Abstract
Background
The bacterium Streptococcus pneumoniae commonly causes severe bacterial illnesses in both children and adults. S. pneumoniae is the most fatal bacterial pathogen, causing 50% of the deaths from lower respiratory infections in people of all ages.
Objectives
This study was aimed to determine a nasopharyngeal carriage rate of S. pneumoniae, related risk factors, and antibiotic susceptibility among prisoners in Hawassa Central Prison Institute Hawassa, Ethiopia
Methods
From July to August 2021, a cross-sectional investigation was carried out in Hawassa prison. All randomly chosen adult volunteers were a part of the study. In this study, about 330 different people were included. Within 3 h of collection, the swab specimen was inoculated onto 5% sheep blood agar; the inoculated medium was then incubated for 18-24 h at 35-37 °C in a CO2-enriched environment. Following incubation, the S. pneumoniae colonies were suspected, but the usual colony shape, alpha-hemolysis, Gram staining, optochin susceptibility, and bile solubility test allowed for confirmation. Antimicrobial susceptibility testing was carried out in accordance with the Clinical and Laboratory Standards Institute's guidelines.
Results
Overall carriage rate of S. pneumoniae among adult individuals were 41 (12.7%). The carriage rate of S. pneumoniae was affected by age classes that 39-48 years (OR = 4.82, 95%CI = 1.49-15.56, p = .009), 49-58 years (OR = 5.27, 95%CI = 1.27-21.89, p = .022), and greater than 58 years (OR = 4.36, 95%CI = 1.13-16.90, p = .033); cigarette smoking (OR = 3.41, 95%CI = 1.16-10.01, p = .026); and sharing beds (OR = 3.91, 95%CI = 1.27-12.07, p = .018). The majority of the isolates are susceptible for clindamycin (87.8%) and resistant for chloramphenicol (56%). Multidrug resistance was observed in 36.7%.
Conclusions
The overall carriage rate of S. pneumoniae was 12.7%. Age, cigarette smoking, and sharing beds in the same prison room had a substantial impact on this nasopharyngeal carriage rate. Chloramphenicol (56%), erythromycin (41.5%), tetracycline (39%), and co-trimoxazole (34.1%) resistance were seen in the majority of S. pneumoniae isolates. Early detection, hygiene maintenance, and appropriate treatment are necessary.
Introduction
Streptococcus pneumoniae was originally described in 1881 in France by Louis Pasteur; and, it is a Gram-positive, elongated (Lanceolate) diplococcus, nonmotile, capsulated, and forming short chain following culture. 1 S. pneumoniae (also called pneumococcus) is not only an opportunistic commensal that frequently colonizes the human upper respiratory tract but also a pathogen that causes invasive diseases such as pneumonia, sepsis, and meningitis. 2
S. pneumoniae is the most fatal bacterial pathogen, causing 50% of the deaths from lower respiratory infections in people of all ages. 3 Of the 2,377,697 lower respiratory infection-related deaths in individuals of all ages reported in 2016 worldwide, 1,189,937 (or 50%) were caused by S. pneumoniae. This is more than all other bacterial pathogens combined. 4 Furthermore, according to the study done by Wahl B and his collogue, showed that S. pneumoniae was the cause of about 50% of all pneumococcal deaths that occurred in Africa in 2015. 5
The prevalence of S. pneumoniae colonization among old-aged patients in Sheik Hassan Yebere Referral and Karamara General Hospitals in Ethiopia was 13.8%, 6 Felege Hiwot was 35.8%, 7 and Gondar Ethiopia 39.4%. 8 Also the prevalence of S. pneumoniae in presumptive tuberculosis adult cases was 21.4%. 9 Also the colonization rate of S. pneumoniae among children was 21.5% 10 and 20.68%. 11
The upper respiratory tract is the ecological niche for the pneumococcus and colonization is established during the first months of life. 12 Although colonization is typically asymptomatic, in some people it may progress into noninvasive or invasive pneumococcal disease (IPD). 13 The development of illness from carriage is influenced by a variety of risk variables, including age, inflammatory diseases, geography, socioeconomic status, genetics, tissue damage to the airways, and immune system. Infants, the elderly, and immunocompromised individuals14,15 are more at risk for IPDs than the general population. 16 Additionally, as carriage and bacterial spread are associated, S. pneumoniae bacteria are acquired widely in the population. 17
