Abstract
Poor binaural hearing in children was hypothesized to contribute to related cognitive and academic deficits. Children with unilateral hearing have normal hearing in one ear but no access to binaural cues. Their cognitive and academic deficits could be unique from children receiving bilateral cochlear implants (CIs) at young ages who have poor access to spectral cues and impaired binaural sensitivity. Both groups are at risk for vestibular/balance deficits which could further contribute to memory and learning challenges. Eighty-eight children (43 male:45 female, aged 9.89 ± 3.40 years), grouped by unilateral hearing loss (n = 20), bilateral CI (n = 32), and typically developing (n = 36), completed a battery of sensory, cognitive, and academic tests. Analyses revealed that children in both hearing loss groups had significantly poorer skills (accounting for age) on most tests than their normal hearing peers. Children with unilateral hearing loss had more asymmetric speech perception than children with bilateral CIs (p < .0001) but balance and language deficits (p = .0004, p < .0001, respectively) were similar in the two hearing loss groups (p > .05). Visuospatial memory deficits occurred in both hearing loss groups (p = .02) but more consistently across tests in children with unilateral hearing loss. Verbal memory was not significantly different than normal (p > .05). Principal component analyses revealed deficits in a main cluster of visuospatial memory, oral language, mathematics, and reading measures (explaining 46.8% data variability). The remaining components revealed clusters of self-reported hearing, balance and vestibular function, and speech perception deficits. The findings indicate significant developmental impacts of poor binaural hearing in children.
Keywords
Introduction
Children with unilateral hearing loss and children who are deaf and use cochlear implants (CIs) experience altered auditory space due to impaired binaural hearing yet the relative outcomes in these two groups of children are not known. The current study aimed to examine the effects of impaired binaural hearing in early development due to unilateral hearing loss and bilateral CI use on visuospatial memory as well as language, cognition, and learning.
Children with hearing loss are at risk for poor perception of auditory space because they have difficulties hearing where sounds are coming from around them (Selz et al., 1996). Present standard of care for children with bilateral hearing loss includes early identification and rapid provision of hearing prostheses which aims to maintain and develop hearing while avoiding abnormal cross-modal take-over of the auditory system (Nicholas & Geers, 2007; Papsin & Gordon, 2008; Polonenko et al., 2018; Sarant et al., 2014). Cross-modal reorganization of the auditory cortices occurs with deafness in early life (Finney et al., 2001; Lomber et al., 2010), limiting auditory development and making it difficult for children to achieve spoken language (Bavelier & Neville, 2002; Lee et al., 2001). On the other hand, cross-modal changes with early onset auditory deprivation may support some aspects of spatial perception. Indeed, adults with early onset deafness who use manual forms of communication show enhanced visuospatial abilities (Bellugi et al., 1990) including, mental rotation (Emmorey et al., 1993; Talbot & Haude, 1993), face recognition (Emmorey et al., 1993), mental image generation (Emmorey & Kosslyn, 1996), and motion detection (Shiell et al., 2014), and peripheral vision is enhanced in deaf white cats (Lomber et al., 2010). Cross-modal plasticity could also facilitate a reallocation of attentional resources toward the remaining intact senses (Occelli et al., 2013). However, cross-modal changes may not completely compensate for abnormal spatial perception of sound in individuals with hearing loss (Gordon et al., 2014; Kan & Litovsky, 2015). It is also possible that the primary sensory deficits expand into other modalities as shown in adults with visual impairment who have deficits in sound localization despite normal hearing (Gori et al., 2014; Lessard et al., 1998; Richards, 2018). Spatial abnormalities in the intact sense might occur because of inconsistencies between visual and auditory spatial maps (Occelli et al., 2013). This means that hearing loss may affect children's visuospatial abilities in addition to contributing to their abnormal perception of sounds in space.
Efforts to give children access to spatial hearing begins with providing bilateral hearing (Avan et al., 2015; Litovsky & Gordon, 2016) because subtle differences in sound level and timing reaching the two ears (binaural hearing) code spatial locations of sound (Avan et al., 2015). The timing of bilateral hearing is key; children with asymmetric hearing in early life develop an aural preference for the better hearing ear which is difficult to change later (Gordon et al., 2015) and likely disrupts their potential to develop binaural/spatial hearing (Gordon et al., 2014; Kan & Litovsky, 2015; Litovsky & Gordon, 2016). Despite this evidence, children with unilateral hearing loss are often left without access to sound in the impaired ear (Fitzpatrick et al., 2017; Ontario Health (Quality), 2020; van Wieringen et al., 2019). This means that they have normal hearing sensitivity from their hearing ear but no access to binaural cues (Avan et al., 2015; Kumpik & King, 2019), impairing sound localization (Bess & Tharpe, 1984; Reeder et al., 2015) and speech understanding in noise (Lieu, 2004; Lieu et al., 2012; Noh & Park, 2012; Ruscetta et al., 2005). Early results of cochlear implantation in children with profound unilateral hearing loss of limited duration suggest potential benefits (Deep et al., 2021; Lee et al., 2020), but this treatment is not standard of care in all countries.
Provision of bilateral CIs simultaneously and with limited interimplant delay in children promotes development of pathways from both ears in tandem and symmetric bilateral hearing (Kan & Litovsky, 2015; Papsin & Gordon, 2008; Polonenko et al., 2018). Yet, even children receiving bilateral simultaneous CIs early do not gain normal binaural/spatial hearing (Gordon et al., 2014; Kan & Litovsky, 2015). Sound localization and detection of binaural cues remains impaired (Gordon et al., 2014; Steel et al., 2015), likely reflecting abnormalities in neural encoding of binaural input (Easwar et al., 2017; Smieja et al., 2020) and mismatched processing by bilateral devices (Goupell et al., 2013; Kan et al., 2013). Similar findings in children who use bilateral hearing aids (Gorodensky et al., 2019) suggest that present fitting methods for hearing devices are not sufficient for normal development of binaural function in children with hearing loss. On the other hand, providing bilateral devices in early life promotes development along the bilateral auditory pathways, and this will be required if novel bilateral fitting paradigms are able to better support acquisition of binaural hearing in children with hearing loss.
