Abstract
Fabry disease (FD) is an X-linked, lysosomal storage disorder caused by a mutation in the alpha galactosidase (GLA) gene leading to a deficiency in α-galactosidase A enzyme (α-Gal A) activity, which in turn results in accumulation of glycosphingolipids in different cells. The 2 major clinical phenotypes are the classic severe phenotype and the milder, later onset phenotype. In severe affected males with little or no α-Gal A activity, the onset of acroparesthesias, hypohidrosis, angiokeratomas, and corneal dystrophy is typically observed in childhood or adolescence. With advancing age, progressive multisystem microvasculopathic disease culminates in renal failure, cardiomyopathy, and/or cerebrovascular disease. Patients with later onset have residual enzyme activity and lack of vascular endothelial glycolipid accumulations; thus, they do not present with the early manifestations of the classic phenotype and typically present cardiac or renal disease in the fourth to seventh decade. Although the pathogenesis of cerebral vasculopathy in FD is poorly understood, it can be hypothesized that white matter changes may reflect the pathophysiology of the disease.
Introduction
Fabry disease (FD) is a rare and highly debilitating storage disorder that results from a total lack of or deficiency in the lysosomal enzyme α-galactosidase A (α-Gal A) because of mutations in the GLA gene. 1 Fabry disease is inherited as an X-linked trait; many of the male patients develop a classic severe phenotype with early onset of symptoms, whereas heterozygous females exhibit phenotypes ranging from absence of symptoms to severe involvement of vital organs. Indeed, as recently demonstrated, the existence of skewed X chromosome inactivation in females with FD is the key molecular explanation of the wide range of severity in clinical manifestations. 2 The deficiency in α-Gal A causes accumulation of globotriaosylceramide (GL3), globotriaosylsphingosine (lyso-GL3) and related glycosphingolipids within the lysosomes of multiple cell types throughout the body. 1 Patients with later onset FD (also called type 2 phenotype, whereas the classic form is called type 1 phenotype) have residual α-Gal A activity and lack vascular endothelial GL3 accumulation; thus, they do not present with the early clinical manifestations of the severe phenotype. In the later onset phenotype, patients typically present with predominantly cardiac or renal disease in the fourth to seventh decade, indicating delayed onset and slower progression. 1
Stroke is a common and serious clinical manifestation of FD. Recent evidence, from the literature on the natural history of FD and postmarketing surveillance databases of patients with FD treated with enzyme replacement therapy (ERT), has indicated that stroke may appear in young patients 3,4 and has been reported, in some patients, as the first disease’s event.
The mechanisms underlying the stroke pathogenesis in FD have not been unraveled yet. Early pathophysiological changes are believed to predominantly involve microvasculature, but recently the compromise of large brain vessels has been reported even before the appearance of white matter lesions (WMLs) in brain magnetic resonance imaging (MRI). 5 Although ischemic strokes and transient ischemic attacks (TIAs) are the most prevalent types of overt cerebrovascular events in patients with FD, intracerebral hemorrhages, microbleeds, neurocognitive decline, and depression have also been reported. 6
Prevalence
Silent WMLs are the most common findings in FD, before the onset of the “clinical” stroke, even though there are no reports on the frequency of other silent cerebrovascular events (ie, lacunar strokes and microbleeds). Several studies have estimated the incidence of stroke in various small cohorts of patients. Vedder et al 7 reported that 12 (48%) of 25 males and 13 (32%) of 41 females had experienced a cerebrovascular accident or lacunar stroke, at a median age of 46 and 52 years, respectively. Another group 8 reported that 4 (7%) of 54 female patients with Fabry had experienced strokes, at a median age of 51 years, and Mehta et al 4 found that 24 (11%) of 216 males and 27 (16%) of 172 females had experienced either a TIA or a stroke. Grewal et al reviewed various clinical and neuroradiological records and reported that 8 of 33 Fabry patients (24%) had experienced strokes at a median age as low as 26.5 years. 9 An analysis of a large cohort of 2446 patients in the Fabry Registry reported that stroke occurs in 6.9% of men and 4.3% of women. 3 Of these, 87% of first strokes were found to be ischemic, with hemorrhagic stroke reported in 13% of cases. Although the incidence of stroke increases with age in patients with FD, as in the general population, analysis of the Fabry Registry has indicated that the incidence of stroke among patients with FD is markedly higher than that observed in the general US population across all age categories. 3 From the Fabry Outcome Survey, the overall prevalence of ischemic stroke or TIAs was 13%. 4 Cerebrovascular events tending to occur at an earlier age with 12 times higher number of ischemic strokes in the male 25- to 44-year age-group compared to what could be expected in the general population. 6
Fellgiebel et al reported that the frequency and severity of WMLs were similar for both sexes, with a prevalence of 31% in male and 36% in female patients. 10 In another study, the corresponding proportions were 27% (4 of 15) for women and 34% (11 of 32) for men. 11 Reisin et al described 36 adults with FD (14 men and 22 women) who were evaluated by MRI; 44% of them showed WMLs. 12 Among the 16 patients with abnormal MRI, 12 were women. The latter study is a further evidence confirming that women are not just carriers of FD and should be followed clinically and evaluated comprehensively to monitor for disease burden and progression.