S. pneumoniae is most commonly carried in the nasopharynx in children under 2 years old, and this rate decreases as children get older and their immune systems develop. 18 In adults of all ages, alcoholism, chronic heart, lung, kidney, or liver disease, cochlear implant, cerebrospinal fluid leak, diabetes, HIV infection, cancer, solid organ transplant, or immunosuppressive conditions, Nephrotic syndrome, Sickle cell disease, a damaged or no spleen are all significant risk factors for the development of pneumococcal disease. 19 Attendance at daycare center, having siblings in the family, immunization status are risk factor for S. pneumoniae infection among children. 20 The prevalence of S. pneumoniae nasopharyngeal (SPNP) carriage is likewise impacted by these risk factors. 21
Furthermore, age, smoking, environmental exposures, malnutrition, previous community-acquired pneumonia, chronic bronchitis/chronic obstructive pulmonary disease, asthma, functional impairment, poor dental health, immunosuppressive therapy, oral steroids, and treatment with gastric acid-suppressive drugs were risk factors for CAP. 22 In the adults, the S. pneumoniae carrying rate was between 0% and 38.8% using classical microbiological analysis and 2.7% and 22.7% using molecular methods. However, the pooled prevalence using microbiological and molecular methods was 9%. 23
Pneumococcal pneumonia infection caused by S. pneumoniae is a global health concern and vastly infects both children and adults with preexisting health conditions. 24 In enclosed environments, it can also lead to epidemics of community-acquired pneumonia. 25 Worldwide, particularly in developing nations, infections caused by S. pneumoniae most frequently manifest as community-acquired pneumonia, meningitis, otitis media, endocarditis, osteomyelitis, pericarditis, pyogenic arthritis, soft tissue infection, neonatal septicemia, lower respiratory infection, and bacteremia in humans. 26
Different sorts of laboratory procedures can be used to identify S. pneumoniae, ranging from the quick and easy Gram stain method to the complex method of identifying the organism at the gene level. The culture approach, which isolates living organisms and does further identification for an antibiotic susceptibility test, is the gold standard.27,28 However, all patients with pneumococcal illness were empirically treated with antibiotics like penicillin, 29 which may result in antibiotic resistance.
Diseases caused by pneumococci can be prevented by vaccination. To stop pneumococcal illnesses, many polysaccharide pneumococcal conjugate vaccines (PCV) have been created. The S. pneumoniae bacterium's sugars from its capsule are coupled to a carrier protein to make up the polysaccharide pneumococcal conjugate vaccine. The vaccines in question include the 10-valent pneumococcal conjugated vaccine (PCV-10), the 13-valent pneumococcal conjugated vaccine (PCV-13), and the 23-valent pneumococcal conjugated vaccine (PCV-23). For vulnerable populations, such as the elderly and those with impaired immune systems, PCV-23 is used. Because they are specific to that serotype, antibodies made against it are unable to create immunity to other serotypes. 28 Therefore, the aim of this study was to determine a nasopharyngeal carriage rate of S. pneumoniae, related risk factors, and antibiotic susceptibility among prisoners in Hawassa Central Prison Institute Hawassa, Ethiopia
Methods
Study Design
The study was carried out in Hawassa, Ethiopia, which serves as both the Sidama National Region's and the South Nations, Nationalities, and People Region's seat. 275 kilometers separate the city from Addis Ababa, the Ethiopian capital city, which is situated on the shores of Lake Hawassa in the Great Rift Valley. In the Dalle woreda of Yirgalem town and Hawassa city, respectively, there are two jails in the Sidama region. At the Hawassa Central Prison Institute, there were roughly 1,835 inmates.
Study Setting
From July 1 to August 30, 2021, a cross-sectional study was carried out at the Hawassa Central Prison Institute. All inmates who were arrested at Hawassa Central Prison Institute and were older than 18 years old made up the research population. A rigorous random sample procedure was used to choose the voluntary convicts for the study's subject matter. Dependent variables include antibiotic susceptibility and the prevalence of S. pneumoniae in the nasopharynx. Age, sex, residences, vaccination history, drunkenness, smoking history, previous history of chronic diseases (asthma, heart disease, kidney disease, diabetes mellitus), housing conditions like crowded/ventilated rooms, and length of time in prison are independent variables.
