Abstract
Glycogen storage disease type 1 (GSD1) is an inherited disorder caused by impaired glucose 6-phosphatase activity. This impairment translates into the inhibition of endogenous glucose production and the subsequent accumulation of cellular glucose 6-phosphate. Excess glucose 6-phosphate enhances glycolysis, increases the production of fatty acids, uric acid, and lactate, causes hepatomegaly due to glycogen and lipid accumulation, and finally results in liver tumor development. Although the exact mechanisms of tumorigenesis in patients with GSD1 remain unclear, GSD1 hepatocytes undergo a Warburg-like metabolic switch. The consequent hyperactivation of specific metabolic pathways renders GSD1 hepatocytes susceptible to tumor development, presumably by providing the building blocks and energy required for cell proliferation. In addition to this, enhanced apoptosis in GSD1 may promote mitotic activity and hence result in DNA replication errors, thereby contributing to tumorigenesis. Increased carbohydrate responsive element-binding protein (ChREBP) and mammalian target of rapamycin (mTOR) activity and impaired AMP-activated protein kinase (AMPK) function likely play key roles in these pro-oncogenic processes.
Keywords
Introduction
Glycogen storage disease type 1 (GSD1) is a rare genetic disease, due to an impairment in glucose 6-phosphatase (G6Pase) 1,2 activity. Glucose 6-phosphatase is a key enzyme for energy and metabolite production that catalyzes the conversion of glucose 6-phosphate (G6P) to free glucose, the final step in both glycogenolysis and gluconeogenesis. 3,4 Therefore, it is a crucial enzyme in the maintenance of blood glucose levels in the postabsorptive phase and upon fasting. The G6Pase complex is composed of 2 subunits, that is, a catalytic domain (G6PC) and a transport protein (G6PT). G6PT is expressed ubiquitously, whereas G6PC is exclusively expressed in the 3 gluconeogenic organs: the liver, kidneys and intestine. 3,4 G6PC mutations lead to GSD type 1a (GSD1a), 5 -7 whereas GSD type 1b (GSD1b) is caused by mutations in SLC37A4, the gene encoding G6PT. 2 Both GSD1a and GSD1b are characterized by hepatic, renal, and intestinal pathologies and exhibit largely the same metabolic symptoms. 8,9 Because G6Pase activity is impaired in patients with GSD1, blood glucose levels are low upon fasting, resulting in hypoglycemic episodes. On the other hand, cellular G6P storage and utilization are strongly increased, thereby activating specific metabolic pathways, including glycolysis, as well as uric acid, lactate, fatty acid, and cholesterol synthesis. Besides fasting hypoglycemia, the metabolic symptoms of GSD1 therefore involve hyperuricemia, lactic acidosis, hypertriglyceridemia, and hypercholesterolemia. Glucose 6-phosphatase-deficient hepatocytes are chronically stressed due to abnormal glycogen and lipid accumulation, which induce hepatomegaly and liver steatosis. 8,9 Similarly, patients with GSD1 and animal models exhibit nephromegaly, 8 -10 leading to tubular injury, glomerulosclerosis, and a progressive loss of renal function, characterized by albuminuria, proteinuria, and at the last stage, by renal failure. 11,12 Strikingly, almost all patients with GSD1 develop hepatocellular adenoma (HCA) during adulthood, with a 10% incidence of transformation to hepatocellular carcinoma (HCC). 13,14 Liver tumor formation in GSD1 induces significant morbidity and impairment of quality of life, and these patients frequently require partial hepatectomy or liver transplantation. 15,16 The European Study on GSD1 showed that HCA can be observed from 15 years of age on, and up to 75% of patients bear at least 1 HCA at the age of 25. 14 The molecular mechanisms of tumor development in GSD1 are unclear. It has been hypothesized that the early switch to a Warburg-like metabolism in nontumoral G6Pase-deficient hepatocytes promotes oncogenesis by providing substrates for cell proliferation, such as lipids and nucleotides. It has been proposed that tumor development in GSD1 may be a rectilinear process, characterized by the initial appearance of HCAs, which may subsequently transform into HCC. 17 To our knowledge, development of HCC in the absence of preexisting/coexisting HCA has not yet been reported for GSD1.