Spatial perception in children with hearing loss could be further affected by concurrent vestibular/balance impairments. A high incidence of vestibular dysfunction in children with hearing loss has been reported in children with both bilateral (Cushing et al., 2013) and unilateral (Sokolov et al., 2019) hearing loss, likely reflecting the shared embryological origin as well as anatomical proximity and similar physiology of these two inner ear components (Cushing et al., 2008; Selz et al., 1996). Vestibular input is important for spatial perception and self-motion perception (Cullen, 2012) as it controls gaze and balance and enables spatial representations to be encoded into memory (Dieterich & Brandt, 2015).
It is also important to consider the effects of increased listening effort in children with hearing loss which can tax available cognitive resources (Hornsby et al., 2016). Increased listening effort has been reported in children with unilateral hearing loss (Bess et al., 2020), children with mild to moderate hearing loss (Hornsby et al., 2017), and children using bilateral CIs (Steel et al., 2015). It is thought that these challenges underlie reduced verbal working memory capacity (AuBuchon et al., 2015; McCreery et al., 2019) and academic delays (Sarant et al., 2015). Of note, these concerns are not entirely mitigated by normal hearing in one ear as reports of grade failures and behavioral problems are also common in children with unilateral hearing loss (Lieu et al., 2012).
The developmental consequences of limited binaural hearing in children who received bilateral CIs with minimal delay and an absence of binaural hearing in children with unilateral hearing loss were examined in the present study by testing the hypothesis that children with limited to no binaural hearing have deficits in spatial perception, language, cognition, and academics.
Methods
Participants
Three groups of children were included in this study: (1) 32 children with bilateral deafness (mean(SD) age of 9.12(3.01), range = 4.10–16.30 years) who received bilateral CIs at young ages with minimum delay and who had minimum 2.68 years of bilateral implant experience (bilateral CI group); (2) 20 children (mean(SD) age of 10.83(4.08) years, range = 5.30–17.90 years) with prelingual onset of hearing loss in one ear (8 left and 12 right; 4 with conductive moderately-severe hearing loss, 16 with profound sensorineural hearing loss) and normal hearing in the other ear (averaged hearing thresholds at 500, 1000, and 2000 Hz ≤ 20 dB HL in 19 children; 1 child had fluctuating conductive hearing loss in the better ear) and little to no hearing aid use (unilateral HL group); and (3) 36 typically developing peers with normal hearing (mean(SD) age of 10.03(3.25) years, range = 5.10–15.20 years) (averaged hearing thresholds at 500, 1000, and 2000 Hz ≤ 20 dB bilaterally) (normal hearing group). Exclusion criteria included cognitive or motor delays that would impede understanding of test instructions and/or completion of the test battery. Demographic details for all three groups are provided in Table 1.
Participant Demographics.

There was no significant difference in age between the three groups (F(2,85) = 1.63, p = .20, η2 = 0.04). Obtaining consent, recruitment and all study procedures were conducted in adherence with the protocol approved by the Research Ethics Board at the Hospital for Sick Children (REB #100058925). Questions regarding gender and ethnicity were not part of the approved protocol. Data are available upon request.
Procedures
Each participant completed a battery of tests which included assessments of sensory ability, spatial ability, memory, and learning.
Hearing
Phonemically Balanced Kindergarten Word List (PBK)
Audio recordings of two PBK word lists (Haskins, 1949) (25 monosyllabic words each) were presented by insert earphones in two conditions (left and right ear). The line-in feature of the CochlearTM Wireless Mini Microphone 2 + was used to wirelessly transmit the audio output from a laptop computer at 65 dB SPL to each CI in bilateral CI users. Words were presented at 65 dB SPL through insert 3A earphones in the unilateral HL and normal hearing groups. Accuracy was recorded as the number of correctly repeated words per ear (maximum = 25). The best score and asymmetry in accuracy between ears in each child were used in the analyses.
Speech, Spatial, and Qualities (SSQ) of Hearing Scale
The SSQ is a questionnaire with three subsections that assess abilities in: (1) speech in quiet and noise, (2) spatial hearing, and (3) the quality of auditory information detected (Galvin & Noble, 2013). Each child rated their detection of auditory information on the three subsections of the SSQ questionnaire: (1) speech (maximum score = 10), (2) spatial hearing (maximum score = 13), and (3) quality of auditory cues (maximum score = 10). The questionnaire was completed by 72 of the 88 (81.81%) children. There were 16 children (six with normal hearing, six with bilateral CIs, and four with unilateral HL) who were unable to provide responses and thus had no available SSQ data for analyses. This was likely due to their young age (mean(SD), range) in each group (normal hearing: 5.57(0.86), 5.10–7.30 years; bilateral CI: 5.27(0.68), 4.10–5.90 years; unilateral HL: 6.45(1.10), 5.30–7.70 years).
Spoken Language
Clinical Evaluation of Language Fundamentals® (CELF)—Fifth Edition Screening Test
The CELF—Fifth Edition Screening Test (Wiig et al., 2013) was used to assess knowledge of oral language morphology, syntax, and semantics. The test is comprised of 45 items divided into eight sections which increase in difficulty. Each section includes language-based tasks. Children may be asked, for example, to complete a sentence or choose particular pictures. Accuracy on this standardized test was measured with maximum accuracy scores of 26 (ages ≤ 8 years) or 45 (ages 9–21 years).
Balance/Vestibular Function
Balance: Bruininks-Oseretsky test of Motor Proficiency 2nd Edition Balance Subtest (BOT-2)
The BOT-2 test (Bruininks & Bruininks, 2005) involves seven tasks that assess static and dynamic balance such as standing on one leg and walking forward heel-to-toe on a line. Tasks were completed both with eyes open and closed. The scores for each task reflected the duration of time the child could maintain the posture (static tasks) or complete dynamic tasks. Total score was the average of scores on each task. Scores were standardized based on age and gender using normative values provided with the test (maximum = 35).