Sample Size
The predicted prevalence (p) of nasopharyngeal carriage of S. pneumoniae among inmates who have previously lived in the research area is not known. Using the method for a cross-sectional study and a 50% estimate of predicted prevalence (p), a 5% margin of error, and a 95% confidence interval (CI) (z = 1.96), we can get the sample size using: n = Z2 p (1 − p)/d2; where np = sample size for prevalence; Z = 95% CI = 1.96 which is a constant, P = estimated prevalence for unknown population = 50%, d = margin of error = 5%; and, the sample size for associated risk factors was also calculated using the prevalence among elders was 26%, 30 among immunocompromised individuals such as sickle cell disease patients was 10% 31 and among HIV infected adults was 17%. 32
The study's sample size with a 5% nonrespondent rate was determined as ni = np + (5%np) = 384 + 19,403, with 384 being the biggest sample size. Using the formula nf = [ni/(1 + ni/N)] and taking into account the finite population correction, where total population (N) = 1,835, which is less than 10,000, the final sample size was 330 inmates chosen by recruiting all voluntarily submitted convicts until the necessary sample size was reached. Except for those who are seriously ill patients and have been receiving antibiotic medication for the past 2 weeks in prison, all voluntary prisoners chosen through a systematic random sample approach were included in this study.
The developed questionnaire was pretested using people who were Adare General Hospital outpatients, who made up 5% of the final sample size estimate. The information gathered was examined, and the questionnaire was modified to capture the information wanted from convicts. Using these pretested questionnaires, trained data collectors gathered the sociodemographic information of the study participants over the study period.
Inclusion Criteria
All randomly selected voluntary adult individuals in Hawassa prison were included.
Exclusion Criteria
All adult individuals who are severely ill patients and on antibiotic therapy for last 2 weeks in prison were excluded.
Sample Collection and Laboratory Diagnosis
Each of the adult volunteers who had been chosen at random to participate in the study had their nasopharyngeal swabs taken using flexible, sterile swabs in accordance with the sample collection procedures. A sterile swab was softly pushed through the nostril along the nasal cavity's floor until it touched the nasopharynx's back wall, rotated at 180-3,600, or was left inside the nasal cavity for 5 s before being carefully removed. A qualified laboratory technologist took one nasopharyngeal swab each person. Thermo scientific™ Oxoid™ product was used to transmit the swab specimen to the South Nations, Nationalities and People Region Public Health Institute as soon as possible (SNNPR-PHIL)
Within 3 hours of collection, a swab specimen was streaked onto 5% sheep blood agar (a Thermo Fisher™ Oxoid™ product) by rolling the swab over a small section of the plate. The inoculated media was incubated for 18-24 h at 35-37 °C in a CO2-enriched environment. Gram staining was done on all suspected alpha-hemolytic colonies, which had a greenish color. All Gram-positive diplococci were then subcultured on 5% sheep blood agar with 5 g of Optochin discs and incubated for 24 h at 35-37 °C in a CO2-enriched atmosphere. After overnight incubation, the sensitivity of Optochin was checked by measuring the diameter of the inhibition zone. If the diameter was ≥14, it was assumed to be S. pneumoniae; if the diameter was <14, bile solubility tests (tube method) were performed by using 2% sodium deoxycholate.
All S. pneumoniae isolates were subcultured in Mueller-Hinton Agar supplemented with 5% defibrinated sheep blood and incubated for 18-20 h at 35-37 °C in a CO2-enriched atmosphere. The modified Kirby-Bauer disc diffusion method was used to determine the antibiotic susceptibility pattern. Tetracycline (30 μg), cotrimoxazole (23.75 μg), oxacillin (1 μg), chloramphenicol (30 μg), erythromycin (15 μg), and clindamycin (2 μg) made up the majority of the antibiotics used for AST, according to the national standard treatment guideline (STG) of Ethiopia, the clinical and laboratory standard institute (CLSI) guideline, and the availability of all required.
Pre-structured questionnaires were used to gather the study's sociodemographic data. Before beginning data collecting and microbiological analysis, a pretest was conducted early in the study to determine the validity of the questionnaire. All swab specimen transportation and microbiological culture media made for microbiological analysis were autoclaved. Prior to use, the microbiological culture media were additionally tested for sterility by incubation at 35-37 °C for 24 h in a 5-10% CO2-enriched environment. As a positive control for microbiological testing, S. pneumoniae ATCC 49619 quality control strain was employed.
Data Management and Analysis
SPSS version 20 was used to examine the data that was gathered. To choose potential variables for a multivariate logistic regression analysis, descriptive statistics, and bivariate analysis were used. Explanatory variables and outcome variables were associated. Odds ratio (OR) with 95% CIs was generated using the multivariate logistic regression controlling for potential confounding and was used to establish the existence and strength of the link between dependent and independent variables using p-values <.05 which statistically significant.
