Abstract
Introduction
Osteoporosis is a condition characterized with an increased risk of fracture and low bone mass and microarchitectural deterioration of bone tissue, thereby leading to enhanced bone fragility and a consequent increase in fracture risk. The prevalence of osteoporosis in the lumbar spine at the age of 40 or more is 3.4% for men and 19.2% for women and that in the femoral neck is 12.4% for men and 26.5% for women (Yoshimura et al., 2010; Yoshimura, Muraki, Oka, Mabuchi, En-Yo, et al., 2009). The incidence is 0.76% per year in the lumbar spine and 1.8% per year in the femoral neck; therefore, osteoporosis is considered a common disease (Yoshimura, Muraki, Oka, Mabuchi, Kinoshita, et al., 2009). Furthermore, it has been reported that fractures caused by osteoporosis, particularly femoral neck fractures, not only reduce mobility and living ability but also increase mortality (Nguyen et al., 2007; Suzuki & Yoshida, 2010). However, calcium and vitamin D intake can suppress the onset of osteoporosis, and hip protectors have been reported to prevent femoral neck fractures (Koike et al., 2009; Ondrak & Morgan, 2007). Furthermore, drugs that increase bone density, such as bisphosphonates, have also been used in clinical practice; the diagnosis, prevention, and treatment of osteoporosis in daily medical care are extremely important (Bone et al., 2004). A marked decrease in bone density with age has also been reported (Steiger et al., 1992). Therefore, osteoporosis is considered a very important disease from the geriatrics point of view.
Bone density is defined as bone mineral density (g/cm3) per unit volume, which may be high in the case of fractures, deformities, and osteosclerotic changes (Fukunaga et al., 2006). In other words, the risk that lumbar bone density being high in patients with lumbar compression fracture exists, and therefore, a diagnosis of osteoporosis might be avoided or missed. However, to date, there are no studies evaluating the association between the presence of lumbar compression fracture and lumbar bone density.
In this study, we retrospectively analyzed bone density measured at our hospital and evaluated a possible correlation between the presence of lumbar compression fracture and lumbar bone density.
Methods
We extracted data from patients whose bone density was measured by Dual-energy X-ray absorptiometry (DXA) at Kennan Hospital (Tsuchiura city, Ibaraki, Japan) from July 2017 to June 2019. We included patient in whom the presence or absence of lumbar compression fracture (L1–4) at that time could be confirmed by an image (X-ray, computed tomography [CT], or magnetic resonance imaging [MRI]). We excluded patients in whom both bone density of lumbar spine and femoral neck were not measured.
Bone density was measured using a Horizon DXA System (Hologic, Inc. Massachusetts, USA). Good image quality and accuracy of this device have previously been reported (Hangartner, 2007). The DXA system of the Kennan Hospital regularly undergoes accuracy control using phantoms. Furthermore, the data under the same conditions, reproducibility of patient positioning, and reproducibility of the analysis area are recorded and the measurement accuracy is obtained.
Bone density was measured three times using DXA during the period in seven patients, and none of the patients underwent four measurements. The short-term
The data of interest were as follows: age, sex, lumbar bone density (standard deviation value compared to the bone density in healthy young adult – T-score, g/cm3), femoral neck bone density (T-score, g/cm3), presence or absence of lumbar compression fracture, number of lumbar compression fracture, old or new lumbar compression fracture, and grade of lumbar compression fracture.
New fractures were defined as fractures within 6 months of injury compared to previous images or showing high intensity on MRI-short tau inversion recovery. The grade of lumbar vertebral fracture was defined according to the Semi-quantitative method (Genant et al., 1993). That is, vertebra were graded on visual inspection and without direct vertebral measurement as normal (Grade 0), mildly deformed (Grade 1, approximately 20%–25% reduction in anterior, middle, and/or posterior height and a reduction of area 10%–20%), moderately deformed (Grade 2, approximately 25%–40% reduction in any height and a reduction in area 20%–40%), and severely deformed (Grade 3, approximately 40% reduction in any height and area).
We compared bone density of fractured lumbar compression vertebral body and nonfractured lumbar compression vertebral body, correlation of lumbar spine T-score and femoral neck T-score, evaluation of bone density of lumbar spine by number of lumbar compression fracture, comparison of bone density by fracture grade, and comparison of bone density of new and old fractures.
All values are expressed as means ±
Results
In the evaluation period, DXA bone density was measured in 510 patients. Of these, 20 patients were excluded because only femoral neck or lumbar spine was measured, and a further 305 patients were excluded as it was not possible to confirm presence or absence of lumbar spine fracture at the time. In total, 185 patients were included in the analysis; 20 males and 165 females, with mean age of 76.9 ± 7.5 years. Detailed data of each vertebral body are shown in Table 1.
Number of Fractures Per Vertebral Body According to Grade and New or Old (

