Abstract
A prospective clinical trial evaluated the effectiveness of cochlear implantation in adults with asymmetric hearing loss (AHL). Twenty subjects with mild-to-moderate hearing loss in the better ear and moderate-to-profound hearing loss in the poorer ear underwent cochlear implantation of the poorer hearing ear. Subjects were evaluated preoperatively and at 1, 3, 6, 9, and 12 months post-activation. Preoperative performance was evaluated unaided, with traditional hearing aids (HAs) or with a bone-conduction HA. Post-activation performance was evaluated with the cochlear implant (CI) alone or in combination with a contralateral HA (bimodal). Test measures included subjective benefit, word recognition, and spatial hearing (i.e., localization and masked sentence recognition). Significant subjective benefit was reported as early as the 1-month interval, indicating better performance with the CI compared with the preferred preoperative condition. Aided word recognition with the CI alone was significantly improved at the 1-month interval compared with preoperative performance with an HA and continued to improve through the 12-month interval. Subjects demonstrated early, significant improvements in the bimodal condition on the spatial hearing tasks compared with baseline preoperative performance tested unaided. The magnitude of the benefit was reduced for subjects with AHL when compared with published data on CI users with normal hearing in the contralateral ear; this finding may reflect significant differences in age at implantation and hearing sensitivity across cohorts.
Keywords
Listeners with significant differences in the unaided hearing thresholds between ears, known as asymmetric hearing loss (AHL), experience poor speech recognition and reduced spatial hearing (Firszt et al., 2017; Rothpletz et al., 2012) and report a poorer quality of life (Wie et al., 2010) than listeners with normal hearing bilaterally. Poorer performance and quality of life are likely due to the limited ability or inability to use the binaural cues that support spatial hearing. Historically, treatment options for AHL have included bilateral hearing aids (HAs), a bone-conduction HA (BCHA), contralateral routing of the signal (CROS) HA, or bilateral microphones with CROS (Bi-CROS) HA. Listeners may discontinue the use of an HA on the poorer hearing ear due to limited speech recognition benefit. Use of technology that routes the signal to the better hearing ear may provide similar (Agterberg et al., 2019) or worse (Mertens et al., 2017) spatial hearing as the information from both the ears is transmitted via one auditory pathway, limiting access to binaural cues. Cochlear implantation of the poorer hearing ear may improve speech recognition and spatial hearing by stimulating the affected auditory pathway to provide the listener with binaural information. Preliminary investigations of cochlear implant (CI) use in subjects with AHL or unilateral hearing loss (UHL; normal to near-normal hearing in the contralateral ear) demonstrate significant improvements compared with preoperative abilities; however, the magnitude of the benefit and time course of acclimatization varies widely—with some studies observing significant benefits as early as 1 month after CI activation (Buss et al., 2018; Dillon et al., 2017a, 2017b) and others not demonstrating benefit until 12 or more months after CI activation (Gartrell et al., 2014; Mertens et al., 2017). As indications for cochlear implantation expand, there is a need to understand the magnitude of the benefit and time course of acclimatization with the CI alone and with bimodal stimulation (CI plus contralateral HA) in CI recipients with AHL.
Predicted benefits of cochlear implantation in cases of AHL include improvements in sound source identification (also known as localization) and masked speech recognition with spatially distributed sources. These tasks are described as reflecting spatial hearing because they rely on auditory abilities required to process sound from different locations in space. When a sound source is off the listener’s midline, there is a difference in time of arrival at the two ears and a difference in level of high-frequency energy at the two ears, due to the acoustic shadow of the head. For listeners with normal hearing, auditory localization relies on a combination of interaural time and level differences (Blauert, 1997). In contrast, CI users do not appear to use low-frequency interaural time differences, relying instead on interaural level differences for localization (Dirks et al., 2019; Dorman et al., 2015). Similarly, for most conditions, the spatial release from masking (SRM) observed when comparing masked speech recognition for colocated and spatially separated sources can be explained by reduced masker energy in the high frequencies contralateral to the noise source (Dirks et al., 2019; Williges et al., 2019). This has led some researchers to argue that the binaural benefit associated with SRM experienced by CI users is really just a monaural effect, indicating reliance on cues from the side with the better signal-to-noise ratio (SNR). However, SRM can be observed in the absence of head shadow under some conditions, such as when the masker is itself composed of speech (Bernstein et al., 2016). Regardless of the cues underlying binaural benefit for different listeners, having independent input to the two sides is thought to be important for spatial hearing. Although a CROS HA or a BCHA presents information from both sides of the head to one ear, using a CI in the poorer hearing ear may support better spatial hearing in the bimodal condition by providing access to binaural cues. In the context of AHL, bimodal stimulation may support better spatial hearing than alternative options by stimulating both auditory pathways, which in turn may support greater subjective benefit (Firszt et al., 2018; Thompson et al., 2020).
The benefits of bimodal stimulation have been demonstrated in CI recipients who meet the traditional candidacy criteria for cochlear implantation: bilateral moderate-to-profound sensorineural hearing loss and poor aided speech recognition. Although most traditional CI recipients demonstrate better masked speech recognition when listening in a bimodal condition when compared with a monaural condition (Dunn et al., 2005; Gifford et al., 2007; Potts et al., 2009), others experience better masked speech recognition with the CI alone (Dunn et al., 2005). Individual differences in the ability to integrate cues from the CI and contralateral HA could be related to the ability to fuse the information provided by the two devices (Kong & Braida, 2011; Reiss et al., 2016). Bimodal listeners with a better aided pure-tone average (PTA; e.g., <55 dB HL) demonstrate better performance on masked speech recognition than bimodal listeners with a poorer aided PTA (Yoon et al., 2012). Considering this, CI recipients with AHL may experience large improvements on spatial hearing tasks because of their relatively good unaided hearing thresholds in the acoustic-hearing ear compared with participants in previous investigations of bimodal hearing.
Investigations of the effectiveness of CI use in listeners with UHL have demonstrated improved speech recognition with the CI alone, spatial hearing in the best aided condition, and subjective benefit when compared with preoperative abilities evaluated unaided or with alternative options such as a BCHA (Arndt et al., 2011; Buss et al., 2018; Dillon et al., 2017a, 2017b; Firszt et al., 2012b; Galvin et al., 2019; Gartrell et al., 2014). The significant subjective benefit, better word recognition, and improved spatial hearing performance observed within the initial weeks of CI use in a UHL cohort by Dillon et al. (2017a, 2017b) and Buss et al. (2018) may have been related to the normal hearing in the acoustic-hearing ear (i.e., ≤35 dB HL from 125 to 8000 Hz). One question of interest is whether the subjects with mild-to-moderate hearing loss in the acoustic-hearing ear would experience a similar magnitude of benefit and time course of acclimatization when tested under the same conditions. Considering the degraded sound quality associated with bilateral hearing loss and the need to fuse the signals from the CI and contralateral HA, it is possible that CI recipients with AHL may experience a more prolonged period of acclimatization than those with UHL. Although prior studies have evaluated spatial hearing in bimodal CI recipients with a range of auditory thresholds in the acoustic-hearing ear (e.g., Firszt et al., 2012a), those protocols have not typically sampled performance across the post-activation period frequently enough to characterize the time course of acclimatization.
