Abstract
Gaucher disease (GD) is an autosomal recessive lipid storage disorder, caused by deficient activity of the lysosomal enzyme β-glucocerebrosidase, resulting in accumulation of glucocerebroside in tissue macrophages. HGT-GCB-068 was an open-label study designed to explore the efficacy and safety of velaglucerase alfa in children and adolescents with type 3 GD, a neuronopathic form of the disease. Six treatment-naive patients received infusions of velaglucerase alfa every other week at 60 U/kg over 12 months. Velaglucerase alfa demonstrated a favorable tolerability profile, and 1 infusion-related reaction (headache) was the only drug-related adverse event reported. Numerical increases from baseline in hematological parameters and decreases in visceral parameters were seen at 12 months. http://ClinicalTrials.gov identifier NCT01685216.
Introduction
Gaucher disease (GD) results from an inherited deficiency of the lysosomal enzyme β-glucocerebrosidase. 1 More than 90% of patients with GD are affected by type 1 disease, which primarily affects the hematological, visceral, and skeletal systems, 2 while a minority of patients experience neuronopathic disease characterized by central nervous system involvement. 3 Neuronopathic GD is classically categorized into type 2 GD, the acute form of the disease, and type 3 GD, the chronic form. 2 Neurological manifestations of GD are diverse and can range from cognitive impairment, developmental delay, learning disabilities, saccadic eye movement abnormalities, auditory processing defects, seizures, muscle weakness, ataxia, and in some cases, a progressive myoclonic epilepsy (type 3a) or early onset of horizontal supranuclear gaze palsy (type 3b). 4,5 Type 2 and 3 GD have been found to be more prevalent in Middle Eastern (excluding Israel) and Asian countries (including China and Japan) compared with type 1 disease, which is more prevalent in Western countries and in Ashkenazi Jewish populations. 6 –9
Type 3 GD typically manifests in childhood, with patients displaying neurological abnormalities in addition to systemic disease manifestations, such as anemia, thrombocytopenia, and hepatosplenomegaly, which are characteristic of type 1 disease. 4 Enzyme replacement therapy and substrate reduction therapy for GD can improve hematological and visceral disease parameters associated with type 1 and 3 GD, 10 -12 but no effects on neurological manifestations have been observed to date. 13 –15 Agents such as the pharmacological chaperone ambroxol are being investigated for the management of neuronopathic symptoms in GD, and the results of a Japanese pilot study show promise in patients with type 3 and type 2 disease. 16 In this study, we report results of an open-label study that aimed to explore the efficacy and safety of velaglucerase alfa in the treatment of naive children and adolescents with type 3 GD.
Materials and Methods
Patients and Dosing
HGT-GCB-068 was a phase 1/2 study (http://ClinicalTrials.gov identifier NCT01685216) conducted to fulfill a postmarketing approval regulatory request to evaluate the efficacy and safety of velaglucerase alfa in children or adolescents with type 3 GD. The study was conducted in compliance with the International Council on Harmonisation Good Clinical Practice guidelines; written informed consent for participation was provided for all patients and assent obtained where appropriate.
Patients meeting the study inclusion criteria were 2 to 17 years of age, naive to GD treatment, and had clinical signs and symptoms consistent with type 3 GD. Patients were also required to have GD–related anemia and at least one of the following: moderate splenomegaly, readily palpable hepatomegaly, or GD-related thrombocytopenia. Confirmation of diagnosis by the glucocerebrosidase gene (GBA) genotype was not required for inclusion in the study. Blood samples were collected for GBA genotyping at screening and results were obtained after the study had begun. All patients were to receive velaglucerase alfa at 60 U/kg body weight by intravenous infusion over 60 minutes every other week for 12 months (26 infusions).
Study End Points and Analyses
The primary end point of the study was the change from baseline to 12 months in hemoglobin concentration. Changes from baseline to 12 months in platelet count, liver volume, spleen volume, and neurological signs, as well as safety end points, were secondary. Changes from baseline to 12 months in chitotriosidase and chemokine (C-C motif) ligand 18 (CCL18) were tertiary. All patients who received velaglucerase alfa were included in the safety and efficacy analyses. For efficacy parameters, individual patient listings were generated, and observed values and changes from baseline were descriptively summarized. No imputation of missing values was performed.
