Abstract
It has been argued that a main limitation of the cochlear implant is the spread of current induced by each electrode, which activates an inappropriately large range of sensory neurons. To reduce this spread, an alternative stimulation mode, the all-polar mode, was tested with five participants. It was designed to activate all the electrodes simultaneously with appropriate current levels and polarities to recruit narrower regions of auditory nerves at specific intracochlear electrode positions (denoted all-polar electrodes). In this study, the all-polar mode was compared with the current commercial stimulation mode: the monopolar mode. The participants were asked to judge the sound dissimilarity between pairs of two-electrode pulse-train stimuli that differed in the electrode positions and were presented in either monopolar or all-polar mode with pulses on the two electrodes presented either sequentially or simultaneously. The dissimilarity ratings were analyzed using a multidimensional scaling technique and three-dimensional stimulus perceptual spaces were produced. For all the conditions (mode and simultaneity), the first perceptual dimension was highly correlated with the position of the most apical activated electrode of the electrical stimulation and the second dimension with the position of the most basal electrode. In both sequential and simultaneous conditions, the monopolar and all-polar stimuli were significantly separated by a third dimension, which may indicate that all-polar stimuli have a perceptual quality that differs from monopolar stimuli. Overall, the results suggest that both modes might successfully represent spectral information in a sound processing strategy.
Introduction
The cochlear implant (CI) is a biomedical device that can restore functional hearing for a large portion of people with severe to profound hearing loss (Blamey et al., 2013). Despite this great success, the sound quality produced by the device needs to be improved to help CI users better understand speech in noisy environments and to enjoy music. In the most common signal processing strategies, the input signal is band-pass filtered, and the envelope of the output of each filter is extracted to amplitude-modulate a fixed-rate electric pulse train on the electrode assigned to that filter band. To avoid uncontrolled current interactions, only one electrode is activated at a time (sequential interleaved stimulation). In monopolar (MP) mode, each singly activated intracochlear electrode is paired with an extra-cochlear return electrode.
Recently, an alternative stimulation mode has been designed to better control current interactions and to create independent and focused places of electrical stimulation along the cochlea. In this mode, called all-polar (AP or Phased Array; van den Honert & Kelsall, 2007), all the electrodes are activated simultaneously. The current levels and polarities on each electrode are set so that the sum of all potentials will confine the current field to specific and independent places within the cochlea. The electrode, around which a focused current field is created in AP mode by activating all the electrodes simultaneously, will be referred to as an AP electrode. Likewise, the term MP electrode designates the single intracochlear electrode activated in MP mode.
Recent behavioral experiments (Marozeau, McDermott, Swanson, & McKay, 2015) have shown that AP mode produces less current summation when two AP electrodes are simultaneously activated compared with two MP electrodes. However, when the stimuli were matched in loudness, no significant advantage over MP mode in terms of spread of neural excitation was found for the AP mode. This study aimed to use a multidimensional scaling technique to investigate the difference between MP and AP modes in the perceptual space generated by two MP or AP component electrodes activated sequentially (interleaved; Experiment 1) or simultaneously (Experiment 2). Five participants judged the similarity between pairs of stimuli composed of two-electrode pulse-train stimuli presented in either MP or AP mode. The distance between the two activated electrodes, as well as their overall position within the array differed across stimuli. The stimuli were designed to test whether the different levels of channel interactions that might exist within AP and MP stimuli can have an effect on the sound quality of such stimuli. In particular, we were interested in whether the reduced interactions would make it easier to distinguish or resolve stimulation at multiple sites in the cochlea, as required, for example, in resolving vowel formant frequencies for speech understanding.
Different hypotheses can be formulated regarding the configurations of the perceptual spaces based on the theoretical effects of current focusing and previous research in this area. Each will be tested in the experiments performed. Hypothesis 1: Similar perceptual spaces for sequential AP and MP dual-electrode stimuli (Experiment 1). Hypothesis 2: Additional dimension linked to the stimulation mode (Experiments 1 and 2). Hypothesis 3: Larger perceptual distances between the AP stimuli compared to the MP stimuli for simultaneous dual-electrode stimuli (Experiment 2).