Data from listeners with AHL reviewed in the present report were collected using the same procedures as the previous study on CI recipients with UHL; those procedures include evaluation of subjective benefit (Dillon et al., 2017b), word recognition (Buss et al., 2018), and spatial hearing (Buss et al., 2018; Dillon et al., 2017a). Comparisons between these datasets are of interest since subjects met similar inclusion criteria, received the same electrode array, and were mapped with the same procedures, yet differed in the hearing sensitivity in the acoustic-hearing ear. Performance was assessed preoperatively and at 1, 3, 6, 9, and 12 months post-activation to characterize the time course of acclimatization. The study was designed with three main goals. First, we compared outcomes of cochlear implantation to alternative options for patients with AHL. The hypotheses were that performance would be similar when listening in an unaided condition when compared with a BCHA and that performance would be superior when listening in a bimodal condition when compared with the alternative options. Second, we assessed the magnitude of the benefit and time course of acclimatization for CI recipients with AHL. The hypothesis was that CI recipients with AHL would experience significant subjective benefit, better word recognition, and improved spatial hearing performance within the initial months of CI use. Third, we compared the magnitude of the benefit and time course of acclimatization of CI recipients with AHL to published data collected on CI recipients with UHL. The hypothesis was that the magnitude and time course of the observed improvements in subjects with AHL would be reduced or prolonged compared with subjects with UHL.
Methods
Subjects
Twenty subjects (11 females) with AHL were enrolled and underwent cochlear implantation as part of a clinical trial assessing the effectiveness of CI use in cases of UHL and AHL over a 12-month follow-up interval. The study procedures were approved by the Food and Drug Administration as part of an Investigational Device Exemption and by the study site Institutional Review Board. Subjects provided consent to participate prior to undergoing cochlear implantation. Results from the UHL cohort (
Subjects with AHL met the following inclusion criteria: (a) an unaided PTA (500, 1000, and 2000 Hz) of ≥70 dB HL in the ear to be implanted and between 35 and 55 dB HL in the contralateral ear, (b) aided word recognition on consonant-nucleus-consonant (CNC) words (Peterson & Lehiste, 1962) of ≤60% correct in the ear to be implanted and ≥80% correct in the contralateral ear, (c) previous listening experience with an alternative technology option for AHL (e.g., HA or Bi-CROS HA), (d) a passing score on the Mini Mental State Examination (Folstein et al., 1975), normed for age and education level, (e) native English proficiency, and (f) tinnitus severity in the ear to be implanted of moderate or less, quantified using the Tinnitus Handicap Inventory (THI; Newman et al., 1996). The majority reported an onset of hearing loss in the ear to be implanted that was sudden in nature (

Individual and Mean Preoperative Unaided Hearing Thresholds for the Ear to be Implanted and the Acoustic-Hearing Ear. Unaided hearing thresholds were obtained behaviorally for pulsed pure tones via insert earphones. Individual results are displayed with thin gray lines, and mean results are displayed with a thick black line.
Subjects underwent cochlear implantation with the MED-EL Synchrony Standard electrode array
1
(31.5 mm; MED-EL Corporation, Innsbruck, Austria), inserted using a round window surgical approach. The age at implantation ranged from 52 to 79 years, with a mean of 70 years (
CI Mapping
Activation of the CI was completed approximately 2 to 4 weeks after cochlear implantation. Subjects were fit with a SONNET ear-level processor. The CI was programmed with the omnidirectional microphone option for consistency with the UHL cohort in the published dataset, who listened with the Opus2 processor that features an omnidirectional microphone only (see Buss et al., 2018). Mapping was completed with the FS4 coding strategy and default electric frequency filters of 100 to 8500 Hz using the procedures described by Dillon et al. (2019). Briefly, mapping included the behavioral measurement of threshold and comfort levels on active channels, as well as loudness balancing using the adjacent-reference method (Throckmorton & Collins, 2001; Zwolan et al., 1997) and the reference method (Throckmorton & Collins, 2001) with the acoustic-hearing ear plugged. For one subject, the frequency of the lowest filter was adjusted from 100 to 70 Hz at the 3-month interval and returned to 100 Hz by the 9-month interval in an attempt to improve the sound quality. Subjects completed a 1-hr aural rehabilitation session with a speech-language pathologist at the CI activation and 1-month intervals (see Evans & Dillon, 2019).
Electrode contacts were deactivated when deemed necessary by the clinical audiologist. At initial CI activation, the most basal electrode (electrode 12) was deactivated for four subjects due to limited loudness growth (
HA Programming: Acoustic-Hearing Ear
Subjects listened with either their own HA or a loaner HA (Phonak Bolero, Aurora, IL, USA) on the acoustic-hearing ear over the study period. Fitting was accomplished with real-ear measures, and the HA output was verified using the NAL-NL1 prescriptive method (Byrne et al., 2001) at each interval. The directionality and SoundRecover features were deactivated for the loaner HAs. Subjects who listened with their own HA were asked to return to their programming audiologist to have these features deactivated.
Subjective Benefit
Subjective benefit was assessed with three questionnaires: Abbreviated Profile of Hearing Aid Benefit (APHAB; Cox & Alexander, 1995), Speech, Spatial, and Qualities of Hearing Scale (SSQ; Gatehouse & Noble, 2004), and THI. At the preoperative interval, subjects responded based on perceived ability or difficulty in their familiar listening condition (i.e., unaided, HA, BCHA, or Bi-CROS). At the post-activation intervals, subjects responded based on perceived ability or difficulty in the bimodal condition.
The APHAB was used to measure perceived difficulty in different auditory environments over the study period and was evaluated for the four subscales: Ease of Communication, Background Noise, Reverberation, and Aversiveness. The SSQ was administered to measure the perceived ability in different auditory scenarios and was scored on the three subscales: Speech Hearing, Spatial Hearing, and Qualities of Hearing. The THI was included as a measure of tinnitus severity in the CI ear; a reduction in tinnitus severity with CI use may influence subjective benefit.