Ethical Approval and Consent Participate
The institutional review board of the Hawassa University College of Medicine and Health Science received the proposal, assessed it, and gave its approval on June 8, 2021 (Ref No. IRB/207/13). The Sidama National Region, Hawassa Central Prison Institute was then contacted for authorization. After receiving the go-ahead from those relevant bodies, the project was then carried out. By gaining the consent of the study participants, the data and sample were gathered. Using codes, all participant data and results were kept confidential. Testing is free. Test results were sent to healthcare providers working in the prison for further diagnosis and treatment.
Results
A total of 323 selected prisoners were enrolled in this examination, which ran from July through August of 2021. The remaining 7 (2.1%) were nonrespondents. Two hundred seventy-seven (85.8%) of the participants in the study were men and 46 (14.2%) were women. The participants in the research ranged in age from 18 to 90 years, with an average age of 30.4 years. Approximately 178 of the participants were under the age of 29, making up 55.1% of the total, while 19 were above the age of 58, making up 4.9%.
Of the study's participants, about 211 (65.3%) and 112 (34.7%) were from urban and rural homes, respectively. There were around 31 (9.6%) individuals who had no formal education, followed by 203 (62.8%) who had gone to elementary school, 82 (25.4%) who had gone to high school, and just 7 (2.2%) who had enrolled in and successfully completed tertiary education (college/university). According to the research subjects, 99 (30.7%) were chosen to be farmers, 32 (9.9%) to be government employees, 62 (19.2%) to be trade or commercial workers, 90 (27.1%) to be students, 12 (3.7% to be drivers), 18 (5.6% to be daily laborers, and 10 (3.0% to be electricians and woodworkers). About 46 (14.2%) of study participants had ever smoked cigarettes and 90 (27.9%) routinely drank alcohol (Table 1).
Sociodemographic Characteristics of the Inmates from Hawassa Central Prison Institute from July 2021 to August 2021 (N = 323).

The study's participants’ health and health-related information indicated that 323 (100%) had not had the pneumococcal disease immunization because it had only recently been introduced.
From the total number of participants in the study, 45 (13.9%) had a history of hospitalization and 19 (4.9%) had chronic illnesses, including 10 (3.1%) renal diseases, 4 (1.2%) cardiac diseases, and 5 (1.5%) cases of diabetes mellitus. Additionally, 60 (18.6%) of the study participants admitted that they coughed or sneezed at least once throughout the period of study (Table 2).