The AP stimuli are designed to create a focused current field and therefore reduce the direct current summation that occurs when more than one AP electrode is activated simultaneously (Marozeau et al., 2015). In the simultaneous MP mode, the current summation will be larger than in simultaneous AP mode and will therefore activate more neurons located between the two electrode positions, filling in the “dip” between the two electrodes and potentially making the two electrode positions more difficult to distinguish (as suggested by Srinivasan, Shannon, & Landsberger, 2012). If the two main perceptual dimensions are related to the two electrode positions, it follows that the simultaneous MP stimuli will be separated by less perceptual distance than the simultaneous AP stimuli. Therefore, this will result in larger perceptual distances between the AP stimuli compared with the MP stimuli.
Methods
Participants
Five CI users participated in the experiment (three women and two men). Their ages ranged from 44 to 82 years with duration of deafness before first implantation ranging from 9 to 31 years. All the participants were unilateral CI users who had received a second research implant on the contralateral side. At the time of the study, no commercial device that could implement AP mode was available for research purposes in Australia. Therefore, the experiments were undertaken using an experimental stimulator provided by Cochlear Ltd equipped with 22 independent current sources connected to standard Cochlear Contour Advance electrode array via a percutaneous connector (for more information, see Marozeau et al., 2015). During an 18-month period, they participated in a number of experiments (e.g., Marozeau et al., 2015). While not participating in experiments, the participants connected their research implant to a standard sound processor programmed with the Advanced Combined Encoder (ACE) strategy (McDermott, McKay, & Vandali, 1992; Vandali, Whitford, Plant, & Clark, 2000) via a wearable adaptor (van den Honert & Kelsall, 2007). After the research period, participants had the research device explanted and were reimplanted with a standard commercial CI. This project conformed to The Code of Ethics of the World Medical Association (Declaration of Helsinki) and was approved by the Royal Victorian Eye and Ear Hospital Human Research Ethics Committee (Project 11-993H). Recruitment was conducted through the Cochlear Implant Clinic at the Royal Victorian Eye and Ear Hospital and the Hearing CRC. Each participant took part in both Experiments 1 and 2.
Stimuli
The stimuli were generated by an experimental stimulator that was able to activate all the 22 intracochlear electrodes simultaneously to produce AP stimuli. AP electrodes were created by first measuring the impedances between all possible pairs of electrodes. Then, a weight matrix that defined the relative current amplitudes across the array predicted to produce the focused current field at each AP electrode was derived by inverting the impedances matrix (Marozeau et al., 2015; van den Honert & Kelsall, 2007).
For each experiment, a set of 20 dual-electrode stimuli were created: 10 in AP mode and 10 in MP mode. Each MP stimulus was a 500-ms pulse train, with two biphasic pulses per period of 10 ms (one pulse to each electrode). Each biphasic pulse had a phase width of 100 μs and an interphase gap of 20 μs. The current level of each AP or MP component electrode was adjusted so that each electrode contributed equally to the overall loudness, and all the dual-electrode stimuli were adjusted to have an equal comfortable loudness (using a loudness balance method described in Marozeau et al., 2015). The electrodes were selected in order to produce different electrode separations and different average electrode positions: 17/15, 17/13, 17/11, 17/9, 15/13, 15/11, 15/9, 13/11, 13/9, and 11/9 (the electrodes were identified by the Cochlear Ltd convention in which Electrode 22 is the most apical electrode and Electrode 1 the most basal). Stimuli presented in AP mode were similar to the MP stimuli in all aspects other than the mode and were loudness balanced to the MP stimuli. The stimuli were shifted basally by two electrode positions for one participant in order to avoid high AP threshold regions. The relative electrode positions were identical to those of the other subjects.
In Experiment 1, the two pulses in each period were presented sequentially with an onset to onset delay of 232 μs to the two different MP or AP electrodes. In the Experiment 2, the two pulses were presented simultaneously with the same stimulus repetition period of 10 ms. The simultaneous AP stimuli were created by summing the weights associated with the two AP electrodes. The order of the two experiments was counterbalanced between subjects.