Because the primary consequence of GSD1 is a metabolic derangement, it is of interest to understand via what mechanisms perturbed intrahepatic metabolism may initiate and/or drive tumor formation. In this review, we discuss the metabolic reprogramming, stress-inducing, and cellular death pathways in G6Pase-deficient hepatocytes that may initiate and/or facilitate HCA and HCC development.
Tumor Metabolism
Tumor initiation and progression may be considered as a rather anarchical process; however, several metabolic pathways are very frequently hyperactivated in tumors. These pathways provide tumor cells with energy (ATP), carbon skeletons, lipids, amino acids, nucleotides, and other metabolites required for biomass gain, growth, and progression. 18,19
The Warburg Effect
In normal quiescent cells, ATP is derived mostly from glycolysis, subsequent Krebs cycle activity, and mitochondrial oxidative phosphorylation (OXPHOS). 20 This oxygen-dependent metabolic profile ensures an efficient ATP production.
On the contrary, in cancer cells or rapidly proliferating cells, metabolism is orientated toward increased glucose uptake and aerobic glycolysis, resulting in a high lactate production, independent of cellular oxygen availability. This metabolic shift is known as the Warburg effect. Initially, the Warburg effect was considered as the driving force for cancer development. 21 Nowadays, it is generally believed that this effect is rather the result of metabolic reprogramming induced by oncogenic mutations. Otto Warburg’s research and other studies suggest that cancer cells shut down their mitochondrial activity, hence decreasing OXPHOS, and concluded that a hypoxic tumor environment selects cells that are dependent on anaerobic metabolism. However, it has been confirmed that most cancers exhibit normal mitochondrial activity and normal rates of OXPHOS, suggesting that other mechanisms drive metabolic reprogramming of cancer cells toward aerobic glycolysis. 18 Several regulatory proteins, including hypoxia-induced factor 1α (HIF1α), 22 -24 avian myelocytomatosis viral oncogene homolog (MYC 24,25 ), and tumor protein p53 (P53 25,26 ), are known to promote this metabolic switch. Most tumors are not very well vascularized during initial growth and oxygen supply is therefore not optimal. However, the Warburg effect has also been observed in lung tumors 27 that have sufficient oxygen supply, indicating that the Warburg effect is not necessarily related to impaired oxygen availability. Moreover, in aerobic tissues, HIF1α is stabilized by high presence of lactate, 22 indicating that this pro-oncogenic protein also operates when oxygen supply is not limiting. The energy yield of the glucose catabolism by aerobic glycolysis is much lower (2 ATP/glucose) compared to mitochondrial OXPHOS (38 ATP/glucose), yet it can generate ATPs at a higher rate. However, high ATP synthesis rate associated with aerobic glycolysis may be of minor relevance to tumor growth, since energy alone is not enough for proliferation. More importantly, in order to increase their biomass and growth rate, proliferating cells need carbon skeletons, amino acids, lipids, and cholesterol to synthesize biomembranes and nucleotides. This accelerated metabolism results in increased reactive oxygen species (ROS) production. Even though ROS can be highly oncogenic and therefore promote tumor development, they can also render tumor cells vulnerable to oxidative and energetic stress. 28 In order to combat ROS, but also to increase anabolic activity, cancer cells increase nicotinamide adenine dinucleotide phosphate (NADPH) production via the pentose phosphate pathway (PPP), as well as other reducing equivalents. This allows not only the detoxification of cancer cells but also the synthesis of macromolecules required for proliferation. 