Abstract
Introduction
Obesity in an older adult population leads to functional impairment (Schaap, Koster, & Visser, 2013), morbidity, and institutionalization (Zizza, Herring, Stevens, & Popkin, 2002). Current interventions are largely limited to research settings focusing on testing strict dietary and in-person coaching in intensive exercise strategies that are unlikely to be practical in usual clinical settings (Villareal et al., 2011). These interventions are especially challenging for older obese adults in rural settings due to major barriers associated with transportation, mobility, and access (Iezzoni, Killeen, & O’Day, 2006). Commercially available wearable sensors and activity-tracking devices potentially provide real-time feedback and support for health behavior change in this older population (McMahon et al., 2016; Mercer et al., 2016). Fitbit is one of many commercial devices available that has demonstrated potential in improving adherence and physical activity in different populations (Evenson, Goto, & Furberg, 2015). However, little is known about its usability and acceptability in elderly adults with obesity, and we are unaware of any studies examining its use in a rural setting. Elders are among the fastest growing subgroups using smartphone devices (Pew Research Center. Internet, Science & Tech., 2014) that readily link to commercially available activity-tracking devices and provide real-time feedback and support for health behavior change. We describe a pilot study that determined the feasibility and acceptability of using Fitbit for supporting a lifestyle intervention among rural obese older adults. We also assessed patient activation as a means for changing behavior and increasing physical activity, and used semi-structured interviews to assess potential barriers specific to rural areas.
Method
The overarching goal was to determine whether Fitbit has the potential to be used to achieve behavioral activation in older adults. Our local institutional review board approved the study.
Participant Characteristics
A convenience sample was recruited from a geriatrician’s primary care practice at a rural academic medical center from December 2014 to September 2015. In advance of the visits, participants’ medical records were reviewed to target recruitment. All participants included in the sample were aged 65 to 80 years, were English speaking, had body mass index (BMI) ≥30kg/m2, and had a medical need to lose weight. We included participants who owned and used a compatible smartphone device. Participants with cognitive impairment based on the six-item Callahan screen (Callahan, Unverzagt, Hui, Perkins, & Hendrie, 2002), a nursing or hospital facility admission within the past 6 months, or with severe comorbidities (Batsis, Singh, & Lopez-Jimenez, 2014) were not asked to be part of the study. Other exclusions consisted of current smokers, a weight loss ≥4.5 kg in the past 6 months, or previous history of bariatric surgery. Individuals on medications inducing weight loss were also excluded.
Study Intervention
Eligible participants were recruited, and eligibility criteria, VES-13, and Callahan questions were assessed by phone. Participants required a score of <4 on the Vulnerable Elder Survey–13 (VES-13) as a measure of frailty (Saliba et al., 2001). Participants were invited to clinic and explained the study. Following informed consent, study measures were completed. A Fitbit Zip was configured and provided; the application was installed on their device following a brief demonstration. A 10-min phone call was performed at 1 week to encourage and troubleshoot. At 4 weeks, participants returned their Fitbit device and completed questionnaires. All were offered a monetary incentive, but none accepted it.
Measures
Height and weight were measured at baseline using a stadiometer and a calibrated digital scale by trained medical assistants. Medications, laboratory values, and socio-demographic information were abstracted from the medical record. Three outcomes were assessed at pre/post intervention with a satisfaction survey at study conclusion. The Self-Efficacy for Exercise Behaviors survey (Sallis, Pinski, Grossman, Patterson, & Nader, 1988) consists of 12 questions classifying one’s ability to “stick to it” or “make time” and has been extensively validated in health-promotion research. Self-reported quality of life was evaluated using a 5-point Likert-type scale (1 = high, 5 = low). The Patient Activation Measure (PAM; Hibbard, Stockard, Mahoney, & Tusler, 2004) assessed knowledge, confidence, and skills for managing health using a 13-question scale. A Fitbit satisfaction survey targeting ease of use and acceptability was developed for this study, adapted from a prior study (Naslund et al., 2016).
Qualitative Analysis
A semi-structured interview was conducted to expand upon participants’ responses to the survey and to understand the experience with Fitbit. Interviews were digitally recorded, transcribed for thematic analysis, and reviewed. Transcripts were coded independently. A list was prepared of all comments and grouped together according to broad categories (e.g., incentives, motivation, satisfaction, challenges, awareness, self-monitoring, group support, feedback, usefulness). Both researchers reviewed and grouped the categories forming four overarching themes. Disagreements were resolved through consensus.
Statistical Analysis
Continuous variables are presented as mean ± standard deviation, and categorical variables as counts (%). Questionnaires were assessed per guidelines and information imputed accordingly. Two-sample paired t tests compared baseline/follow-up assessments. Analyses were conducted using STATA v.13 (College Station, Texas). A p value of <.05 was considered statistically significant.