Word Recognition: CI Ear
Word recognition in the CI ear was evaluated using the CNC words test. Subjects were seated 1 m away from the sound source in a double-walled soundbooth. Recorded materials were presented at 60 dB sound pressure level (SPL). Audibility at the acoustic-hearing ear was restricted by presenting speech-shaped noise, with a constant spectrum level from 250 to 1000 Hz and a 12-dB/octave roll-off from 1000 to 6000 Hz (GSI 61, Grason-Stadler Inc., MN, USA). This noise was presented at 30 to 40 dB sensation level using an insert earphone; a circumaural earphone was placed over the ear for additional attenuation. Subjects listened with an HA in the ear to be implanted at the preoperative interval and with the CI at the post-activation intervals. Performance was calculated as the percent of correctly repeated words (50 words per list).
Spatial Hearing Tasks
Spatial hearing was assessed in the soundbooth with subjects seated in the center of a 180° arc of 11 loudspeakers spanning ±90° and facing the middle loudspeaker. The spatial hearing tasks included localization and masked sentence recognition.
The localization task has been described previously (see Buss et al., 2018; Dillon et al., 2017a, 2019). Briefly, subjects were presented with 200-ms speech-shaped noise bursts that were bandpass filtered at 126 to 6000 Hz and gated using 20-ms raised-cosine ramps. The stimulus was presented from one of the 11 loudspeakers at 52, 62, or 72 dB SPL. Subjects were instructed to report the loudspeaker number corresponding to the perceived sound source. The intensity level and loudspeaker location were randomized, with each loudspeaker presenting the stimulus four times at each intensity level, for a total of 132 trials. Level was roved to prevent monaural loudness from serving as a reliable cue to location. Performance was characterized as the root-mean-squared (RMS) error in degrees.
For masked sentence recognition, the target sentence was presented from the center loudspeaker (0° azimuth), and the masker was colocated with the target at 0° (SoNo), 90° to the CI ear (SoNci), or 90° to the acoustic-hearing ear contralateral to the CI (SoNcontra). Subjects were instructed to face the center loudspeaker and repeat the target sentence. Recorded materials were presented at 60 dB SPL. The test battery included Bamford–Kowal–Bench sentences in noise (Bench et al., 1979) and AzBio sentence test (Spahr et al., 2012). Due to the limited number of Bamford–Kowal–Bench sentences in noise test lists, subjects heard the same lists across sequential intervals; therefore, the results were not included in the present report. The AzBio sentences were presented in a 10-talker masker at a fixed SNR. The specific SNR for each subject was determined using a decision tree, where the SNR was adjusted in 5 dB steps from 10 dB SNR to 0 dB SNR until the subject scored ≤50% in the SoNo spatial configuration. The decision tree was conducted at the preoperative and 1-month intervals, and the SNR determined at the 1-month interval was used at the subsequent post-activation intervals. This procedure could result in different SNRs for testing at the preoperative and post-activation intervals. Performance is quantified as the percent of words repeated back correctly for one list (20 sentences per list). Subjects did not hear the same sentence list twice before the 6-month interval.
Procedures
The test battery (i.e., subjective benefit, word recognition, and spatial hearing) was completed at the preoperative interval and at 1, 3, 6, 9, and 12 months after CI activation. Subjects completed the APHAB and the SSQ on their own and discussed their responses with the researcher. The researcher administered the THI. All subjective questionnaires were completed prior to the behavioral hearing assessments. Unaided behavioral pure-tone detection thresholds were measured for each ear at each interval. Word recognition and spatial hearing tasks were completed with subjects listening to their familiar maps.
Three listening conditions were evaluated on the spatial hearing tasks. Subjects listened with the HA on the acoustic-hearing ear for all conditions. At the preoperative and 12-month intervals, subjects were evaluated without technology on the ear to be implanted (unaided) and with a BCHA that was fit acutely. At all post-activation intervals, subjects were evaluated in the bimodal condition.
The majority of subjects completed the protocol at all intervals. Two subjects did not complete testing at the 9-month interval, and one subject withdrew after the 6-month interval after moving out of state. Two subjects did not complete the BCHA condition at the 12-month interval due to time limitations and fatigue.
Data Analysis
Data were analyzed with linear mixed models (LMM) implemented using R statistical software (R Core Team, 2019) with a random intercept for each subject, as described later. LMMs accommodate missing data when compared with other models, such as repeated-measures analysis of variance (Oleson et al., 2019). A logit transformation was applied to the proportion correct data for the LMMs assessing word recognition and masked sentence recognition. The logit transformation imposed a minimum of 0.1% and a maximum of 99.9%. Post hoc comparisons were completed with either reduced models or paired
Compare Outcomes of Cochlear Implantation to Alternative Options for AHL on Spatial Hearing Tasks
The first goal was to compare outcomes of cochlear implantation to alternative options for AHL (i.e., unaided and BCHA) for the localization and masked sentence recognition tasks. Performance in the unaided and BCHA conditions was compared between the preoperative and 12-month intervals to determine if undergoing cochlear implantation influenced performance with alternative options for AHL. Performance in the unaided condition at the preoperative interval was also compared with performance in the bimodal condition at the 12-month interval to assess whether the use of a CI provides a performance benefit. It was hypothesized that performance would be similar or poorer with the BCHA when compared with the unaided condition; thus, the unaided condition was selected as the comparison condition to the bimodal condition.
Determine the Magnitude of the Benefit and Time Course of Acclimatization for CI Recipients With AHL
Next, the magnitude of the benefit and the time course of acclimatization were assessed for CI recipients with AHL on measures of subjective benefit, word recognition, and spatial hearing. The LMMs assessed performance over all study intervals. For cases with a significant main effect of interval, a reduced model or paired
Compare the Magnitude of the Benefit and Time Course of Acclimatization for CI Recipients With AHL to a Cohort of CI Recipients With UHL
Last, the magnitude of the benefit and time course of acclimatization was compared between the present AHL cohort and published data from the UHL cohort on measures of subjective benefit, word recognition, and spatial hearing. The models used to assess the magnitude of the benefit and time course of acclimatization for the AHL cohort described earlier were expanded to include cohort (i.e., AHL and UHL). The UHL cohort presented with unaided air-conduction thresholds of ≤35 dB HL from 125 to 8000 Hz in the contralateral ear, while the AHL cohort presented with a PTA of 35 to 55 dB HL.
An exploratory analysis of SRM was also evaluated, comparing the performance between cohorts to quantify the spatial hearing benefit. SRM was calculated as the difference in percent correct scores between the spatially separated configurations (SoNci or SoNcontra) and the colocated spatial configuration (SoNo).
Results
The SNR used for evaluating masked sentence recognition was determined at the preoperative and 1-month test intervals using the procedure described earlier. At the preoperative interval, 1 subject was assessed at 10 dB SNR, 6 subjects were assessed at 5 dB SNR, and 13 subjects were assessed at 0 dB SNR. The SNR determined at the 1-month interval—and used over all post-activation intervals—was 10 dB SNR for 1 subject, 5 dB SNR for 3 subjects, and 0 dB SNR for 16 subjects. Seventeen subjects were assessed with the same SNR at the preoperative and post-activation intervals. Two of the three subjects who were evaluated at 5 dB SNR at the preoperative interval and at 0 dB SNR at the post-activation intervals repeated the masked sentence recognition task at the 12-month interval at 5 dB SNR; these additional data points supported a comparison of preoperative and post-activation performance for these subjects.