29,30 Altogether, metabolic reprogramming allows the cells to maintain sufficient energy levels, but it also provides them with substrates for growth. One of the main players providing cancer cells with these substrates is pyruvate kinase (PK), the enzyme responsible for the conversion of phosphoenolpyruvate (PEP) to pyruvate and ATP, the final step of glycolysis. This enzyme exists under different isoforms (M1, M2, L, and R) and can form dimers or tetramers, which makes it the key switch between OXPHOS and aerobic glycolysis. Because the M2 dimer isoform has a low affinity for PEP, glycolytic metabolites upstream of PEP accumulate when this isoform is predominant in the cell, and metabolism is redirected to nucleotide, lipid, and amino acid synthesis. 31 This phenomenon is observed in almost every cancer type. 32,33 Many important signaling pathways, such as MYC, 34 HIF1α, 33,35,36 STAT3 (for Signal Transducer and Activator of Transcription), 36 and Epidermal Growth Factor Receptor (EGFR), 37 are responsible for the upregulation of pyruvate kinase isoform M2 (PKM2) in cancer cells. Interestingly, recent studies indicate that PKM2 is not only involved in the regulation of glucose metabolism but also plays a role in cancer metastasis in HCC. 38,39
Pro-Oncogenic Changes in Lipid Metabolism
Nonalcoholic fatty liver disease (NAFLD) 40 is commonly used to describe liver pathologies characterized by abnormally high lipid content in the liver, in the absence of alcohol consumption. It is mostly observed in developed countries and frequently associates with obesity, diabetes, and metabolic syndrome. Multiple studies have established a link between NAFLD and increased risk of HCC. 41,42 Multiple factors, such as increased lipogenesis, oxidative stress, cytokine-induced signaling pathways, and environmental and genetic components, have been proposed to contribute to liver tumor formation in patients with steatosis. 43,44 Increased de novo lipogenesis (DNL) requires high concentrations of NADPH, rendering NADPH a limiting factor in tumor growth. Nevertheless, as mentioned earlier, an increase in PPP activity is frequently observed in tumors, thereby facilitating NADPH production. 29 The activity of malic enzyme also generates NADPH synthesis, and the repression of its expression has been shown to inhibit tumor progression. 45,46 Furthermore, increased DNL promotes malonyl-CoA production, which in turn inhibits carnitine palmitoyltransferase 1 (CPT1) activity, thereby reducing intramitochondrial fatty acid availability for β-oxidation. Thus, under conditions of increased DNL, the degradation of fatty acids is reduced, which further enhances steatosis. 43 High lipid concentrations induce the production of ROS in hepatocytes. Reactive oxygen species subsequently lead to lipid peroxidation, inflammatory cytokine (interleukin [IL] 6, 47 tumor necrosis factor α 48 ) release, and DNA and protein damage. For example, the trans-4-hydroxy-2-nonenal, a product of lipid peroxidation, causes mutations in the tumor suppressor P53 in HCC. 49,50 In addition to this, ROS-induced DNA damage can also result in oncogene activation and/or tumor suppressor inactivation, thereby driving carcinogenesis. 28 Once neoplastic lesions are formed, accumulated lipids are highly favorable for tumor growth, as they are crucial for the assembly of biomembranes for new tumor cells. A wide range of DNL-inhibiting drugs are currently being developed to decrease hepatic stress and even halt tumor development. Most of these drugs target the fatty acid synthase (FASN), a key enzyme in DNL. 51,52 As this enzyme is frequently overexpressed in cancer cells, its inhibition has been proven successful to attenuate cancer progression and metastasis. 51,52
Nucleotide Provisions
Besides synthesizing biomembranes, tumor cells need to produce nucleotides for DNA replication during cell division. In order to increase nucleotide synthesis, the high glycolytic flux associated with the Warburg effect is directed toward PPP. 53 PKM2 is mainly responsible for the redirection. 32 The PPP produces ribose 5-phosphate, which serves as a substrate for de novo nucleotide synthesis. 54 Interestingly, yet not surprisingly, HIF1α 55 and C-MYC 56 also promote nucleotide production by increasing the PPP activity, thereby likely potentiating tumor development and growth.