In 2005, the first selective screening study in patients with cryptogenic stroke was published and reported a high number of presumed FD diagnoses: 4.9% of studied males and 2.4% of studied females. 13 However, other authors have failed to corroborate these relevant figures. Indeed, more recent studies indicate that the frequency of FD in cryptogenic stroke is likely less significant than initially suggested, probably below 1%. 14
One possible explanation could be that in the study of 2005, there was the inclusion of intronic mutations, mutations of unknown clinical significance, or genetic variants/polymorphisms. It should be pointed out that the relationship between various types of GLA gene variants, the residual α-Gal A activity, and the clinical manifestations is complex and still under investigation. According to Schiffmann et al, we can classify GLA gene variants/mutations into 3 main groups: (1) benign genetic variants, not directly responsible for the clinical findings (eg, D313Y); (2) risk factors, genetic variants (eg, A143T) that concur with other risk factors for the clinical findings; (3) causative mutations, directly responsible for the disease. 15
Pathophysiology
Historically, the cerebral pathology of FD has been considered a secondary manifestation of a primary endotheliopathy. Progressive accumulation of GL3 within the endothelium of intracranial blood vessels is thought to play a primary role in vasculopathy and risk of ischemic stroke. However, involvement of smooth muscle cells (SMCs) in the vessel wall has also been demonstrated, suggesting a more complex pathogenesis. The damage of SMCs in the medial vessel layer is the result of the storage of GL3 and derivatives, as a consequence of released hypertrophic factors such as Lyso-GL3 and sphingosine 1 phosphate. 16,17 This last mechanism has been confirmed by in vitro studies of cardiomyocytes and SMCs in culture. 18 A recent study showed that the removal of stored glycosphingolipid from the endothelial cells, as identified by conventional histological examination, does not prevent the progression of vascular disease. 19 Thus, the traditional concept that the storage in endothelial cells is the only mechanism responsible for the vascular dysfunction cannot be held. Indeed, other factors, such as the presence of a prothrombotic state, 20 abnormalities in cerebral blood flow velocity, autonomic dysfunction, 21 and increased production of reactive oxygen species, have also been identified to contribute to the development of stroke in patients with FD. 22 Another relevant mechanism contributing to stroke occurrence may be due to cardiac emboli and arrhythmias, 23 which are important and common cardiac manifestations in both genders of patients with Fabry.
The role of vascular or autonomic dysfunction as a pathogenic mechanism for stroke in patients with Fabry by compromising cerebral blood flow velocities and cerebral autoregulation has been studied by Hilz et al. 21 They investigated, by means of transcranial Doppler sonography, 22 patients with Fabry and 24 controls and assessed the resistance index, pulsatility index, cerebrovascular resistance, spectral powers of oscillations in electrocardiographic RR-intervals intervals, mean blood pressure, and mean cerebral blood flow velocities. The tested hypothesis was that the decreased cerebral blood flow velocities might result from downstream stenoses of resistance vessels and dilation of the insonated segment of the middle cerebral artery due to reduced sympathetic tone and vessel wall pathology with decreased elasticity. Furthermore, the augmented gain between blood pressure and cerebral blood flow velocity oscillations indicated an inability to dampen blood pressure fluctuations by cerebral autoregulation. The authors’ conclusion was that, both reduced cerebral blood flow velocities and impaired cerebral autoregulation were likely to be involved in the increased risk of stroke in patients with FD.
