Abstract
Cochlear-implant (CI) users rely heavily on temporal envelope cues to understand speech. Temporal processing abilities may decline with advancing age in adult CI users. This study investigated the effect of age on the ability to discriminate changes in pulse rate. Twenty CI users aged 23 to 80 years participated in a rate discrimination task. They attempted to discriminate a 35% rate increase from baseline rates of 100, 200, 300, 400, or 500 pulses per second. The stimuli were electrical pulse trains delivered to a single electrode via direct stimulation to an apical (Electrode 20), a middle (Electrode 12), or a basal location (Electrode 4). Electrically evoked compound action potential amplitude growth functions were recorded at each of those electrodes as an estimate of peripheral neural survival. Results showed that temporal pulse rate discrimination performance declined with advancing age at higher stimulation rates (e.g., 500 pulses per second) when compared with lower rates. The age-related changes in temporal pulse rate discrimination at higher stimulation rates persisted after statistical analysis to account for the estimated peripheral contributions from electrically evoked compound action potential amplitude growth functions. These results indicate the potential contributions of central factors to the limitations in temporal pulse rate discrimination ability associated with aging in CI users.
Introduction
Temporal processing, related to the perception of pitch, is important for music appreciation, speech understanding, and the perceptual segregation of sound sources (Oxenham, 2008). Cochlear implants (CIs) are poor at conveying pitch spectrally because electrical current spread limits frequency selectively and because CIs do not represent the harmonic structure of a sound (e.g., Azadpour & McKay, 2012; Goupell et al., 2008). CI sound processing strategies typically remove the temporal fine structure of a signal while preserving the slow-varying temporal envelope (Moon & Hong, 2014); thus, CI users must rely on temporal envelope patterns of an input signal for perceptual tasks (Feng & Oxenham, 2018; Goupell et al., 2017; Loizou, 2006; Won et al., 2015). The ability to use temporal pitch cues, however, may be limited in CI users (e.g., Carlyon, Lynch, et al., 2010).
CI listeners’ ability to utilize temporal information in simpler, non-speech tasks, has been previously measured via temporal rate discrimination, a perceptual task that involves discriminating small changes in electrical pulse rate presented to a single electrode (e.g., Gaudrain et al., 2017). Discrimination performance drops rapidly as the standard rate increases beyond 200 to 300 pulses per second (pps) in most CI users (McDermott & McKay, 1997; Townshend et al., 1987; Zeng, 2002; Zhou et al., 2019). This finding has led to the argument of an upper limit of temporal pulse rate discrimination at about 300 pps for CI users. There is also a minority of CI users who can detect changes from substantially higher rates of 500, 900, or even 3,520 pps (Goldsworthy & Shannon, 2014; Kong & Carlyon, 2010; Kong et al., 2009). These findings, particularly the variability in performance and the upper limit of pulse rate discrimination across CI users, raise questions about the mechanisms underlying the limited ability of CI users to discriminate temporal rate changes.
Multiple factors may constrain temporal pulse rate discrimination performance in CI users. Peripheral factors such as the loss of synchrony from auditory nerve fibers, possibly due to poor neural survival (Carlyon & Deeks, 2015), may play a role. Central auditory factors beyond the level of the auditory nerve may also account for some of the limitation. For example, selective activation of neurons in the central nucleus of the inferior colliculus (Middlebrooks & Snyder, 2010) and the auditory cortex (Kirby & Middlebrooks, 2012) are important for accurate representation of temporal aspects of sound, such as electrical pulse rate. Hearing impairments, however, may result in diminished encoding of temporal features at subcortical and cortical levels (Anderson et al., 2013, 2020; Walton et al., 1998).
The number of older adults who use a CI to restore hearing and speech understanding is rapidly increasing (Lin et al., 2012). Natural aging processes negatively affect peripheral neural health (e.g., the loss of spiral ganglion cells and auditory nerve fibers; Sergeyenko et al., 2013) and central auditory processing abilities (Clinard et al., 2010; Gaskins et al., 2019), which may contribute to declines in temporal processing in CI users (Committee on Hearing, Bioacoustics, and Biomechanics, 1988; Gordon-Salant et al., 2011; Jin et al., 2014). Indeed, age-related deficits in auditory temporal processing have been documented in older CI listeners on measures of word identification based on silence duration cues (Xie et al., 2019), amplitude modulation detection (Shader et al., 2020a), gap detection (Shader et al., 2020b), and voice emotion recognition (Chatterjee et al., 2015; Christensen et al., 2019). However, it remains unclear whether aging impacts the ability to discriminate pulse rate changes in CI users.
Age-related deficits in temporal processing for CI users likely involve both peripheral and central mechanisms. However, their relative contributions are still debated (Bao et al., 2020; Humes et al., 2012). CIs allow us to mostly bypass cochlear effects (e.g., the distortion introduced by damaged or missing inner and outer hair cells, reduced responses due to age-related reductions in the endocochlear potential) and to characterize some of the peripheral contributions with relative ease (Turner et al., 2010). For example, the electrically evoked compound action potential (ECAP) has been proposed as a promising measure of the quality of peripheral functioning and the electrode-to-neural interface in CI users (DeVries et al., 2016; Schvartz-Leyzac et al., 2020; Smith & Simmons, 1983). Specifically, the amplitude growth function (AGF) of the ECAP, the change in ECAP amplitude as a function of current level, has been used as an indicator of peripheral neural survival (Pfingst et al., 2015; Scheperle, 2017). Steeper ECAP AGFs are expected near regions of good peripheral neural survival due to the greater spread of current in response to higher stimulation levels (Goldwyn et al., 2010; Hall, 1990; Pfingst et al., 2015, 2017; Ramekers et al., 2014, 2015; Scheperle, 2017; Smith & Simmons, 1983). If a region along the electrode array has good neural survival, then more neurons will be recruited in response to stimulation compared with a region with poor neural survival. Steeper ECAP AGFs suggest that a greater number of neurons are being activated as current level increases compared with shallower ECAP AGFs (Goldwyn et al., 2010; Hall, 1990; Pfingst et al., 2015, 2017; Ramekers et al., 2014, 2015; Scheperle, 2017; Smith & Simmons, 1983).