The average daily CI use between post-activation intervals was obtained via datalogging provided by the CI processor. Datalogging revealed variability both across and within subjects in the average hours of daily CI use. Seven subjects listened consistently with the CI for ≥8 hr per day over the study period. Three subjects listened with the CI for approximately 5 hr per day prior to the 1-month interval and ≥8 hr per day in subsequent intervals. Seven subjects fluctuated between more or less than 8 hr of CI use per day in the postactivation period. One subject listened with the CI ≤7 hr per day over the study period. Datalogging was not available for one subject.
Compare Outcomes of Cochlear Implantation to Alternative Options for AHL on Spatial Hearing Tasks
Spatial Hearing: Localization
Figure 2 plots the RMS localization error over the study period in each of three listening conditions: unaided (open), BCHA (diagonal hatching), and bimodal (solid). A lower value indicates better performance. Localization performance was similar for the unaided and BCHA conditions and similar at the preoperative and 12-month intervals. An LMM assessed the main effects of listening condition (i.e., unaided and BCHA) and interval (i.e., preoperative and 12 months), and their interaction on RMS error. There were no significant main effects of listening condition,
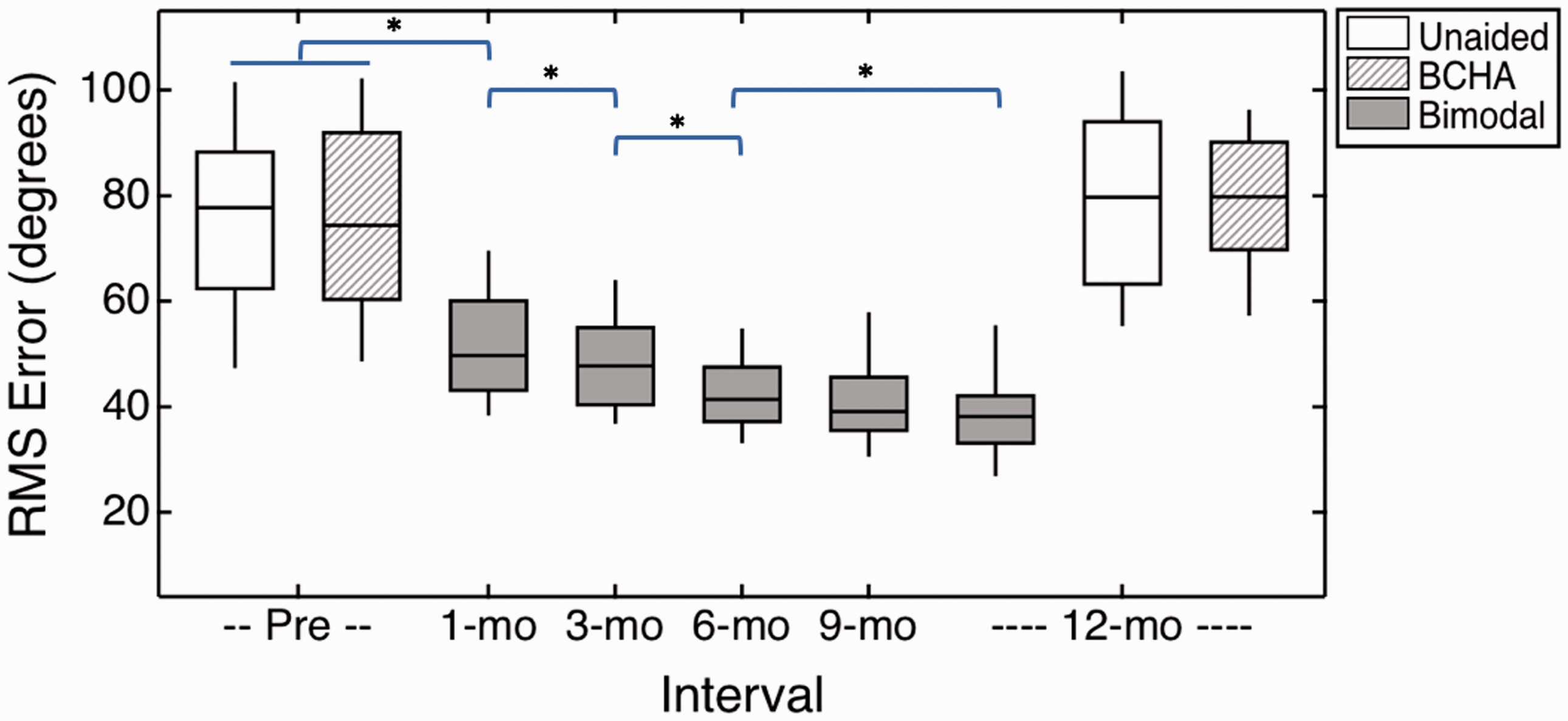
Results on the Localization Task Over the Study Period. Performance is reported as root-mean-squared (RMS) error, where a smaller value indicates better sound source identification. Box shading reflects the listening condition, which was unaided (open), BCHA (diagonal hatching), or bimodal (solid fill). Horizontal lines indicate median scores, boxes span the 25th to 75th percentiles, and vertical lines indicate the 10th to 90th percentiles.
Localization was then compared between the unaided condition at the preoperative interval and the bimodal condition at the 12-month interval, using a paired
Spatial Hearing: Masked Sentence Recognition
Figure 3 plots the masked sentence recognition results on the AzBio sentence test in percent correct. The left column includes the results from the 17 subjects assessed at the same SNR at the preoperative and post-activation intervals, with plotting conventions following those of Figure 2. Results are shown separately for the three spatial configurations (i.e., SoNo, SoNci, and SoNcontra). As observed in localization data, masked sentence recognition was similar in the unaided and BCHA conditions for both the preoperative and 12-month intervals. An LMM assessed main effects of listening condition (i.e., unaided and BCHA), SNR (i.e., 10, 5, or 0 dB SNR), interval (i.e., preoperative and 12-month), and spatial configuration (i.e., SoNo, SoNci, and SoNcontra), and the interaction between listening condition and interval. There were no significant main effects of listening condition,

Masked Sentence Recognition Measured With AzBio Sentence Test Over the Study Period. Panels on the left show the distribution of responses over time for the 17 subjects evaluated at the same SNR at all intervals, with listening condition defined in the legend. Boxplots follow the convention defined in Figure 2. Panels on the right show results for individual subjects; symbol shape and line type indicate the SNR, as defined in the legend. Results are reported in percent correct, with higher values indicating better performance.