Pathology studies regarding central nervous system (CNS) involvement in FD are rare. Nevertheless, a specific neuronal involvement has been reported. 24 -27 The selective nature of the storage phenomenon is in accordance with the other disorders of the nervous system where accumulation of the substrate in nerve cells results from a metabolic disorder, as, for example, in gangliosidoses and metachromatic leucodystrophy. 28,29 Sites of selective storage in the CNS are amygdala, hypothalamus (supraoptic and paraventricular nuclei), substantia nigra, pontine reticular formation, the dorsal efferent nucleus of vagus, salivary nuclei, nucleus ambiguous, the mesencephalic nucleus of the fifth nerve, intermedial lateral columns, anterior horns, and dorsal root ganglia. 24 -26 Numerous deposits of GL3 were visualized in the cytoplasm of the SMCs of the parenchymal and leptomeningeal vessels. 25,26 Other findings usually reported are the presence of segments of subarachnoidal medium-sized arteries with definite luminal stenosis due to intimal fibrosis, severe undulation of the internal elastic membrane, and stiffening due to subtotal or total replacement of medial SMCs by fibrosis. 26
Brain Imaging Findings
The cerebrovascular involvement can be visualized on conventional MRI as (1) multiple lesions located in the deep white matter and in the subcortical gray matter of both hemispheres; (2) ischemic lesions, which can be silent lacunar strokes or clinically relevant strokes (Figure 1). The cerebral WML burden visible on brain MRI increases with age and can precede the onset of neurological symptoms. 30 The usual pattern of WMLs in FD shows a symmetric distribution, resembling a “vascular leukodystrophy.” Nevertheless, the load of lesions and the pattern of distribution can be different according to aging, raising the question of differential diagnosis with multiple sclerosis. A recent study on a large cohort of patients with Fabry showed that the fourth decade of life is critical in the progression of WMLs. 31
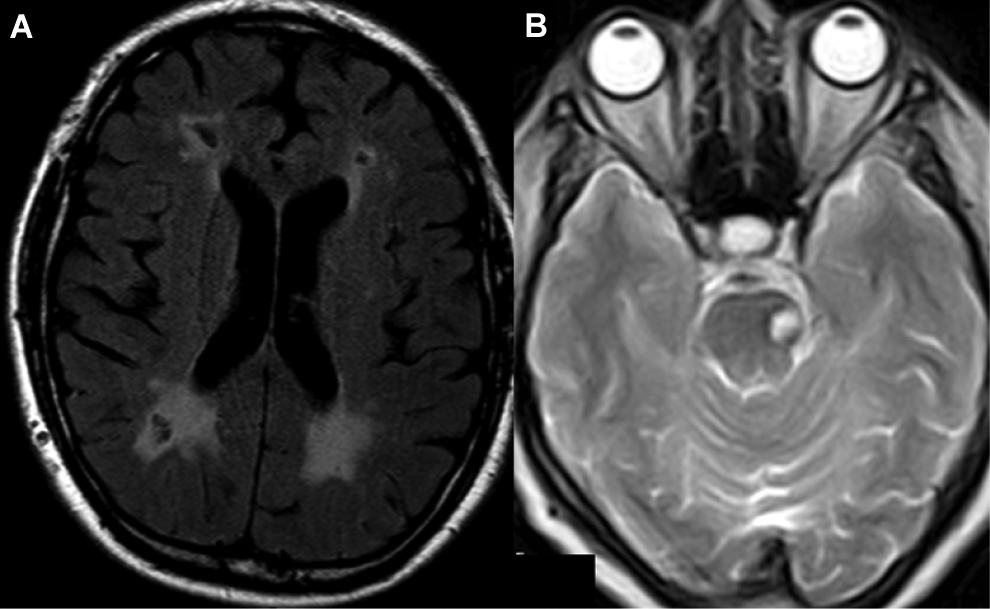
A, Periventricular white matter lesions in a 51-year-old male patient with Fabry disease. B, Ischemic lesion at the left pons level in a 42-year-old female patient with Fabry disease.
More recent studies, using proton emission tomography 32 and proton magnetic resonance spectroscopy, 33 have suggested that metabolic abnormalities can be found even in the absence of cerebral lesions in patients with FD.