The main goal of this study was to determine the extent to which chronological age affects temporal pulse rate discrimination abilities in adult CI users. We hypothesized that temporal rate discrimination would be negatively affected by aging in CI users because of poorer peripheral and central processing. A second goal of this study was to characterize peripheral contributions with ECAP AGFs in relation to age-related changes in temporal pulse rate discrimination in CI users. We hypothesized that steeper ECAP AGFs (i.e., possibly related to better peripheral encoding) would predict better temporal pulse rate discrimination performance.
Methods
Overview
Participants completed testing monaurally in the implanted ear for unilaterally implanted CI users and in the self-reported preferred ear for bilaterally implanted CI users. First, each participant was programmed with a Cochlear Nucleus 6 research processor to obtain threshold, most comfortable loudness level (MCL), and uncomfortable loudness level (UCL) for three test electrodes (4, 12, and 20) at each of five baseline rates (100, 200, 300, 400, and 500 pps). Then, ECAP AGFs were recorded from individual participants at each of the same three electrodes to estimate peripheral neural survival near these electrodes. Finally, participants completed a temporal pulse rate discrimination task. Participants completed all procedures in a single session for approximately 6 h. They were encouraged to take frequent breaks and were compensated for their time. Later, we provide a detailed description of participants and the tasks: level setting, ECAP recording, loudness balancing, and temporal pulse rate discrimination.
Participants
Twenty adult CI users aged 23 to 80 years (mean = 50.4 ± 19.4 years) participated in this study. At least two participants were included within each age decade. Participant demographics are provided in Table 1. All participants were native speakers of English. They had Cochlear-brand Nucleus electrode arrays to minimize differences across devices. For this study, duration of deafness (DoD) was determined by subtracting the patient’s chronological age at the time of testing by the chronological age at which they were identified with severe-to-profound hearing loss (Lundin et al., 2014). The age at which the participant was identified with a severe-to-profound degree of hearing loss was self-reported as part of an intake questionnaire. In cases where the answer to this question was unknown, participants were asked to report the age at which time their hearing loss progressed to such a degree that hearing aids no longer benefited them.
Participant Demographics.

Note. DoD = duration of deafness; MoCA = Montreal Cognitive Assessment.
Written informed consent was obtained from all participants. All materials and procedures were approved by the Institutional Review Board at the University of Maryland.
Cognitive Screening
Participants completed the Montreal Cognitive Assessment (MoCA) screener (Nasreddine et al., 2005) to exclude those at risk for mild cognitive impairment (≤22 of 30 points; Cecato et al., 2016). All participants passed the MoCA screening except for one participant (S4: MoCA = 19).
We included the MoCA primarily to screen for mild cognitive impairment in our sample of older participants. Surprisingly, one younger participant (S4) performed poorly on this screening tool. We decided to include this participant in the experiment due to the need to include younger CI users in the study and the challenge of recruiting CI users within a specific age bracket. The MoCA is normed for individuals aged 65 years and older who are typically thought to be at a greater risk of cognitive decline; therefore, the validity of the MoCA as a screening measure for participants below 65 years of age is unclear. Participant S4 is not suspected to have Alzheimer’s disease or other age-related dementias due to their chronological age of 33 years.
Level Setting
Equipment
The task and response collection were controlled by custom scripts in MATLAB (MathWorks, Natick, MA). Stimuli were presented through the research processor using monaural direct stimulation via the Nucleus Interface Communicator (NIC version 4) software provided by Cochlear Ltd (Sydney, Australia).
Procedure
We adopted an ascending method of adjustment (Landsberger & McKay, 2005) to program the research processor in individual participants. We obtained threshold, MCL, and UCL for the three test electrodes (4, 12, and 20) at each of the five baseline rates (100, 200, 300, 400, and 500 pps). To follow the best practices for direct stimulation in CI research, the researcher presented initial stimulation below the level of audibility and gradually increased the current to minimize discomfort and avoid the possibility of biological damage (Litovsky et al., 2017). During threshold assessment, this procedure involved increasing the stimulation level of a constant-amplitude pulse train until the stimulus is perceived. The stimulation level was then steadily increased until the participant indicated MCL and subsequently UCL. Participants pointed to a loudness scaling chart to indicate the perceived level to the tester. The subjective levels of the loudness scale included Inaudible, Very Quiet, Quiet, Comfortable, Loud, and Too Loud. For the purposes of this study, UCL was defined as the current level that was just below the perceived level of Too Loud on the loudness scaling chart. The level setting procedure was conducted independently on each combination of electrode and baseline rate. Electrodes 4, 12, and 20 were chosen to span the length of the internal electrode array, thus capturing a basal, middle, and apical electrode location. The values of threshold, MCL, and UCL were used for later loudness balancing and temporal pulse rate discrimination tasks.
ECAP AGFs
Equipment
ECAPs were measured using Cochlear N5 research processors through the Custom Sound EP software version 4.4 (Cochlear Ltd).