Results
Of the eight participants, mean age was 73.4 ± 4.0 years (50% female). Four participants took part in the study between December and January, one in June, and two in August. Baseline weight and BMI were 96.6 ± 23.9 kg and 34.5 ± 4.5kg/m2, respectively, which remained the same at 30 days (96.9 ± 23.2 kg and 34.5 ± 4.5kg/m2, ps = .66 and .96). Comorbid conditions included hypertension (n = 6 [75%]), dyslipidemia (n = 5 [63%]), diabetes (n = 3 [38%]), osteoarthritis (n = 4 [50%]), and sleep apnea (n = 4 [50%]). Quality of life increased, but was not significant (2.38 vs. 3.00; p = .32). Decreases were observed at follow-up in exercise confidence (sticking to it: 34.5 ± 3.3 to 30.9 ± 4.3, p = .04, and in making time for exercise: 18.9 ± 1.3 to 17.0 ± 2.6, p = .03). No changes were observed in patient activation (45.4 ± 4.3 vs. 45.0 ± 3.9, p = .63) for scores representing “lacks confidence and knowledge to take action.” Table 1 presents satisfaction data. There were positive trends toward using Fitbit and that participants would recommend Fitbit (100%). Ease of use (n = 7 [87.5%]) and useful feedback (n = 5 [37.5%]) were observed. Qualitative data are presented in Table 2. No adverse events were observed. The one participant who was very dissatisfied noted the following specific comment: “I’m not sure what I was planning to get out of it. I was hoping to improve my health somewhat by doing it [Fitbit] . . .. I was hoping to get some weight loss.”
Satisfaction Survey of Using Fitbit.
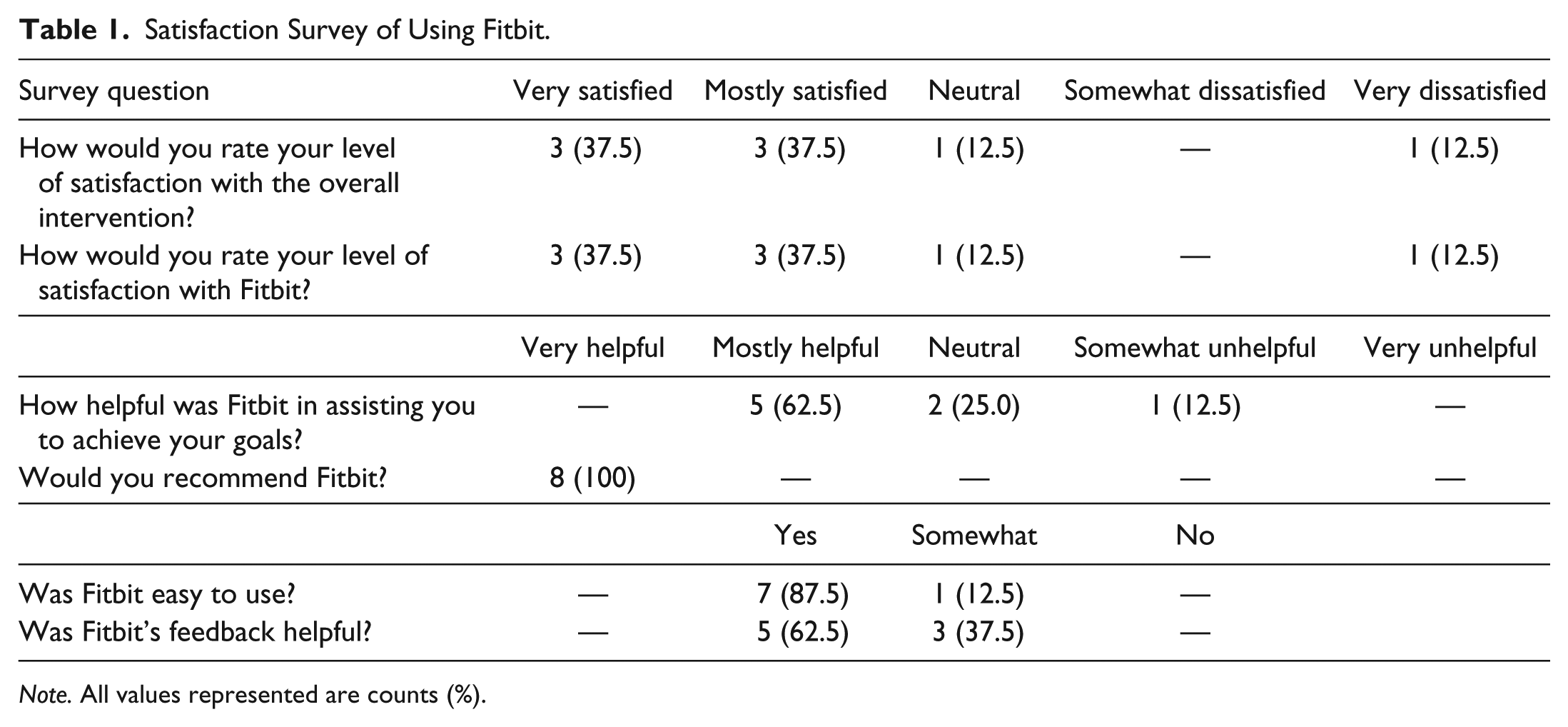
Note. All values represented are counts (%).