Masked sentence recognition was then compared between the unaided condition at the preoperative interval and the bimodal condition at the 12-month interval, using an LMM that assessed the main effects of listening condition, SNR, and spatial configuration, and the interaction of listening condition and spatial configuration. There was a nonsignificant main effect of listening condition,
The right column in Figure 3 plots the individual masked sentence recognition scores for the 17 subjects tested with the same SNR at the preoperative and 12-month intervals. Open circles indicate the results for subjects assessed at 0 dB SNR, filled triangles for subjects assessed at 5 dB SNR, and filled squares for subjects assessed at 10 dB SNR. There are individual differences in all spatial configurations; however, the majority of subjects experience an improvement in masked sentence recognition in the SoNcontra configuration, which is the evidence of the head shadow effect in the bimodal condition. Paired
Determine the Magnitude of the Benefit and Time Course of Acclimatization for CI Recipients With AHL
Next, the magnitude of the benefit and time course of acclimatization for CI recipients with AHL was assessed during the first year of bimodal listening experience on measures of subjective benefit, word recognition, and spatial hearing.
Subjective Benefit
Figures 4 and 5 plot the results from the APHAB and SSQ questionnaires at the preoperative (open boxes) and post-activation (filled boxes) intervals, respectively. Better performance is indicated by a lower value on the APHAB (Figure 4) and a higher value on the SSQ (Figure 5). The calculation for the total score on the APHAB includes the responses from the communication subscales (i.e., Ease of Communication, Background Noise, and Reverberation) only, as described by Cox (1997). Overall, the subjects reported an improvement of benefit in the bimodal condition over preoperative experiences within the initial months of CI use.

Results From the Abbreviated Profile of Hearing Aid Benefit (APHAB) Across Test Intervals, Indicated on the Abscissa. Scores reflect the percentage of difficulty, ranging from 0% to 100%, with lower values indicating less perceived difficulty. Responses are stratified by the total score and by subscales (i.e., Ease of Communication, Background Noise, Reverberation, and Aversiveness). The total score is the mean across responses from the Ease of Communication, Background Noise, and Reverberation subscales. The open boxes indicate the results from the preoperative interval with the individual’s familiar listening condition (i.e., unaided, HA, BCHA, or Bi-CROS), and the filled boxes indicate the results from the post-activation intervals with the bimodal condition. Boxplots follow the convention defined in Figure 2.

Results From the Speech Spatial and Qualities of Hearing Scale Questionnaire Over the Study Period. Responses are stratified by the total score and for the individual subscales (Speech, Spatial, and Qualities of Hearing). Scores reflect the perceived ability, from 0 to 10, with higher values indicating better perceived abilities. Plotting conventions are the same as in Figure 4.
The individual LMMs for the APHAB and SSQ included main effects of interval and subscale, and the interaction of interval and subscale. Total scores on the questionnaires were not included in the analyses because the total score is derived from the subscale scores. When comparing responses over the study period, there were significant main effects of interval and subscale for both the APHAB,
Next, LMMs compared the responses across post-activation intervals to assess acclimatization. There were significant main effects of subscale for the APHAB,
Figure 6 plots the reported tinnitus severity in the CI ear over the study period. An LMM assessed the main effect of interval on the reported level of tinnitus severity. Subjects reported a significant reduction in tinnitus severity over the study period,

Reported Tinnitus Severity in the CI Ear Using the Tinnitus Handicap Inventory Over the Study Period. Scores reflect severity of tinnitus, from 0 to 100, with lower values indicating less severe tinnitus. The horizontal dashed line represents the tinnitus severity candidacy criterion. Potential candidates whose reported tinnitus exceeded this level were excluded. Plotting conventions are the same as Figures 4 and 5.
Word Recognition
Figure 7 plots the aided CNC word scores at the preoperative interval with an HA fit on the ear to be implanted (open boxes) and at the post-activation intervals with the CI alone (filled boxes). An LMM assessed the main effect of interval on word recognition over the study period. There was a significant difference in word recognition over the study period,
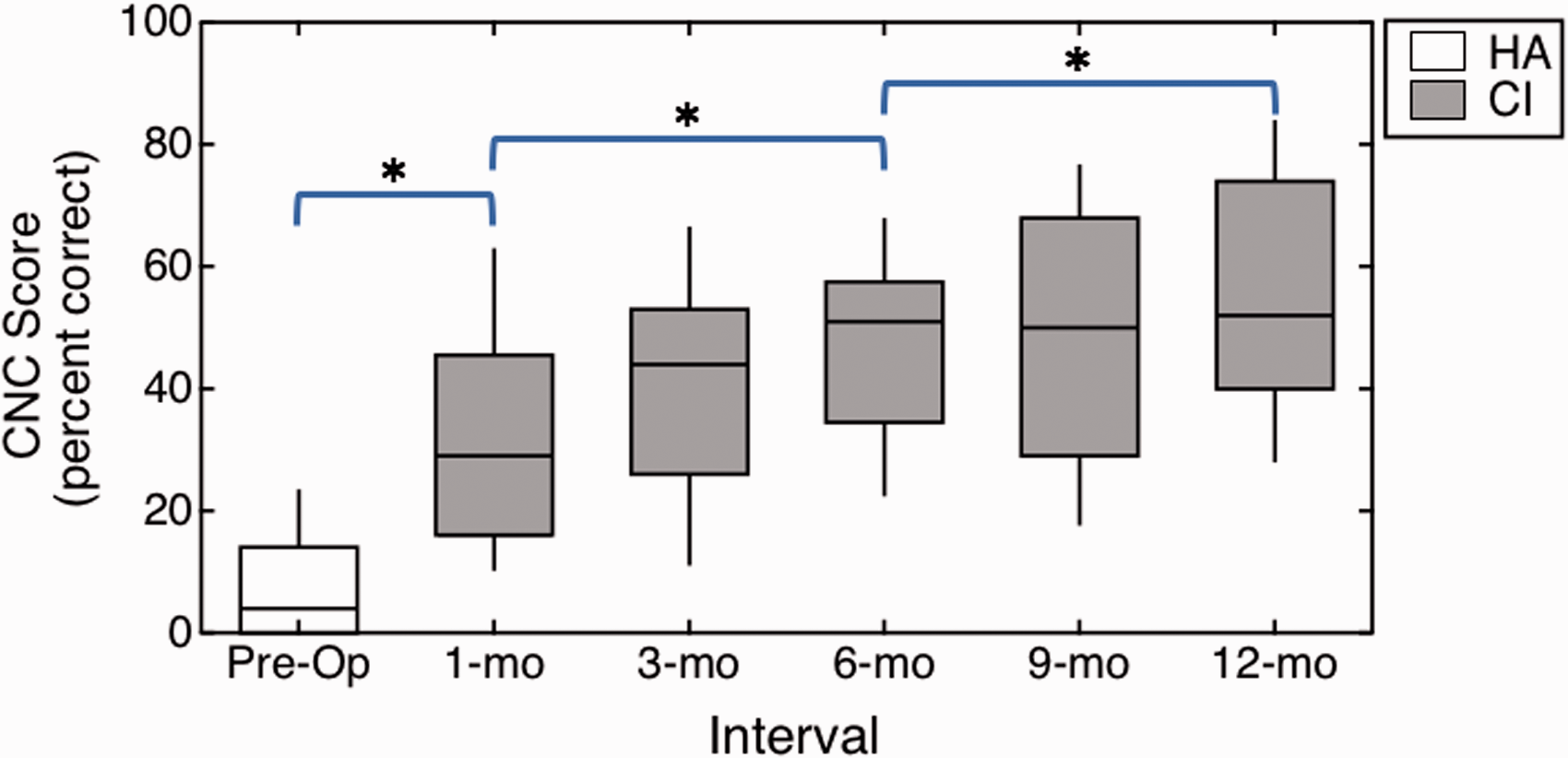
Aided CNC Word Recognition in Quiet for the CI Ear Over the Study Period. Masking noise was presented to the acoustic-hearing ear. Subjects listened with an HA at the preoperative interval and with their CI alone at the post-activation intervals. Results are reported in percent correct, with higher values indicating better performance. Boxplots follow the convention defined in Figure 2.