Vertebrobasilar dolichoectasia (VBD) is a vascular condition that may cause severe morbidity due to ischemic or compressive dysfunction in the posterior fossa (ie, paroxysmal vertigo due to the compression of the VIII cranial nerve has been reported in a patient with FD). 34 Vertebrobasilar dolichoectasia may not be readily recognized by many neurologists or radiologists owing to the “normal” variation in the tortuosity or diameter of the vertebral and basilar arteries in healthy individuals. The study of dolichoectasia has been hampered by the lack of standardized diagnostic criteria. In 1996, Mitsias and Levine reviewed 53 cases and found elongated, ectatic, and tortuous vertebral and basilar arteries as the most common angiographic and pathologic features. 35 Recently, Politei et al reported that VBD was present in 55.5% of men and 34.8% of women (Figure 2) using Smoker’s criteria. 5 The definitive prevalence and incidence of VBD is still unknown in the general population. According to the literature, the diagnostic frequency of VBD has been noted as 4.4% among 1440 magnetic resonance angiography records, 3.7% among 780 patients with posterior circulation ischemic stroke, 6% among 381 autopsies of fatal stroke, and 18.8% in 96 patients with isolated pontine infarction. 5 Pico et al reported that intracranial arterial dolichoectasia, including VBD, is found in 12% of patients with stroke. 36 It is important to emphasize that the median age in all the previous series is more than 50 years. The role of atherosclerosis in dolichoectasia is unknown, and whether atherosclerosis is the inciting event or an innocent bystander remains to be determined. The results from FD studies did not show a clear relationship between cardiovascular risk factors, cerebral ischemia, and the presence or absence of VBD or cerebral ischemia. 5 Currently, the different mechanisms involved in the cerebrovascular damage in FD have not indicated atherosclerosis as a major contributing risk factor. On the contrary, Doppler ultrasound studies showed an increase in the intima–media thickness without the presence of atherosclerotic plaques. 37,38 This finding has been reported in brain and coronary arteries in some autopsies. 39 The primary cause of dolichoectasia seems to be aberrant vascular remodeling. Dilation is associated with a large increase in the levels of early growth response factor 1 in endothelial cells, and the expression of this transcription factor is closely linked to the expression of matrix metalloproteinases (MMPs), 40 especially MMP-9. 41 Evidence indicates that MMPs break down the elastic lamina, and degradation of this component of the arterial wall is thought to aid the migration of SMCs, residing in the tunica intima, to the stressed medial layer. 38,42 Levels of MMP-9 were significantly higher in patients with FD than in controls and there was a positive correlation between MMP-9 levels and the Mainz Severity Score Index, a validated severity score for FD. 43 Dolichoectasia can affect both the anterior and the posterior circulations, but the posterior circulation and the basilar artery are most frequently affected in FD, one explanation could be that posterior circulation has less sympathetic innervation than the anterior circulation (Figure 2A). 5 Indeed, this asymmetry in trophic support that the sympathetic nerves confer on the vessel walls may render the posterior circulation more susceptible to deformity upon exposure to increases in blood flow. In 2009, Fellgiebel et al reported results of brain MR angiography of 25 patients with FD, in comparison with a control group. 44 The principal finding of this study was that the basilar artery diameter is clearly superior to the WML load for the differentiation of FD from controls. However, as recently reported by Üçeyler et al, the increased basilar artery of males with FD cannot distinguish patients with FD from patients with stroke. 45
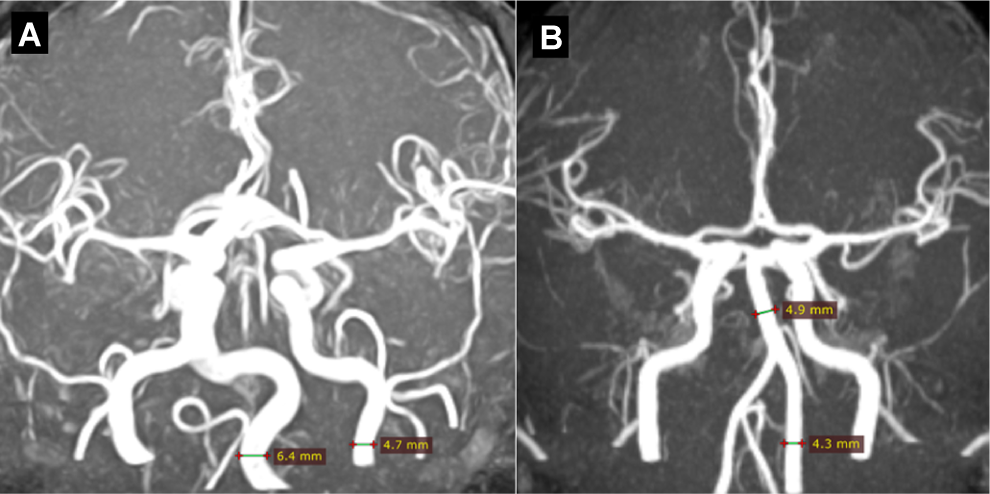
Vertebrobasilar dolichoectasia. A, 55-year-old male patient with Fabry disease, the diameter of the left vertebral artery is increased more than the homolateral internal carotid artery. B, 46-year-old female patient with Fabry disease; both diameters of the left vertebral artery and the basilar artery are increased.