Task
Loudness balancing
All AP and MP single-electrode stimuli that were components of the dual-electrode stimuli were loudness balanced using a similar method as described in Marozeau et al. (2015). In short, the stimulus on the AP electrode with the highest threshold was selected as the reference stimulus and was set to a comfortable level. Then, the level on each electrode used in this experiment was set to match the loudness of the stimulus on the reference electrode. The stimulus to be balanced and the reference were presented in random order with an inter-stimulus interval of 500 ms. The participant was asked to indicate which sound was louder using a two-interval two-alternative forced choice task. If the participant selected the reference, then the level of the sound to be balanced was increased or vice versa with a one-up one-down adaptive rule. The initial step size was 0.6 dB in MP mode and 1.2 dB in AP mode. After two reversals, the step size was halved. The test stopped after a total of eight reversals and the level of the balanced stimulus was derived as the average of the final four reversal points. The trial was repeated with a different initial level for the stimulus to be balanced: in one the level was higher than the expected balanced level, and in the other lower. To counterbalance any bias related to the varying stimulus, the role of the reference and the to-be-balanced stimulus were reversed, and two new trials were performed. The differences between the levels of the reference and balanced stimulus in the four trials were averaged and used to set the final level of the stimulus to be balanced. A similar procedure was used to loudness balance all the dual-electrode stimuli.
Dissimilarity rating
After loudness balancing was completed, participants were first presented with each of the 20 stimuli in random order to acquaint them with the range of perceptual differences in the set of stimuli. They were allowed to hear them as many times as they wanted. Then, they were informed that the goal of the experiment was to estimate the similarity in sound quality between pairs of sounds. Any small differences in loudness were to be ignored. They were presented with every possible pair of the 20 stimuli in random order, totaling 380 pairs (excluding same-stimulus pairs). In each trial, the participants were instructed to judge how similar the pairs were, and to respond by moving a cursor on a slider bar labeled from most similar to least similar mapped to continuous scale ranging from 0 to 1. Participants could listen to the pair as many times as they wanted, by pressing a “listen again” button. When they were satisfied with their judgment, they pressed a “validate” button, and the next trial began. Participants had a break between the sessions for the two experiments.
Results
Experiment 1—Sequential stimuli
A multidimensional scaling (MDS) solution was derived based on the dissimilarity scores averaged across the five participants. The scores were first normalized so that the average dissimilarity ratings were equal across participants and then analyzed using a metric MDS procedure, implemented according to the SMACOFF algorithm (Scaling by Majorizing a Complex Function, Borg & Groenen, 1997). A three-dimensional solution was selected because the solution reached a satisfactory stress level (Stress = 0.12, the correlation between the dissimilarity matrix and the distance matrix, R2, was equal to .87) and higher dimensional solutions decreased the stress of the model only marginally. As the MDS solution is rotationally undetermined, the solution was rotated with a procrustean procedure in order to maximize the correlation between the MDS dimensions and some physical descriptors (described later).
Figure 1 shows the three-dimensional solution. The MP and AP stimuli that shared the same activated component electrodes are linked by an arrow. Each MP stimulus is represented by a square and each AP stimulus is represented by the end of the arrow. The two numbers next to each stimulus indicate the activated electrodes (the same ones in AP and MP stimuli). The projection on the first dimension is highly correlated with the positions of the apical electrode (R2 = .84, df = 19, p < .0001). The second dimension is significantly correlated with the positions of the basal electrode (R2 = .74, df = 19, p = .001). No simple and physiologically relevant physical correlate could be found on the third dimension. However, two features can be observed: First, the stimuli with Electrode 15 as the most apical (15/13, 15/11, and 15/9) are separated in that dimension from the other stimuli; second, the AP stimuli are consistently separated from the MP stimuli (i.e., the arrows are always pointing upward).
MDS solution for the stimuli presented sequentially (Experiment 1). Each MP stimuli is represented by a square and each AP stimuli is represented by the end of the arrow. The two numbers next to each stimulus indicate the “AP” and “MP” electrodes activated. Each MP and AP stimulus that shared the same activated electrodes are linked by an arrow.