Fractured Compression Vertebral Bodies Had Higher Bone Density Than Nonfractured Vertebral Bodies
The bone density of fractured compression vertebral body was significantly greater (0.830 ± 0.229 g/cm3, number of vertebral bodies = 132) than nonfractured vertebral body (0.765 ± 0.178 g/cm3, number of vertebral bodies = 608,

Fractured vertebral bodies had significantly higher bone density than nonfractured vertebral bodies (
The bone density of fractured compression vertebral body, even in males only, was significantly higher (0.790 ± 0.203 g/cm3, number of vertebral bodies = 110) than that of nonfractured vertebral body (0.752 ± 0.171 g/cm3, number of vertebral bodies = 550,

Fractured vertebral bodies had significantly higher bone density than nonfractured vertebral bodies regardless of gender or age: (A) a group with only females (
The bone density of fractured compression vertebral body was significantly higher (0.857 ± 0.260 g/cm3, number of vertebral bodies = 21) than that of nonfractured vertebral body even for those younger than 75 years (0.789 ± 0.162 g/cm3, number of vertebral bodies = 271,
Therefore, fractured vertebral bodies had significantly higher bone density than nonfractured vertebral bodies regardless of gender or age.
As the Number of Vertebral Compression Fractures Increased, the Correlation Between Lumbar Spine T-Score and Femoral Neck T-Score Weakened
Pearson’s correlation coefficient was calculated to evaluate the correlation between lumbar spine and femoral neck T-scores. The correlation coefficient between lumbar spine and femoral neck T-scores was 0.782 in the nonvertebral compression fracture group (

The correlation between lumbar spine T-score and femoral neck T-score weakens as the number of vertebral fractures increases: (A) a group without lumbar vertebral fracture (
Average Bone Density of L1–4 Did Not Increase Significantly as the Number of Vertebral Compression Fracture Increased
The average bone density of L1–4 was 0.792 ± 0.151 g/cm3 in the nonvertebral compression fracture group (
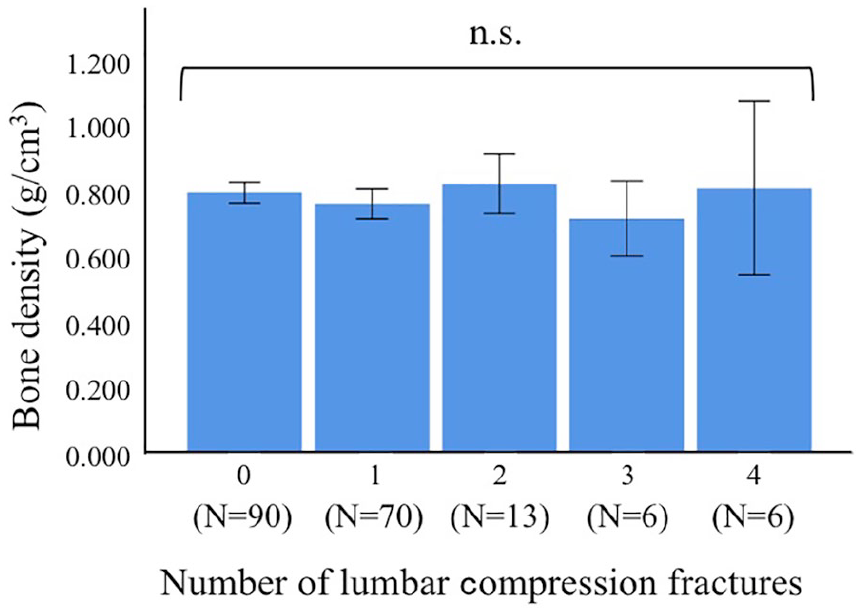
The average bone density of L1–4 did not increase significantly even with increasing number of vertebral fractures (
There Was No Significant Difference in Bone Density Depending on the Grade of Fracture
The bone density of vertebral fracture was 0.888 ± 0.253 g/cm3 in Grade 1 (number of vertebral bodies was 35), 0.786 ± 0.191 g/cm3 in the Grade 2 (number of vertebral bodies was 48), and 0.830 ± 0.236 g/cm3 in the Grade 3 (number of vertebral bodies was 49). There was no significant difference between grade of fracture groups (

No significant difference in bone density between grades of lumbar vertebral fracture (
There Was No Significant Difference in Bone Density in Old Versus New Fractures
The bone density of new fractures (0.766 ± 0.162 g/cm3, number of vertebral bodies was 34) were not significantly different from the old fractures (0.779 ± 0.193 g/cm3, number of vertebral bodies was 49,