Stimuli
ECAPs were measured in response to biphasic stimulus pulses with a phase duration of 25 μs, interphase gap (IPG) of 7 μs, pulse duration of 500 ms, and pulse rate of 80 pps as described in McKay et al. (2013). In the case that ECAPs could not be elicited with the specified parameters, the following configurations were attempted in the following order: increasing phase duration to 50 μs, increasing IPG to 30 µs, and reducing pulse rate to 30 pps. A longer phase duration of 50 μs is typically utilized to offset voltage compliance limitations (He, Teagle, et al., 2017). Longer IPGs may result in lower ECAP thresholds and thus may allow AGFs to be measured at lower stimulation levels compared with shorter IPGs (Hughes et al., 2018). A slower pulse rate of 30 pps was attempted when ECAP thresholds could not be obtained prior to reaching the participant’s tolerable upper limit of their dynamic range, as faster ECAP stimulation rates have been found to produce higher ECAP thresholds (Hughes, 2012). Six electrodes’ ECAP recordings across four participants (S4, S9, S12, and S15; 6 of a total of 60 ECAP recordings = 20 Participants × 3 Electrodes; 10% of total recordings) were obtained with different stimulation parameters (phase duration and IPG) than the specified default. An ECAP AGF could not be obtained on a total of seven electrodes (12% of total recordings) across six participants (S9, S11, S12, S17, S18, and S19). In these instances, stimulation was terminated prior to obtaining the minimum required number of data points due to loudness intolerance or compliance limitations.
Procedure
ECAPs were recorded on all test electrodes in monopolar mode in order to obtain AGFs. Stimulus presentation level started at 100 current units (CU) and increased in 5 CU steps. Participants were instructed to inform the tester when the stimulus became uncomfortably loud, at which point stimulation was terminated immediately.
Data Analysis
Peak-to-peak amplitudes were automatically derived from the linear portion of each waveform through the software and markers were automatically set at the most positive and negative peak beyond the noise floor (McKay et al., 2013). Erroneous markings of peak-to-peak ECAP amplitudes due to artifacts were manually discarded prior to computing AGFs. A minimum of two non-zero ECAP amplitudes were required to fit the slope. Prior to calculating ECAP AGFs, input logarithmic CU values were converted into a linear charge scale (nC). The slope of the input-output function was calculated through the Statistical Package for the Social Sciences (IBM) software for each test electrode. We calculated R2 for each ECAP AGF to assess goodness of fit post hoc. The R2 values ranged from .72 to .99 for our data. In cases that the ECAP N1-P1 amplitude slope started to plateau as the input level increased, points along the function after the plateau were removed so that a linear fit would be more appropriate. The linear slope function was used for all electrodes, including linear and nonlinear growth functions. The input-output functions were only repeated once, as ECAP slope has not been shown to significantly change across multiple runs within a session (Hughes & Stille, 2010). Figure 1 includes examples of acceptable waveforms (Panel A), an example of a linear curve fitting (Panel B), as well as a comparison of ECAP AGF slope between older and younger participants (Panel C).

Comparison of ECAP AGF Between Age Groups. A: Examples of acceptable ECAP waveforms recorded from an individual electrode. B: An example of a linear curve fitting used to derive ECAP AGF slope from an individual electrode using ECAP amplitude values such as those illustrated in (A). C: Boxplots comparing the ECAP AGF slope values between older and younger CI users. Younger was defined as below the median age (53.5 years); older was defined as at the median age or above. The boxes represent the first to third quartile. The line within the box represents the median for each group. The filled square and triangle within the boxes denote the mean for each group. The whiskers denote 1.5 times the interquartile range. Outliers outside of this range are not represented in the boxplots. ECAP AGF = electrically evoked compound action potential amplitude growth function.
Loudness Balancing
Equipment
The task and response collection were controlled by custom scripts in MATLAB. Stimuli were presented through a Nucleus 6 research processor programmed earlier to participants using monaural direct stimulation via the NIC version 4 software provided by Cochlear Ltd.
Stimuli
Stimuli consisted of 300-ms biphasic pulse trains with individual pulses of 25 µs/phase and an IPG of 8 µs. The reference sound was fixed at the participant’s MCL for Electrode 12 at 100 pps. The test sound was randomly selected from a pool of five rates (100–500 pps), three electrodes (4, 12, or 20), and two rate changes (0% or 35%), resulting in 29 test sounds (excluding the reference sound itself). Those stimuli including the reference sound, after loudness balancing, were used in the following temporal pulse rate discrimination task. The decision to use a fixed rate change of 35% higher than baseline was based on data reported by Zeng (2002), who found that average frequency difference limens for CI users increases monotonically with increasing baseline rate. Kong et al. (2009) suggested that a fixed 35% rate change would capture an above-chance performance level for pulse rate discrimination within the range of 100 to 500 pps based on the average frequency difference limens reported in the CI literature.
Procedure
Loudness balancing was achieved using the two-interval two-alternative forced-choice adaptive procedure (Landsberger & McKay, 2005). The 29 test sounds were loudness balanced to the reference sound separately. For each stimulus pair, participants heard a test sound and a reference sound. The order of the two sounds was randomized per trial. Their task was to judge whether the first or the second sound was louder. If the participant indicated that the reference sound was louder, the level of the test sound would increase. If the participant indicated that the test sound was louder, the level of the test sound would decrease. Initially, the test sound was set at a level randomly selected from 50% of the dynamic range of the test electrode ±5 CU. Each loudness procedure terminated after 10 reversals. For the first reversal, the step size was set at 4 CU. After the first reversal, the step size decreased to 2 CU. The level for matched loudness was calculated as the arithmetic mean of the levels for all the trials across the 6th to 10th reversal. In the case that the participant reached UCL before 10 reversals, MCL was used as the matched level. The MCL was used as the matched level for 5% of the total pulse rate discrimination data (15 of 300 trials across all participants). Participants initiated each loudness balancing procedure by clicking a button on the screen. The loudness balancing procedure was completed over the course of approximately 2h.
