Abstract
Untreated vitamin B12 deficiency manifests clinically with hematological abnormalities and combined degeneration of the spinal cord and polyneuropathy and biochemically with elevated homocysteine (Hcy) and methylmalonic acid (MMA). Vitamin B12 metabolism involves various cellular compartments including the lysosome, and a disruption in the lysosomal and endocytic pathways induces functional deficiency of this micronutrient. Gaucher disease (GD) is characterized by dysfunctional lysosomal metabolism brought about by mutations in the enzyme beta-glucocerebrosidase (Online Mendelian Inheritance in Man (OMIM): 606463; Enzyme Commission (EC) 3.2.1.45, gene: GBA1). In this study, we collected and examined available literature on the associations between GD, the second most prevalent lysosomal storage disorder in humans, and hampered vitamin B12 metabolism. Results from independent cohorts of patients show elevated circulating holotranscobalamin without changes in vitamin B12 levels in serum. Gaucher disease patients under enzyme replacement therapy present normal levels of Hcy and MMA. Although within the normal range, a significant increase in Hcy and MMA with normal serum vitamin B12 was documented in treated GD patients with polyneuropathy versus treated GD patients without polyneuropathy. Thus, a functional deficiency of vitamin B12 caused by disrupted lysosomal metabolism in GD is a plausible mechanism, contributing to the neurological form of the disorder but this awaits confirmation. Observational studies suggest that an assessment of vitamin B12 status prior to the initiation of enzyme replacement therapy may shed light on the role of vitamin B12 in the pathogenesis and progression of GD.
Keywords
Introduction
Lysosomal storage disorders (LDs) are a broad group of more than 50 rare, life-threatening diseases characterized by abnormal degradation of glycans, carbohydrates, lipids and proteins, and lysosomal transporter and trafficking. 1
The concept of LDs or lysosomal disorders was developed in the early 1960s, after the discovery that Pompe disease was caused by a deficiency in the lysosomal enzyme α-glucosidase. 2 Lysosomal storage disorder may be caused not only by defective enzymes but also by enzyme activator proteins (eg, Prosaponin [PSAP] deficiency), membrane proteins (eg, Danon disease), transporters (eg, cystinosis), or enzyme signaling (eg, mucolipidosis type II). Lysosomal storage disorders are characterized by an abnormal storage of a variety of molecules, including triglycerides, sterols, sphingolipids, sulfatides, sphingomyelin, gangliosides, and lipofuscins. 3 The buildup of substrates within lysosomes results in impaired function of the affected organs (eg, liver, spleen, bone, and nervous system), causing a wide and diverse range of clinical features. In addition, the release of lysosomal acid hydrolases into the cytoplasm will cause cellular damage, which may worsen disease progression. Also, dysregulation of apoptosis may cause disease manifestations in some LDs. Indeed, increased apoptosis has been noted in a number of the sphingolipidoses and in neuronal ceroid lipofuscinoses. 4 Since different mechanisms related to apoptosis, cholesterol metabolism dysregulation, inflammation, and alteration in signal transduction are also related to the pathogenesis of these conditions, we prefer to use the term “lysosomal disorder” than “lysosomal storage disorder.” 5 To date, more than 60 different proteins were identified as causing LDs. 6 Individually, LDs are rare, inherited disorders with an estimated frequency from 1 in 25 000 to 1 in 250 000 live newborns, but the overall incidence of all LDs is estimated to be 1 in 7000 live newborns, which makes them a relevant public health issue. The frequency may be underestimated because nowadays more individuals with mild disease and/or adult-onset forms of the diseases are being identified. 7
Gaucher disease (GD) is an autosomal recessive, multi-organ disorder caused by mutations in the lysosomal enzyme β-glucocerebrosidase (GCase, OMIM: 606463; EC 3.2.1.45, gene: GBA1), which catalyzes the conversion of the glycolipid glucosylceramide to ceramide and glucose. 8 The GBA1 gene is located on chromosome 1q21, comprises 7.6 kb of genomic DNA, and it is divided into 11 exons. 9 The GBA1 messenger RNA (mRNA) has approximately 2 kb and produces a mature protein of 497 amino acids with 56 kDa. 10,11 The expression levels of mRNA produced from GBA1 varies considerably between different cell types and has no direct correlation with GCase enzyme activity. 10 GBA1 has a pseudogene (GBAP) of approximately 5 kb, which is highly homologous (96%) with the functional gene. GBAP has an identical genomic organization and is located 16 kb downstream of GBA1. 9,10 The high degree of homology between the gene and the pseudogene must be taken into account in the investigation of mutations in patients with GD, since some of the mutations found in patients are also present in the GBAP sequence. 10 -12
A dysfunctional or absent β-GCase protein leads to the buildup of glucosylceramide in the lysosome, in particular within macrophages of the reticuloendothelial system, and to a defective production of ceramide, the hydrophobic membrane anchor for all sphingolipids in the cell. 13
This review covers clinical and molecular aspects of GD, the second most prevalent inborn error of LD, with an emphasis on lysosomal metabolism and the potential occurrence of abnormalities in vitamin B12 status.