Moore et al 46 studied neuroradiologic images of 104 hemizygous patients with FD. Overall, 22 patients (23%) demonstrated pulvinar hyperintensity on T1-weighted images (the so-called the pulvinar sign). They concluded that hyperintensity in the pulvinar on T1-weighted images is a common, pathognomonic finding in FD, likely reflecting the presence of calcification, secondary to cerebral hyperperfusion and selective vulnerability of the pulvinar and adjacent thalamic nuclei. In the same issue of the American Journal of Neuroradiology, Takanashi et al reported the presence of T1 hyperintensity in 7 out of 9 male patients with Fabry. 47 This finding was later confirmed by Burlina et al 48 who investigated the pulvinar sign and its relationship with other clinical findings in a total of 36 patients from 14 families (Figure 3). The pulvinar sign was found in 5 severe affected male patients (with severe renal and cardiac involvement) but not in any of the investigated female patients. Furthermore, Burlina et al demonstrated that the pulvinar sign is not a pathognomonic neuroradiological sign of FD, being present under other pathological conditions. 48 Recently, the detection of a faint pulvinar sign in 2 female patients with no significant cardiac and renal involvement indicates that this sign is not restricted to advanced stages of FD. 49
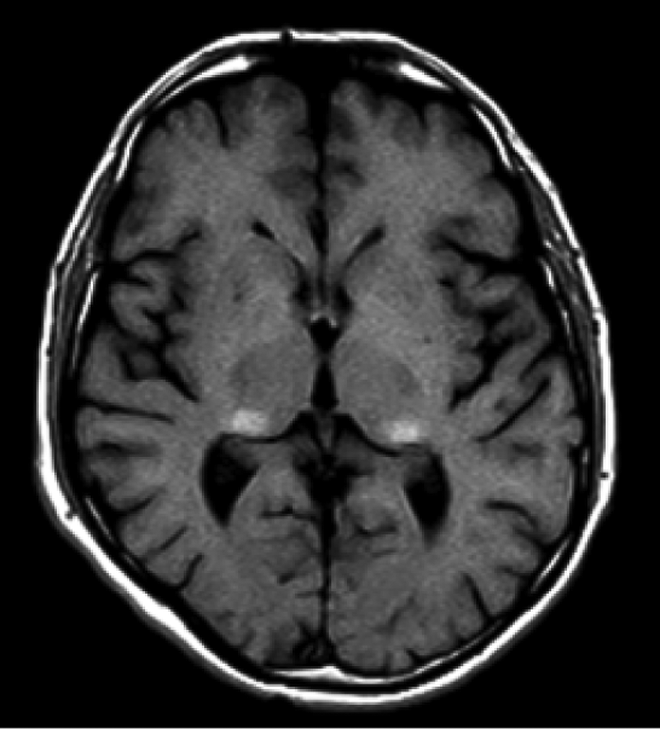
T1-axial image showing a well-defined symmetric hyperintensity of both pulvinar nuclei of a 46-year-old male patient with Fabry disease (Adapted from Burlina et al,48 with permission from Springer).