Pulse Rate Discrimination Task
Equipment
The task and response collection were controlled by custom scripts in MATLAB. The equipment for stimulus delivery was identical to the above loudness balancing task.
Stimuli
Stimuli for the current task were the loudness-balanced stimuli from the above described loudness balancing task. These included 30 stimuli specified by the following three parameters: five baseline rates (100, 200, 300, 400, or 500 pps), three electrodes (4, 12, or 20), and two rate changes (0% or 35%).
Procedure
Pulse rate discrimination performance was measured with a three-interval two-alternative forced-choice task using a “mixed-block (pp. 1652)” procedure as described in Kong et al. (2009). Each block included stimuli of all five baseline rates at all three electrodes. Each trial consisted of three stimuli with the same baseline rate presented at the same electrode but with different rate changes: 0% as the standard and 35% as the target. The first stimulus was always the standard stimulus. In half of the trials, the second stimulus was the standard and the third was the target. In the other half of the trials, the second stimulus was the target and the third was the standard. The task was to indicate (by clicking on the screen) whether the second or the third sound was different than the first (standard) stimulus. Participants initiated each trial by clicking a button on the screen. Each combination of baseline rate and electrode (15 combinations) was repeated 10 times, resulting in 150 trials in a block. A total of 10 blocks (100 trials for each combination of baseline rate and electrode) were administered to each participant. Trial order was randomized in each block in individual participants. No feedback was provided.
Statistical Analysis
A multi-level generalized linear (logistic) mixed-effects model, implemented with glmer function of the lme4 package (Bates et al., 2014) in R Studio version 1.2 (RStudio Team, 2018), was used to examine the effects of chronological age at time of testing, ECAP AGF slope, and baseline rate on pulse rate discrimination performance while controlling for the effects from DoD and chronological age at onset of deafness. Pulse rate discrimination performance was quantified as a dichotomous variable (correct or incorrect) for individual trials. Chronological age, ECAP AGF slope, DoD, and chronological age at onset of deafness were treated as continuous variables and were standardized (z-scores) before being entered into the models. We subsequently calculated the correlation matrix of these variables. The variables of chronological age at time of testing and chronological age at onset of deafness were highly intercorrelated (r = −.89). Thus, in an attempt to reduce multicollinearity, chronological age at onset of deafness was not included as a subject-level predictor. Age at onset of deafness was removed, rather than chronological age, because age at time of testing was the primary variable of interest in this study. Baseline rate and electrode location were recoded (Baseline rate: 100 pps = 1, 200 pps = 2, 300 pps = 3 [reference level], 400 pps = 4, 500 pps = 5; electrode location: Electrode 4 = −1, Electrode 12 = 0 [reference level], Electrode 20 = 1) and were treated as categorical variables. A model building approach was utilized following the procedure described by Hox et al. (2017).
The model building started with an intercept-only model. The random effects for the intercept-only model were structured to represent a two-level model in which the multiple electrode locations were nested within subject. This structure allowed electrode results to remain clustered within a participant and alerts the model that measurements obtained across electrode within a participant are not independent measures.
Then, baseline rate and electrode location were added as Level-1 predictors to the fixed effect structure. A Chi-squared significant test (α level = .05) was used to test whether these fixed-effect variables yielded a significant improvement in model fit. Next, the main effects and interactions for all Level-2 predictors (chronological age, ECAP AGF slope, and DoD) were added to the fixed effects. However, the model failed to converge after 2,000,000 iterations. We progressively dropped the Level-2 predictors that accounted for the least variance in the model. We further removed non-significant Level-2 predictors to reduce the risk of overfitting.
Then, random slope variation for the Level-1 predictors (baseline rate and electrode location) was added to the model. The model failed to converge when the random slope of the baseline rate was added and when the random slope of electrode location was added; thus, the random effects for the final model included the intercept variation for participants and electrodes nested within participants.
Finally, cross-level interactions (interactions between fixed Level-1 and Level-2 predictors) were added to the fixed effects structure. A Chi-squared test was performed to examine if those interactions significantly improved model fit. Non-significant predictors were removed to reduce the risk of overfitting. However, if an interaction was significant, the main effects and lower level interaction of the related predictors were kept in the model even if they were not significant. The simplest, best-fitting model is reported in the “Results” section.
Results
Figure 2 displays pulse rate discrimination accuracy data across the three test electrodes (4, 12, and 20) in individual listeners across the five baseline rates: 100, 200, 300, 400, and 500 pps. Table 2 displays the final model for pulse rate discrimination performance. The effect of rate was significant (p < .001) for all rate conditions compared with the reference condition (300 pps), demonstrating that pulse rate discrimination ability decreased (worsened) as stimulation rate increased. For example, participants were more likely to accurately discriminate changes in rate with a 100-pps compared with a 300-pps signal and were less likely to accurately discriminate changes in rate with a 500-pps compared with a 300-pps signal. There was also a significant effect of ECAP slope (p < .01), demonstrating that with every one standard deviation (SD) increase in ECAP slope, participants with an average age were less likely to accurately detect a rate change for the 300-pps reference condition. The main effects of age and electrode location were not significant (both ps > .05).

