Abstract
Introduction
Promoting and distributing evidence-based high value care, which may reduce variation in health care spending, remains a challenge. Medicare spending varies across the United States, with some areas spending 60% more per Medicare beneficiary than other areas without corresponding improvements in health outcomes (Fisher et al., 2003a, 2003b; Skinner, Fisher, & Wennberg, 2005). Although some of the variation in spending reflects demographic differences in a region, there remains substantial variation after demographic adjustments which may be indicative of inefficient health care delivery (Fisher, Bynum, & Skinner, 2009; Orszag, 2008; Singer & Lowy, 1992). Medicare spending per beneficiary was 52% higher in geographic regions in the highest spending quintile than in regions in the lowest quintile (Zuckerman, Waidmann, Berenson, & Hadley, 2010). After adjustment for demographic and health characteristics, the difference was only reduced from 52% to 33%, thus substantial variation in geographic spending remains unaccounted for (Zuckerman et al., 2010). Our goal was to assess whether physician preferences for end-of-life care were associated with variation in health care spending.
Physician and patient preferences for care may be an important source of variation in health care services and spending that are frequently unmeasured. Patient preferences appear to account for a small percentage of variation in health care costs in comparison to factors such as physician supply and patient health (Baker, Bundorf, & Kessler, 2014). Although physician preferences and knowledge of end-of-life care may be quite variable (Malhotra, Chan, Zhou, Dalager, & Finkelstein, 2015), physician preferences are less well understood. If preferences for aggressive care are correlated within geographic regions, then one would observe a pattern in which the intensity of care varies (though there need not be any corresponding variation in health outcomes; Chandra & Staiger, 2007). Cutler and colleagues used data from a cross-sectional survey of physicians to demonstrate that physician beliefs for providing “more aggressive” care were associated with higher health care spending (Cutler, Skinner, Stern, & Wennberg, 2013). Such a finding supports policies such as Choosing Wisely that aim to alter physician behavior (Cassel & Guest, 2012), though effects of the program have been modest (Rosenberg et al., 2015; Schwartz, Chernew, Landon, & McWilliams, 2015).
Spending on end-of-life care (defined as costs of care for persons aged 65 years and older in the last 6 months of life) has been used as a measure of the local intensity of health care and seems correlated to the quality of health care (Barnato et al., 2007; Fisher et al., 2003a, 2003b). Our data are from the Johns Hopkins Precursors Study, which began in 1948 and has collected detailed health and preference data from over 1,300 graduates of the Johns Hopkins School of Medicine in the ensuing years. In 1999, questionnaires asked participants to consider what types of treatment they would want if they suffered irreversible brain damage that left them unable to speak understandably or to recognize anyone. In prior publications, we have characterized the responses to the desire for 10 interventions such as cardiopulmonary resuscitation (CPR) and mechanical ventilation into “most,” “intermediate,” and “least” aggressive categories (Gallo et al., 2003; Straton et al., 2004; Wittink et al., 2008).
Building on the recognition that physician preferences may be associated with health care spending, we studied the association between a physician’s stated preferences toward various life-sustaining treatments and area-level health care spending. To do so, we linked physician data to area-level data on health care spending from the Dartmouth Atlas of Health Care in the physician’s location (www.dartmouthatlas.org). We took two approaches to examine the association between preferences and costs. First, we arrayed all hospital-related regions (HRRs) in the United States into quartiles of cost and then compared the proportion of physicians in the “most” aggressive category in lowest and highest cost quartiles. Second, we estimated hospital-related regional costs associated with specific services for physicians in the “most,” “intermediate,” and “least” categories, where we chose hospital-related costs since those costs are most likely to be under the control of the physician. We hypothesized that health care spending, especially in the last 6 months of life, would be associated with physician preferences for end-of-life care.
Method
The Johns Hopkins Precursors Study
The Johns Hopkins Precursors Study was designed in 1946 by Caroline Bedell Thomas to identify characteristics associated with premature cardiovascular disease and death (Thomas, 1951). All 1,337 students who matriculated into the graduating classes of 1948 to 1964 of the Johns Hopkins University School of Medicine were eligible. Precursors Study procedures are reviewed on a regular basis and approved by the Johns Hopkins University School of Medicine Institutional Review Board (IRB00051731).