Spatial Hearing: Localization
As described earlier, Figure 2 plots the performance on the localization task over the study period. An LMM assessed the main effect of interval on RMS error. Subjects demonstrated a significant improvement in localization over the study period,
Spatial Hearing: Masked Sentence Recognition
As described earlier, Figure 3 plots the masked sentence recognition results on the AzBio sentence test over the study period, with results shown separately for each of the three spatial configurations (i.e., SoNo, SoNci, and SoNcontra).
As reported earlier, masked sentence recognition for the SoNo and SoNci spatial configurations was not significantly different for the unaided condition at the preoperative interval and bimodal condition at the 12-month post-activation interval, but performance did improve significantly in the SoNcontra configuration. In light of this finding, the SoNcontra configuration was selected to evaluate the magnitude of the benefit and acclimatization in the bimodal listening condition. An LMM assessed the main effects of interval and SNR on performance in the SoNcontra configuration over the study period. There was a significant main effect of interval,
A reduced model omitting the preoperative interval indicates that masked sentence recognition continued to significantly improve over the post-activation period in the bimodal condition,
Compare the Magnitude of the Benefit and Time Course of Acclimatization for CI Recipients With AHL to a Cohort of CI Recipients With UHL
The post-activation performance of CI users with AHL was then compared with published data for CI users with UHL who were tested following the same protocol. The analyses used previously to assess performance on measures of subjective benefit, word recognition, and spatial hearing were expanded to include cohort (i.e., AHL, UHL) as a main effect and the interaction of cohort with the other main effects on post-activation performance.
Subjective Benefit
Table 1 lists the results from the LMMs assessing the main effects of cohort, post-activation interval, and subscale and their interactions for the APHAB and SSQ. Briefly, there were nonsignificant main effects of cohort on the APHAB,
Results From the Linear Mixed Models Assessing the Main Effects of Cohort, Post-activation Interval, and Subscale and Their Interactions on Subjective Benefit Reported on the Abbreviated Profile of Hearing Aid Benefit (APHAB) and the Speech, Spatial, and Qualities of Hearing Scale (SSQ).

Word Recognition
An LMM assessed the main effects of interval and cohort, and their interaction on word recognition over the post-activation period. For word recognition in quiet in the CI ear, there was a significant main effect of interval,
Spatial Hearing: Localization
For localization performance, the LMM found significant main effects of cohort,
Spatial Hearing: Masked Sentence Recognition
An LMM assessed the main effects of cohort, interval, SNR, and spatial configuration, and the two-way and three-way interactions of cohort, interval, and spatial configuration on masked sentence recognition across post-activation intervals, with results listed in Table 2. As a reminder, the SNR used over the post-activation intervals for the AHL cohort was 10 dB SNR for 1 subject, 5 dB SNR for 3 subjects, and 0 dB SNR for 16 subjects; all subjects with UHL (
Results From the Linear Mixed Model Assessing the Main Effects of Cohort, Post-activation Interval, Signal-to-Noise Ratio (SNR), and Masker Condition, and Two-Way and Three-Way Interactions of Cohort, Interval, and Masker Condition on Masked Sentence Recognition.

Performance for the Three Spatial Configuration (i.e., SoNo, SoNci, and SoNcontra) on the Masked Sentence Recognition Task at the 1-Month and 12-Month Intervals.

aOne subject was withdrawn prior to the 12-month interval.
Spatial Release From Masking
An exploratory analysis of SRM for subjects with AHL or UHL was conducted to gain insight into the possible effects related to hearing sensitivity in the acoustic-hearing ear. SRM is typically computed as the difference between speech recognition scores when the target and masker are colocated when compared with when they are spatially separated; in this case, SRM was calculated separately for the SoNci and SoNcontra spatial configurations. Figure 8 shows the SRM for masked sentence recognition at the 12-month interval; subjects with AHL were tested in the bimodal condition and subjects with UHL listened with the CI and their acoustic-hearing ear. Results are plotted as a function of low-frequency PTA (LFPTA; 125, 250, and 500 Hz); PTA (500, 1000, and 2000 Hz); and high-frequency PTA (HFPTA; 4000, 6000, and 8000 Hz) in the acoustic-hearing ear. The horizontal dashed line indicates no change in percent correct with the introduction of spatial separation; positive values indicate reduced masking (i.e., SRM), and negative values indicate increased masking. It appears that better LFPTA, PTA, and HFPTA may be associated with greater SRM for the SoNci spatial configuration, but this trend is not evident in the SoNcontra spatial configuration.

Spatial Release From Masking for the AzBio Sentences in a 10-Talker Masker (0 dB SNR) for the Subjects With AHL Listening With the Bimodal Condition (Closed Squares) and the Previously Reported Subjects With UHL Listening With the CI Plus Acoustic-Hearing Ear Condition (Open Circles; Buss et al., 2018). Results are presented as the difference in performance in the colocated configuration (S0N0) and the spatially separated configurations (SoNci and SoNcontra). The left column plots the results from SoNci spatial configuration, and the right column plots the results from the SoNcontra spatial configuration. Results are stratified by LFPTA (top row), PTA (middle row), and HFPTA (bottom row). The horizontal dashed line at 0% differentiates those who benefited from spatially separated babble (positive values) and those who did not (negative values).