Metabolic Reprogramming in GSD1 Hepatocytes
Hepatocytes have different physiological roles and converge many metabolic pathways. They are involved in protein, glucose, and lipid metabolism, and their functioning has systemic effects. Glycogen storage disease type 1 hepatocytes display an abnormal metabolic phenotype, primarily caused by G6P accumulation. Metabolic pathways that use G6P as a substrate, such as glycolysis, DNL, glycogen synthesis, and degradation, are hyperactivated in GSD1 livers. 57,58
Hyperactive Glycolysis and Warburg-Like Metabolism
It has been established that excess G6P forces metabolism toward glycolysis 57 -59 in G6Pase-deficient nontumoral hepatocytes. Increased glycolysis leads to increased pyruvate levels and subsequent lactate production. Lactate production due to enhanced glycolysis explains the lactic acidosis observed in both GSD1 animal models and in patients with GSD1. 60,61 This metabolic phenotype resembles the Warburg effect in cancer cells (Figure 1). In normal hepatocytes, lactate can be converted back to glucose in the liver via the Cori cycle, thereby preventing lactic acidosis. On the contrary, it has been hypothesized that G6Pase-deficient hepatocytes cannot convert lactate back to glucose, since they cannot dephosphorylate G6P, the final step of the Cori cycle. An impaired Cori cycle in GSD1 may therefore lead to a further increase in circulating lactate concentrations. Although the expression of the liver isoform of PK (PKLR) is markedly increased in GSD1, 62,63 an induction of PKM2 has not yet been reported in G6Pase-deficient hepatocytes. However, from a therapeutic perspective, it would be interesting to assess its enzymatic activity in GSD1, as many PKM2-inhibiting drugs are currently being developed. In case PKM2 is activated in GSD1, such drugs may be effective to reduce nucleotide, lipid, and amino acid synthesis, 31 and hence perhaps impair tumor development in GSD1. 64,65
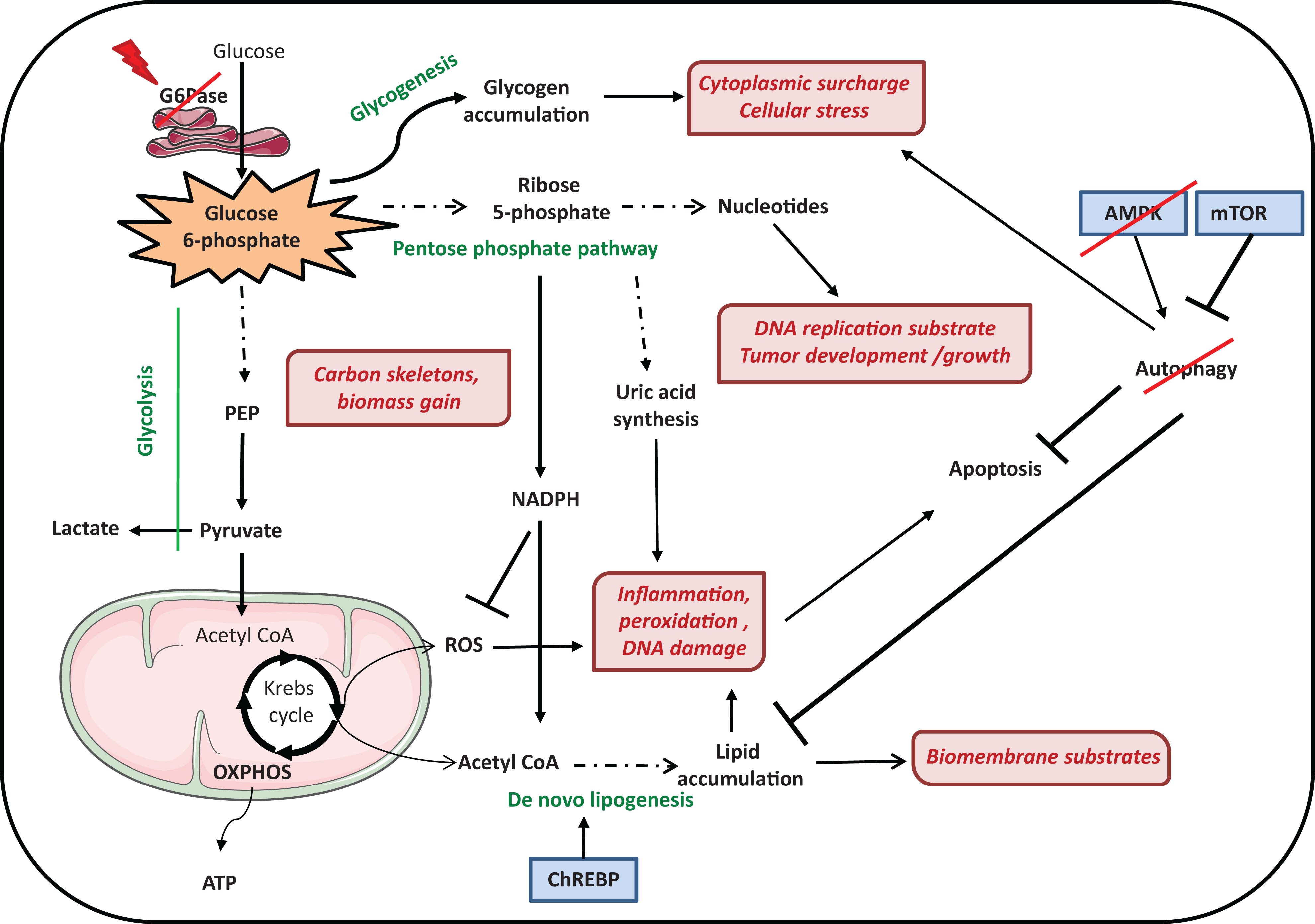
Metabolic deregulation and pro-tumorigenic pathways in the GSD1 liver. Images were drawn by Sevier Medical Art.