Assessment of Preferences for Aggressive Care
In 1999, questionnaires asked participants to consider what types of treatment they would want if they suffered irreversible brain damage that left them unable to speak understandably or to recognize anyone (Gallo et al., 2003; Straton et al., 2004; Wittink et al., 2008). In the scenario, based on the Medical Directive developed by Emanuel and Emanuel (1989), the participants were told that they had no terminal illness and might remain in this brain-injured state for a long period of time. We selected the scenario of irreversible brain damage without terminal illness because previous research demonstrated that this scenario provided the greatest variability in responses compared with other vignettes in the Medical Directive (Emanuel & Emanuel, 1989; Fischer, Alpert, Stoeckle, & Emanuel, 1997). Participants were asked to state their wishes regarding the use of 10 medical interventions: CPR, mechanical ventilation, intravenous fluids, surgically placed feeding tube for nutrition, dialysis, chemotherapy, major surgery, invasive diagnostic tests, blood or blood products, and antibiotics (Gallo et al., 2003; Wittink et al., 2008). The responses for each intervention were coded a priori into a dichotomous variable as either reject (No, I would not want) or accept (yes, undecided, or trial of treatment), consistent with previous work (Gallo et al., 2003; Wittink et al., 2008). The dichotomization reflects common clinical practice in which “treatment trial” and “unsure” would translate into providing life-sustaining treatment to incompetent patients, at least initially (Emanuel, Emanuel, Stoeckle, Hummel, & Barry, 1994; Fischer et al., 1997; Straton et al., 2004). We considered wanting CPR in the context of brain injury with poor quality of life as “aggressive” treatment (Gallo et al., 2003; Wittink et al., 2008).
Area-Level Health Care Spending
We obtained area-level health care spending for the years 1999 to 2009 for the location of each physician from the Dartmouth Atlas of Health Care (www.dartmouthatlas.org). Total Medicare spending per capita in the HRR consists of spending on six components: hospitals and skilled nursing facilities, physician services, outpatient facilities, home health, hospice, and durable medical equipment. We also obtained variables related to spending specific to the last 6 months of life and to the hospitalization in which death occurred.
Analytic Strategy
Our analysis proceeded in three phases. First, to study potentially life-sustaining treatments as a pattern or set of treatments (latent “classes” or “profiles”) in contrast to a focus on individual interventions, we applied the latent class model (Collins & Lanza, 2009), as in prior work on preferences for end-of-life treatment (Wittink et al., 2008). For estimation of the latent classes, analysis was performed using Mplus version 7.1 and PROC LTA in SAS version 9.4 (SAS Institute Inc., Cary, NC), which both use an efficient estimation-maximization algorithm for maximum likelihood estimation (Dempster, Laird, & Rubin, 1977). Model choice, in terms of the number of latent classes, was determined through examination of fit indices and model identification (e.g., homogeneity and latent class separation) as well as in relation to clinically interpretable results. Bayesian information criteria (BIC; Schwarz, 1978) were used to compare nonnested models that differed in the number of latent classes. A smaller value of BIC indicates a better model fit. The four-class model yielded the best fit over two- and three-class models using statistical criteria (BIC2 = 5369.85, BIC3 = 4975.21, and BIC4 = 4922.63). However, the more parsimonious model was chosen because of good latent class separation with the latent classes appearing conceptually distinct overall (Collins & Lanza, 2009). For this study, we assigned each physician to the latent class with the highest probability of class membership.