Dynamic Visual Acuity (DVA)
Ability to maintain gaze during horizontal head oscillations was recorded as a measure of the vestibulo-ocular reflex (VOR) (Peters et al., 2012). The DVA score reflects the difference in the number of the line on the Snellen visual acuity chart the child was able to read between when their head was stable and during lateral oscillations. A difference in the line number >3 suggested a possible VOR deficit.
Subjective Visual Vertical (SVV) Test
The SVV test is a subjective assessment of perceived gravitational vertical. The SVV was measured using the Visual Vertical iOS app (Holmes, 2014) which has been shown to be as effective as the bedside bucket test in assessing SVV (Dai et al., 2020). Using the Visual Vertical iOS app, each child oriented a red line displayed on an iPhone to their perception of vertical and the angle (°) difference from true gravitational vertical was measured.
Visuospatial Memory
Dot Matrix Task and Corsi Block Tapping Tests
Memory for sequences of two-dimensional (2D) and three-dimensional (3D) visual objects was assessed using the Dot matrix (Alloway et al., 2004) and Corsi block tapping test (Berch et al., 1998), respectively. Each participant was asked to reproduce a sequence of 2D dot (Dot matrix) or 3D cube (Corsi block) positions. In the Dot matrix task, a red dot was presented for 2 s in one square of a 4 × 4 square matrix that was displayed on a computer screen. The position of the dot then changed to a different square. The child was instructed to indicate the sequence in the same order as the dot presentation (testing short-term memory) and in backward order (testing working memory). Sequences increased from 1 dot to a maximum of 9 (forward order/short-term memory) or maximum of 6 (backward order/working memory) in 1 dot step size if the child was able to accurately replicate the sequence in 4 of 6 possible trials at a given length. The Corsi block tapping test involved a fixed array of 3D blocks on a table which did not change in position. The examiner tapped a block with a pencil and the child had to tap the same block. The sequence of blocks tapped started with 1 block and progressed to a maximum of 9 blocks or 6 blocks in 1 block step size, depending on whether short-term or working memory was being assessed. Sequences increased from 1 block to a maximum of 9 (forward order/short-term memory) or maximum of 6 (backward order/working memory) if the child was able to accurately replicate the sequence in 4 of 6 possible trials at a given length. The number of total trials correct was recorded for the Dot matrix and Corsi block test in both the forward and backward order (forward maximum = 36 and backward maximum = 24).
Verbal Memory:
Digit Span Test
Verbal memory was tested by measuring the span of a random sequence of digits (1–9) that each child could repeat both in forward (testing short-term memory) and backward (testing working memory) order (Alloway et al., 2004). The digits were presented by live-voice by an examiner who sat facing the child. The digits were spoken at an approximate frequency of 1 digit per 1 s. Sequences increased from 1 digit to a maximum of 9 (forward order/short-term memory) or maximum of 6 (backward order/working memory) in 1 digit step size if the child was able to accurately replicate the sequence in 4 of 6 possible trials at a given length. The number of total trials correct was recorded (forward maximum = 36 and backward maximum = 24).
Academics
Wechsler Individual Achievement Test 3rd Edition (WIAT-III)
Academic peformance was assessed using subtests from the WIAT-III, a validated and standardized measure of cognitive ability (Breaux & Miller, 2010). Each child received a total score on the early reading (maximum = 34), word-reading (maximum = 75), pseudo-word decoding (maximum = 52), and mathematic problem-solving (maximum = 72) subtests of the WIAT-III. The mathematic problem-solving questions were word problems that assessed understanding of numbers, counting, addition and subtraction, fractions, proportions, measurement, and geometry.
Analyses
Analyses were completed in R (R-studio version 1.0.153, R version 3.5.2). Linear mixed effects models (Kuznetsova et al., 2017) were performed to assess the fixed effects of age, group, measure, and the interaction of group and measure with random intercepts by participant to account for repeated measures on: (1) working memory (# correct trials on backward span tests); (2) short-term memory (# correct trials on forward span tests); and (3) academics (standardized scores on WIAT-III subtests). Linear regression (nlme package version 3.1–137) (Pinheiro et al., 2021) was used to assess effects of age and group on language (CELF test), hearing (PBK test), and balance/vestibular function (BOT-2, SVV, DVA tests). Model effects were described by Type III Analysis of Variance Tables using Satterthwaite's method. Least-squares means were used for post-hoc comparisons of factors in the mixed models (Kenward-Roger method to correct degrees of freedom and Tukey method for comparing a family of three estimates to adjust p-values). Model details and results are provided in Supplemental Table 1. Relevant descriptive statistics and model estimates are provided in the main article. A principal component analysis (PCA) was used to identify relationships between test measures (factoextra package 1.0.7 in R). The PCA was run on all measures after scaling to unit variance and missing data points were removed (no rotation). All nine tests were completed in 43/88 children; missing data were limited to one or two tests in 38 children (14 bilateral CI; six unilateral; 18 NH) and to three to five tests in seven children (four bilateral CI; one unilateral; two NH). Most missing tests were the SVV and DVA (20 and 17 children, respectively). Linear regression was used to assess effects of age and group on each PCA component. Model effects were described by Type III Analysis of Variance Tables using Satterthwaite's method. Least-squares means were used for post-hoc comparisons of factors in the mixed models (Kenward-Roger method to correct degrees of freedom and Tukey method for comparing a family of three estimates to adjust p-values). Model details and results are provided in Supplemental Table 1. Residual diagnostics were performed for each analysis. Inspection of residual versus fitted values plots, normal QQ plots, hat value (leverage) plots, square root of standardized residual versus predicted value (scale-location), and plots of residuals versus values of each covariate and variable in the model did not reveal obvious or large deviations from homoscedasticity or normality, nor extreme outliers.