Assessments
Blood samples were collected for determination of hemoglobin concentration and platelet count every 6 weeks and for evaluation of chitotriosidase activity, CCL18, and anti-velaglucerase alfa antibodies every 12 weeks. Liver and spleen volumes were determined by quantitative abdominal magnetic resonance imaging (MRI) every 6 months. Neurological manifestations were assessed every 12 weeks on an individual basis with a limited examination appropriate for age and developmental stage. Standardized examination tools were not expected to be used uniformly across all patients, but neurological examinations during the treatment phase and end-of-study visit were to be conducted in the same manner as the baseline examination.
Adverse events were monitored continuously throughout the study. Treatment-emergent adverse events were adverse events that occurred any time from the first study infusion to 30 days after the last study infusion. Infusion-related reactions were adverse events that began within 12 hours of the start of an infusion and were deemed possibly or probably related to velaglucerase alfa.
Results
Patients
Six treatment-naive patients (5 male), 2 to 14 years of age, were enrolled into the study and all received velaglucerase alfa according to the treatment schedule (26 infusions over 50.1-50.6 weeks; Table 1). All patients presented with systemic and neurological features indicative of type 3 GD at baseline as assessed by their treating physician (Table 1). Hypotonia was the most common abnormality, reported in 4 patients each. Horizontal ophthalmoplegia or oculomotor apraxia was reported in 3 patients.
Summary of General and Neurological Symptoms at Baseline.
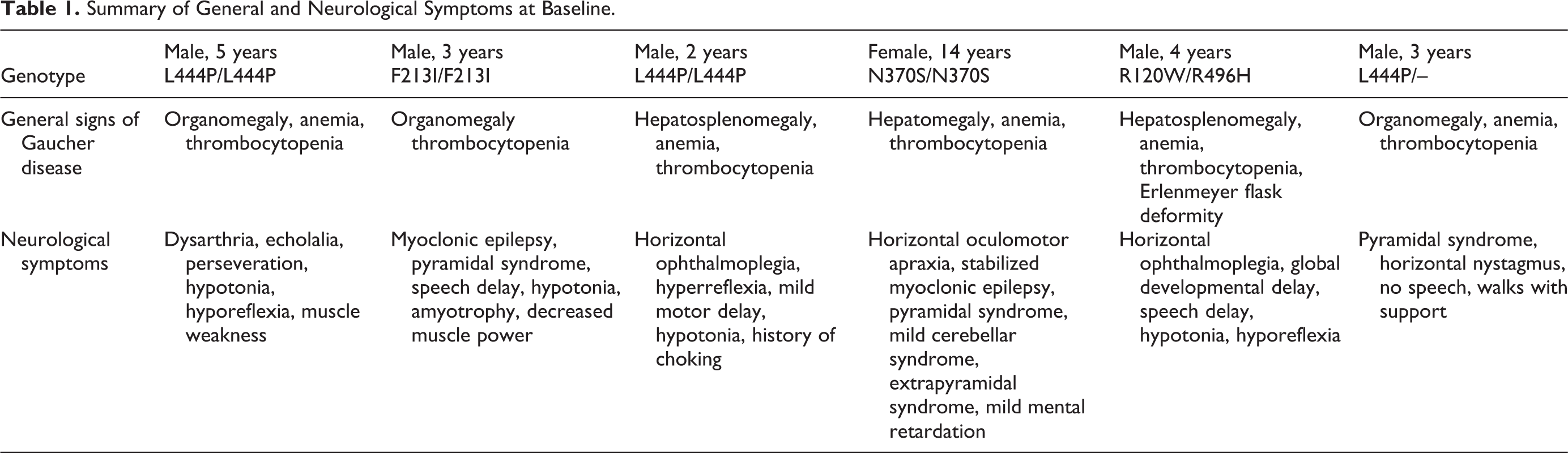
GBA genotypes were obtained after the start of the study and were consistent with classical genotypes associated with a type 3 GD diagnosis in 3 patients (2 patients with L444P/L444P and 1 patient with F213I/F213I genotypes). One patient reported an incomplete genotype (L444P/unidentified), although with 1 confirmed allele associated with type 3 disease; 1 patient was identified with N370S/N370S genotype, consistent with type 1 GD; and 1 patient was confirmed with R120W/R496H, both rare alleles, the former of which has been reported in type 2 and (rarely) type 1 GD, whereas the latter is associated with mild disease. No patients were removed from the study subsequent to obtaining genotype results.