Increased Hepatic Lipid Synthesis Contributing to Liver Steatosis
In GSD1, increased pyruvate production via glycolysis due to excess G6P results in increased production of acetyl-CoA, which subsequently enters the Krebs cycle. Besides generating energy via OXPHOS, citrate produced in the Krebs cycle can be transported to the cytosol, converted back to acetyl-CoA, and serve as a substrate for the synthesis of fatty acids and cholesterol. Bandsma et al observed a 40-fold increase in de novo synthesized palmitate in very low density lipoprotein (VLDL) from patients with GSD1 (Figure 1), in parallel to a 7-fold increase in cholesterol synthesis and a slower conversion of VLDL to low density lipoprotein (LDL). 66 An increased expression of genes involved in mitochondrial citrate export and DNL, such as ATP citrate lyase (ACLY), acetyl CoA carboxylase (ACACA), FASN, sterol CoA desaturase (SCD1), and fatty acid desaturase 1 and 2 (FADS1 and 2), has already been confirmed in GSD1 livers. 58,59 These enzymes are physiologically induced in the fed state by elevated insulin and glucose levels. Insulin and glucose act via 2 transcription factors, that is, sterol regulatory element binding protein 1c (SREBP1c) and carbohydrate responsive element binding protein (ChREBP). 67 In a mouse model of GSD1b, the induction of glycolytic and lipogenic genes in the liver was found to be mediated by ChREBP. 62 Increased DNL likely represents an important pathophysiological process in GSD1 hepatocytes, as it contributes to the accumulation of lipids and potentially lipotoxicity in the liver, causing NAFLD-like complications in both patients with GSD1 and GSD1 animal models. 60,68 Several studies suggested that dietary treatment can partially reduce lipid levels in plasma and liver, although hepatic lipid accumulation cannot be completely alleviated. 68,69 The β-oxidation pathway, responsible for fatty acid degradation, is likely reduced in GSD1, as it was shown that the expression of peroxisome proliferator-activated receptor α is significantly decreased in hepatocyte-specific G6PC null mice. 70 As mentioned earlier, excessive malonyl-CoA production caused by DNL induction may result in the inhibition of CPT1 and a consequent reduction in fatty acid β-oxidation. Finally, a decreased activity of AMP-activated protein kinase (AMPK) in GSD1 nontumoral hepatocytes 71 may contribute to impaired fatty acid oxidation and increased fatty acid and cholesterol synthesis 72 (Figure 1). AMP-activated protein kinase regulates these processes by decreasing malonyl-CoA production via ACC inhibition and by controlling SREBP1 73 and ChREBP 74 activities.
As mentioned earlier, ChREBP is the key transcription factor responsible for the elevated hepatic lipogenic gene expression in GSD1a and GSD1b mouse models. 62,71 Increased levels of ChREBP have already been shown to promote tumorigenesis. 75,76 This transcription factor not only promotes lipogenesis but also increases glucose uptake and glycolysis, cell proliferation, and glucose-dependent de novo nucleotide synthesis in hepatocytes. 77 Carbohydrate responsive element binding protein–deficient cells exhibit increased OXPHOS, suggesting that ChREBP is yet another potential metabolic switch in GSD1, deviating the metabolism from OXPHOS toward aerobic glycolysis. In the same study, ChREBP deficiency was shown to activate P53-dependent cell cycle arrest, indicating that this transcription factor interferes with tumor suppression.
