Abstract
Anderson-Fabry disease (AFD) is a rare inherited X-linked disease, caused by mutations of the gene encoding the α-galactosidase A enzyme, that leads to a deficiency or absence of its activity with consequent accumulation of globotriaosylceramide (Gb3) and other glycosphingolipids in the lysosomes of several cells types in the organism, mainly the endothelial, nervous system, cardiac, and renal cells. Its heterogeneous and nonspecific presentation, similar to other common pathologies, delays the diagnosis and leads to incorrect therapy. In the presence of attenuated phenotypes with predominant involvement of an organ, it is even harder to identify patients with AFD. It is highly important to be aware of this diagnosis, since enzyme replacement therapy is currently available. This review aims to approach the clinical manifestations of AFD and the phenotypes related to the differential diagnosis for each manifestation and the frequency of follow-up recommended.
Keywords
Introduction
Anderson-Fabry disease (AFD), also known as Fabry disease or angiokeratoma corporis diffusum, is an X-linked, rare, progressive, and systemic lysosomal storage disorder. Pathogenic mutations affecting the GLA gene that encodes α-galactosidase A enzyme cause absent or markedly deficient activity of this lysosomal enzyme leading to a lysosomal accumulation of globotriaosylceramide (Gb3) and other glycosphingolipids in a wide range of cells types, especially in endothelial, nervous system, cardiac, and renal cells. 1 -3
More than 550 mutations of GLA gene have been described. Most of them are private (each family has its own mutation), and about 3% to 10% of the patients have de novo mutations (mutations that are not present in their parents). 4 Even in the same family, clinical manifestations are heterogeneous and can vary widely from no symptoms to the classical clinical picture, with intermediate states affecting one or more organs. 2,5 -7 Residual activity of α-galactosidase A enzyme, together with genetic, epigenetic, and environmental factors, seems to affect the phenotype. Although some genotype–phenotype correlations are known, for example, null mutations in males associated with classic phenotype, in general, the genotype–phenotype correlation is poor in FD. 8
The prevalence of AFD ranges from 1:40 000 to 1:117 000 in the United States and Australia, 9 and although more common in Caucasians, all races can be affected by the disease. These data likely underestimate the true prevalence of the disease, since some newborn screenings conducted in North Italy, Taiwan, and Missouri showed prevalence of between 1:1250 and 1:3100 (most of which are associated with attenuated/late onset phenotypes). 10 -12
The classical phenotype affects mainly hemizygous males during childhood/adolescence. Female patients can be as severely affected as males, although more frequently later in life and with an insidious course of the disease, which could be related to X-chromosome inactivation. 2,7,8,13,14
In adulthood, cardiac, renal, and cerebrovascular manifestations are prominent. 14 Some patients present attenuated phenotypes characterized by later onset and predominant commitment of one organ (cardiac, renal, and cerebrovascular-attenuated phenotypes). 2,15
Since AFD clinical manifestations are nonspecific and common to other frequent pathologies such as diabetes, hypertension, and atheromatosis (phenocopies), the diagnosis is frequently established later in life: 13.7 years in male and 16.3 years in female patients after initial symptoms, 16 earning the title of “the new great impostor,” previously reserved to syphilis. 15
Compared to the general population, survival of untreated patients with AFD is reduced by about 20 years in males and 15 years in female patients. 17,18 Chronic kidney disease (CKD)—a prominent feature of the classical form of the AFD—was the main cause of premature death before the advent of dyalisis. 2 After the introduction of renal replacement therapy, cardiovascular events became the leading cause of death. 19 A 10-year study documents the long-term effectiveness of ERT in patients with classic AFD. However, ERT did not completely prevent the occurrence of severe clinical events, and stroke was most frequently reported in patients under 40 years of age. In fact, ERT did not prevent the occurrence of cerebral white matter lesions, and the effect of ERT on the risk of consecutive strokes remains unknown. Recombinant lysosomal enzyme has not been shown to cross the blood–brain barrier.
Renal disease progression after 10 years under ERT seems to be related, at least in part, to the severity of the disease before treatment, suggesting that ERT should be initiated before the development of significant glomerulosclerosis and proteinuria. 20 Similarly, patients initiated on agalsidase beta at a younger age had a favorable cardiac treatment response. 21
Enzyme replacement therapy has been associated with a delay in the progression of kidney disease, reduction in left ventricular mass, and improvement in neuropathic pain, with consequent improvement in quality of life. 22,23 The most favorable treatment responses were observed in younger patients who had less organ damage at the beginning of treatment. This underlines the importance of early diagnosis and treatment to prevent disease progression and irreversible damage.