Frequency Distribution of Health and Health-Related Variables of the Inmates from Hawassa Central Prison Institute from July 2021 to August 2021 (N = 323).

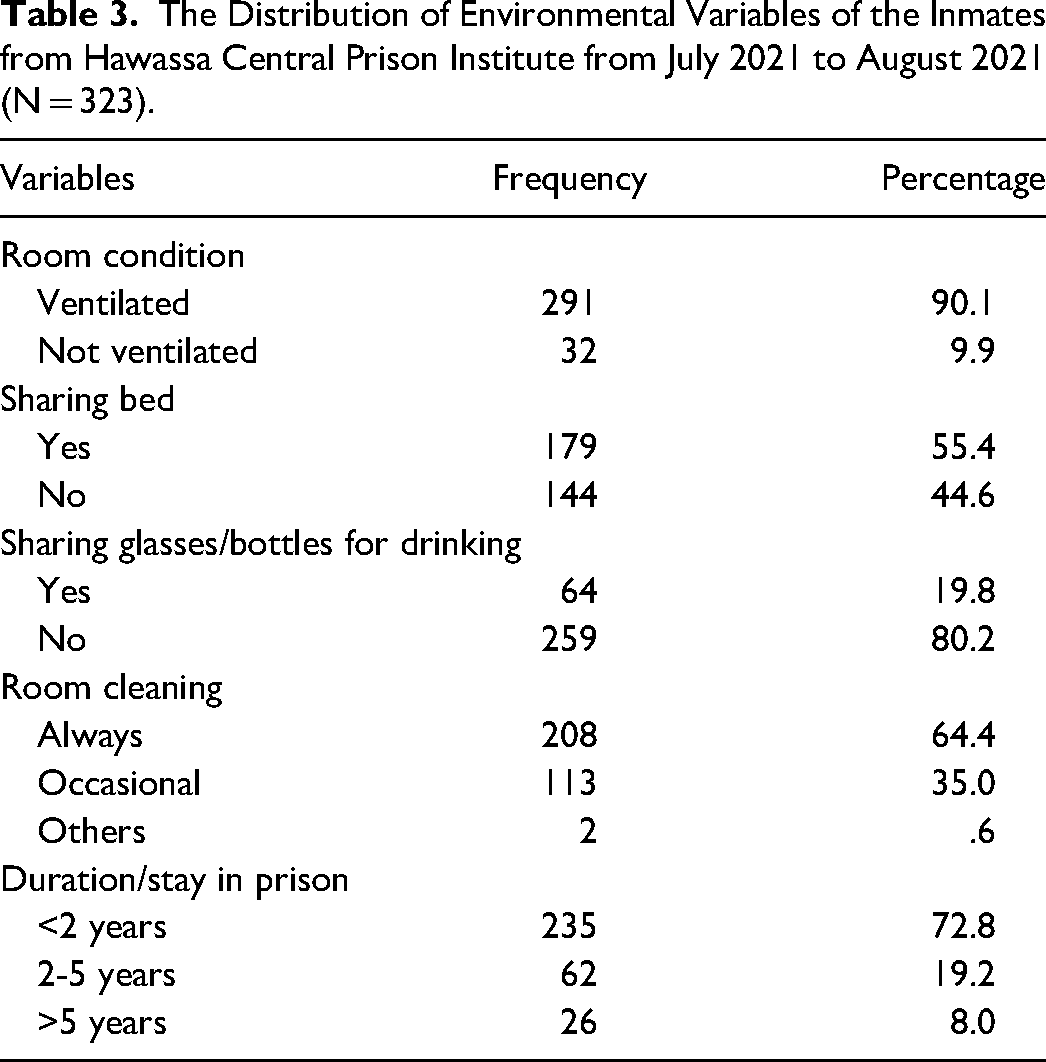
A total of 291 individuals (or 90.1% of the research participants) were living in ventilated housing while imprisoned and 208 (64.4%) of them maintained their rooms clean at all times. But exclusively 113 of them, or 35%, did occasionally. About 179 (55.4%) of the study participants shared a bed in the same room, and 64 (19.8%) of them shared drinking glasses and plastic bottles. The majority of research participants—235 or 72.8%—stayed in jail for less than 2 years. Only 26 (8%) stayed more than 5 years, while the remaining 62 (19.2%) stayed for between 2 and 5 years (Table 3).
The Distribution of Environmental Variables of the Inmates from Hawassa Central Prison Institute from July 2021 to August 2021 (N = 323).
Nasopharyngeal Carriage Rate
S. pneumoniae was detected in 41 (or 12.7%) of the 323 nasopharyngeal swab samples collected and assessed from prisoners.
Risk Factors
The bivariate model was used to examine the data in order to identify the variables that were related to the prevalence of S. pneumoniae in the nasopharynx. At the same time, variables with scores below 0.25 were chosen as potential multivariate model candidates. A p-value <.05 was considered statistically significant.
The results of this investigation demonstrated that three distinct risk variables had a substantial impact on the prevalence of S. pneumoniae in the nasopharynx. Age of the participants was one of these, and it was associated with the prevalence of S. pneumoniae in the nasopharynx in the age groups of 39-48 years (OR = 4.82, 95% CI = 1.49-15.56, p = .009), 49-58 years (OR = 5.27, 95% CI = 1.27-21.89, p = .022), and over 58 years (OR = 4.36, 95% CI = 1.13-16.90. The second factor, smoking cigarettes, had a significant impact on the incidence of S. pneumoniae carriage in the nasopharynx (OR = 3.41, 95% CI = 1.16-10.01, p = .026). Last but not least, sleeping in the same bed had a significant impact on the incidence of S. pneumoniae carriage in the nasopharynx (OR = 3.91, 95% CI = 1.27-12.07, p = .018) (Table 4).
Risk Factors That Affect the Nasopharyngeal Carriage Rate of S. pneumoniae among Inmates in Hawassa Central Prison Institute from July 2021 to August 2021 (N = 323).