Figure 2 shows the average difference of the projection on each dimension between the position of the MP stimuli and their AP counterparts. On average, on the third dimension, the AP stimuli are separated from the MP stimuli (t(9) = 4.79, p < .0001). No significant differences can be observed on the first dimension (t(9) = −1.17, p = .273) nor on the second dimension (t(9) = −1.21, p = .257).
Results for the Experiment 1. Average difference of the projection on each dimension between the position of the MP stimuli and their AP counterparts. The error bars show the 95% confident interval.
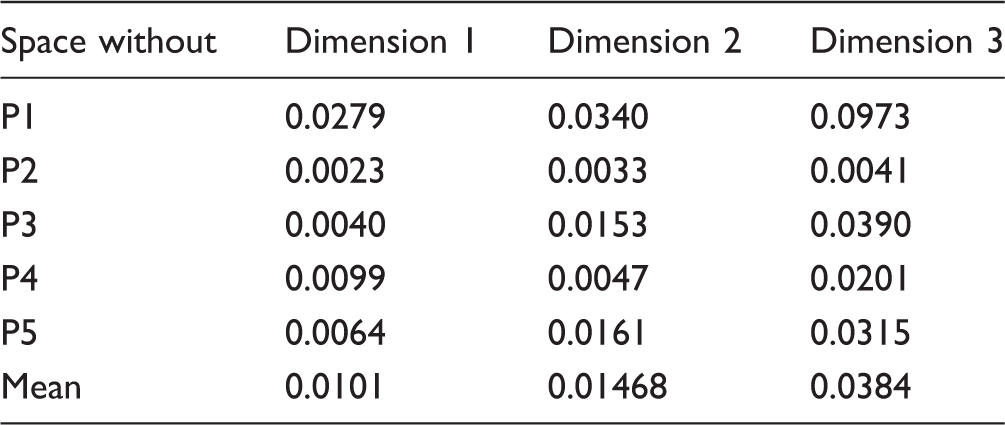
Given the high variability seen among the five participants in other auditory tasks (Marozeau et al., 2015), it is interesting to estimate the individual contribution of each participant toward the averaged space as plotted in Figure 1. Unfortunately, given the low number of participants and the dimensionality of the solution, three-way MDS models like Individual Differences Scaling (INDSCAL) were not appropriate to produce a stable and reliable solution. Therefore, the contribution of each participant was evaluated by a jackknife resampling analysis technique. Five new spaces were produced with a subset of four participants out of the five, leaving out for each new space a different participant. If one of these new spaces differs substantially from the other ones, this will indicate that the participant that was left out had a large contribution on the other spaces. Figure 3 shows the five solutions rotated to the same orientation. Although some minor differences of position can be observed for some stimuli, the main outcome described earlier is still valid for each of the five spaces: The first dimension is correlated with the positions of the most apical electrode activated and the second dimension with the positions of the most basal electrode; AP stimuli are consistently separated from the MP stimuli along the third dimension. This indicates no outlier among the five participants and a reliable space averaged across the five participants. Table 1 shows the mean squared errors, MSE, between each of the four-participant averaged spaces and the five-participant spaces for each dimension. On average, the MSE of the first dimension was smaller by a factor of 2 compared with the other two dimensions indicating that the first dimension was more consistent across participants.
Five MDS solutions for the sequential condition (Experiment 1) averaged across four out of five participants realigned to have the same orientation as the space averaged across all five participants. Mean Squared Errors Between Each of the Four-Participant Averaged Space and the Five-Participant Space for Each Dimension in the Sequential Condition (Experiment 1).