This review aims to approach the clinical manifestations of AFD and its several phenotypes, addressing the major differential diagnoses for the different manifestations and recommendations for follow-up of patients with AFD.
Methods
Extensive bibliographic review was conducted using the PubMed and Clinical Key databases on the AFD by a group of experts from nephrology, neurology, cardiology, and internal medicine. The search terms used were “Fabry disease,” “nephropathy,” “hypertrophic cardiomyopathy,” “stroke,” “cerebrovascular,” “polyneuropathy,” “angiokeratoma, chronic diarrhea,” “persistent abdominal pain,” “acroparesthesia,” and “hypohidrosis.” After analysis of all relevant publications, consensus meetings were held with all the coauthors.
Clinical Manifestations
The progressive accumulation of Gb3 leads to lysosomal and cellular dysfunction, triggering an inflammatory response, leading to fibrosis and irreversible end-organ damage. 2 There is not any defined sequence order for organ involvement and disease progression. Therefore, the affected organs should be evaluated, treated, and monitored separately. 2
The classical picture of AFD is characterized by the appearance of angiokeratomas, acroparesthesias, and gastrointestinal symptoms during childhood/adolescence. 2,13,14 Angiokeratomas, one of the early signs of AFD, are purplish punctate lesions that do not disappear with vitropression. They may gradually increase in number and size with age and are predominantly distributed on the hips, back, belly, and buttocks. They can also appear on the oral mucosa and conjunctiva. 2 Heat and/or cold and physical exercise intolerance, due to hypo- or hyperhidrosis, are some other nonspecific symptoms, which may start during child and teenage with exacerbations in the beginning of the adult life. 24,25
The diagnosis may be confirmed by determining α-galactosidase A activity and/or by sequencing of the GLA gene. In male, the determination of the α-galactosidase A activity is the initial laboratory method to confirm diagnosis. Male patients with classic phenotype usually have α-galactosidase A activity <5% of normal, which is diagnostic, but in those with attenuated phenotype, the residual enzymatic activity could be considerable, requiring GLA gene sequencing for definitive diagnosis. 7 Likewise, female patients (heterozygotes) can have significant levels of enzyme activity and, for that, it’s a mandatory GLA gene sequencing to screen mutations in women. 2
GLA gene mutations include missense and nonsense mutations, small deletions or insertions, and large gene rearrangements. Some mutations, for example N215S, are associated with a residual enzyme function and an attenuated phenotype. The pathogenicity of novel gene alterations must always be evaluated, and it is important to be aware that some mutations are nonpathogenic, such as the inert exonic polymorphism p.D313Y substitution, associated with low plasma enzyme activity measure but with high residual lysosomal enzyme activity in vivo and no pathologic excretion of urinary Gb-3. The p.D313Y substitution is an example of α-Gal A pseudodeficiency. 8,26
Cardiac Involvement
Cardiac manifestations occur in 40% to 60% of the patients. Schiffmann et al reported that 49% of males and 35% of females had an event by a mean age of 36.2 and 44.4 years, respectively, and the leading cause of death in AFD is cardiovascular. 3,27 –29 Cardiac involvement results from the accumulation of Gb3 in the cardiomyocytes, vascular endothelium, vascular smooth muscle cells, fibroblasts, conduction system, and valve apparatus. 30 Given that Gb3 deposits constitute only about 1% of the myocardial mass, additional mechanisms such as inflammation, neurohumoral dysregulation, collagen deposition, and oxidative stress have been proposed to explain the severity of hypertrophy. 30
Anderson-Fabry disease may cause left ventricular hypertrophy (LVH), aortic and mitral valves insufficiency, conduction abnormalities, coronary artery disease (mainly microvascular), aortic root dilatation, and right ventricular hypertrophy. 31,32 These changes may be clinically silent or cause angina, dyspnea, palpitations, or even syncope, usually in the third to fourth decade of life in men and a decade later in women. 31 In more advanced stages of the disease, ventricular chamber enlargement and contractile dysfunction may occur. 2 The attenuated cardiac phenotype, in which cardiac manifestations are predominant, is more common among women 33 with presentation between the fifth and eighth decades of life. These patients may also have proteinuria. 6