Participants’ Views on the Benefits of Using a Wearable Activity-Tracking Device to Support a Physical Activity Program.

Discussion
This is the first pilot study evaluating the impact of Fitbit in rural, older adults with obesity. We demonstrated feasibility, acceptability, and usability of Fitbit.
Prior studies focused on mHealth use in younger populations. Older adults’ are among the fastest growing users of smartphones, providing a significant opportunity to couple these devices with commercially available activity sensor technologies. Fitbit satisfaction was promising; signifying it could be helpful in achieving individual goals. The interviews demonstrated enthusiasm in the use of activity feedback, self-monitoring, and motivation. Our results were consistent with others that used accelerometers in elders (Beevi, Miranda, Pedersen, & Wagner, 2016; Tiedemann, Hassett, & Sherrington, 2015).
Although we observed usability and satisfaction with Fitbit, contrary to our hypothesis, exercise confidence decreased and patient activation was no different pre/post in our pilot sample. These results suggest that the device and its information may not lead to activation on its own. Our qualitative data emphasized the importance of social interaction and group engagement in older adults to motivate and inspire behavioral change. Providing individuals with a standalone device with or without mobile connectivity may be sufficient in younger populations, yet without a human component (peers, coaches, clinicians) to allow the interpretation of the information, Fitbit should be considered an adjunct in older adults. Although Fitbit does have a Web-based and application-based platform, we deliberately did not obtain quantitative data because in this population, its inter-person validity requires further examination (Gomersall et al., 2016). Without guidance, one’s confidence in engaging in a physical activity regimen is reduced. Studies should focus on person-level interactions that integrate individual feedback.
Participants were sedentary at baseline and had no experience using activity-tracking devices. Our results suggested challenges with regard to finding time that may also influence confidence. Although speculative, participants may have started becoming more engaged only to realize the difficulty of pursuing a regular program. A lack of patient activation provides some useful information in that the impact of a commercial health device may be more limited than what is qualitatively described by participants.
For rural older adults with obesity, our results provided information that can be helpful in designing future studies. First, there was interest in both individual face-to-face meetings and group meetings. Group appointments can increase patient satisfaction (Heyworth et al., 2014) and provide peer support in obesity care, with potential to alter behavior. Second, rural barriers were clearly evident due to participants’ mobility limitations and geographic and home restrictions. Third, frequent sessions and longer duration of contact were advocated. Fourth, we ascertained challenges in conducting this study in the wintertime. Future studies should be designed with this in mind and/or collaborate with community-based settings that will allow this population to become more active during this timeframe. Last, peer mentoring was suggested as an important phenomenon that was lacking considering the distance participants lived apart from their neighbors.
Several strengths should be noted. Few studies have evaluated this subgroup, and even fewer have used Fitbit or other devices in older adults (Takacs et al., 2014; Tiedemann et al., 2015; Wang et al., 2015). Our study demonstrated the feasibility and value of providing an intervention in a rural, academic setting, whose demographic composition differs from urban, research settings. Our use of mixed methods provides valuable insight into the behavioral change challenges facing rural, older adults. Qualitative research is a powerful tool that allows investigators to gain insight into a phenomenon such as older adult obesity. Future work should expand on the use of such devices in rural, older populations with obesity.
Our findings should be considered exploratory and interpreted with caution. The intent of pilot studies with small sample sizes is to evaluate feasibility rather than to formally test hypotheses or provide generalizable knowledge as in fully powered randomized, controlled trials. They are also important in conducting reliability testing of survey instruments and generating information from semi-structured interviews. A convenience sample has a number of advantages (fast, easy to conduct) and allows extrapolations of theories from data. This type of recruitment allows the execution of pilot studies to enhance an understanding of the research question. However, we recognize that very limited generalizations can be made and that the sample is not chosen at random. Our data imply that larger studies could be conducted. The short 1-month time frame prevents us from noting any effect on sustainability. We gained knowledge and expertise to allow intervention and measure refinement. We also were unable to adjust for confounding variables in our analyses. Whether standalone fitness devices with or without additional applications/Web connectivity are needed requires further exploration.
Conclusion
Although Fitbit is acceptable to older adults with obesity, a short-term drop in exercise confidence occurs suggesting a need to include a human component in behavioral change in this high-risk population.
Footnotes
Authors’ Note
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health (NIH). The findings and conclusions in this journal article are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Batsis’ research reported in this publication was supported in part by the National Institute on Aging of the National Institutes of Health (NIH) under Award Number K23AG051681. He also received funding from the Department of Medicine, Dartmouth–Hitchcock Medical Center, and the Dartmouth Centers for Health and Aging. Research reported in this publication was supported by the Dartmouth Clinical and Translational Science Institute, under Award Number UL1TR001086 from the National Center for Advancing Translational Sciences (NCATS) of the NIH. This work was also supported by the Dartmouth Health Promotion and Disease Prevention Research Center (Cooperative Agreement Number U48DP005018) from the Centers for Disease Control and Prevention.