Discussion
Bone density is useful for fracture risk assessment, particularly at the age of 65 and more (Cummings et al., 1993; Johnell et al., 1995). It is reported that DXA of the spine is one of the most useful evaluations for high-risk patients (Cummings et al., 1995; Kanis et al., 2001; Torgerson et al., 1996). In addition, it has been recommended that bone density be measured at both the lumbar spine and the femoral neck (Leib et al., 2004).
Bone density measured by DXA is the bone mineral density (g/cm3) of a unit volume; in the case of fracture, deformity, and osteosclerotic change, a high value of bone density may be shown (Fukunaga et al., 2006). However, we could not find previous reports evaluating between the presence of lumbar compression fracture and the lumbar bone density.
In this study, fractured compression vertebral bodies had significantly higher bone density than nonfractured vertebral bodies. Furthermore, the correlation between lumbar spine and femoral neck T-scores became weak with an increasing number of lumbar vertebral compression fractures. Previous reports suggest that the bone density of the lumbar spine and femoral neck should have a correlation of approximately 0.7 (Wilson, 1977). The volume of the vertebral body is reduced by the compression fracture of the lumbar spine; however, the total amount of mineral components such as calcium in the vertebral body is not reduced. Therefore, bone density and T-score are considered to increase. On the other hand, because lumbar vertebral fractures do not affect the bone density of the femoral neck, it is considered that dissociation occurs between the lumbar spine and femoral neck
The World Health Organization (WHO) collaboration center for metabolic bone diseases recommends the use of
Because lumbar compression fracture increases bone density, we expected that the higher the number of lumbar compression fractures or higher grade of fracture would lead to higher bone density. However, we found no significant difference in bone density depending on the number and grade of compression fractures. This might be explained by a marked decrease in bone mineral density due to aging and osteoporosis in cases of multiple fractures and/or severe fractures. That is, although the bone density was increased due to multiple or severe fractures, the increase or decrease in bone mineral density was offset since bone density was reduced secondary to aging and osteoporosis. Thus, proper evaluation must take into account the number and degree of lumbar compression fractures without judging bone fragility by bone density or T-score alone. To evaluate the presence or absence and degree of lumbar compression fracture at the time of bone density measurement, additional imaging such as X-ray, CT, or MRI is necessity.
We found no significant difference in bone density between old and new lumbar body fractures. In other words, it can be said that temporal changes in bone density reflect clinically important changes such as aging, fractures, and so on. Therefore, it is important to repeat bone density measurements.
This study refers only to lumbar compression fractures because the measurement site of lumbar vertebral bone density is L1–4 or L2–4 (Leib et al., 2004). However, the most common site of vertebral fracture is the thoracolumbar junction, which includes the thoracic spine (Cooper et al., 1992). Therefore, it is important to evaluate compression fractures of not only the lumbar spine but also the lower thoracic spine in the evaluation of bone fragility.
This study has a potential limitation in that multiple radiologists made their own measurement. This might affect accuracy and repeatability. In addition, we were not able to adjust the confounding factors of body mass index (BMI). All the subjects were Japanese and rarely suffered from obesity nor were extremely skinny; therefore, we could not collect the BMI of all subjects. Because this is a retrospective study, we did not record the BMI of all subjects. In future, further prospective studies that can eliminate the confounding factors are necessary. Furthermore, because the number of repeated measurements during the period is small, the credibility of the values of short-term
In conclusion, our data suggest that lumbar compression fractures increases bone density. This is important information for nonspecialists and patients for proper diagnosis of osteoporosis, which should not be done by bone density or T-score alone. In addition, bone fragility can be more accurately evaluated by taking images for fracture evaluation and repeatedly measuring bone density. Such warnings will help nonspecialists to correctly diagnose osteoporosis.
Footnotes
Acknowledgements
We would like to thank all staff at the Kennan Hospital, who helped with this research. The authors would like to thank Enago (![]() ) for the English language review. The first author would also like to extend his indebtedness to Yumiko Takahashi for her endless love, understanding, support, encouragement, and sacrifice throughout his work.
) for the English language review. The first author would also like to extend his indebtedness to Yumiko Takahashi for her endless love, understanding, support, encouragement, and sacrifice throughout his work.
Author Contributions
Toshihide Takahashi designed the study, analyzed and interpreted of data, and wrote the initial draft of the manuscript. Tomoya Takada assisted in the preparation of the manuscript. All other authors have contributed to data collection and interpretation, and critically reviewed the manuscript. All authors approved the final version of the manuscript, and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
This study was approved by the Kennan Hospital ethics committee (The approval number is 2019-001).