Efficacy
From baseline to 12 months, hemoglobin concentration increased numerically in all 6 patients, and platelet count increased in 5 (Table 2). Liver and spleen volumes decreased numerically at 12 months compared with baseline in all 5 patients with available data (1 patient had only 1 postbaseline MRI assessment at 6 months). Chitotriosidase and CCL18 levels decreased over time in 4 patients (Table 2). One patient (genotype L444P/L444P) was chitotriosidase deficient, and in another patient (genotype L444P/unidentified), chitotriosidase and CCL18 were not elevated at baseline.
Mean Change From Baseline and Individual Observed Values at Baseline and 12 Months for Efficacy Parameters.
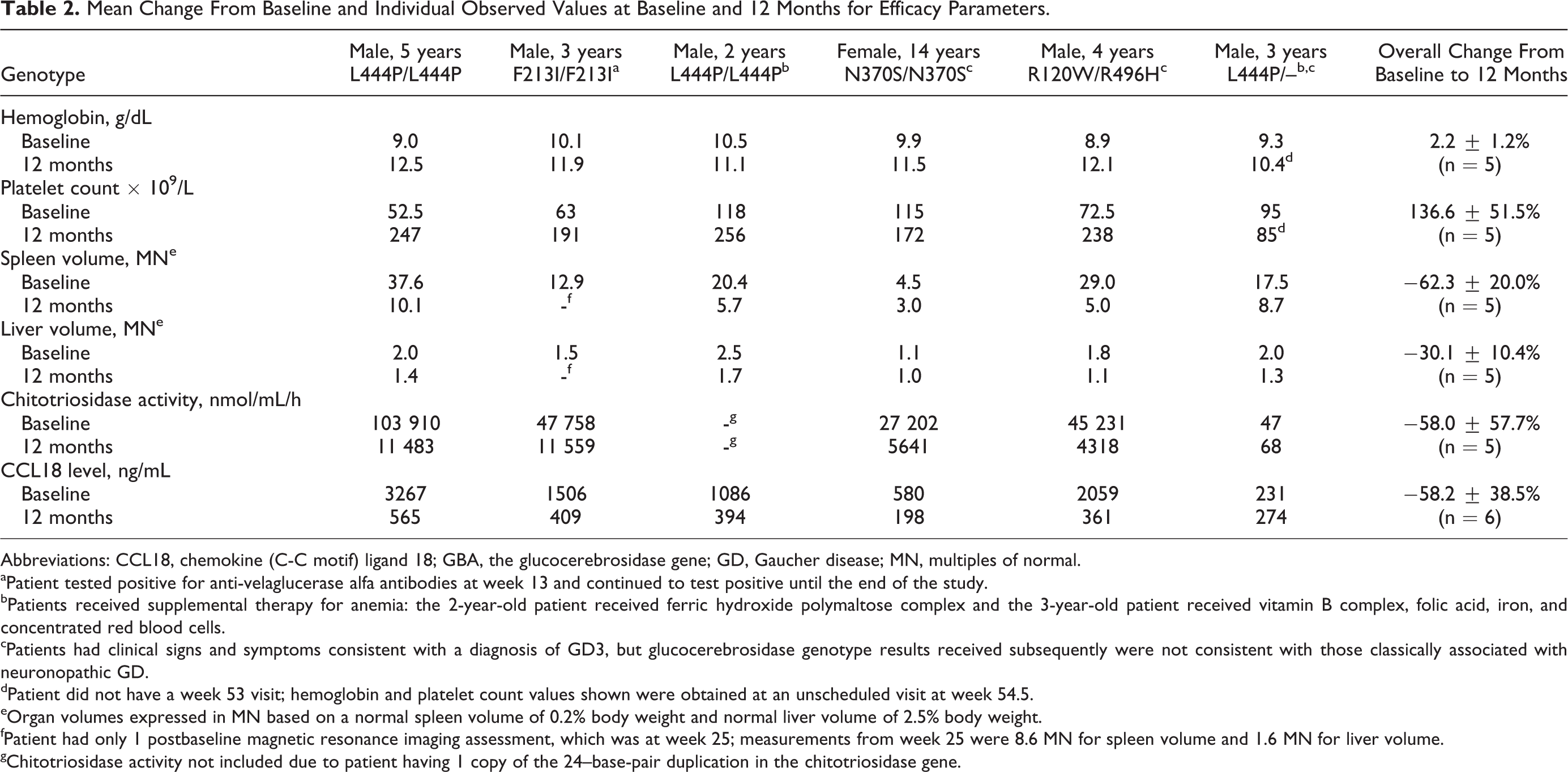
Abbreviations: CCL18, chemokine (C-C motif) ligand 18; GBA, the glucocerebrosidase gene; GD, Gaucher disease; MN, multiples of normal.