Gaucher Disease
Gaucher disease has been classified into 3 major types based on the absence (type 1) or presence (types 2 and 3) of neurological impairments. 14 The prevalence of GD in the general population has been estimated to be about 1:60 000 15 ; however, as seen in other autosomal recessive disorders, GD exhibits ethnical preference.
At the cellular level, GD is characterized by the presence of macrophages with an altered morphology due to abnormal lipid storage, also known as “Gaucher cells.” Gaucher cells are typically found in affected organs including spleen, liver, and bone marrow. 16 In addition to exhibiting an abnormal morphology, macrophages from GD have a distinct pattern of expression of pro-inflammatory effectors. 17,18
At the subcellular level, GD features abnormalities in lysosomal pathways, which is accompanied by mitochondrial dysfunction and accumulation of α-synuclein in the mitochondrion. 19 Lysosomal lipid storage in GD reduces the efficiency of lysosomes to fuse with autophagosomes, thereby impairing cellular clearance of unnecessary substrates, protein aggregates, and dysfunctional mitochondria. 16 This triggers an inflammatory response that ultimately leads to cellular death. 16 Interestingly, mitochondrial dysfunction and increased deposition of α-synuclein are 2 features of GD that are shared with other peripheral neuropathies such as Parkinson and Alzheimer diseases. 20
The molecular mechanism underlying GD pathogenesis remains elusive. A few metabolic hallmarks have been identified in GD, including increased chitotriosidase (EC 3.2.1.14), angiotensin-converting enzyme (EC 3.4.15.1), C-C Motif Chemokine Ligand 18 (CCL18) (P55774), tartrate-resistant acid phosphatase (EC 3.1.3.2), and serum ferritin (P02792 and P02794), and some of these continue to be utilized as diagnostic and prognostic tools in clinical practice. 21 An interesting aspect of GD is the presumptive abnormality in vitamin B12 status. 22 -26 The earliest observations linking GD with vitamin B12 metabolism were documented about 4 decades ago by Gilbert and Weinreb 26 and by Rachimilewitz and Rachimilewitz. 27 These studies demonstrated elevated levels of circulating holotranscobalamin (holo-TC) in GD patients, which was not associated with decreased serum levels of vitamin B12 or any other vitamin B12 binders. 26 The levels of holo-TC were directly proportional to the severity of GD. 26 Very few follow-up studies were conducted thereafter, and to this date, the associations between GD and vitamin B12 metabolism are open for investigation, as also will be shown below.