Altogether, as a result of metabolic reprogramming and deregulated intracellular signaling, GSD1 livers present the full spectrum of NAFLD, including increased lipid synthesis, hepatic steatosis, and lipotoxicity, which are associated with tumorigenesis.
Enhanced Nucleotide Metabolism
Because G6P serves as a substrate for the PPP, it can be assumed that its accumulation in GSD1 promotes de novo nucleotide synthesis. Although an actual increase in PPP activity in GSD1 has not yet been reported, an increased flux through this pathway potentially provides the nucleotides required for neoplastic cellular proliferation and reducing equivalents for DNL. On the other hand, when inorganic phosphate is depleted, for example, due to the increased retention of phosphate in G6P, cells start to degrade nucleotides, resulting in uric acid production. 78 Hyperuricemia is a very well-documented feature of GSD1 animal models and patients with GSD1. 14,60 Uric acid has pro-inflammatory properties contributing to tumor development and growth. 79 Nevertheless, patients with GSD1 are frequently treated with drugs to lower circulating uric acid concentrations, hence protecting against uric acid–induced inflammation. Because these treatments do not prevent tumor development in patients with GSD1, it seems unlikely that hyperuricemia is the sole oncogenic factor in GSD1.
Cellular Defense and Death Mechanisms in GSD1 Livers
Oxidative Stress
Mitochondrial and peroxisome activities generate ROS that are degraded by cellular antioxidant enzymes such as catalase, superoxide dismutase, and glutathione peroxidase. Failure to neutralize excess ROS results in disturbed redox levels, causing serious tissue injury. Hepatic steatosis renders the liver more susceptible to ROS. 41 Reactive oxygen species not only damage intracellular molecules but also alter the activities of cellular signaling pathways. It has been reported that markers of oxidative stress and antioxidant defense systems are both increased in the plasma of patients with GSD1, 80 while no data are currently available on ROS levels in G6Pase-deficient livers. Accumulated ROS in G6Pase-deficient hepatocytes can react with the nucleic acids, proteins, lipids, and other metabolites, thereby impairing organelle functions and inducing membrane damage as well as chromosomal instability and mutations. All of these processes are favorable to tumor development 28 (Figure 1).
Autophagy
Reactive oxygen species–induced cellular stress is frequently accompanied by mitochondrial dysfunction, because these organelles are not only the generator but also a target of ROS. 81 Mitochondrial turnover via autophagy, also referred to as mitophagy, is tightly linked to oxidative stress. 82 Autophagy enables the elimination of nonfunctional proteins, organelles, and metabolites, providing the cell with energy and recycled metabolites for reuse. The autophagic substrates can be, in part, a consequence of increased ROS concentrations. The autophagy-related (ATG) proteins initiate the formation of and serve as building blocks for autophagic vesicles. This initiation and progression is regulated by an orchestrated network that includes the AMPK and the AKT/mTOR (for Serine/Threonine Kinase 1/mammalian target of rapamycin) pathways, which are both key proteins involved in energy and nutrient sensing. 83 Although AMPK activation promotes autophagy, mTOR is known to suppress it.
Recently, it was reported that autophagy is decreased in G6Pase-deficient livers. 71 This study showed that AMPK activity was impaired, while mTOR was activated, translating in decreased autophagy (Figure 1). Furthermore, it was shown that mTOR inhibition by rapamycin restored autophagy and reduced hepatic lipid and glycogen content in GSD1 livers. Autophagy depletion in the early stages of cancer development is a frequently observed phenomenon that generates a tumorigenic environment, as free radical accumulation, nonfunctional proteins, and organelle persistence increase chromosome instability. 84,85 For example, a mouse model of impaired autophagy induced by ATG5 ablation developed HCA, suggesting a tumor suppressive role for autophagy. The HCAs from these animals showed damaged mitochondria, oxidative stress, and genomic damage responses. 86 Almost the same phenotype, including liver tumors, was observed in ATG7-deficient mice. 86 Thus, autophagy appears to have an important anti-oncogenic role in the liver, and its impairment in a pretumor stage may facilitate oncogenesis in GSD1 livers as well.