Results
Sensory Deficits in Children with Hearing Loss
The sensory tests identified auditory and balance deficits in both groups of children with hearing loss. As shown in Figure 1A, children with bilateral CI and unilateral HL had poorer balance than the normal hearing group as measured by the BOT-2 (bilateral CI: −3.16(SE = 0.97), t(83) = 3.26, p = .005; unilateral HL: −3.98(SE = 1.11), t(83) = 3.59, p = .002). Children with unilateral HL had similar speech perception accuracy on the PBK with their better hearing ear as the normal hearing group (−4.05(SE = 3.22)%, t(81) = 1.26, p = .42). None of the children were able to accurately repeat any of the words presented at 65 dB SPL to the ear with hearing loss. Although children with bilateral CIs had good accuracy (mean = 71.61%, 95%CI = 65.30%–77.92%) on the PBK test, this was poorer than either of the other groups (contrast to normal hearing group: −26.91(SE = 2.81)%, t(81) = 9.59, p < .0001 and to unilateral HL group: −22.86(SE = 3.32)%, t(81) = 6.88, p < .0001). As expected, children with unilateral HL had highly asymmetric hearing as measured by absolute differences between PBK accuracy (mean = 94.95%, 95%CI = 90.74%–99.16%) and children with bilateral CI showed slightly larger performance differences between ears than children with normal hearing (−13.40(SE = 2.16%, t(81) = 6.23, p < .0001). Self-reported hearing measured across three sections of the SSQ, shown in Figure 1B, revealed poorer than normal scores in both groups of children with hearing loss (bilateral CI: −1.53(SE = 0.49), t(68) = 3.12, p = .003; unilateral HL: −1.79(SE = 0.57), t(68) = 3.15, p = .002). The differences between the three SSQ sections were experienced similarly in the three groups (main effect: (F(2, 136) = 22.15, p < .0001), with no interaction with group: F(4, 136) = 1.16, p = .33), indicating that these self-reported challenges relative to normal were experienced by children with hearing loss to a similar extent in each of the measured hearing domains.

(A) Sensory findings in children with hearing loss: the bilateral CI and unilateral HL groups showed poorer balance than the normal hearing group; bilateral CI had poorer hearing measured by speech perception accuracy on PBK than the other groups and slightly more asymmetric performance on PBK (absolute differences) between ears than the normal hearing group. The unilateral HL group had similar hearing to normal hearing group with their better ear, but highly asymmetric hearing. (B) Self-reported scores on the SSQ reveal greater subjective hearing challenges in both groups of children with hearing loss. Significant findings of least square means contrasts of regression models accounting for age are indicated: .0001 “***”; .001 “**”; .01 “*.”
Two additional tests commonly used in vestibular testing, DVA and SVV, were conducted and no group differences were found in regression models accounting for age (DVA: F(2, 67) = 0.73, p = .48; SVV: F(2,64) = 0.39, p = .68).
Visuospatial Working Memory Deficits in Children with Hearing Loss
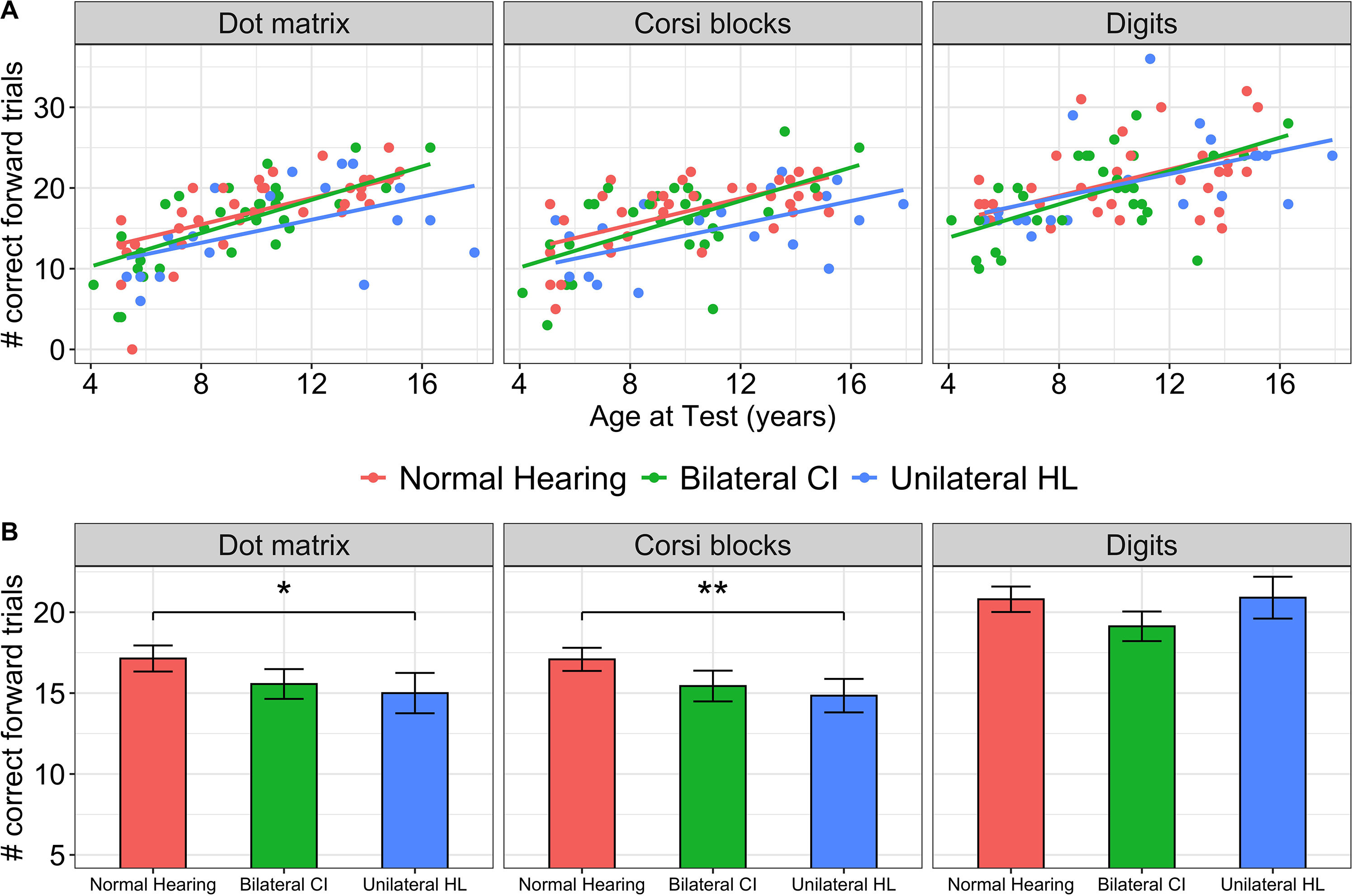
Results of two visuospatial and one verbal working memory tests are shown in Figure 2. As shown in Figure 2A, working memory, measured by correct number of trials in which children recalled sequences in backward order, increased across all tests with age (F(1,84) = 110.17, p < .0001) with a main effect of group (F(2,81) = 4.06, p = .02). Specific group differences in each test are shown in Figure 2B (bars = mean data, symbols = individual data) across age for each test. Contrasts of the model revealed poorer performance on the Dot matrix test for both groups of children with hearing loss (bilateral CI: −2.84(SE = 0.91), t(216) = 3.13, p = .002; unilateral HL: (−2.64(SE = 1.03), t(216) = 2.57, p = .01) and a trend for poorer performance by the unilateral HL group on the Corsi block test (−1.79(SE = 1.05), t(219) = 1.71, p = .09) compared to the normal hearing group. There were no significant differences between the normal hearing and either hearing loss group on the digit working memory test (bilateral CI: −0.81(SE = 0.90), t(216) = 0.90, p = .37; unilateral HL: (−1.47(SE = 1.03), t(216) = 1.42, p = .16). Of note, there was a significant difference in scores between these memory measures (F(2, 161) = 25.53, p < .0001)) across the groups; more correct trials occurred for the Corsi block test than the Dot matrix (1.46(SE = 0.50), t(166) = 2.93, p = .004) and fewer correct trials occurred for digits than the Dot matrix (−2.17(SE = 0.49), t(165) = 4.45, p < .0001). Lower scores on backward Digit span compared to the Dot matrix or Corsi block tests is consistent with previous reports (Alloway et al., 2006; Nadler & Archibald, 2014).