Left ventricular hypertrophy is a key feature of AFD cardiomyopathy and it is observed in about 50% of men and 33% of women affected by the disease. 34 In women, the loss of contractile function and development of fibrosis, which can be shown by late gadolinium enhancement in cardiac magnetic resonance imaging (cardiac MRI), often occurs without evidence of hypertrophy, while in men, LVH often precedes fibrosis and ventricular dysfunction. 34 It is estimated that the prevalence of AFD is set between 0.9% and 3.9% in men and 1.1% to 11.8% in women with LVH. 35 Echocardiography is the complementary diagnostic test recommended for LVH evaluation. 36 Usually, hypertrophy is concentric, but it can also be asymmetric (with predominant septal involvement) and rarely with outflow tract obstruction of the left ventricle (more common in sarcomeric hypertrophic cardiomyopathy). In 40% of the cases, papillary muscles and right ventricle hypertrophy are observed. 33,37 The evaluation of myocardial contraction and relaxation velocities by tissue Doppler allows an early diagnosis of diastolic dysfunction even before the appearance of ventricular hypertrophy. 38,39 The strain/strain rate evaluation in the echocardiogram enables the identification of regional fibrosis, similar to the late gadolinium enhancement in the cardiac MRI. 40
In the Fabry Registry, from the 1448 nontreated patients, 14% of men and 20% of women developed ventricular arrythmias. 41 These arrhythmias were responsible for the increase in early morbidity and mortality of this disease. In early stages of the disease, the electrocardiogram shows a typical short PQ interval. With the disease progression, LVH voltage criteria and repolarization abnormalities appear that can be related to left ventricular overload and ischemia progressing to prolonged PR interval and more advanced degree of atrioventricular block and ventricular arrhythmias, including ventricular tachycardia, which can cause sudden cardiac death.
Although angina is common in AFD, the incidence of acute coronary syndromes is low. Myocardial ischemia is the result of various pathophysiological changes, but atherosclerosis is not the main one. Microvascular coronary disease, endothelial dysfunction, autonomic dysfunction, and dysregulation of vascular reactivity with vasospasm phenomena cause imbalance between oxygen supply and consumption in the hypertrophied myocardium and are mainly responsible for myocardial ischemia. 13
The cardiac valves, mainly the mitral valve, could show a diffuse thickening that can be observed early in childhood. 13 The aortic root dilatation is more frequent in male patients, although surgical repair is rarely need. 13
Renal Involvement
Gb3 deposits accumulate in interstitial, endothelial, epithelial tubular, and glomerular cells. 13 The first manifestations of renal injury and dysfunction are tubular isosthenuria and hyperfiltration. 5,8 With the increasing accumulation of deposits, proteinuria and a progressive decline in glomerular filtration rate (GFR) appear, generally in the second to third decade of life, in classically affected males. Urinary Gb3 levels can be used as a diagnostic and a follow-up marker after enzyme replacement therapy, but there is no evidence of its value in ERT efficacy evaluation or renal prognostic B. Glomerular filtration rate can be measured by creatinine or cystatin-C, a protein produced by all nucleated cells and freely filtered by the glomerulus. Torralba-Cabeza et al suggested that cystatin-C concentration is a superior and more sensitive marker than serum creatinine in detecting early renal involvement in patients with AFD of both genders. 42 The same results were published by Feriozzi et al in patients with Fabry nephropathy under ERT. 43
The presence of albuminuria/proteinuria is one of the first signs of AFD nephropathy and is the current gold-standard biomarker in Anderson-Fabry nephropathy. Proteinuria may vary from trace to nephrotic and, in 18% of cases, are associated with nephrotic syndrome. 14 However, it is important to notice that the absence of proteinuria does not exclude nephropathy, which, in these cases, may be of ischemic nature due to impairment of renal arteries. 44
The progression of CKD in AFD is comparable to diabetic nephropathy, with a GFR average decrease, in men of 12 mL/min/yr. 16 The progression toward terminal CKD is more frequent in males (8:1) and usually occurs on the fourth or fifth decade of life in classical phenotypes. 27
To both genders, proteinuria is the most important prognostic for renal disease progression. Besides proteinuria, the male gender, the degree of glomerulosclerosis, a higher baseline creatinine with estimated GFR bellow 60 mL/min/1.73 m2 in untreated patients, and hypertension are other risk factors for renal disease progression. 30