Predicted Probability (Log Odds) of Correct Rate Discrimination as a Function of Standardized Chronological Age at Time of Testing. Data points represent the average performance across all trials from individual electrodes (4, 12, and 20) in individual listeners for the five baseline rates: 100, 200, 300, 400, and 500 pps. The blue lines represent linear regression lines.
Final Multilevel Model for Pulse Rate Discrimination Performance.
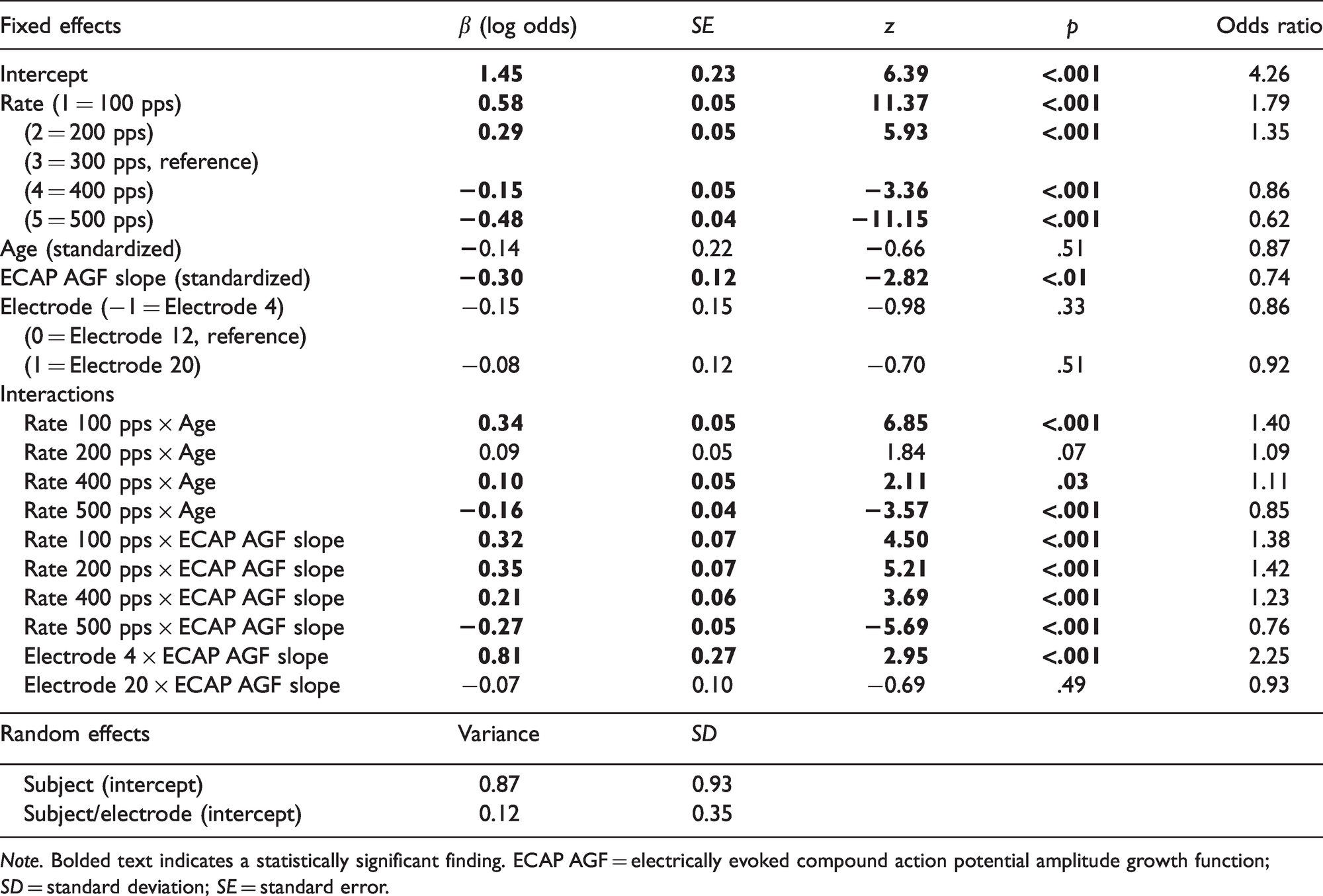
Note. Bolded text indicates a statistically significant finding. ECAP AGF = electrically evoked compound action potential amplitude growth function; SD = standard deviation; SE = standard error.
Significant two-way interactions were identified between age and rate in the 100-pps rate condition, the 400-pps rate condition, and the 500-pps rate condition, demonstrating that the effect of age on rate discrimination performance was different at these rates compared with the reference rate (300 pps). These interactions, highlighted in Figure 2, were further investigated post hoc by releveling the final statistical model to utilize each rate as the baseline (reference) and noting the effect of age for each baseline rate condition. The main effect of advancing age on pulse rate discrimination ability was not significant at any baseline rate condition. The presence of statistically significant interactions between age and rate, in the absence of a main effect of age, suggests that the interaction between these two variables is more influential on rate discrimination performance than the effect of age alone. It appears that age was acting as a moderator of the effect of baseline rate on rate discrimination performance. Results demonstrated that older participants performed better than younger participants in the lowest rate condition (e.g., 100 pps), but that older participants performed worse than younger participants at higher rate conditions (e.g., 500 pps).
