholism, and those who smoke cigarettes or who have more than one chronic medical condition (National Comprehensive Cancer Network, 2020).
The Indian recommendations outlined in our review are in line with the ACIP recommendations. ICS—NCCP guidelines 2020 guidelines are different from the ACIP guidelines, in terms of recommendation regarding vaccination with PCV13 vaccine. The ACIP guidelines are based on the fact that in the United States, PCV13 is being administered to all children since the year 2010, and therefore, there is herd immunity, which prevents adults from being affected by the 13 serotypes covered in PCV13. PCVs are frequently part of a national immunization program, resulting in high levels of use in the infant population in developed countries. In countries with mass childhood vaccination through national immunization program, a reduction in pneumococcal disease is seen in elderly due to herd effect. In these countries due to reduction in disease caused due to vaccine type serotypes from elderly, the coverage difference between PCV13 and PPSV23 is increasing. Such effect is not seen in countries without mass childhood immunization coverage like India. In India, pneumococcal vaccination in the pediatric immunization schedule has only been incorporated recently. Thus, the concept of herd immunity is not applicable. Hence, in the current guidelines, for individuals aged 19 to 64 years and with health conditions as stated above, we recommend vaccination with PCV13 followed by PPSV23 (Dhar et al., 2020).
Among international vaccination guidelines, the National Comprehensive Cancer Network (NCCN) 2020 recommends that an individual with cancer or hematopoietic cell transplantation (HCT) should not be immunized if he/she is allergic to vaccines, has abnormal white blood cell counts, taking immunosuppressive drugs, undergoing chemotherapy, or suffering from any infection (Table 8).
• Pneumococcal vaccines should be considered and encouraged for adult cancer survivors, when there is a health condition that weakens the immune system.
• PCV13 administered at least 8 weeks before PPSV23.
• PPSV23 once, then again after 5 years, if first administered when aged <65 years.
While the 2018 IMA guidelines do not recommend any vaccination for patients with HCT, the Indian oncology societies are yet to develop formal recommendations for pneumococcal vaccines (Tandon et al., 2018).
Recommendations for Vaccination in Patients With HCT.

Source. National Comprehensive Cancer Network (2020).
Note. HCT = hematopoietic cell transplantation.
The Portuguese Respiratory Society (2014) recommended the sequential administration of PCV13 and PPSV23 in adults ≥50 years including patients with chronic asthma under inhaled corticosteroid (Sociedade Portuguesa de Pneumologia, 2014). The Spanish Society of Pneumology and Thoracic Surgery (2013) in a joint consensus document by several medical societies recommends the use of PCV13 in chronic respiratory diseases (including COPD, asthma, and interstitial lung disease; Picazo et al., 2013).
The PREVENT expert opinion of Saudi Arabia states that pilgrims in mass gatherings are vulnerable to pneumococcal infections due to age and previous illnesses, intense crowding, and air pollution, and hence, they should be advised to receive pneumococcal vaccines (Rashid et al., 2013). The mass gatherings consensus statement from India provides reference of the PREVENT expert opinion (Mathai et al., 2016).
In the United States, CDC strongly recommends hospitals, long-term care facilities, pharmacies, and other health care settings, to implement standing orders for ACIP recommended vaccines, including pneumococcal vaccines in elderly population (Power Pak, 2017). A similar practice is necessary in India for prevention of pneumococcal disease in the elderly.
Conclusion
Elderly people are at a significantly higher risk of suffering from pneumococcal disease and associated complications. The different scientific bodies in India recommend pneumococcal vaccines. The recommendation is to begin with PCV13 followed by PPSV23 after a specific time interval.
Immunization with available pneumococcal vaccines is a cost-effective preventive measure to combat pneumococcal infections and reduce mortality rate and disability among the elderly. The need of the hour is to integrate adult vaccination for pneumococcal diseases into public and private access programs, especially in old-age homes where the aging population aspires for a disease-free life.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that there is no conflict of interest. The authors were employees of Pfizer India during the development of the manuscript and are no longer employed with Pfizer India.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This review was funded by Pfizer Vaccines.