Working memory measured by correct number of trials in which children recalled sequences in backward order: (A) increased with age (regression lines for each group from the mixed model regression) and (B) were different between groups (bars = mean, errorbars = SE). Significant findings of least square means contrasts of regression model accounting for age are indicated: .0001 “***”; .001 “**”; .01 “*”; .05 “.”.
Short-Term Visuospatial Memory Deficits in Children with Unilateral Hearing Loss
Results of two visuospatial and one verbal short-term memory tests are shown in Figure 3. As shown in Figure 3A, short-term memory, measured by correct number of trials in which children recalled sequences in forward order, increased across all tests with age (F(1,85) = 70.95, p < .0001) with a trend for group effects (F(2,85) = 2.72, p = .07). Specific group differences in each test are shown in Figure 3B (bars = mean data, symbols = individual data) across age for each test. Contrasts of the model revealed poorer performance on the Dot matrix test and Corsi blocks for only the unilateral HL group compared to normal hearing (Dot matrix: −2.58(SE = 1.16), t(199) = 2.23, p = .03; Corsi blocks: −3.07(SE = 1.16), t(201) = 2.64, p = .009). There were no significant differences or even trends to suggest differences between the unilateral HL and normal hearing groups on the Digit test or between the bilateral CI group and the normal hearing group on any of the short-term memory tests. Of note, there was a significant difference in scores between measures (F(2, 168) = 49.00, p < .0001); more correct trials in forward order occurred for the digits than either the Corsi block (4.51(SE = 0.51), t(168) = 8.78, p < .0001) or Dot matrix (4.27(SE = 0.51), t(168) = 8.33, p < .0001) tests. Higher scores on forward Digit span than the Dot matrix or Corsi block tests is consistent with previous reports (Alloway et al., 2006; Nadler & Archibald, 2014).
Short-term memory measured by correct number of trials in which children recalled sequences in forward order: (A) increased with age (regression lines for each group from the mixed model regression) and (B) were poorer in children with unilateral HL (bars = mean, errorbars = SE). Significant findings of least square means contrasts of regression model accounting for age are indicated: .0001 “***”; .001 “**”; .01 “*.”
Learning Deficits in Children with Hearing Loss
Results of four WIAT subtests (three reading tests and one math test) are shown in Figure 4. As shown in Figure 4A, scores on standardized tests increased across all tests with age (F(1,75) = 140.87, p < .0001) with significant differences between groups (F(2,75) = 6.07, p = .004). Specific group differences in each test are shown in Figure 4B (bars = mean data, symbols = individual data) across age for each test. Contrasts of the model revealed that both groups of children with hearing loss performed more poorly than the normal hearing group on the tests of Word reading (bilateral CI: −4.40(SE = 2.22), t(209) = 2.03, p = .04; unilateral HL: −6.68(SE = 2.34), t(205) = 2.85, p = .005), the Pseudo-word reading (bilateral CI: −4.75(SE = 2.23), t(211) = 2.13, p = .03; unilateral HL: −6.57(SE = 2.35), t(206) = 2.79, p = .006) and Math (bilateral CI: −5.92(SE = 2.17), t(201) = 2.73, p = .007; unilateral HL: −5.50(SE = 2.33), t(203) = 2.36, p = .02). There were no significant differences between the two hearing loss groups on any of the measures.

Scores on the standardized WIAT subtests: (A) increased with age (regression lines for each group from the mixed model regression) and (B) were poorer in children with bilateral CI and unilateral HL compared to the normal hearing group (bars = mean, errorbars = SE). Significant findings of least square means contrasts of regression model accounting for age are indicated: .0001 “***”; .001 “**”; .01 “*.”
Language Deficits in Children with Hearing Loss
As shown in Figure 5A, CELF screener test scores increased across all tests with age (F(1,79) = 51.98, p < .0001) with significant differences between groups (F(2,79) = 10.46, p < .0001). Specific group differences are shown in Figure 5B (bars = mean data, symbols = individual data) across age. Contrasts of the model revealed that both groups of children with hearing loss performed more poorly than the normal hearing group (bilateral CI: −4.01(SE = 1.18), t(79) = 3.38, p = .003; unilateral HL: −3.22(SE = 1.31), t(79) = 2.46, p = .04). There was no significant difference in CELF scores between the hearing loss groups (0.80(SE = 1.37), t(79) = 0.58, p = .83).