Figure 3 displays pulse rate discrimination performance as a function of ECAP AGF slope across the three test electrodes (4, 12, and 20) in individual listeners across the five baseline rates: 100, 200, 300, 400, and 500 pps. The interaction between ECAP AGF slope and baseline rate was significant (p < .001), showing that the effect of a steeper ECAP slope associated with poorer rate discrimination was larger at 500 pps compared with 300 pps, but this effect was smaller at 100, 200, and 400 pps compared with 300 pps (Figure 3). We further investigated these interactions post hoc by releveling the final statistical model to utilize each rate as the baseline (reference) and noted the effect of ECAP slope for each baseline rate condition. Results showed a significant main effect of ECAP slope on pulse rate discrimination ability for the 300- and 500-pps baseline rate conditions, suggesting small but significant decreases in rate discrimination at these baseline rates as ECAP AGF slope increased. The main effect of ECAP slope on pulse rate discrimination ability was not significant for the 100-, 200-, or 400-pps baseline rate conditions.

Pulse Rate Discrimination Performance as a Function of ECAP AGF Slope. Data points represent the average across all trials from individual electrodes (4, 12, and 20) in individual listeners for the five baseline rates: 100, 200, 300, 400, and 500 pps. Unfilled black squares denote younger CI users, and filled red triangles denote older CI users. Younger was defined as below the median age (53.5 years); older was defined as at the median age or above. ECAP AGF = electrically evoked compound action potential amplitude growth function.
The two-way interaction between ECAP AGF slope and electrode location was significant (p = .003), demonstrating that the effect of ECAP slope depended on the electrode location. Releveling of the final statistical model to utilize each electrode as the reference electrode revealed that there were significant effects of ECAP slope on pulse rate discrimination ability at Electrodes 12 and 20, but not at Electrode 4. The ECAP slope effect was not significantly different between Electrodes 12 and 20.
Finally, as can be shown in Figure 3, there were some data points with extreme ECAP AGF slopes (e.g., >2 SD) relative to the majority of data points. To test whether the above effects were driven by these relatively extreme data points, we reran the optimal model by only focusing on data with ECAP AGF slopes in the range of −2 to +2 SDs. The general pattern of results, including the effects and interactions between age and baseline rate, still hold. It was found that the estimated main effect of ECAP AGF slope was significant using baseline rates of 100, 200, and 400 pps as the reference level (all ps < .01). However, the main effect of ECAP AGF slope was not significant using baseline rates of 300 or 500 pps as the reference level (all ps > .05). While the ECAP AGF slope by electrode location interaction was still significant, the main effect of electrode location was not significant using any baseline rate as the reference level (all ps > .05). The interaction was driven by that, for example, the estimated effects of ECAP AGF slope were significantly different between certain electrode comparisons (e.g., 4 vs. 12, p = .007) but not at other electrode comparisons (e.g., 12 vs. 20, p = .93).
The effect of DoD on rate discrimination performance was removed during final model selection. A post-hoc Pearson’s correlation was computed to examine the relationship between standardized DoD and rate discrimination performance in our sample for each baseline rate. A weak negative correlation was found between standardized DoD and rate discrimination performance at 100 pps (r = −.45, p = .04). The negative correlation between standardized DoD and rate discrimination performance at all other baseline rates was not statistically significant (200 pps, r = −.19, p = .43; 300 pps, r = −.09, p = .68; 400 pps, r = −.29, p = .22; 500 pps, r = −.25, p = .28).
Discussion
The purpose of this study was to investigate the effect of chronological age and peripheral neural survival on pulse rate discrimination ability in adult CI users. We hypothesized that older CI users would have poorer pulse rate discrimination performance compared with younger CI users because of poorer peripheral and central processing. In general agreement with our hypothesis, we found age-related declines in pulse rate discrimination performance at higher stimulation rates (e.g., 500 pps; Figure 2) compared with lower stimulation rates. We also hypothesized that steeper ECAP AGF slopes (i.e., possibly reflecting better peripheral encoding) would be associated with better pulse rate discrimination performance. In contrast to such a hypothesis, we found that steeper ECAP AGF slopes were not predictive of better pulse rate discrimination performance in the majority of the conditions.
Effect of Baseline Rate on Pulse Rate Discrimination Performance
Consistent with previous studies (McDermott & McKay, 1997; Townshend et al., 1987; Zeng, 2002), on average, pulse rate discrimination performance decreased with increasing baseline rate (Figure 2). For instance, when we convert the effect estimates (i.e., β) to odds ratios (Table 2), on average, participants were 1.79 times more likely to accurately discriminate a change in rate for a 100-pps standard rate compared with a 300-pps standard rate on the reference electrode (i.e., Electrode 12). In addition, participants were 0.62 times less likely to accurately discriminate a change in rate for a 500-pps standard rate compared with a 300-pps standard rate on the reference electrode.
However, an inspection of the raw data shows that some participants did not show a rapid decrease in pulse rate discrimination performance with increasing stimulation rate. The high variability in pulse rate discrimination performance above the upper limit (about 300 pps) of pulse rate discrimination in CI users has been previously documented in the literature. For example, Kong et al. (2009) found that the majority of the CI participants tested performed well above chance on pulse rate discrimination at the highest rate tested (500 pps). Carlyon, Deeks, et al. (2010) also documented variability in pulse rate discrimination performance from 100 to 500 pps, such that one of their six listeners performed near the ceiling for the highest rate tested while the majority of other listeners performed at chance level for 400 and 500 pps stimuli.