Second, we assigned hospital referral regions to aggressiveness category based on the latent class level of the physicians located in the zip codes of the HRR. Of 306 HRRs, 201 included at least one Precursors physician. The generalized linear mixed effects model was used with specified gamma distribution and log-link function (Fitzmaurice, Laird, & Ware, 2011). Third, we sorted all 306 HRRs in the United States into quartiles year by year, based on spending for intensive care unit (ICU) care in the last 6 months of life and, separately, for spending during the hospitalization in which death occurred, to examine whether more physicians with aggressive preferences were to be found in the HRRs with the most spending. Year and clustering effects at the HRR level were taken into account during analyses. Results were considered statistically significant if the two-sided p value was less than .05. Statistical analyses were performed using SAS, version 9.4 (SAS Institute Inc.).
Results
Sample Characteristics
Of the 1,016 participants to whom questionnaires were sent in 1999, 796 (78.3%) responded with their preferences for end-of-life care in response to the brain injury scenario. Among them, we excluded 59 physicians with incomplete information on zip code, leaving a sample size of 737. The mean (SD) age of the study sample at baseline was 68.0 (5.2) years. Reflecting medical school enrollment between 1948 and 1964, the study sample was 92.2% White men.
Distribution of Physician Preferences Within All HRRs Arrayed by Spending
We arrayed all HRRs into quartiles in 1999 (the year closest to when the preferences were assessed) and compared the distribution of physician preferences for end-of-life care (Table 1). The prevalence of physicians with the most aggressive preferences was significantly higher in the most costly HRRs for ICU care in the last 6 months of life (top half of Table 1). Similarly, the prevalence of physicians with the most aggressive preferences was significantly higher in the most costly HRRs for ICU care during the hospitalization in which death occurred (bottom half of Table 1). Separate analyses for years 2000 to 2009 were similar to the results in Table 1.
All HRRs Arrayed According to Quartile of Spending in 1999, Showing Distribution of Precursors Physician Preferences for End-of-Life Care According to Quartile of Total ICU Spending in the Last 6 Months of Life (Top Half of Table) and ICU Spending in the Hospitalization During Which Death Occurred (Lower Half of Table).

Source. Data from the Johns Hopkins Precursors Study (1999) and the Dartmouth Atlas of Health Care (1999-2009).
Note. Spending estimates are age, sex, and race adjusted. HRR = hospital-related region; ICU = intensive care unit.
Physician Preferences and Total Area-Level Spending
We examined the association between physician preferences and annualized area-level Medicare spending (for hospital and skilled nursing facility, physician, outpatient, home health, hospice, and durable medical equipment spending) according to preferences for end-of-life treatment in 1999. For hospital and skilled nursing facility spending, physician spending, and total spending, the HRRs with physicians who expressed the most aggressive preferences had few differences in spending compared with physicians in intermediate or least aggressive categories.
Care in the Last 6 Months of Life
Table 2 focuses attention on health care spending and services use in the last 6 months of life and during the hospitalization in which death occurred. ICU charges in the last 6 months of life in HRRs of physicians with the most aggressive profile of preferences for their personal end-of-life care were significantly higher compared with those HRRs with physicians with the least aggressive profile (US$9,318 in HRRs of most aggressive physicians compared with US$7,511 in HRRs of least aggressive physicians, p = .01). Inpatient spending was higher in HRRs of physicians with the most aggressive profile but did not reach conventional levels of statistical significance.
Association of Physician Preferences With Area-Level Hospital and Inpatient Utilization During the Last 6 Months of Life and During the Hospitalization in Which the Death Occurred, Expressed as a Difference From the Mean (n = 737).