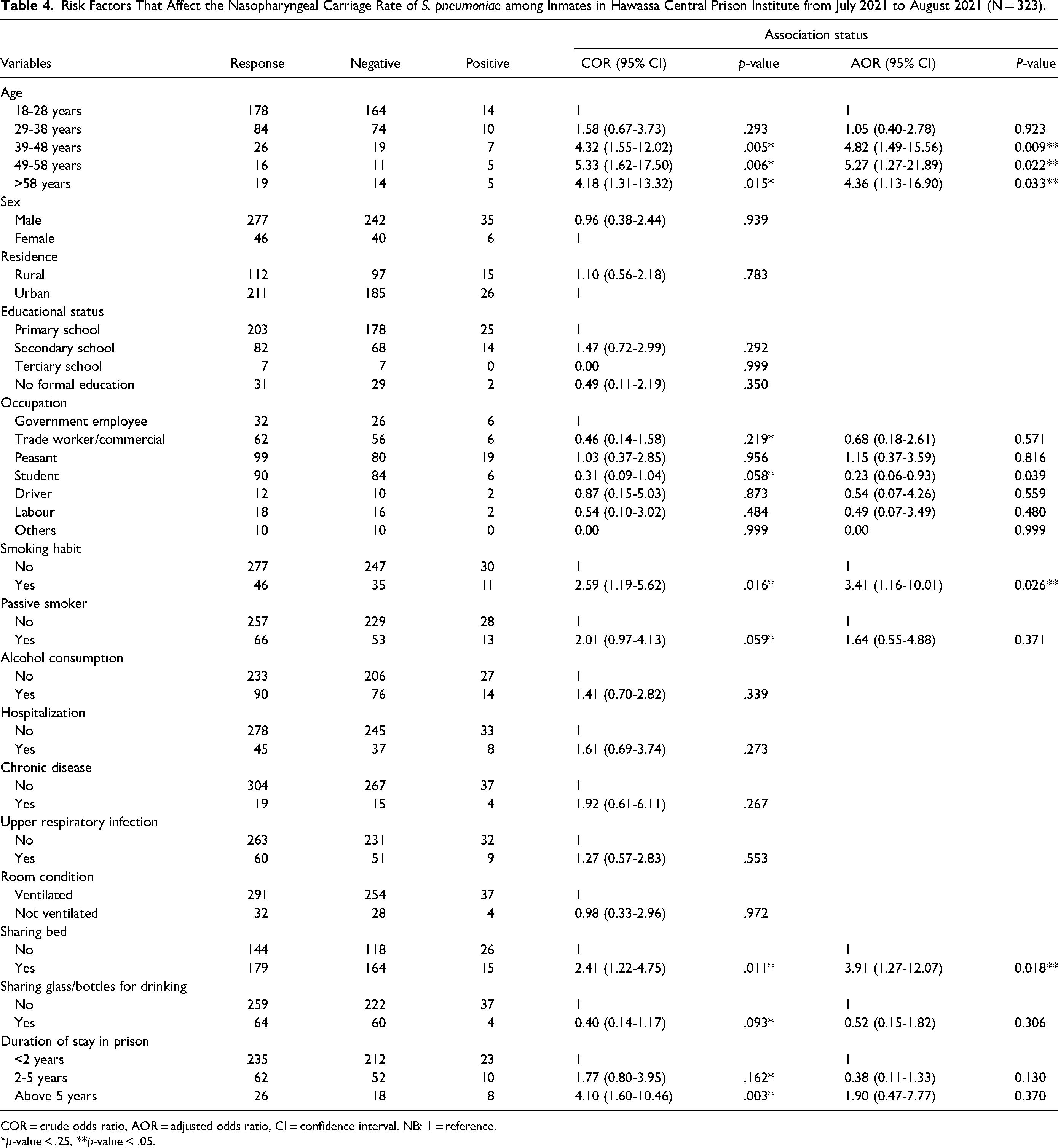
COR = crude odds ratio, AOR = adjusted odds ratio, CI = confidence interval. NB: 1 = reference.
*p-value ≤ .25, **p-value ≤ .05.
Antimicrobial Susceptibility Pattern
Using the disc diffusion method and seven different antibiotic discs, including oxacillin (1 µg), clindamycin(2 µg), co-trimoxazole(23.75 µg), rifampin(5 µg), chloramphenicol(30 µg), erythromycin (15 µg), and tetracycline (30 µg), the antimicrobial susceptibility profiles of all S. pneumoniae culture isolates were determined. Only 5 (12.2%) of the total number of S. pneumoniae isolates were intermediately resistant to clindamycin, leaving 36 (87.8%) isolates susceptible to the antibiotic. About 16 (39%) of the isolates of S. pneumoniae were resistant and 12 (29.3%) were intermediate resistant to tetracycline, 14 (34.1%) of the isolates of S. pneumoniae were resistant, and 6 (14.6%) of the isolates were intermediate resistant to co-trimoxazole. About 23 (56%) of the isolates of S. pneumoniae were resistant to chloramphenicol (Table 5).
Antimicrobial Susceptibility Pattern of S. pneumoniae Isolates from Inmates in Hawassa Central Prison Institute from July 2021 to August 20021 (n = 41).