Experiment 2—Simultaneous Stimuli
Figure 4 shows the three-dimensional solution of Experiment 2. The same analysis as in the Experiment 1 was applied to the results of this experiment. The projection on the first dimension is also correlated with the positions of the most apical basal electrode (R2 = .83, df = 19, p < .0001). The second dimension is weakly correlated with the positions of the most basal electrode (R2 = .22, df = 19, p = .0389). Furthermore, as was observed on the third dimension in Experiment 1, the stimuli with Electrode 15 as the most apical electrode are separated in that dimension from the other stimuli. The configuration of the first two dimensions (Figure 3 upper panel) strongly suggests that the stimuli are located around a curve (the so-called horse shoe configuration). This configuration is commonly seen in similar experiments (McKay et al., 1996) and suggests that two dimensions represent only one perceptual dimension (represented position around the curve). The third dimension is correlated with the stimulation mode (R2 = .54, df = 19, p = .0002).
MDS solution for the stimuli presented simultaneously (Experiment 2). See caption of Figure 1.
Figure 5 shows the average difference of the projection on each dimension between the position of the MP stimuli and their AP counterparts. As for Experiment 1, the AP stimuli are separated from the MP stimuli in the third dimension (t(9) = 5.77, p = .0002). No significant differences can be observed on the first (t(9) = −1.20, p = .262) and second dimension (t(9) = −1.25, p = .241). It is worth noting that the scales of the two experiments are independent, and therefore, the magnitude of differences found on the third dimension cannot be compared across experiments.
Results for the Experiment 2. Average difference of the projection on each dimension between the position of the MP stimuli and their AP counterparts. The error bars show the 95% confident intervals.
Figure 6 shows the five spaces averaged across four participants using the jackknife technique. Although the spaces are all fairly similar, the space created without P1 seems to vary systematically, especially on the third dimension. Table 2 shows the MSE between each of the four-participant averaged space and the five-participant space for each dimension. On average, the MSE on the third dimension was 2 to 3 times larger than the other two, indicating that this dimension was more variable across participants.
Five MDS solutions for the simultaneous condition (Experiment 2) averaged across four out of five participants realigned to have the same orientation as the space averaged across all five participants. Mean Squared Errors Between Each of the Four-Participant Averaged Space and the Five-Participant Space for Each Dimension in the Simultaneous Condition (Experiment 2).
To test the third hypothesis, that a larger dissimilarity would exist between the AP stimuli compared with the MP stimuli, a subset of the raw dissimilarity scores including only the pairs with the same stimulation mode (the pairs including only AP vs. AP and MP vs. MP) was modeled using a mixed linear model with the stimulation mode (AP’s pair vs. MP’s pair) and the pairs as fixed factors; the participants were added as random factors. All possible interactions were considered. The effect of pair was expected to be always significant. A significant main effect of the mode would support Hypothesis 3, while a significant interaction between the mode and the pair will indicate an idiosyncratic effect of the mode on the pairs. The effect of electrode pair was significant (F(44,176) = 8.79, p < .0001) as well an effect of the interactions between mode and pairs (F(44,176) = 1.98, p = .001). However, no significant main effect of mode was found (F(1,4) = 0.991, p = .376).
Discussion
These experiments report two three-dimensional spaces that describe the perceptual distances between dual-electrode AP and MP stimuli, with pulses on component electrodes interleaved sequentially (Experiment 1) or presented simultaneously (Experiment 2). Overall, the two spaces were fairly similar. In both spaces, the first dimension was correlated with the positions of the apical activated electrode position, and the second to the positions of the basal electrodes.
The main aim of these experiments was to study the effect of stimulation mode (AP vs. MP) on the perceptual space of dual-electrode stimuli. Overall, the AP stimuli and their MP counterparts are always located close to each other on the first two dimensions of the perceptual spaces, as proposed in the first hypothesis (similar perceptual space for AP and MP sequential stimuli). However, as the AP and MP stimuli differ significantly on the third dimension, this hypothesis cannot be fully supported.
The effect of mode can be seen in the third dimension, as proposed in the second hypothesis (additional dimension linked to the stimulation mode). It is unknown how the stimuli vary perceptually on the third dimension. However, a recent study by Padilla and Landsberger (2016) showed that subjective scaling of verbal descriptors such as Clean or Dirty and Pure or Noisy correlated with the reduction in spread of excitation induced by a tripolar stimulation mode. This suggests that the third dimension in our study might be associated with similar descriptors.