Neurologic Involvement
The earliest neurological manifestations of AFD translate dysfunction of small-diameter peripheral nervous fibers. In the classical form of the disease, they could appear in the first decade in male patients and 1 decade later in females. 45 These symptoms—neuropathic pain, paresthesia, dysesthesia, and decreased thermic sensitivity—are classically known as acroparesthesias. 46 Neuropathic pain can be chronic, described as burning sensation, but in the first years of disease, it is typically presented as recurrent paroxysmal episodes of excruciating pain—the painful Fabry crises—induced by fever, temperature changes, exercise, fatigue, or stressful situations. 45,46 Pain and sensitivity changes initially affect, symmetrically, the soles of the feet and progress proximally involving the palms. The painful crises can be accompanied by arthralgia, fever, or elevated sedimentation rate. 47 The involvement of the autonomic nervous system results in hypo/anhidrosis or hyperhidrosis, gastrointestinal dysmotility, cold, warm and exercise intolerance, and orthostatic hypotension. 48
The conventional electromyography is normal in patients with small-fiber neuropathy, except in those with renal dysfunction, associated with small-fiber neuropathy. For a better characterization and quantification of the neuropathic involvement, noninvasive autonomic and sensitive fiber tests are recommended (quantitative sensory testing, quantitative sudomotor axon reflex, test pain-related evoked potentials, electrochemical skin conductance, among others). Skin biopsy could also demonstrate reduced intraepidermal nerve fiber density.
Cerebrovascular events of AFD include transient ischemic attacks (TIAs), ischemic stroke, hemorrhagic stroke, and cerebral white matter lesions. The typical pattern of vascular injury is a small vessel type, 49 and there seems to be a predilection for involvement of the vertebrobasilar territory.
Fabry Registry data indicate a prevalence of stroke of 6.9% in males and 4.3% among females, with an average age at the first event of 39 years in men and 46 in women. 50 About 25% of the patients have their first stroke before 30 years. 51 Most of these patients had no history of cardiac or renal disease, and in 50% of men and 38.3% of women, AFD diagnosis has not yet been established. The presence of changes in the cerebral white matter in young adults of both sexes, identified by brain computed tomography or MRI, has been consistently reported in patients with AFD. Other frequently described imaging aspects are dolichoectasia of intracranial arteries, namely the basilar artery and the pulvinar hyperintensity on MRI. 52
Other Signs and Symptoms
Gastrointestinal symptoms that may arise in patients with AFD are postprandial abdominal pain, early satiety, bloating, intermittent diarrhea, chronic nausea, and vomiting. Many children have failure to thrive and are short-statured. These symptoms typically appear in adolescence. 2
The cornea verticillata is common. This corneal abnormality rarely causes changes in visual acuity and it is characterized by pale cornea streaks. 2 Retinal vessels are commonly affected with arteriolar narrowing, venous dilatation, and irregularity and excessive tortuosity of retina vessels. 2 The tear production may be reduced. 2
Late pulmonary involvement can be manifested with chronic cough, shortness of breath with exercise, and sibilance 2 due to obstructive lung disease. 53 The otologic involvement is characterized by progressive hearing loss (especially for frequencies above 4000 Hz), sudden deafness (often asymmetrical), and vestibular symptoms (vertigo). 2
Recommendations for Diagnostic Work-Up
After the diagnosis in the proband, it is important to make up a pedigree in order to identify all relatives at risk. Although AFD is an X-linked disease with full penetrance, it is important to remember that it can skip a generation due to the variable phenotypic expression. 8 The diagnosis should be proposed to all at-risk family members.
In this article, we present a practical approach to differential diagnosis for the main manifestations of AFD. Children with AFD are often diagnosed with rheumatism, viral infections, growing pain, bone problems, psychogenic or cryptogenic pain, food poisoning, or nonspecific gastrointestinal pain. 54 In Table 1, we present the most common differential diagnosis and the Fabry symptoms that can help to differentiate those pathologies.
Most Common Differential Diagnosis in Patients With AFD.