Multiple Antibiotic Resistance Phenotypes of S. pneumoniae
Antimicrobial resistance demonstrated by S. pneumoniae to at least one medication in three or more categories of antibiotics is known as multiple antibiotic resistance (MAR). In this work, MAR phenotypes of S. pneumoniae isolates from nasopharyngeal swabs of inmates at Hawassa Central Prison Institute were determined using four categories of antibiotics. For S. pneumoniae isolates from this research location, Cot-CAF-Clin 3 (7.3%) and Tet-Cot-CAF-Rif 2 (4.9%) were the two most common MAR phenotypes (Table 6).
The Predominant Multiple Antibiotic Resistant Phenotypes for S. pneumoniae Isolates from Nasopharyngeal Swabs of Prisoners in Hawassa Central Prison Institute from July 2021 to August 2021 (n = 41).

Ox: oxacillin; Ery: erythromycin; Tet: tetracycline; Cot: co-trimoxazole; CAF: chloramphenicol; Rif: rifampin; Clin: clindamycin.
Antibiotic Resistance Profiles of S. pneumoniae Isolates
S. pneumoniae isolates from nasopharyngeal swabs of study participants were resistant to chloramphenicol, erythromycin, tetracycline, and co-trimoxazole in around 56.5%, 41.5%, 39%, and 34.1% of the cases, respectively. Clindamycin (12.2%) showed the least amount of resistance (Table 7).
Antibiotic Resistance Profiles of S. pneumoniae Isolated from Nasopharyngeal Swabs of Prisoners in Hawassa Central Animates Institute from July 2021 to August 2021 (n = 41).

Ox: oxacillin; Ery: erythromycin; Tet: tetracycline; Cot: co-trimoxazole; CAF: chloramphenicol; Rif: rifampin; Clin: clindamycin.
Discussion
S. pneumoniae nasopharyngeal carriage rates in adult populations have been estimated by a variety of research in a variety of study regions; nevertheless, adult populations have received less attention than children in terms of nasopharyngeal carriage rates. In this investigation, the nasopharyngeal carriage rate of S. pneumoniae among adult inmates in the confined or access-restricted environment of Hawassa Central Prison was 41 (12.7%). The results of the investigation are considerably higher than those of a 2018 study done in Alabama, USA, which were 3.1%, 33 North Carolina, USA among retired community 1.9%, 34 noninstitutionalized USA adults 1.8%, 35 and UK healthy adults 6.5%. 36 However, the study done in a military setting is also lower than our finding which is 1.6% in Gambia, 37 1.5% in Michigan, USA, 38 1.2% in US Marine Corps trainees, 39 and 10.8% in Israel. 40 Even though the repeated contact with several people, particularly as a result of poor hygiene, overcrowding, sharing saliva through drinking cups or glasses, and crowded environments is a related factor, the discrepancy of this study is might be due to the study setting, study method, and investigation technique.
This finding is lower than the studies conducted in Marine Corps cadets in the US Army 41 15.7%, in the Aboriginal population 30 26%, in the Liguria administrative region 42 18.7%, in Sheik Hassan Yebere Referral and Karamara General Hospitals Ethiopia 6 13.8%, in Felege Hiwot, Ethiopia 7 35.8%, and in Gondar Ethiopia 8 39.4%. This may be due to sample size, seasonal variation, and methods that we used. However, the close proximity in settings with restricted access, socioeconomic factors, environmental, and personal hygiene, may contribute occurrence of S. pneumoniae.
The findings of this study are comparable to those of a study conducted in Semarang, Java Island, Indonesia, among children and adults aged 45-75 years that were pneumonia prone (11%), 43 and to those of a study conducted in Milan, Italy, among older adults aged 65 and older (9.8%). 44 The study's findings might be the result of the subjects’ older age.
The results of this study are more significant than the findings from Israel to compare the S. pneumoniae carriage rate among adults and children less than 6 years old in the same community (4%) 45 and the results of pneumococcal carriage rate among elderly adults in Portugal (2.3%). 46
The study's findings are much lower than the S. pneumoniae carriage rate among Gambian villagers older than 40 (51%) years. 47 However, the results of this study are comparable to those of a study that found that S. pneumoniae was carried by 10% of patients with sickle cell disease in Accra, Ghana. 31 This is because the study participants are getting older and have more illness.