Metformin is a drug that is commonly used to treat diabetes. 87 This hypoglycemic agent reduces circulating glucose and lipid levels and induces autophagy by activating AMPK. 73 Aside from its antidiabetic properties, metformin was shown to exert anticancerous actions as well. 88 Although it is tempting to evaluate whether metformin treatment activates autophagy and subsequently impedes tumorigenesis in the GSD1 liver, a putative decrease in blood glucose levels upon metformin treatment in these patients poses a potential risk. However, metformin acts mainly on the liver and reduces hyperglycemia by inhibiting hepatic glucose production. 89 Given that G6Pase-dependent glucose production is absent in GSD1, the risk for metformin-induced hypoglycemia in these patients may be negligible, making this treatment appealing for the prevention/treatment of HCA in patients with GSD1.
Apoptosis
Once autophagy fails to improve the metabolic and energy state of the cell, molecular mechanisms induce a switch from autophagy to apoptotic or even necrotic cell death. 90 G6Pase–deficient mice display increased apoptosis in the liver 57 (Figure 1). Caspase3/7 protein levels were significantly increased in the livers of these mice, implying a Fas-mediated apoptosis. The authors considered that apoptosis in GSD1a mice was activated through glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and further promoted by steatosis-derived inflammation. 57 The expression of GAPDH, an enzyme that is primarily involved in activated glycolysis, was elevated as well. Interestingly, GAPDH has recently been associated with increased apoptosis. 91,92 Increased apoptosis in G6Pase-deficient cells results in low viability of isolated primary hepatocytes (A. Gautier-Stein, PhD, unpublished data 2012). Although apoptosis is commonly regarded as an anti-oncogenic mechanism, it has been shown that high apoptotic rates can also promote tumorigenesis. 93 To retain proper functioning of the liver, apoptotic hepatocytes must be replaced rapidly. Thus, an increase in apoptosis in the G6Pase-deficient liver may trigger hepatocyte proliferation to compensate for hepatocytes loss. Rapid and constant cell division and growth of mitotic G6Pase-deficient hepatocytes may consequently induce DNA replication errors and tumor formation.
Necrosis
Hepatocellular necrosis has also been observed in G6Pase-deficient livers. 94 Necrotic foci, neutrophil infiltration, and elevated expression of chemokines were documented, indicative of liver injury in mice and patients bearing HCA but also in liver material in a pretumor stage. Glycogen 95 and lipid accumulation 96 are known to induce inflammation. Taken together, the origin of this inflammatory response needs to be investigated, as it may significantly contribute to the tumor development in GSD1.
GSD1 Metabolism Drives Adenoma Formation: Similarities With HNF1A-Mutated Tumors
Genome sequencing and subsequent classification of HCAs from non-GSD1 patients has identified 3 main groups of mutations associated with HCA. 97 In HCAs from the first group, hepatic nuclear factor 1α (HNF1α), a transcription factor involved in glucose and lipid metabolism, is mutated. Hepatic nuclear factor 1α is considered as a tumor suppressor and a key factor that maintains hepatocytes in a differentiated state. 98,99 In HNF1A-mutated HCAs, the expression of the fatty acid–binding protein 1 (FABP1) gene, a direct target of HNF1α, is downregulated, leading to steatosis and lipotoxicity. 100 Tumors bearing a HNF1A mutation have an increased proliferation rate due to the subsequent activation of the Phosphoinositide 3-kinase/AKT pathway. 101,102 The second group represents inflammatory HCAs. These HCAs are characterized by mutations that activate the IL-6 and JAK (Janus kinase)/STAT pathways and are the most prevalent HCAs, representing 50% of all HCAs. 103 The third group is β-catenin (CTNNB1)-mutated HCAs. Hepatocellular adenomas with an activating mutation of β-catenin represent around 10% to 15% of all HCAs 103 and are associated with poor prognosis, since they represent a great risk of malignant transformation. 104
In order to investigate the origin of tumorigenesis in GSD1, HCAs from patients with GSD1 were subjected to gene expression and DNA sequencing analysis. This analysis showed that 52% of the HCAs bared inflammatory mutations, 28% were CTNNB1 mutated, and 20% were unclassified. 59 Intriguingly, none of the GSD1 HCAs were HNF1A mutated, whereas all of the GSD1 HCAs, even the ones in the inflammatory and β-catenin groups, had a decreased expression of FABP1. Since HNF1α is the main regulator of FABP1 expression, one could infer that HNF1α could be decreased in all of the GSD1 HCAs. As HNF1α regulates G6PC and G6PT 105 -107 expression, HNF1α and G6Pase-mutated hepatocytes may display a similar phenotype. Consequently, comparison of GSD1 nontumoral liver tissue and HNF1A-inactivated HCAs revealed similarities with regard to the expression of glycolytic, gluconeogenic, and lipogenic enzymes. In both GSD1 nontumoral tissue and HNF1A-invalidated HCAs, gluconeogenesis was repressed, and glycolysis and lipogenesis were induced. 100 Decreased Hnf1a messenger RNA expression was observed in the liver-specific G6pc knockout mice as compared to their wild-type littermates (M. Gjorgjieva, PhD student, and F. Rajas, PhD, unpublished data 2015). To summarize, GSD1 nontumoral liver displays striking phenotypic similarities with HNF1A-inactivated HCAs, indicating that even in the pretumor stage, GSD1 livers exhibit a tumor-like metabolism.