aPatient tested positive for anti-velaglucerase alfa antibodies at week 13 and continued to test positive until the end of the study.
bPatients received supplemental therapy for anemia: the 2-year-old patient received ferric hydroxide polymaltose complex and the 3-year-old patient received vitamin B complex, folic acid, iron, and concentrated red blood cells.
cPatients had clinical signs and symptoms consistent with a diagnosis of GD3, but glucocerebrosidase genotype results received subsequently were not consistent with those classically associated with neuronopathic GD.
dPatient did not have a week 53 visit; hemoglobin and platelet count values shown were obtained at an unscheduled visit at week 54.5.
eOrgan volumes expressed in MN based on a normal spleen volume of 0.2% body weight and normal liver volume of 2.5% body weight.
fPatient had only 1 postbaseline magnetic resonance imaging assessment, which was at week 25; measurements from week 25 were 8.6 MN for spleen volume and 1.6 MN for liver volume.
gChitotriosidase activity not included due to patient having 1 copy of the 24–base-pair duplication in the chitotriosidase gene.
Velaglucerase alfa treatment had no impact on neurological abnormalities in 4 patients, including 2 patients with the L444P/L444P genotype, consistent with type 3 GD, and 1 each with the R120W/R496H and N370S/N370S genotypes. One patient with the genotype L444P/unidentified showed improvements in hyperreflexia, saccadic eye movements, speech, and ability to walk, while another patient with the genotype F213I/F213L showed motor and developmental regression and worsening convulsions.
Safety
Treatment-emergent adverse events were reported in all patients. One event of headache was the only adverse event considered related to velaglucerase alfa and the only infusion-related reaction reported. No patients received premedication to prevent an infusion-related reaction. One serious adverse event of inguinal hernia was reported, requiring hospitalization to repair, but was not deemed related to velaglucerase alfa. No deaths or life-threatening adverse events were reported. One patient tested positive for anti-velaglucerase alfa antibodies at week 13 and continued to test positive until week 53, although his antibody titer decreased over time.
Discussion
Few clinical studies report on the efficacy of treatment in patients with type 3 GD. In this study, we describe the clinical characteristics and response to velaglucerase alfa of a cohort of 6 treatment-naive children and adolescents diagnosed with type 3 GD.
Overall, numerical increases in hemoglobin concentration and platelet count, and decreases in liver and spleen volumes, were seen over the course of the study in response to treatment with velaglucerase alfa. There were no reported effects on neurological symptoms in 5 of the 6 patients, consistent with previous reports of type 3 GD treated with enzyme replacement therapy, 14,17 –19 although 1 patient, a 3-year-old male with the L444P/unidentified genotype, reported some improvements in hyperreflexia, nystagmus, speech, and walking over time, suggesting a need for longer follow-up in patients with type 3 GD. Over the course of the study, this patient’s spleen and liver volumes decreased, consistent with a response to velaglucerase therapy; however, his hematological parameters responded poorly to treatment.