Abbreviation: AFD, Anderson-Fabry disease.
Cardiac
Left ventricular hypertrophy is not pathognomonic of AFD, and several other conditions can be responsible for this feature. The identification of LVH should lead to a systematic approach to the exclusion of other causes of LVH that are summarized in Table 2. 33
LVH Differential Diagnosis.
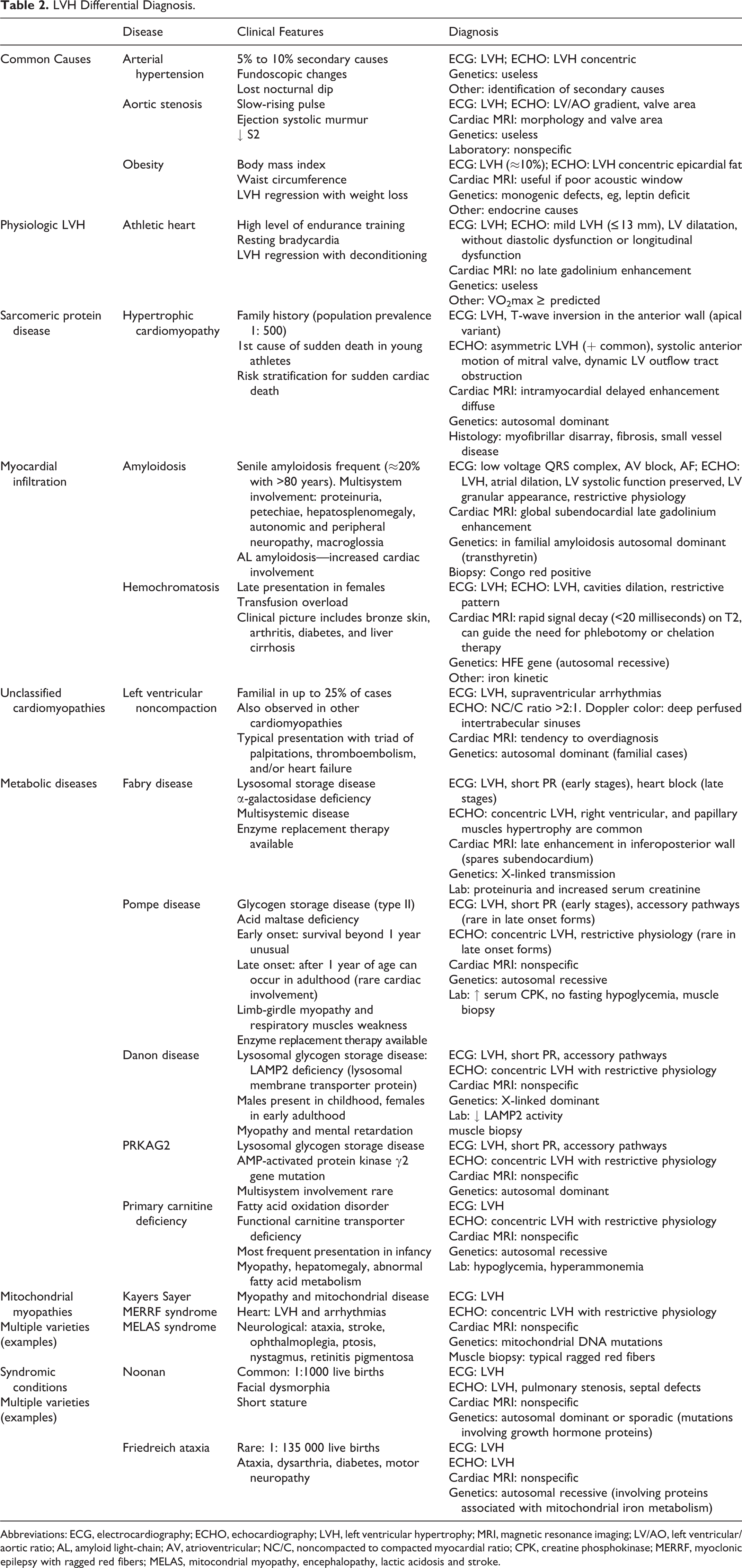
Abbreviations: ECG, electrocardiography; ECHO, echocardiography; LVH, left ventricular hypertrophy; MRI, magnetic resonance imaging; LV/AO, left ventricular/aortic ratio; AL, amyloid light-chain; AV, atrioventricular; NC/C, noncompacted to compacted myocardial ratio; CPK, creatine phosphokinase; MERRF, myoclonic epilepsy with ragged red fibers; MELAS, mitocondrial myopathy, encephalopathy, lactic acidosis and stroke.