Clinical Manifestations
Gaucher disease is classically divided into 3 clinical types based upon the severity and onset of neurological involvement; however, overlap is often seen among the phenotypes. Gaucher disease type 1 (OMIM 230800), the nonneuronopathic type, is the most common form of the disorder in the western hemisphere and the most prevalent type overall (90%-95% of the patients), with an incidence of 1 in 70 000 live newborns worldwide. 28 In Ashkenazi Jews, the incidence is 1 in 400 live newborns. 29 Type 1 is characterized by multi-organ involvement, especially hepatic, splenic, bone, hematologic, and pulmonary systems. The life expectancy depends on the time of diagnosis, severity of visceral involvement, and treatment. Patients with GD type 1 are treated with specific therapies such as enzyme replacement therapy (ERT) or substrate reduction therapy. Most treated patients usually have a normal life expectancy. The absence of early-onset primary central nervous system (CNS) is essential for the diagnosis of GD type 1. 30 During the last 2 decades, population studies have shown an association between GD and Parkinson disease. Carriers of GBA1 mutations are also at risk of developing parkinsonism. 31 Also, GD type 1 patients are at an increased risk of developing cholelitiasis 32 and hematological malignancies as multiple myeloma. 33
Gaucher disease type 2 (OMIM 230900), the acute neuronopathic type, is the less frequent phenotype, with an incidence of 1 in 100 000 live newborns and is characterized by cholestasis, hepatosplenomegaly, and early-onset and rapidly progressive CNS manifestations with bulbar involvement. Hydrops fetalis and collodion baby may be present in the most severe forms. The life expectancy varies from hours to a few months. 34 There is no specific treatment for patients with GD type 2.
Gaucher disease type 3 (OMIM 231000), the subacute or chronic neuronopathic type, is particularly prevalent in Asian and Arab countries. 14 Gaucher disease type 3 is characterized by an intermediate phenotype with visceral manifestations as GD type 1 and CNS manifestations are less severe than GD type 2. Patients may have severe bone involvement, with kyphoscoliosis, ataxia, myoclonic epilepsy, strabismus, horizontal gaze palsy, and dementia. Some patients may present corneal clouding and cardiac valvular calcifications. Enzyme replacement therapy is indicated to treat the visceral signs and symptoms of GD, but it fails to alleviate CNS manifestations. The life expectancy is 20 to 30 years. 35
Diagnosis
Clinical manifestations, such as hepatosplenomegaly, bone lesions, hematologic changes, and/or CNS involvement, are important signs that would suggest the presence of GD. 23,36 However, the diagnosis of GD should not be based exclusively on the clinical evaluation of the patient. There are other LDs that may present with symptoms similar to GD, which may complicate the establishment of a precise diagnosis.
The standard diagnostic method for GD is the evaluation of β-GCase (acid β-glucosidase) activity in dried blood spots, peripheral blood leukocytes, cultured skin fibroblasts, or other nucleated cells. Molecular analysis of GBA1, which encodes GCase, and the identification of 2 disease-causing mutations may assist the patient’s clinical classification into a determined subtype or at least make it possible to distinguish between neuronopathic and nonneuronopathic forms. 37 Genetic testing enables the confirmation and a better characterization of the patient’s condition and is considered an essential tool for GD diagnosis. 38 Up to date, more than 400 different disease-causing mutations have been described in the GBA1 gene (www.hgmd.cf.ac.uk 37,39 ). GBA1 mutations may alter GCase stability and/or impair its catalytic function. 37,38
The identification of disease-causing mutations in GBA1 may be challenging due to the GBAP. The most accurate method for mutation analysis in GD is full-gene sequencing of GBA1. 39 In order to describe a recombinant allele, a combination of direct sequencing along with an additional method, such as Southern blot or qPCR, is strongly recommended. 40,41 The occurrence of deletion and/or duplication of any region of GBA1 can be specifically addressed by multiplex ligation-dependent probe amplification. 40,41
Vitamin B12 Metabolism
Vitamin B12 is an essential micronutrient synthesized only by a select group of bacteria and archaea. Humans completely rely on a dietary intake of minimally 2 to 3 μg of vitamin B12 per day, 42 which is indispensable to support the activities of cytosolic methionine synthase (MS) and mitochondrial methylmalonyl-CoA mutase (MCM). Dietary vitamin B12 is absorbed in the lower portions of the ileum after sequential relay by the dedicated transporters haptocorrin, intrinsic factor, and transcobalamin. 43,44 Vitamin B12 bound to transcobalamin, that is, holo-TC is distributed via systemic circulation to all cells in the body. Cells take up holo-TC via receptor-mediated endocytosis, aided by the transcobalamin receptor (CD320), 45,46 which shuttles vitamin B12 into lysosomes. The protein binder TC undergoes degradation in the lysosome, liberating vitamin B12 that is subsequently exported out of this compartment using the transporters LMBR1 Domain Containing 1 (LMBRD1) 47,48 and ATP Binding Cassette Subfamily D Member 4 (ABCD4). 49,50 Once in the cytosol, newly internalized vitamin B12 undergoes processing and trafficking by proteins, CblC 51 -57 and CblD, 58 -63 respectively, to finally reach acceptor proteins, MS in the cytosol and MCM in the mitochondrion.