Conclusion
Glycogen storage disease type 1 is a complex metabolic disease that exhibits all the hallmarks of NAFLD. Liver steatosis and hepatic glycogen accumulation are already present during infancy, resulting in hepatomegaly. The hepatic accumulation of lipids and glycogen induces severe stress and causes chronic inflammation and ROS production, which potentially contributes to the development of liver tumors. The abnormal intrahepatic G6P content activates metabolic pathways that support tumor growth, such as aerobic glycolysis, lipid synthesis, uric acid and lactate production, glycogen storage, and nucleotide synthesis. Moreover, autophagy is impaired, which may further promote accumulation of nonfunctional proteins and organelles, and possibly translates into cellular stress and DNA and protein damage. ChREBP and mTOR activation and AMPK inhibition likely represent key molecular events that orchestrate metabolic reprogramming in GSD1. The rates of apoptotic cell death in advanced GSD1 are elevated, probably due to a switch from autophagy to apoptosis, as well as increased GAPDH concentrations. Increased levels of ROS and inflammatory cytokines may also contribute to the increase in apoptosis. As increased hepatocellular apoptosis may trigger cell division to repopulate the liver, continuous mitosis in G6Pase-deficient hepatocytes may lead to DNA replication errors, hence inducing HCA development. In addition, hepatocyte necrosis represents another mechanism activated that likely contributes to inflammation and cell death in GSD1 livers. Finally, by comparing HNF1α-deficient HCAs and G6Pase-deficient nontumoral tissue, many similarities were found, despite a lack of HNF1A mutations. It was suggested that both groups exhibited HNF1α deficiency, accompanied by lipotoxicity, due to the reduced FABP1 expression. HNF1α is considered as a tumor suppressor, and therefore, its impairment in G6Pase-deficient hepatocytes could lead to dedifferentiation and further susceptibility to tumor development. 101,108
These data accentuate the need for strict metabolic control of patients with GSD1 in order to slow down the tumorigenesis process. The consumption of “fast sugars” such as fructose and galactose represents an important source of the substrate for the activation of glycolysis, glycogen synthesis, and DNL, therefore potentiating tumor formation. Even though a strict dietary regimen does not appear to fully prevent HCA/HCC formation, the European study of GSD1 has clearly shown an increase in life expectancy in patients who followed the nutritional guidelines. 14
Altogether, different metabolic and cellular adaptations render GSD1 hepatocytes prone to tumor formation and growth. Although the exact contribution of these different features needs to be mechanically elucidated, nontumoral G6Pase-deficient hepatocytes may be considered as preneoplastic cells that develop to GSD1 HCA, which in some cases even progress to HCC.
Footnotes
Acknowledgments
The authors would like to thank the “Ligue Nationale Contre le Cancer” and the “Ligue Régionale contre le cancer” for the financial support attributed to the project for analyzing the mechanisms involved in GSD1 hepatic tumor development.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Maaike H. Oosterveer holds a Rosalind Franklin Fellowship from the University of Groningen. The research is financially supported by the “Ligue Nationale Contre le Cancer,” “Ligue Régionale contre le cancer,” and “Stichting Metakids.”