To date, it remains less clear about the factors contributing to variabilities in pulse rate discrimination among CI users. Training has been shown to significantly improve pulse rate discrimination performance in CI users across electrode and stimulation rate, which may contribute to variability on this task (Goldsworthy & Shannon, 2014). In addition, participants who did not show strong upper limits in temporal pulse rate discrimination might have relied on loudness cues rather than pitch cues to complete the task, as level roving was not utilized in this experiment (Goldsworthy & Shannon, 2014). Our current findings suggest that responses from individuals with older chronological age (Figure 2) tend to exhibit relatively lower limits of pulse rate discrimination.
Effect of Age on Pulse Rate Discrimination Performance
The current finding of age-related declines in temporal pulse rate discrimination at higher stimulation rates compared with lower stimulation rates (Figure 2) agrees with emerging evidence of age-related changes in temporal processing in CI users in a variety of tasks such as word identification based on silence duration cues (Xie et al., 2019), amplitude modulation detection (Shader et al., 2020a), gap detection (Shader et al., 2020b), and voice emotion recognition (Christensen et al., 2019). The aging effects on temporal processing (e.g., temporal pulse rate discrimination) are likely to be associated with age-related changes at peripheral (Sergeyenko et al., 2013) and central processing (Anderson et al., 2020; Clinard et al., 2010; Gaskins et al., 2019). However, the relative contributions of those factors are still debated (Bao et al., 2020; Humes et al., 2012). These converging findings regarding age-related changes in temporal processing in CI users may be the result of a common mechanism (Hopkins & Moore, 2011). This common mechanism could potentially arise from an age-related reduction in neural synchrony at the level of the auditory periphery (Ozmeral et al., 2016), a decrease in inhibitory signals in the auditory cortex (Roque et al., 2019), and the slowing of neural oscillatory activity at the level of the cortex (Harris & Dubno, 2017).
Effect of Age on Pulse Rate Discrimination in Electric Versus Acoustic Hearing
Aging effects have been observed in many temporal processing tasks in acoustic-hearing listeners (see Gordon-Salant, 2010). Relevant to this study, pulse rate discrimination performance has been investigated using band limited pulse trains (4-kHz center frequency and 1-kHz bandwidth) presented at rates of 80, 200, and 400 pps (Gaskins et al., 2019). Relative performance (ΔF/F) was better for the younger compared with the older normal-hearing participants. One caveat to this finding is that despite having normal pure tone thresholds, the average amount of hearing loss was greater in the older group and may have contributed to the aging effect. There was also better performance at 400 pps compared with 80 and 200 pps. This may have occurred because the harmonics in the pulse train were resolved at 400 pps (Carlyon & Deeks, 2002). The effects of age and hearing loss on fundamental frequency discrimination has also been measured using acoustic complex tones (which, depending on the phase relationship, produce acoustic pulse trains) that contained only unresolved harmonics (Bianchi et al., 2019). There were no effects of age or hearing loss for these conditions. The lack of aging effects and the discrepancy to other similar studies (e.g., Bianchi et al., 2016; Moore et al., 2012) could be a result of the careful design of the stimuli and procedure, which include the randomization of lowest harmonic in the complex to avoid spectral edge effects. If one ignores the differences between studies, we can broadly say that the stronger rate limitations with increasing age seen in the CI participants in this study appear at odds with data from the acoustic hearing participants using acoustic pulse train stimuli. Of course, this comparison should be done cautiously because subject, stimulus, and procedural confounds appear to severely limit the ability to test rate discrimination in normal-hearing listeners and provide a true comparison group to the CI listeners of this study.
Relationship Between Temporal Pulse Rate Discrimination and Peripheral Neural Status
This experiment utilized ECAP AGFs to infer the status of the neural periphery in participants. We included these values to estimate peripheral encoding to help separate peripheral versus central contributions. Steeper ECAP AGF slopes are thought to reflect better peripheral encoding due to a greater surviving neuronal population, which may result in better temporal processing (Pfingst et al., 2015; Scheperle, 2017). Our results, however, did not consistently support a positive relationship between ECAP AGF slope and temporal processing as indexed by pulse rate discrimination. We speculate this may be partly due to the high variability in pulse rate discrimination performance among CI users. For example, our data showed that test electrodes with shallow ECAP AGFs, falling below the mean slope value, demonstrate a large range of performance on the temporal pulse rate discrimination task across baseline stimulation rates (Figure 3).
It should be noted that the predictive validity of ECAP AGFs to temporal processing appears to be inconsistent with some of the extant literature (Hay-McCutcheon et al., 2005; Hughes et al., 2014; Shader et al., 2020a). However, consistent with our results, Shader et al. (2020a) found that steep ECAP AGF slopes were not predictive of better temporal processing as measured by an amplitude modulation detection task. Future studies may need to include more reliable measures of peripheral encoding to explore its relationship to temporal processing in the context of aging effects, such as accounting for electrode location and distance to the modiolous using imaging techniques (Cakir et al., 2016; Schvartz-Leyzac et al., 2020). Nevertheless, considering the independent effects of age and ECAP AGF slope on pulse rate discrimination in this study, we argue that the aging effects, to some extent, could have resulted from central contributions to limitations in temporal processing with advancing age (Clinard et al., 2010; Gaskins et al., 2019).