While the differences in hearing sensitivity in the acoustic-hearing ear were expected between the AHL and UHL cohorts due to the inclusion criterion, the cohorts also differed significantly in age at implantation,

Spatial Release From Masking for the AzBio Sentences in a 10-Talker Masker (0 dB SNR) for Subjects With AHL Tested in the Bimodal Listening Condition (Closed Squares) and Previously Reported Subjects With UHL Listening With Their CI and Acoustic-Hearing Ear (Open Circles; Buss et al., 2018) Plotted by Age at Implantation. Results are presented as the difference in performance in the colocated configuration (SoNo) and the spatially separated configurations (SoNci and SoNcontra). The top column plots the results from SoNci spatial configuration, and the bottom column plots the results from the SoNcontra spatial configuration. The horizontal dotted line at 0% differentiates those who benefited from spatially separated babble (positive values) and those who did not (negative values).
Discussion
The present findings contribute to the growing evidence supporting cochlear implantation as an effective treatment option for patients with moderate-to-profound hearing loss in one ear and normal-to-moderate hearing sensitivity in the contralateral ear. Generally, performance in the unaided condition was similar to performance in the BCHA condition and poorer compared with performance in the bimodal condition. Subjects with AHL experienced early, significant subjective benefit, better word recognition, and improved spatial hearing performance with the CI when compared with preoperative abilities. While the early, significant subjective benefit remained stable after the 1-month interval, performance on the word recognition and some spatial hearing tasks continued to improve with further listening experience. On the localization task, the absolute localization acuity in the bimodal condition differed significantly between the subjects with AHL tested here and published data on CI recipients with UHL. On the masked sentence recognition task, the magnitude of the benefit observed in the post-activation period differed significantly between subjects with AHL and subjects with UHL. The time course of acclimatization did not significantly differ between cohorts on the subjective benefit, word recognition, and spatial hearing tasks.
Performance on the spatial hearing tasks did not differ significantly between the unaided and BCHA listening conditions, which corroborates the previous data demonstrating that subjects with AHL or UHL do not experience a significant improvement in spatial hearing performance when using prostheses that route the signal from the poorer hearing ear to the better hearing ear (Agterberg et al., 2019; Mertens et al., 2017). The present data also indicate similar performance unaided and with the BCHA before and after 12 months of CI use in subjects with AHL. This indicates that CI recipients may expect similar performance with these alternative treatment options preoperatively and postoperatively, in the event that they become non-users of the CI after implantation. Non-users is reportedly rare in CI recipients with AHL or UHL (Távora-Vieira et al., 2020; Zeitler et al., 2019), but it can occur and may be due to lack of perceived and/or demonstrated benefit with the CI (Távora-Vieira et al., 2020). Although all subjects in the present study continued to use their CI during the study period and demonstrated a significant benefit in the bimodal condition when compared with the unaided condition, it is possible that inclusion criteria for this clinical trial may have excluded candidates with risk factors for non-users (e.g., prolonged duration of moderate-to-profound hearing loss).
Comparisons between data for CI recipients with AHL and published data on CI recipients with UHL demonstrate similar benefit on the subjective measures, with significant improvements demonstrated by the 1-month interval and maintained over the post-activation period (Dillon et al., 2017b). The present report assessed benefit on the SSQ using the traditional Speech Hearing, Spatial Hearing, and Qualities of Hearing subscales. Use of the SSQ pragmatic subscales (described by Gatehouse & Akeroyd, 2006) may be more sensitive to the differences in perceived spatial hearing abilities of CI recipients with AHL versus UHL (Thompson et al., 2020). Newer versions of the SSQ may be more sensitive to the influence of auditory asymmetry (Moulin et al., 2019). For tinnitus severity, the early suppression of tinnitus that is maintained over the study period when listening with the CI corroborates the findings from previous studies of CI recipients with AHL and UHL (Firszt et al., 2012b; Galvin et al., 2019; Mertens et al., 2016; Punte et al., 2011). The tinnitus severity in one subject in the present cohort fluctuated over the post-activation period, which he associated with stress level and treatments for other health issues. While the majority of subjects reported a significant reduction in tinnitus severity, CI candidates with AHL and UHL should be counseled that this does not occur in all cases.
The magnitude of the benefit and time course of acclimatization for word recognition with the CI alone was similar for CI recipients with AHL and UHL (Buss et al., 2018). Buss et al. (2018) reported similar mean word recognition scores for CI recipients with UHL and conventional CI recipients, all of whom were implanted with a 31-mm electrode array (Buchman et al., 2014). The early significant improvements observed in these AHL, UHL, and conventional CI cohorts may be due to the close alignment of the CI default frequency filters with the cochlear place frequency in recipients of long electrode arrays when compared with recipients of shorter arrays (Canfarotta et al., 2020). These data also corroborate the tentative conclusion of Buss et al. (2018) that the hearing sensitivity in the acoustic-hearing ear does not influence word recognition growth with the CI alone.
In contrast to word recognition in quiet with the CI alone, the magnitude of the benefit for spatial hearing tasks differed between the AHL and UHL cohorts. For instance, subjects with UHL experienced better localization when compared with the subjects with AHL, whose performance remained poorer though continued to improve by the study endpoint (12-month interval). This pattern was similar for performance on the masked sentence recognition task. The mechanism influencing these differences in the magnitude of the benefit is of interest to better understand the contributions of patient and device variables in this specific CI recipient population. Consistent with data presented here, poorer hearing sensitivity in the acoustic-hearing ear has been shown to be negatively associated with masked sentence recognition for CI recipients with AHL (Firszt et al., 2018). While CI recipients with UHL appear to experience greater masking release in the SoNci spatial configuration compared with those with AHL, this trend is not evident in the SoNcontra spatial configuration. For the SoNci spatial configuration, listeners rely on the audibility in the acoustic-hearing ear; therefore, better hearing sensitivity supports better performance. For the SoNcontra spatial configuration, listeners rely on the audibility from the CI; hearing sensitivity in the acoustic-hearing ear appears to have little or no influence on performance in this configuration.
Another potential explanation for differences in the magnitude of the benefit on the spatial hearing tasks between the AHL and UHL cohorts is their age at implantation, with a greater mean age observed in the AHL cohort. Advanced age at implantation is associated with poorer speech recognition within the initial months of device use in conventional CI recipients (Chatelin et al., 2004; Waltzman et al., 1995) and in CI recipients with AHL (Firszt et al., 2018). Unfortunately, differentiating effects related to hearing sensitivity in the acoustic-hearing ear and age at implantation is not possible in the present dataset because these effects are confounded in the population; older subjects presented with more hearing loss in the acoustic-hearing ear, which is representative of the older patient population (Pichora-Fuller & Souza, 2003). Another consideration to keep in mind is that 12-month data may not represent asymptotic performance. Older conventional CI recipients demonstrate significant improvements in speech recognition out to 5 years of device use (Dillon et al., 2013), and it is possible that older CI recipients with AHL may require prolonged device use to acclimate to the CI and bimodal stimulation. Long-term (>12 months) data collection is ongoing to assess the magnitude of the benefit and time course of acclimatization in CI recipients with AHL and UHL after prolonged CI use.