Renal
Renal impairment, usually, manifests with albuminuria or proteinuria on the second or third decade of life. The podocytes injury occurs early, and segmental foot process effacement was observed in all young Fabry patients, despite the absence of abnormal albuminuria. 21 For that, in patients at risk of FD, any value of albuminuria observed in children, even if in the “normal” range, should be considered as suspected. 8 Microhematuria is rare. Later hypertension and renal dysfunction can appear. In the absence of familiar involvement, these findings may occur in the presence of other glomerulopathies such as immunoglobulin A nephropathy, focal, and segmental glomerulosclerosis. In the presence of familial renal disease, Alport syndrome could be considered (Table 3).
Nephropathy Differential Diagnosis.
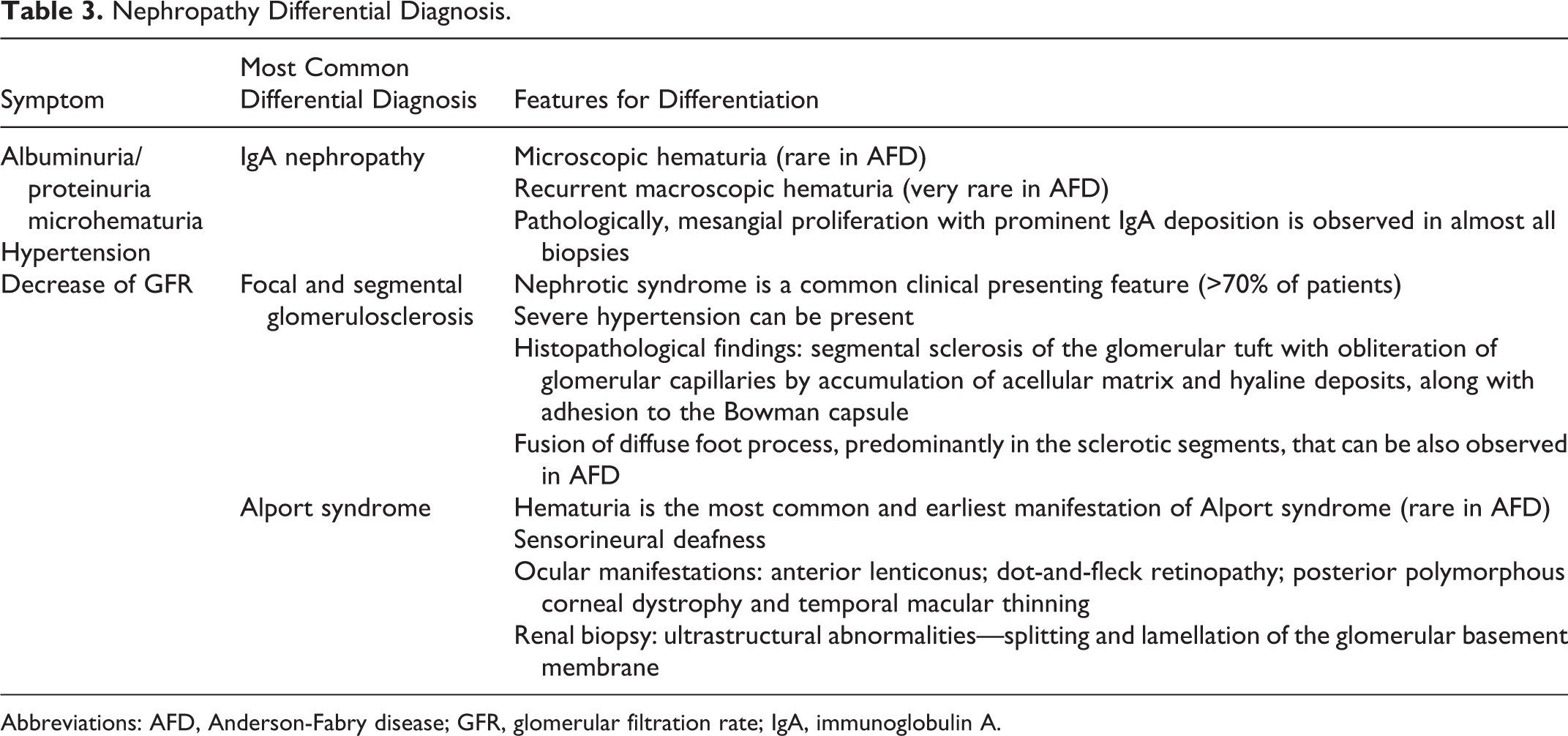
Abbreviations: AFD, Anderson-Fabry disease; GFR, glomerular filtration rate; IgA, immunoglobulin A.