A correlation between peripheral neural status and AGF slope has been reported in the literature (Hall, 1990; Smith & Simmons, 1983). However, temporal refractory measures have also been used to infer peripheral neural health in studies investigating temporal processing in CI users (Tabibi et al., 2019). Neural estimates other than slope may have a greater utility for examining the relationship between temporal pulse rate discrimination and peripheral neural status. It has been shown that larger ECAP refractory periods are correlated with a denser neural network (Botros & Psarros, 2010; Cohen, 2009; Lee et al., 2012; Tabibi et al., 2019). Smaller ECAP refractory periods are thought to be a byproduct of less-excitable auditory neurons or a less dense electrode-to-neural interface (Tabibi et al., 2019). Neural adaptation alone may not account for variability in temporal processing abilities across CI users (Hughes et al., 2014; Zhang et al., 2013). Lee et al. (2012) found that ECAP refractory time constants were equivalent between older and younger CI users; however, perceptual recovery from forward masking was significantly slower in older CI users compared with younger CI users.
Importantly, peripheral neural survival may not directly correlate to the peripheral neural synchrony (i.e., auditory nerve phase locking) required of temporal rate discrimination (Shepherd & Javel, 1997). Future studies investigating the relationship between temporal rate discrimination and indirect measures of peripheral neural health via ECAPs may consider incorporating a phase-locked response, temporal refractory measure, and ECAP AGF slope in order to better capture the complexities of ECAP response patterns.
Limitations With This Study
First, although we included a rigorous loudness balancing procedure before the pulse rate discrimination task, the possibility that some individuals utilized loudness cues to complete the task cannot be completely ruled out. In the loudness balancing procedure, in the event that the participant reached their UCL before 10 reversals, their MCL was used as the matched level. The MCL was used as the matched level for 5% of the total data in the main pulse rate discrimination task (15 of 300 trials across all participants). For these conditions, participants are more likely to utilize loudness cues to complete the pulse rate discrimination task.
Second, the current pulse rate discrimination task used a method of constant stimuli in which there was a constant 35% difference between the baseline and stimulation rates. It may be the case that older CI users can distinguish between larger differences in stimulation rates; however, this study did not utilize an adaptive procedure to target peak pulse rate discrimination performance. Such a procedure would have been impractical given the need for loudness balancing.
Third, following the procedures from previous studies, this study used non-default ECAP parameters to obtain AGFs for a small proportion of data (10% of total recordings; across four participants). Increasing the IPG and pulse width could have inadvertently increased ECAP slope in ways that cannot be accounted for by converting the value to nC (Ramekers et al., 2014, 2015). Although a simple statistical model completed post hoc determined that the effect of variable ECAP parameters on ECAP AGF slope, including pulse width and IPG, were not significant (p = .453 and p = .783, respectively), a more systematic analysis of the effects of ECAP variables on AGFs is warranted in the future. It is important to note that ECAP amplitude, and thus ECAP AGFs, are affected by the apical–basal location of the electrode (Brill et al., 2009; van de Heyning et al., 2016). Specifically, ECAP amplitude is generally higher in the apical region of the cochlea compared with the basal region (Brill et al., 2009; Jahn & Arenberg, 2020). While differences among ECAP AGFs across electrode location within and between participants may be due to differences in peripheral neural conditions, it may also simply reflect the region of the electrode array being stimulated. In addition, ECAP recordings have been shown to be impacted by the type of internal electrode array. Newer internal devices have been shown to have a lower noise floor than older internal devices, which may impact the slope of ECAP AGFs (Kim et al., 2010, 2017).
Fourth, chronological age was highly correlated with age at onset of deafness in our sample (r = −.89). In other words, younger participants tended to have earlier ages at onset of deafness. In an attempt to avoid issues of multicollinearity, only chronological age was included in the final statistical analysis. However, the strong relationship between these variables may confound the comparisons made between chronologically older and younger participants within our sample. Interestingly though, our data show that many participants with early onset of deafness (see Table 1), referring to severe-to-profound hearing loss that was present by the age of three (Centers for Disease Control and Prevention, 2019; Jallu et al., 2019; Zeitler et al., 2012), appeared to perform worse on the temporal pulse rate discrimination task than participants with later onsets of deafness, especially at lower rates. Due to the small sample size of CI participants with early onset of deafness in this study (n = 5), this observation should be interpreted with caution. Future studies should include a larger sample size in order to potentially reduce the confound between chronological age and chronological age at onset of deafness and to further investigate the relationship between onset of deafness and pulse rate discrimination performance.
Implications for CI Clinical Practices
Our current finding of age-related changes in temporal pulse rate discrimination at higher stimulation rates compared with lower stimulation rates may indicate the need to consider age as a factor during CI programming, particularly for stimulation strategies designed to convey temporal pitch cues that utilize a variable pulse rate to track the temporal fine structure of the input signal (e.g., fine-structure processing, Müller et al., 2012). The present data suggest that older CI users, who showed limited pulse rate discrimination abilities at higher stimulation rates, would not have access to possible pitch information conveyed by fine-structure processing strategies such as FS4 and FS4-p (MED-EL, 2020) as the running stimulation rate changes would not be discriminable above a certain pulse rate. This would potentially result in increased difficulty using these strategies to perform pitch-related tasks such as talker gender discrimination, utilizing pitch cues in tonal languages, or melody perception.
Conclusions
Temporal pulse rate discrimination performance declined with advancing age at higher stimulation rates (e.g., 500 pps) when compared with lower rates. Such age-related changes in pulse rate discrimination appear to be independent of ECAP AGF slopes, an indirect measure of peripheral neural survival. These results may indicate a central contribution underlying limitations in temporal pulse rate discrimination ability associated with aging.
Footnotes
Acknowledgments
The authors would like to thank Cochlear Ltd for providing testing equipment and technical support, and to Anna Tinnemore and Kristina DeRoy Milvae for help with data analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the National Institute on Aging of the National Institutes of Health under Award Number R01AG051603 (M. J. G.).