Velaglucerase alfa was well tolerated during the study, and no drug-related serious adverse events were reported. One infusion-related reaction of headache, which occurred following the patient’s first infusion, was the only adverse event deemed to be drug related; the patient continued to receive velaglucerase alfa for the duration of the study and did not experience any further infusion-related events of headache or any other infusion-related reactions. One patient tested positive for anti-velaglucerase alfa antibodies without apparent impact on treatment efficacy or increased risk of infusion-related reactions. The safety profile in these patients is consistent with that observed in clinical trials of velaglucerase alfa in patients with type 1 GD. 10,20
This cohort of 6 patients presented with both systemic and neurological manifestations typically associated with type 3 GD and were included in the study on this basis. A variety of neurological abnormalities were recorded in all patients, with many types of abnormalities being observed in only 1 or 2 patients each, demonstrating a high level of phenotypic heterogeneity. However, GBA genotyping results (received after the study start) were not consistent with classical genotypes associated with type 3 GD for 3 patients, underscoring the value of genotyping in the differential diagnosis of GD. One patient had a GBA genotype of N370S/N370S, which is common in type 1 GD and accepted as being protective against neuronopathic disease. 21 Although type 1 GD is typically non-neuronopathic, neurological manifestations such as parkinsonism, peripheral neuropathy, and other neurological abnormalities have been reported in patients with type 1. 22,23 However, the neurological picture of the patient with the N370S/N370S genotype in our study did not correspond with those reported for patients with type 1 GD and may have been due to an underlying condition unrelated to her GD. Another patient had GBA genotype R120W/R496H. R120W is considered a severe mutation, which has been reported in patients with type 2 GD and shown to be associated with Parkinson disease, 21,24,25 while R496H is reported as a mild mutation, noted for frequently being detected in compound heterozygosity with a severe allele in patients with GD. 26 The R496H allele is thought to preclude neuronopathic GD; however, it is clear that the R120W/R496H patient in this study had signs and symptoms consistent with type 3 GD. 3,26 A third patient, a 3-year-old male, had only 1 GBA mutation allele confirmed (L444P), while a second allele remained unidentified. His β-glucocerebrosidase activity was retested and deficiency confirmed, but his chitotriosidase and CCL18 levels were not elevated at baseline. Therefore, despite the presence of both systemic and neuronopathic symptoms, determination of type 3 disease could not be verified for this patient. All 6 patients remained in the study and received all scheduled doses of velaglucerase alfa.
Conclusion
Velaglucerase alfa was shown to have a good safety profile in this cohort of 6 patients and a positive effect on systemic parameters in 3 patients with a GBA genotype and a clinical picture consistent with type 3 GD. Although this study was limited by the small number of patients, these observations highlight the challenges in confirming a diagnosis of type 3 GD on the basis of clinical presentation alone and may reflect the real-world situation in many cases, particularly where genotyping is not readily available.
Footnotes
Acknowledgments
A.E.B. acknowledges Dr Khaled Eid, of Cairo University Hospital, Cairo, Egypt, for his considerable contribution to the study. Under the direction of the authors, Julia Cope, PhD, and Lindsay Napier, PhD, employees of Excel Medical Affairs, provided writing assistance for this publication. Editorial assistance in formatting, proofreading, copy editing, and fact checking was also provided by Excel Medical Affairs. Quinn Dinh, MD, and Hak-Myung Lee, PhD, from Shire International GmbH, also reviewed and edited the manuscript for scientific accuracy. Shire International GmbH provided funding to Excel Medical Affairs for support in writing and editing this manuscript. A.A.G.T., A.E.B., I.M., A.B., and H.B.T. were investigators in the study, which was sponsored by Shire. Y.Q. conducted statistical analyses. B.M. was the medical monitor for the study. All authors contributed to the development of the manuscript, critically reviewed the manuscript during development, and approved the final draft prior to submission.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.A.G.T. conducts contracted research for Shire and Genzyme. A.E.B. conducts contracted research for Shire and Genzyme and is on the speaker bureau for Genzyme. Y.Q. was an employee of and shareholder in Shire at the time of data analysis. B.M. is an employee of and shareholder in Shire. IM, AB and HBT have no competing interests to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Shire Human Genetic Therapies, Inc.