Other Variables for Consideration
The present study was not powered to assess the influence of all the variables that may influence the magnitude of the benefit and time course of acclimatization with the CI for listeners with AHL, such as the magnitude of frequency-to-place mismatch, duration of daily CI use, and CI and HA fitting methods and settings.
Frequency-to-Place Mismatch
A variable that warrants consideration when assessing the magnitude of benefit and rate of acclimatization is the frequency-to-place mismatch, which is the discrepancy between the CI frequency filters and the cochlear place frequency. Wide variability in frequency-to-place mismatch has been demonstrated even among CI recipients of the same electrode array who are all listening with default frequency filters (Canfarotta et al., 2020; Landsberger et al., 2015), and frequency-to-place mismatch correlates with initial speech recognition with the CI (Canfarotta et al., 2020). Further, CI recipients vary in their ability to acclimate to frequency-to-place mismatches (Reiss et al., 2011, 2015), which could influence the fusion between ears (Reiss et al., 2016) and thus spatial hearing performance. Frequency-to-place mismatch can also negatively influence performance on spatial hearing tasks by introducing interaural frequency mismatches; these effects have been observed in bilateral CI recipients and normal-hearing subjects listening to bilateral CI simulations (Goupell et al., 2013; Kan et al., 2013; Xu et al., 2020). Ongoing investigations are assessing the effectiveness of matching CI frequency filters to the cochlear place frequency on tasks of speech recognition with the CI alone and spatial hearing in the bimodal listening condition.
Duration of Daily Device Use
The magnitude of the benefit and time course of acclimatization may also be influenced by the duration of daily CI use. Recent investigations using data from the datalogging feature available in newer CI processors demonstrate a positive association between the number of hours of daily CI use and speech recognition in adult and pediatric CI recipients (Easwar et al., 2018; Gagnon et al., 2020; Holder et al., 2020; Schvartz-Leyzac et al., 2019). Dillon et al. (2017a) speculated that limited CI use (<8 hr/day) may have contributed to the poorer localization results observed for one subject with UHL at the 1-month interval; that subject who reported more consistent daily CI use at subsequent intervals experienced improved performance. The present AHL cohort listened with a CI processor that provided datalogging regarding duration of daily use, revealing variability across subjects. Although all but one of the CI recipients with UHL reported wearing their CI 8 or more hr per day, datalogging indicates that only 10 out of 20 CI recipients with AHL wore their device this much. One possibility is that less consistent daily CI use contributed to the poorer performance observed in the AHL cohort on the spatial hearing tasks when compared with the UHL cohort. This possibility should be interpreted cautiously, however, as no datalogging records are available to substantiate the self-reported daily use in subjects with UHL.
CI and HA Fitting Methods and Settings
The methods used to individualize the fitting of the CI and HA devices may have also influenced the initial performance observed in the present AHL cohort. Each subject underwent behavioral mapping of threshold and comfort levels, loudness balancing between channels, and loudness balancing with the acoustic-hearing ear as part of fitting of the CI. Individualizing map parameters, such as threshold and comfort levels on each channel, supports good speech recognition with the CI (Dawson et al., 1997; Skinner, 2003). The HA on the acoustic-hearing ear was fit to NAL-NL1 targets using real-ear measures, and the output was verified at each interval. Individualized fitting of the HA is recommended for optimal bimodal performance (Ching et al., 2004; Potts et al., 2009). The fitting practices of bimodal stimulation influence listener performance (Potts et al., 2009). Considering this, the present results may have been influenced by the methods used to individualize the fitting of the CI and HA devices.
Differences in the signal processing applied by CI and HA devices may have also influenced the observed outcomes of the AHL cohort. For example, amplitude compression makes it possible to present the full range of audible cues into a compressed dynamic range, but it can also introduce signal distortion and mismatches in loudness growth across ears. Differences in compression across ears have been shown to hurt performance on spatial hearing tasks for listeners with normal hearing and hearing loss (Moore et al., 2010; Wiggins & Seeber, 2012). Simulations of bimodal hearing suggest that disruptive effects of mismatched compression across ears may also occur for CI users (Wess & Bernstein, 2019). Further, envelope distortion and differences in processing times of the CI and HA devices can also degrade performance in bimodal listeners (Wess & Bernstein, 2019; Zirn et al., 2019). Investigations of device setting manipulations are needed to support the clinical decision making to optimize outcomes in CI recipients with AHL and UHL.
Conclusions
The present work provides evidence of significant subjective benefit, better word recognition, and improved spatial hearing performance for subjects with AHL within the initial weeks of CI use when compared with preoperative performance. Performance at the 12-month interval with bimodal stimulation exceeded performance in an unaided or BCHA condition, supporting cochlear implantation as an effective treatment option for AHL. The magnitude of the benefit and time course of acclimatization was similar on measures of subjective benefit and word recognition with the CI alone between subjects with AHL and UHL. Localization accuracy was poorer in subjects with AHL than subjects with UHL. Although speech recognition with the masker offset contralateral to the CI improved over the post-activation intervals for subjects with AHL, this benefit was smaller than that observed for subjects with UHL. In contrast to the UHL cohort, subjects with AHL did not continue to improve in the post-activation period in the other two spatial configurations (i.e., masker colocated in front or offset to the side of the CI). Ongoing work is assessing the influence of contralateral hearing sensitivity, age at implantation, duration of daily device use, and CI frequency filters on initial and long-term performance in this patient population. The present data substantiate the expansion of candidacy criteria for cochlear implantation to include patients with normal-to-moderate hearing sensitivity in the contralateral ear, and preliminary data highlight the need to better understand the mechanisms that support early, significant improvements in CI recipients.
Footnotes
Acknowledgments
The authors acknowledge the regulatory assistance of the North Carolina Translational and Clinical Sciences Institute, which is supported by the National Center for Advancing Translational Sciences, National Institutes of Health, through Grant Award Number UL1TR002489. The authors would also like to thank Joshua Light for his contributions reviewing data reported here.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E. R. K. serves on the Audiology Advisory Board, and K. D. B. serves on the Surgical Advisory Board for MED-EL Corporation. H. C. P. is a consultant for MED-EL Corporation. B. P. O. is on the Surgical Advisory Board for Advanced Bionics.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: M. T. D., M. A. R., and M. E. R. are supported by a research grant from MED-EL Corporation provided to their university.