Insufficient intake and certain inborn errors of metabolism impairing the cellular transport, trafficking, and utilization of vitamin B12 manifest as functional cobalamin deficiency, with either isolated or combined homocystinuria and methylmalonic aciduria. Untreated vitamin B12 deficiency causes hematological abnormalities, subacute combined degeneration of the spinal cord, and polyneuropathy. 64 However, its precise role in the progression of neurological diseases (measured as the onset of dementia in only 1 large study 65 ) has been debated. 65 It is possible that sufficiency of vitamin B12 is important for preventing nerve deterioration. Vitamin B12 administration only partially reverses clinically established nerve degeneration and neuropathies.
Lysosomal Disorders of Vitamin B12
The lysosome is an essential compartment in cellular vitamin B12 metabolism by connecting uptake and downstream utilization of the micronutrient. Two genetic disorders affecting lysosomal proteins LMBRD1 (cblF) 47,48 and ABCD4 (cblJ) 49,50,66 have been described, leading to trapping of vitamin B12 inside the lysosome and the concomitant onset of functional cobalamin deficiency by inactivation of the B12 acceptors MS and MCM. LMBRD1 and ABCD4 mediate the export of vitamin B12 from the lysosome. 47 -50,66
Apart from these canonical defects of lysosomal vitamin B12 transporters, 2 other reports documented abnormal vitamin B12 metabolism caused by genetic mutations that impair the endocytic and lysosomal pathways independent of vitamin B12 metabolism. These include the occurrence of abnormal lysosome acidification in a patient with Alzheimer disease 67 and impaired endocytosis in a patient with mutations in the rabenosyn-5 gene. 67,68 In both cases, cellular vitamin B12 deficiency was documented. 68
Vitamin B12 Status in GD
The overlap of clinical manifestations of GD and vitamin B12 deficiency concerning neurological impairments suggests shared mechanisms of pathogenesis. One hypothesis is that lysosomal dysfunction in GD leads to functional deficiency of vitamin B12 by disrupting the uptake, intralysosomal degradation of transcobalamin, or the export of free vitamin B12 from the organelle into the cytoplasm, thereby compromising the downstream reactions of MS and MCM. Further, it is unknown whether the abnormal accumulation of glycosphingolipids, in particular N-acyl-sphingosyl-1-O-β-
The earliest assessment of vitamin B12 status in GD reported abnormally high levels of circulating TC, 26,29 which did not correlate with serum levels of vitamin B12 or any other B12-transport protein levels. It was suggested that increased TC levels in GD resulted from a general status of inflammation. Unfortunately, no other biomarkers of vitamin B12 status were measured. 69 Indeed, although holo-TC represents the bioactive fraction of vitamin B12 that is available for cellular uptake, 70 -73 this has limitations as a stand-alone marker of vitamin B12 status in that the majority of circulating vitamin B12 is bound to haptocorrin (80%), and thus, fluctuations in the serum level of holo-TC (representing 6%-20% of total serum vitamin B12 70 -73 ) may not be accurate marker of vitamin B12 sufficiency (reviewed by Djaldetti et al 74 ). For instance, low levels of holo-TC have been determined in patients with several disorders not featuring vitamin B12 deficiency. 75 -78 At present time, it is unknown whether and how holo-TC levels vary in various disease states, and thus, the diagnostic and prognostic value of holo-TC as a first-line test awaits further investigation.