Neurological
All children and teenagers with a suggestive clinical picture of neuropathy—especially if associated with painful complaints and autonomic dysfunction—should be evaluated by a neuropediatrician/pediatrician in order to establish the diagnosis. Small-fiber neuropathy differential diagnosis in young adults and children is described in Table 4.
Young Adults and Children Small Fibers Neuropathy Differential Diagnosis.

Abbreviation: HCV, hepatitis C virus.
Histologic Diagnosis
Renal biopsy is important to confirm AFD diagnosis in cases where the pathogenicity of the mutation is unknown and in order to exclude other pathologies when atypical forms of the disease are presented. 55 It is also important to evaluate the degree of the renal involvement. The presence of global or segmental glomerulosclerosis in more than one half of glomeruli was associated with the progression of the decline of GFR, despite the ERT. The renal biopsy also allows the detection of AFD in patients with proteinuria or chronic renal disease with no other signs or symptoms of AFD. 14
Initial Assessment and Follow-Up Recommendations
After the initial evaluation, the frequency of follow-up depends on the clinical severity and the involvement of different organs (Table 5). The initial baseline assessment should be performed for both sexes. In women with confirmed diagnosis, the organic involvement needs to be determined clinically. Asymptomatic women may be evaluated every 2 years by increasing the frequency to annual in adulthood, but symptomatic women should be monitored every 6 months, as recommended for men. 2
Follow-Up of the Different Clinical Manifestations of Fabry Disease.
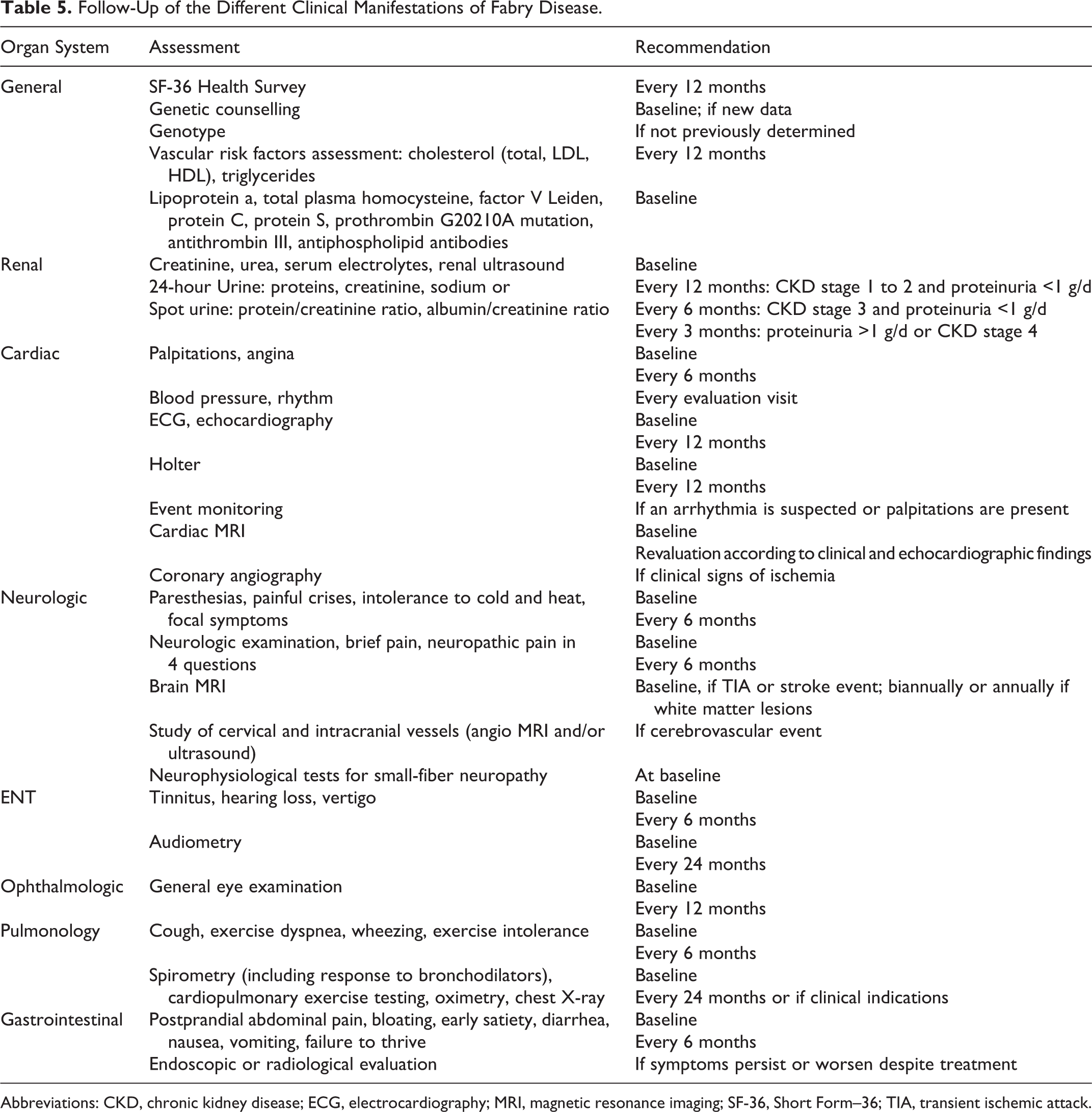
Cardiac MRI is recommended to all patients with AFD as the preferential examination to detect localized forms of hypertrophy and its exact distribution, which sometimes are not detected by echocardiography. 56 Late gadolinium enhancement in cardiac MRI allows the identification and quantification of focal myocardial fibrosis. In AFD, the late gadolinium enhancement is usually observed in intramyocardial and subepicardial regions of basal segments of the inferolateral wall. 57 Although this location is not pathognomonic of the disease, 58 in other forms of hypertrophy, the fibrosis usually has other distribution patterns. 