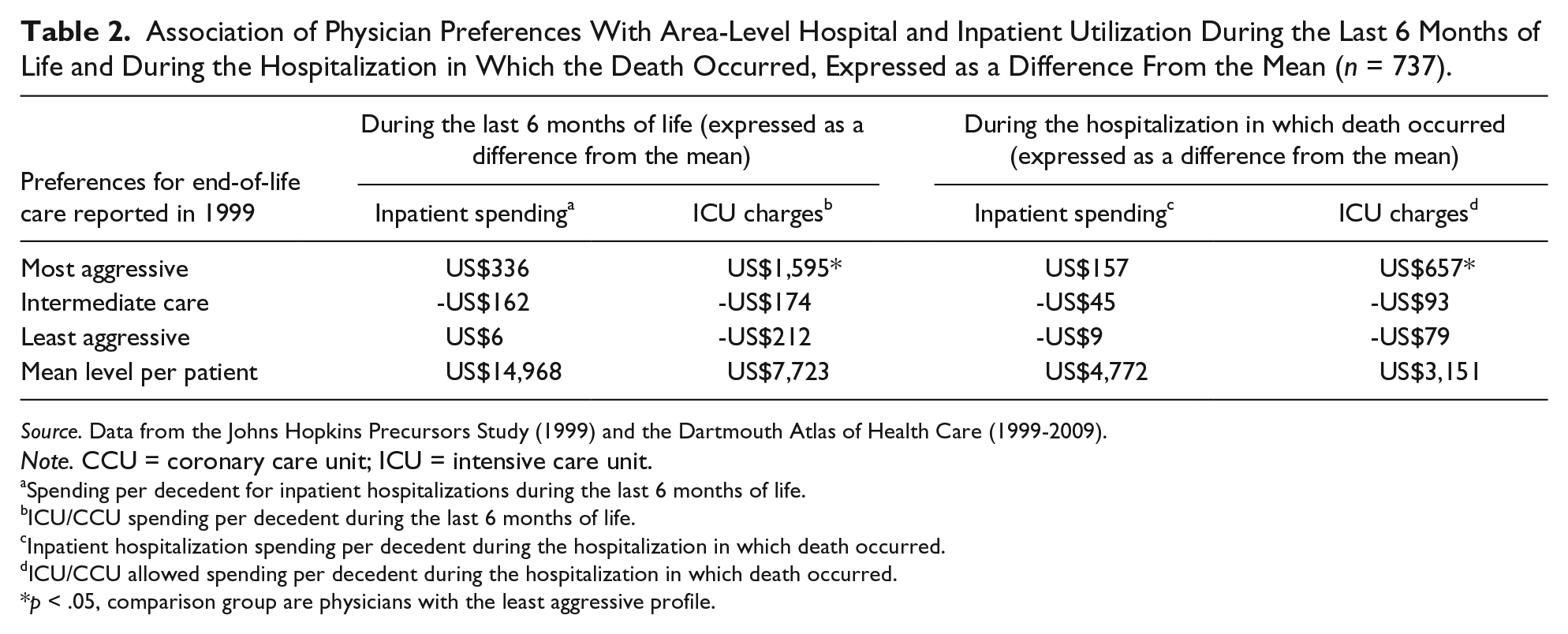
Source. Data from the Johns Hopkins Precursors Study (1999) and the Dartmouth Atlas of Health Care (1999-2009).
Note. CCU = coronary care unit; ICU = intensive care unit.
Spending per decedent for inpatient hospitalizations during the last 6 months of life.
ICU/CCU spending per decedent during the last 6 months of life.
Inpatient hospitalization spending per decedent during the hospitalization in which death occurred.
ICU/CCU allowed spending per decedent during the hospitalization in which death occurred.
p < .05, comparison group are physicians with the least aggressive profile.
Care During the Hospitalization in Which Death Occurred
A similar pattern was observed for the hospitalization in which the death occurred (right side of Table 2). During the hospitalization in which death occurred, ICU charges in HRRs of physicians with the most aggressive profile of preferences for their personal end-of-life care were significantly higher compared with physicians with the least aggressive profile (US$3,808 per person per year in HRRs of most aggressive physicians compared with US$3,072 in HRRs of least aggressive physicians, p < .05). Inpatient spending in the hospitalization in which death occurred was higher among physicians with the most aggressive profile but did not reach statistical significance.