Language measured by the CELF—fifth edition screening test: (A) increased with age (regression lines for each group from the mixed model regression) and (B) were poorer in children with bilateral CI and unilateral HL compared to the normal hearing group (bars = mean, errorbars = SE). Significant findings of least square means contrasts of regression model accounting for age are indicated: .0001 “***”; .001 “**”; .01 “*.”
Memory and Learning Challenges in Children with Hearing Loss Revealed by Principal Component Analyses
A PCA was conducted to assess the relationship between the measures collected in the three groups of children. Four main components (dimensions (Dim)) with eigenvalues >1 were found. The PC loadings (cos2) of each measure to each of the four dimensions are plotted in Figure 6A. Dim.1 represented a cluster of the language, memory, and academic tests and explained the largest portion of variance in the data (46.8%). The self-reported SSQ subtests clustered in Dim.2 which explained a smaller portion of data variance (10.5%). The remaining two dimensions contained clusters of sensory tests; balance and vestibular tests clustered in Dim.3 (8.5% data variance), and the PBK (speech perception) measures clustered in Dim.4 (6.4% data variance). PC scores for each dimension are shown for all children with different symbols and shades/colours by group in Figures 6B and 6C. Larger symbols indicate the group means and elipses denote 95% confidence around the mean.

(A) Measures clustered into four main PCA dimensions. Dim.1 explained the largest portion of variance in the data (46.8%) and contained contributions from the tests of language (CELF Screener), memory (Dot matrix, Corsi blocks, Digit span) and academics (WIAT subtests). Dim.2 (10.5% of data variance) was a cluster of self-reported measures on the SSQ. Dim. 3 (8.5% of data variance), was a cluster of balance (BOT-2) and vestibular (DVA and SVV) tests. Dim.4 (6.4% of data variance), was a cluster of speech perception (best PBK score and PBK asymmetry. PC scores for each dimension are shown for all children with different symbols and shades/colours by group in Figures 6B and 6C. Larger symbols indicate the group means and ellipses denote 95% confidence around the mean. Significant effects of group were found in all four PCA dimensions.
As shown in Figure 6B, and confirmed with regression models that accounted for age, PC scores in Dim.1 and Dim.2 were significantly reduced in the two hearing loss groups compared to normal. Estimated differences in Dim.1 PC scores (bilateral CI: −1.61(SE = 0.45), t(84) = 3.57, p = .002; unilateral HL: −2.03(SE = 0.52), t(84) = 3.93, p = .0005) revealed similar deficits in “memory and learning” in both groups and this was further supported by the lack of significant differences in Dim.1 PC scores between hearing loss groups (bilateral CI and unilateral HL). Dim.2 PC scores, reflecting self-reported hearing, also showed significant deficits in both groups of children with hearing loss compared to the normal hearing group (bilateral CI: −1.35(SE = 0.30), t(84) = 4.52, p = .0001; unilateral HL: −1.34(SE = 0.34), t(84) = 3.92, p = .0005).
PC scores in Dim.3 and Dim.4 are plotted in Figure 6C. PC scores of Dim.3 in the normal hearing group are slightly negative reflecting the opposite polarity of the measures that clustered in this dimension. Good balance measured by high BOT scores occurred with few DVA lines lost and little degree of error on the SVV as expected. The significantly more positive Dim.3 scores in the two hearing loss groups reflects both the poorer BOT scores and increased challenges on the DVA and SVV tests (bilateral CI: 0.81(SE = 0.27), t(84) = 3.00, p = .01; unilateral HL: 1.68(SE = 0.31), t(84) = 5.44, p < .0001). Increased Dim.3 scores in the unilateral HL compared to the bilateral CI group (0.81(SE = 0.32), t(84) = 2.72, p = .02) suggest increased balance/vestibular deficits in the former group despite nonsignificant group differences in the two vestibular tests (DVA, SVV) alone. Dim.4 scores are comprised of two speech perception measures that occur with opposite polarity; children with normal hearing showed high PBK scores and limited asymmetry in PBK scores between the ears, explaining their mean Dim.4 scores = −0.03, SD = 0.35. The bilateral CI group showed significantly lower Dim.4 scores than the normal hearing group (−0.86(SE = 0.17), t(84) = 4.97, p < .0001), reflecting reduced PBK scores and slightly higher PBK asymmetry, whereas the unilateral HL group had significantly elevated Dim.4 scores compared to the normal hearing group (−1.47(SE = 0.18), t(84) = 7.44, p < .0001), reflecting good PBK scores and high PBK asymmetry.
Discussion
The present study is the first, to our knowledge, to compare performance on sensory (hearing and vestibular/balance) function as well as memory and academic measures in children with unilateral hearing loss and children using bilateral CIs. Our hypothesis was that these two groups of children have similar developmental challenges as they both have poor spatial hearing compared to typically developing peers. Results confirmed that the sensory deficits are unique in the two groups of children with hearing loss but that both groups have significant and often similar deficits in language, memory, and learning. The data also revealed that memory deficits in children with poor spatial hearing occurred in the visuospatial domain.
Children with Bilateral and Unilateral Deafness Have Unique Sensory Deficits
Data shown in Figure 1A confirmed that the group of children with unilateral hearing loss had good speech perception in their normal hearing ear but experienced highly asymmetric hearing. Despite differences in degrees of hearing loss, none were able to repeat PBK words presented at 65 dB SPL to the impaired ear. Children provided with bilateral CIs (mostly simultaneously) showed excellent speech perception scores, but their best scores were still significantly poorer than the other two groups and they also showed slightly more asymmetry than their normal hearing peers. Although bilateral CI with limited delay allows for development of both ears in tandem and reduces the “aural preference” found in children receiving bilateral CIs with long interimplant delays (Gordon et al., 2015), the present findings reflect the unique hearing provided by CIs and the potential for each ear to develop slightly differently in each child. Data in Figure 1A also confirmed that children with hearing loss experienced more balance challenges than their normal hearing peers (Cushing et al., 2013). The novel finding in the present study is that balance was as impaired in children with unilateral hearing loss as in the children receiving bilateral CI. This supports prior suggestions that an etiology affecting both auditory and vestibular systems is likely to contribute more to poor balance in children with hearing loss than additional vestibular insults of cochlear implantation (Cushing et al., 2008).