In this study, the nasopharyngeal carriage rate of S. pneumoniae was significantly associated with age classes ([OR = 4.82, 95% CI = 1.49-15.56, P = .009], [OR = 5.27, 95% CI = 1.27-21.89, p = .022], and [OR = 4.36, 95% CI = 1.13-16.90, p = .033]), smoking cigarette (OR = 3.41, 95% CI = 1.16-10.01, p = .026), and sharing bed in the same room (OR = 3.91, 95% CI = 1.27-12.07, p = .018). Similar studies on the prevalence of pneumococcal carriage in adults in an Australian Aboriginal community, 30 in persons 60 and older in the Liguria administrative Region, 42 and in sickle cell patients in Accra, Ghana 31 have found a substantial correlation between age and these results. Similar to Portuguese research that evaluated the incidence of pneumococcal illness in elderly people, smoking cigarettes had an impact on the prevalence of S. pneumoniae in the nasopharynx. 46 A substantial correlation between the rate of S. pneumoniae carriage in the nasopharynx and participants’ use of beds in the same room was also found in this investigation. This may be due to the participants’ close proximity for a prolonged period of time in the same environment.
With regards to the antimicrobial susceptibility pattern of S. pneumoniae, the resistance of oxacillin among isolates in this study is lower than the findings of studies conducted among sickle cell disease patients (the penicillin resistance 37.4%), 31 among patients suspected of having pneumococcal infections (38.6%), 48 and Felege Hiwot. 7 This study's oxacillin resistance finding is higher than the study's findings from Gambian villagers, which showed that 14.3% of isolates there initially exhibited penicillin resistance. 47
The prevalence of erythromycin resistance in this study is higher than that observed in a study of patients with pneumococcal infections at Felege Hiwot Referal Hospital in Northwest Ethiopia (96.7% sensitive), 7 but it is lower than that observed in another Ethiopian study (59.6%). 48
The tetracycline resistance found in this study was higher than that found in a survey of Gambian villagers (32.3%), 47 but less prevalent than that found in a study of patients with pneumococcal infections at Felege Hiwot Referal Hospital in Northwest Ethiopia (45%). 7
This study's co-trimoxazole resistance profile is lower than the studies’ findings of co-trimoxazole resistance profiles (85%) among sickle cell disease patients in Accra, Ghana, 31 and (39%) in Gambian communities. 47 Additionally, patients at Felege Hiwot Referal Hospital in Northwest Ethiopia who had pneumococcal infections showed higher rates of chloramphenicol resistance than those patients did in the research (56%). 7
The majority of S. pneumoniae isolates in this investigation were clindamycin-susceptible (87.8%), which is significantly higher than the result of a previous study among patients with pneumococcal infections in Ethiopia (80.7%). The findings of this study are also relatively bigger than the study conducted in Ethiopia among individuals suspected of having pneumococcal infections, with total MAR being 36.7% and 33.3%, respectively. 48
Limitation
This finding is limited to discussion because adult-related study among prisons and community is very old. Regarding this study, it was done in a single prison.
Conclusion
For families and the entire community, S. pneumoniae colonization is a prelude to pneumococcal disease transmission. Age, cigarette smoking, and sharing beds in the same prison room had a substantial impact on this nasopharyngeal carriage rate. Chloramphenicol (56%), erythromycin (41.5%), tetracycline (39%), and co-trimoxazole (34.1%) resistance was seen in the majority of S. pneumoniae isolates. These studies generally serve as the starting point for other interested researchers to evaluate the prevalence of S. pneumoniae its risk factors, and the antibiotic susceptibility pattern among convicts in access-restricted settings. The spread of pneumococcal illness must also be planned for and managed as much as feasible by public health specialists.
Footnotes
Acknowledgements
We would like to thank the data collector, the prison personnel who made it possible to collect the data, and Hawassa University College of Medicine and Health Sciences. Additionally, we would like to thank everyone who volunteered to take part in this research.
Author Details
School of Medical Laboratory Science, College of Health Science and Medicine, Jimma University, Ethiopia. School of Medical Laboratory Science, College of Health Science and Medicine, Hawassa University, Ethiopia.
Authors’ Contribution
OR conceived the research idea, compiled, analyzed, and interpreted the statistical data. DD participated in the design of the study and in the statistical analysis, and final manuscript preparation. All authors read and approved the final manuscript of this study.
Availability of Data and Materials
Data essential to the conclusion are included in this manuscript and additional data are available upon reasonable request from the first author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Hawassa University, College of Medicine and health Sciences. The assistance includes funding for the study's necessary supplies and paying data collectors. The support did not cover study design, data analysis and interpretation, or publication production.