A study performed within an Ashkenazi Jewish cohort of 85 untreated GD patients and 122 neighbor controls showed a high incidence of low-serum vitamin B12, elevated plasma homocysteine (Hcy), and methylmalonic acid (MMA); however, these findings were not statistically significant with respect to controls, due to an overall low vitamin B12 status among healthy Ashkenazi individuals. 23 The generally low vitamin B12 status in this Ashkenazi Jewish population has been suggested to arise from frequent blood donation that would exhaust blood and liver storages of vitamin B12 as well as to ethnical differences. 79,80 A questionnaire-based study identified a high incidence of neurological complaints in patients with nonneuronopathic forms of GD, with concomitant vitamin B12 deficiency and gammopathies. 81 Unfortunately, no laboratory measurements of marker metabolites Hcy and MMA were performed in these studies, which makes it difficult to ascertain the role of vitamin B12 status in this cohort of GD patients. 81
A 2-year prospective, longitudinal, observational cohort study involving 8 centers across 7 countries in Europe examined vitamin B12 status in adult patients with GD type 1 with (n = 17) and without (n = 86) polyneuropathy. 82 The study found statistically significant elevation of serum Hcy and MMA in patients with polyneuropathy compared to those without neuropathic impairment, with low–normal values of serum vitamin B12 in both groups. 82 However, both groups of patients displayed metabolite levels still within the normal range (MMA, <0.4 μmol/L; Hcy, premenopausal females, 6-15 μmol/L, males, and postmenopausal females, 8-18 μmol/L). 82 Importantly, both groups of patients had received ERT for at last 2 to 3 years. Table 1 summarizes the data that represent the largest examination of vitamin B12 status in GD to date. Patients with polyneuropathy had received a lower dose of ERT compared to patients without polyneuropathy. 82 In the absence of vitamin B12 biomarker (Hcy and MMA) values before ERT, it is difficult to state whether the reported values represent a partially corrected vitamin B12 metabolism. In sum, a disturbed vitamin B12 metabolism in GD is plausible, but the available data beg for additional investigation.