Data in Figure 1B revealed that children with unilateral hearing loss reported similar hearing challenges as their peers using bilateral CIs. This finding supports prior evidence of reduced quality of life in children with unilateral hearing loss (Doshi et al., 2013; Härkönen et al., 2015) and the need to recognize and address challenges of relying on one ear to hear (Lieu et al., 2012; Roland et al., 2016; van Wieringen et al., 2019). Whereas children with bilateral CIs receive therapy and support services for their hearing loss, this is often not the case for children with unilateral HL. Indeed, none of the children in the unilateral HL group used a hearing aid in their poor ear. At the same time, the findings suggest that ratings of hearing by children with bilateral deafness who receive early bilateral CI were not poorer than ratings from peers who can hear normally in one ear; this represents a remarkable benefit of bilateral cochlear implantation in children.
Children with Poor Spatial Hearing Show Deficits in Visuospatial Memory
Data shown in Figures 2 and 3 revealed that both groups of children with hearing loss had deficits in visuospatial working memory and that children with unilateral hearing loss had impaired short-term visuospatial memory. These data confirm the hypothesis that children with spatial hearing loss have cognitive challenges. These data also indicate that cognitive deficits occur across sensory domains (verbal and visuospatial) which is not consistent with expectations of enhanced visual processing due to compensation and/or cross-modal plasticity in individuals with hearing loss (Shiell et al., 2014).
Visual processing deficits have been reported in children with hearing loss (Bharadwaj & Mehta, 2016; Castellanos et al., 2015; Cleary et al., 2001; Marschark et al., 2015), but unlike the present cohorts, these challenges typically occurred with clear concurrent impairments in verbal memory. The resulting impression was that diminished scores on these visual tasks in children with hearing loss related to general cognitive impairments (Bharadwaj & Mehta, 2016; Castellanos et al., 2015; Cleary et al., 2001; Marschark et al., 2015), perhaps stemming from listening fatigue (Bess et al., 2014, 2020; Camarata et al., 2018; Hornsby et al., 2016) and poor phonological processing (Geers & Hayes, 2011; Lyxell et al., 2008; Wass, Ching, et al., 2019; Wass, Löfkvist, et al., 2019). Relatively good verbal memory skills in the present cohort likely reflects their better hearing than prior study groups. Although both children receiving bilateral CIs at young ages and children with unilateral hearing loss experience language-related deficits (Bess et al., 2020; Bess & Hornsby, 2014; Geers & Hayes, 2011; Nittrouer & Burton, 2005; Peelle, 2018; Steel et al., 2015), their cognitive impairments may be less severe than indicated in earlier study cohorts of children with CIs. This could be due to benefits of bilateral CI over unilateral CI use in children. For example, children using bilateral CIs experience reduced listening fatigue (Galvin & Mok, 2015; Hughes & Galvin, 2013) and better phonological processing (Caselli et al., 2012) than children using unilateral CIs. Still, even children with mild hearing loss show reduced cognitive resources (McCreery et al., 2019), which means that, while the severity of deficits in children with hearing loss in the present study may be less than in prior cohorts of CI users, they were likely to have experienced cognitive challenges relative to normal hearing peers.
It is also possible that poor visuospatial processing in the two groups of children with hearing loss in the present study reflect additional consequences of poor spatial hearing. Although deafness-related language delays may be limited by early access to hearing through bilateral CIs or through one normal hearing ear, the remaining deficits in binaural hearing establish ongoing spatial mismatches between auditory and visual input. These mismatches could affect spatial perception in both visual and auditory domains as shown in individuals with partial vision loss (Lessard et al., 1998; Richards, 2018) and might explain why poor visuospatial processing was found in the present cohorts of children with poor or no binaural hearing despite their good vision. In addition, mismatched sensory input likely deteriorates sensory integration with implications for attention and cognitive resources (Liu et al., 2020). These potential effects of poor spatial hearing on cognitive load could explain the sensitivity of the visuospatial memory tasks to deficits in children with hearing loss.
Children With Unilateral and Bilateral Deafness can Achieve Similar Academic and Language Skills
Data shown in Figures 5 and 6 revealed that children with unilateral hearing loss achieved similar academic results and language scores as their peers with deafness who use bilateral CIs. Both groups showed deficits compared to normal hearing peers as previously shown by separate studies of children using CIs (Sarant et al., 2015) and children with unilateral hearing loss (Bess & Tharpe, 1984; Lieu, 2004; Lieu et al., 2012). Children with unilateral hearing loss are known to have poorer hearing and language development compared to children with normal hearing and similar impairments to children with mild to moderate bilateral hearing impairment (Fitzpatrick et al., 2019), indicating that normal hearing in one ear cannot compensate for the loss of bilateral hearing. Novel findings from the present study indicate that bilateral CIs provided to children with bilateral deafness at young ages can promote similar academic and language skills to peers who have much better residual hearing (normal) in one ear. This similarity may seem suprising given that spectral cues of sounds are poorly available to children using CIs (Chatterjee et al., 2015; Gifford et al., 2018; Good et al., 2017; Hopyan-Misakyan et al., 2009) but are accessible in the normal hearing ear of children with unilateral hearing loss. This leads us to hypothesize that these similarities are better explained by the impaired spatial hearing faced by both groups of children.
Children With Impaired Spatial Hearing Have Memory and Learning Deficits
PCA was used to assess potential relationships between the measures conducted in the present study. As shown in Figure 6, the main dimension (Dim.1) was a cluster of the language, memory, and academic test scores, reflecting important relationships amongst these measures. Dim.1 scores were significantly different in both hearing loss groups compared to the normal hearing group, confirming deficits in memory and learning that were similar in children with bilateral CIs and children with unilateral HL. The remaining dimensions reflected self-reported hearing (Dim.2), balance/vestibular outcomes (Dim.3), and speech perception measured by PBK (Dim.4). These three dimensions confirm expected relationships between SSQ subtests, between balance and vestibular function, and between speech perception scores, respectively, and also confirm that both hearing loss groups show abnormalities in all three dimensions.