The Hypothesis 3 (larger dissimilarity between AP stimuli compared with MP stimuli) was tested with a mixed-linear model. As no main effect of mode was found, this hypothesis was not supported. Therefore, it cannot be concluded that the AP mode would systematically increase the perceptual difference between two multielectrode stimuli compared with MP mode or would facilitate the resolving of vowel formant frequencies.
As the MDS solution is rotationally undetermined, it is possible to rotate the spaces toward other physical descriptors such as the average activated electrode position and the distance between the two activated electrodes. In that situation, significant correlations can be found for the first two dimensions of both spaces (R2 = .96 and the .78 between the first dimension and the average electrode position in Experiments 1 and 2, respectively; and R2 = .5 and .21 between the second dimension and the distance between the two electrodes in Experiments 1 and 2, respectively). Similar results for sequential dual-electrode stimuli were previously found by McKay et al. (1996). They asked four CI participants to rate the dissimilarity between pairs of dual-electrode bipolar stimuli that varied in electrode separation and overall position. The separation of electrodes comprising each bipolar pair was also varied with two distances between the active and return electrodes of the bipolar pair compared to test the effect of current spread. They found, for most CI participants, a two-dimensional solution related to the average activated electrode position and the activated electrode separations. The descriptors of average electrode position and electrode separation are analogous to common acoustical descriptors of timbre: the spectral centroid and the spectral spread, respectively (see Marozeau, de Cheveigné, McAdams, & Winsberg, 2003 for a complete description). Therefore, the perceptual dimensions seen in the CI users might be similar to the perception of timbre by normal hearing listeners (Kong, Mullangi, & Marozeau, 2012; Kong, Tansman, Marozeau, & Epstein, 2009; Macherey & Delpierre, 2013). It is interesting to note that the correlation between the second dimension and the distance between activated electrodes is weaker in the simultaneous condition compared with the sequential condition. This may be caused by the increased interaction between component electrodes in the simultaneous condition, which may have reduced the salience of any percept related to the two independent electrode positions.
Additionally, it is unclear why the stimuli 15/13, 15/11, and 15/9 are also separated from the other electrode positions in third dimension of the Experiment 1 and the second dimension in the Experiment 2. However, it is possible that the solutions were distorted. This kind of distortion is often found in MDS studies, where a one-dimensional solution is represented as a horse shoe in a two-dimensional solution (e.g., McKay et al., 1996). If this distortion is ignored, then the third dimension of Experiment 1 clearly separated the MP and AP stimuli. This result would indicate that the AP mode differed from the MP mode along a perceptual dimension that was independent of electrode position and separation.
The implications of this result for potential vowel identification ability are unclear. However, the results indicate that dual-electrode AP stimuli, whether presented sequentially or simultaneously, produce a very similar perceptual space to MP stimuli and might at least produce similar speech perception scores.
Conclusions
Overall, the results indicated that dual-electrode AP stimuli produce very similar perceptual spaces to the MP stimuli. For both spaces (sequential or simultaneous pulses), the MP and AP stimuli were significantly separated by an orthogonal third dimension, which may indicate that AP stimuli have a perceptual quality that differs from monopolar stimuli. Additional experiments using descriptive magnitude estimations will be required to characterize the perceptual quality of this additional dimension. The results suggest that both sequential and simultaneous AP stimuli might be successfully used to represent spectral information in a sound processing strategy.
Footnotes
Acknowledgments
The authors would like to thank the five participants who selflessly dedicated 18 months of their life to this and other studies. The authors would like also to thank Hugh McDermott for his help on the design of the experiment and his comment on the manuscript, Brett Swanson and Cochlear Ltd for technical support, and the Hearing CRC for their clinical support. Finally, the authors would like to thank the two anonymous reviewers for their constructive feedbacks on an earlier version of the manuscript. The Bionics Institute acknowledges the support it receives from the Victorian Government through its Operational Infrastructure Support Program.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by The Garnett Passe and Rodney Williams Memorial Foundation and supported by Cochlear Ltd.