Demographics, Treatment, Vitamin B12 Status, and Clinical Manifestations of Adult GD Type 1 in a Cohort of European Patients.a,b,c
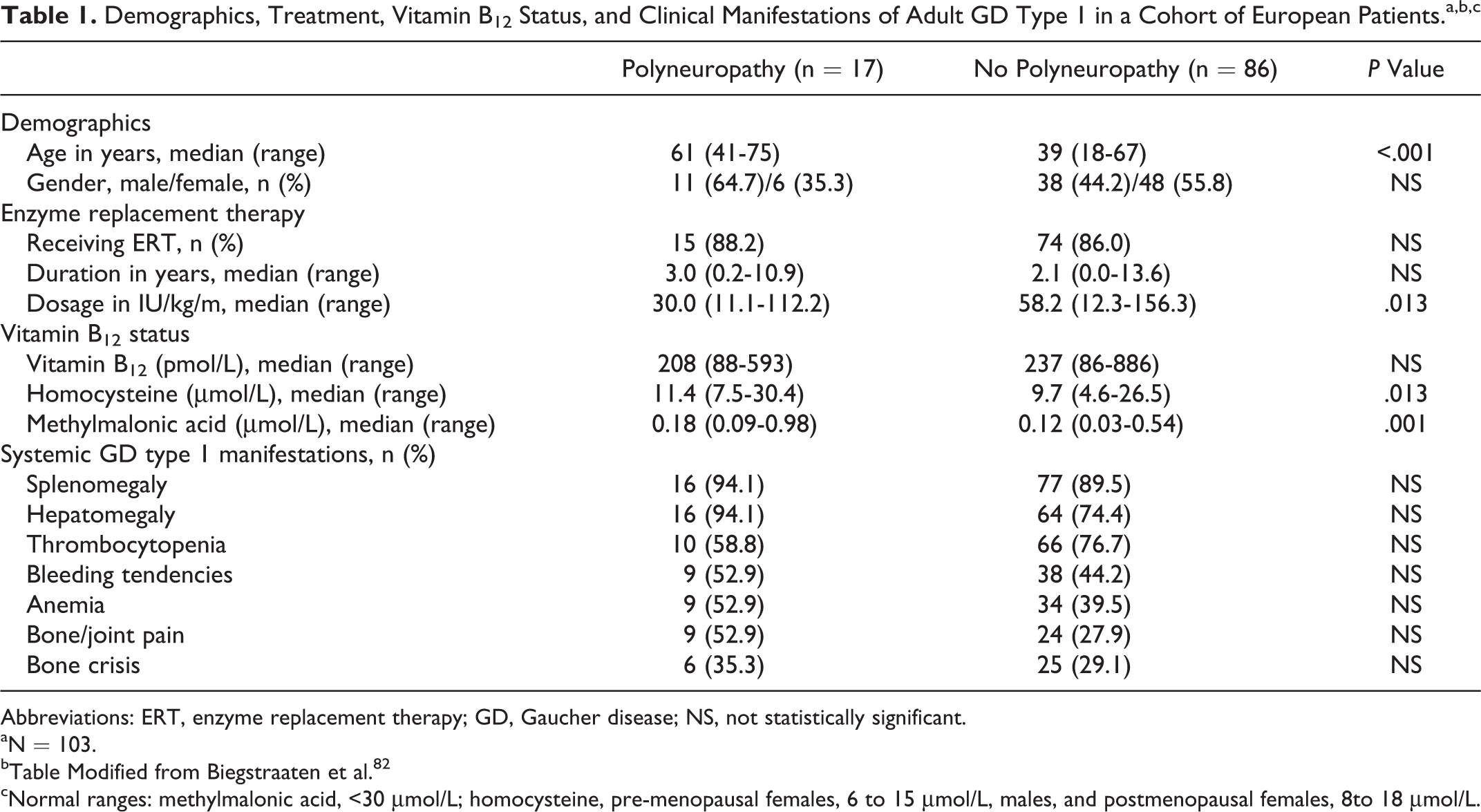
Abbreviations: ERT, enzyme replacement therapy; GD, Gaucher disease; NS, not statistically significant.
aN = 103.
bTable Modified from Biegstraaten et al. 82
cNormal ranges: methylmalonic acid, <30 μmol/L; homocysteine, pre-menopausal females, 6 to 15 μmol/L, males, and postmenopausal females, 8to 18 μmol/L.
Outlook
Lysosomal storage disorders and GD in particular are complex diseases affecting various facets of metabolism. The occurrence of vitamin B12 deficiency as a general manifestation of GD awaits further confirmation, but available studies point to disturbances in vitamin B12 metabolism that may originate from abnormal lysosomal metabolism or a general status of inflammation as it has been reported in 2 other human disorders, 73,74 not intrinsically related to vitamin B12 metabolism. It has been suggested that aging leads to increased impairments in lysosomal metabolism, and that this could explain the concomitant disturbances of vitamin B12 pathways often found in association with diseases featuring peripheral neuropathies, such as Alzheimer and Parkinson disorders. 83,84 This further supports the need for vitamin B12 supplementation in the aging population, beyond the known limitations in gastric absorption of the micronutrient, which also becomes less efficient with aging. Our revision of the available literature points to the importance of assessing vitamin B12 status prior to the initiation of ERT in GD patients, in order to establish involvement of this micronutrient on the onset of symptoms and possibly in its associated peripheral neuropathies.
Footnotes
Acknowledgments
L.H. wishes to acknowledge intramural support from the Department of Pediatrics, Medical Center, University of Freiburg, Freiburg, Germany.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