The cluster in Dim.1 of impaired memory, academic, and language in children with unilateral hearing loss and in bilateral CI users provides further evidence that hearing loss in childhood yields significant developmental challenges. The working memory (backward) tasks provided larger contributions to this dimension than the short-term (forward) tasks as shown in Figure 6. Several models of working memory have been considered in the context of language development in children with hearing loss (McCreery et al., 2019; Nittrouer, 2017). Baddeley's model is particularly appealing because it suggests that stimuli enter into either a visuospatial sketchpad or a phonological loop and that information in these subsystems can be used or stored in long-term memory. The phonological loop has been an area of focus in individuals with hearing loss, given their impaired access to spoken language, but data from the present study suggests that the remaining system, the visuospatial sketchpad, can also be impaired in those with childhood hearing loss.
The loss of normal binaural hearing in children with hearing loss could have particular effects on working memory in both auditory and visuospatial domains. The hearing advantages of providing bilateral input in children with hearing loss in both ears are clear (Godar & Litovsky, 2010; Jacobs et al., 2016; Papsin & Gordon, 2008; Polonenko et al., 2018, 2019; Sarant et al., 2014), but their binaural/spatial hearing remains impaired, in part, because of poor preservation of binaural cues provided by two independent hearing devices (Bierer et al., 2011; Easwar et al., 2017; Litovsky et al., 2012). Without binaural/spatial hearing, children have difficulty listening to one speaker in the midst of many surrounding sounds (Misurelli & Litovsky, 2012). In the present study, binaural hearing deficits were greatest in children with unilateral hearing loss as measured by increased asymmetry in speech perception scores between the ears (Figure 1). In this context, children with unilateral hearing loss were at a greater disadvantage than their peers with severe to profound deafness who used bilateral CIs. Indeed, the children with unilateral hearing loss showed similar deficits in academics (Figure 4) and language (Figure 5) as the CI users but were the only group to show both short-term and working memory visuospatial memory deficits (Figures 2 and 3). We speculate that children with unilateral hearing loss were particularly vulnerable to these changes because they had no access to bilateral hearing and are known to have very poor spatial hearing (Liu et al., 2020). By contrast, children with bilateral CI show some sound localization albeit with less accuracy and reduced sensitivity compared to peers with normal hearing (Bennett & Litovsky, 2020; Killan et al., 2019). If these auditory deficits are more pronounced in children with no bilateral hearing than children using bilateral hearing devices, perhaps similar deficits in other modalities are also more pronounced. The expansion of a primary sensory deficit into another modality has been shown in adults with visual impairment who have poor sound localization despite normal hearing (Gori et al., 2014; Lessard et al., 1998; Richards, 2018) and could reflect inconsistencies between visual and auditory spatial maps (Knudsen & Brainard, 1995; Knudsen & Knudsen, 1990; Occelli et al., 2013).
If children's loss of binaural/spatial hearing compromises spatial perception in other modalities, these deficits could be further exacerbated by poor balance (Figure 1) and could impair cognitive abilities in both domains. Visuospatial memory is a predictor of reading and mathematics ability (Alloway, 2007; Alloway et al., 2009; De Weerdt et al., 2013; Li & Geary, 2017), as well as long-term academic achievement (Sarver et al., 2012) in children with normal hearing. Spatial perception is involved in understanding the concept of relative proportions which is important in mathematics (Andersen, 2014; Mix & Cheng, 2012; Verdine et al., 2017; Wai et al., 2009), and orthographic word recognition may be impaired by visuospatial deficits (Wass, Löfkvist, et al., 2019). Impairments in academic abilities could also be due to increased cognitive load created by reduced perception of auditory space (Andéol et al., 2017; Loughrey et al., 2018; Peelle, 2018). In sum, the data support the hypothesis that binaural/spatial hearing impairments are a major contributor to developmental delays in children.
Conclusion
Novel findings in the present study reveal that children with impaired binaural hearing are at high risk for impaired development. Children with unilateral hearing loss experienced the same degree of visuospatial memory, language and academic deficits as children with bilateral CIs, indicating the importance of binaural auditory input during development. Deficits in working memory in children with bilateral deafness using bilateral CIs and in children with unilateral hearing loss were prevalent in the visuospatial domain and less clear in the verbal domain, perhaps reflecting their good access to sound in early life. In sum, the results further emphasize previous calls to provide children with access to bilateral hearing early in life and to seek improved methods to promote their binaural/spatial hearing. Further, the links between spatial deficits in the auditory and visual domains and their potential effects on cognition and learning in children need further investigation.
Supplemental Material
sj-docx-1-tia-10.1177_23312165211051215 - Supplemental material for Functional Consequences of Poor Binaural Hearing in Development: Evidence From Children With Unilateral Hearing Loss and Children Receiving Bilateral Cochlear Implants
Supplemental material, sj-docx-1-tia-10.1177_23312165211051215 for Functional Consequences of Poor Binaural Hearing in Development: Evidence From Children With Unilateral Hearing Loss and Children Receiving Bilateral Cochlear Implants by Claire McSweeny, Sharon L. Cushing, Jennifer L. Campos, Blake C. Papsin and Karen A. Gordon in Trends in Hearing
Footnotes
Acknowledgments
We would like to acknowledge funding provided by SickKids Foundation Research endowments as well as the participation of the children and their families and our collaboration with the clinical team at the Hospital for Sick Children, Cochlear Implant Program.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KG: speaker for Cochlear Corp, Advanced Bionics, lecturer: Salus University. SLC holds sponsored research agreements with Cochlear Americas, is a speaker for Cochlear Americas and Interacoustics, earns royalties from the book, Balance Disorders in the Pediatric Population, Plural Publishing and is a patent holders for Patents #: 7041–0: Systems and Methods for Balance Stabilization..
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by research endowment funds.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