59 The late gadolinium enhancement has also important prognostic implications, since it correlates with the myocardial mass index and the risk of developing potentially fatal arrythmias. 60 In the near future, this image method may add even more information to the diagnosis, follow-up, and evaluation of the therapeutic benefits with the development of new cardiac MRI techniques (T1 color mapping). 61,62
Patients with AFD can develop arrhythmias, associated with morbidity and mortality. The Holter monitoring is indicated on the patient’s initial assessment and follow-up. If there is high suspicion of rhythm abnormalities with complaints of palpitations or syncope, and if they are undetected by conventional Holter monitoring, event monitoring registers is recommended. 2
Renal evaluation is based in serum creatinine, serum urea, serum cystatin-C, serum electrolytes albuminuria/proteinuria, and renal ultrasound. Patients with CKD stage 1 and 2 should be reevaluated every 12 months, those with CKD stage 3 and proteinuria <1 g/d should be reevaluated every 6 months, and if proteinuria >1 g/d or CKD stage 4, the reevaluation should occur every 3 months.
To characterize the neuropathic involvement, noninvasive autonomic and sensitive fiber tests are recommended (quantitative sensory testing, quantitative sudomotor axon reflex, test pain-related evoked potentials, electrochemical skin conductance, among others).
The brain MRI is important to identify cerebral white matter lesions and other described imaging aspects such as dolichoectasia and the pulvinar hyperintensity, and it should be repeated biannually or annually in the presence of white matter lesions.
Conclusion
Anderson-Fabry disease diagnosis requires high clinical suspicion, particularly in attenuated forms of the disease. The presence of proteinuria and/or unknown etiology CKD, LVH, cerebral white matter lesions, and stroke/TIA especially in young adults is important sign of the disease and requires screening. Given the availability of ERT and its impact on morbidity/mortality, timely diagnosis of AFD is imperative.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Sanofi Pharmaceuticals Ltd and was not conditional on obtaining any specific type of results.