Discussion
We found that preferences for more treatment were associated with higher ICU spending in the last 6 months of life and in the hospitalization in which the death occurred. The methods we used were similar to other studies using cost and services data from the Dartmouth Atlas hospital referral regions (Barnato et al., 2007). Medicare spending in the last year of life is substantial (Hogan, Lunney, Gabel, & Lynn, 2001), with widespread geographic variation (Goodman, Esty, Fisher, & Chang, 2011), and spending may be increasing (Calfo, Smith, & Zezza, 2011). The estimate we provided for the association of physician preferences with spending and services, while a small proportion of total spending per patient, is large when extrapolated to the national level. For example, the excess US$1595 ICU charges associated with the most aggressive physicians in the last 6 months of life would be estimated to an annual cost of over US$33 million nationally. The expressed preferences of physicians for end-of-life care belie an underlying tendency toward an aggressive management style. More research is needed to gain a better understanding of the physician factors that drive use of services and costs at the end of life.
We used data on physician preferences to study the association between physician preferences and health care utilization. Consistent with physician preferences affecting the utilization of health care, we found that preferences of physicians were associated with health care spending at the Hospital Referral Region level and were correlated with measures of hospital and ICU utilization in the last 6 months of life. Several important caveats should be kept in mind. First, the nature of the data is ecological. Physicians are not randomly allocated to hospital referral regions. Rather, our results may reflect the tendency of physicians to adopt their practice style to the community in which they practice (Molitor, 2016). We did not account for hospital characteristics, as often practice styles of individual physicians mirror that of their hospitals/facilities. Second, our data capture preferences for their own care that may differ from a physician’s preferences for providing care to others. However, patients tend to have attitudes about end-of-life care similar to their physicians (Gramelspacher, Zhou, Hanna, & Tierney, 1997; Schneiderman, Kaplan, Pearlman, & Teetzel, 1993). Third, Precursors physicians are not be representative of all physicians in an HRR. We cannot be sure the extent to which physicians in the cohort are fully engaged in practice, whether they practice in the same zip code as their home or what fraction of their patients are covered by Medicare. Finally, while preferences may change over time, the percentage of physicians in a given category does not change substantially over time (Wittink et al., 2008). Future work using individual-level data on physicians and matching to the physician’s specific patients may provide the means to assess these alternative hypotheses and mitigate the limitations of this analysis.
Physician responses to vignettes or scenarios have yielded valuable insights into the role that physician beliefs and preferences play in the patient treatment or care choice (Cutler et al., 2013; Dresselhaus, Peabody, Luck, & Bertenthal, 2004; Lucas, Sirovich, Gallagher, Siewers, & Wennberg, 2010; Mandelblatt et al., 2012; Matlock et al., 2010; Obermeyer, Powers, Makar, Keating, & Cutler, 2015; Peabody, Luck, Glassman, Dresselhaus, & Lee, 2000). Several studies based on physician responses to scenarios have suggested that more aggressive management style was associated with higher costs. Using scenarios about end-of-life care for cardiac patients, Matlock showed that patients of cardiologists who responded to the scenario in ways contrary to guidelines had more health care use in the last 6 months of life (Matlock et al., 2010). Similarly, oncologist “style” of practice, as assessed with vignettes, was associated with rates of use of chemotherapy for older women (Obermeyer et al., 2015). Cutler et al. (2013) elicited physician preferences for aggressiveness of treatment with scenarios, finding that physician beliefs were strongly associated with regional variation in health services use and spending. Cutler found that physician beliefs were not predictable from physician or practice characteristics, implying that direct assessment of physician attitudes and beliefs is necessary, as we have done in our study.
Ours was one of the few studies that have used direct measures of physician preferences to examine the influence on health care spending. Physician preferences may be an important determinant of health care use, and these preferences may be important to address in measures intended to alleviate geographic variations in cost. Strategies intended to better align health care spending with patient values will need to consider individual physician preferences and practice style as well. Choosing Wisely is an effort of the American Board of Internal Medicine Foundation with over 70 professional organizations as partners to encourage conversations between providers and patients to carefully consider the value of tests and procedures that may be of limited benefit to the patient. However, it remains to be seen whether such efforts are sufficient to change physician and patient behaviors and reduce health care spending (Rosenberg et al., 2015; Schwartz et al., 2015).
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Institute of Nursing Research (R01 NR014068, Dr. J.J.G., principal investigator [PI]) and the Agency for Healthcare Research and Quality (R01 HS024106, Dr. R.J., PI).