Chiari type I malformation is a rare condition characterized by a herniation of the cerebellar tonsils, which extend more than 5 mm below the foramen magnum, often associated with syringomyelia. The prevalence has been reported to be lower than 0.24% in the literature. 50 In FD, the prevalence of this malformation was 10% and 12.7% in 2 different series patients, respectively. 51
Neuropsychiatric Symptoms, Cognitive Dysfunction, and Sleep Disorders in FD
In FD, cognitive deficits have been described primarily in attention, executive functions, and psychomotor performance. 52,53 However, studies are inconclusive on the degree of these deficits and whether its development is a result of depressive symptoms or neurological alterations.
Schermuly et al could not find any relationship between depression and WML load in patients with FD. 53 In the same investigation, they found that 60% of the 25 patients with FD enrolled showed clinical depression compared to healthy controls. In fact, depression is by far the most frequently reported psychiatric complication of FD, ranging from 15% to 62.5% in all series.
Depression in FD has been reported to be directly associated with chronic pain. Cole et al 54 found 4 factors to be predictive of increased risk of depression in patients with FD. Two of these were factors related to depression in the general population (relationship status and financial status). The other 2 factors were more directly related to patients’ illness. Firstly, having an additional chronic illness as well as FD increased the risk of depression. However, the strongest predictor of depression was the degree to which FD symptoms interfered with daily life, with both pain symptoms and inability to sweat (hypohidrosis/anhidrosis) being found to strongly predict depression. It is of note that cerebrovascular disease were not found to strongly predict depression.
Recently, 14 patients with Fabry (46.1 ± 10.8 years) participated in a neurocognitive investigation 8 years after the first assessment. 55 Patients were investigated by neuropsychiatric testing, brain MRI and MR angiography to determine WMLs, hippocampal volume, and the diameter of the larger intracranial arteries. While patients with Fabry did not differ in cognitive performance, they showed progressive and significant hippocampal volume loss over the 8-year observation period. None of the aforementioned brain imaging parameters were associated with the neuropsychiatric parameters. The depression frequency reduced from 50% at baseline to 21% at follow-up, but it did not reach significance. The authors concluded that patients with Fabry (10 patients were on ERT) have clinical stability in cognitive function, while pronounced hippocampal atrophy progressed throughout the 8 years.
A common finding in the clinical practice is the complain of excessive daytime sleepiness by patients with Fabry disease. 56,57 Indeed, as reported by Duning et al, excessive daytime sleepiness, with a frequency of 68%, can be considered one of the most common symptoms of FD. 56
Treatment
Enzyme Replacement Therapy
There are 2 drugs available for ERT: agalsidase alfa (administered IV at a dosage of 0.2 mg/kg biweekly) and agalsidase beta (administered IV at a dosage of 1.0 mg/kg biweekly). Besides dosage and other different pharmacologic properties, both drugs share a common feature. They do not cross the blood–brain barrier. Short-term and long-term Fabry studies on ERT efficacy had focused on kidney and heart outcome. 58 There isn’t any study designed to assess stroke prevention or recurrence. Nevertheless, data from the literature indicate that strokes can occur in patients on ERT. 59,60
Recently, the WML burden and the effect of agalsidase beta 1 mg/kg biweekly on WML progression were assessed longitudinally in a phase 4 agalsidase beta placebo-controlled analysis of untreated and treated FD patients with mild to moderate renal involvement. 61 The primary end point was the difference in the number of patients with increased WML burden between the agalsidase beta and placebo groups at the end of treatment. Brain MRI scans from 41 patients with FD were analyzed. Patients with previous or recent strokes (n = 11, 39–68 years) showed an increase in the number of WMLs. A greater proportion of younger patients (≤50 years) on ERT had stable WML burden compared with younger patients in the placebo group (P = .014). Authors concluded that the WML burden in patients on ERT was more likely to remain stable, compared with patients on placebo. Thus, this study showed that agalsidase beta may reduce the progression of WML load, according to MRI analysis.
Concomitant/Supportive Therapy
Prevention of stroke is a critical issue in FD. Antiplatelet agents have been recommended for primary and secondary stroke prevention in FD, but their effectiveness has not been assessed with a specific study. 62 The same holds true for anticoagulants that are necessary if embolic events that stem from cardiac causes are a concern (warfarin is used in the clinical practice, while there are no reports on the use of the new oral anticoagulants). Blood pressure control is mandatory, and statin therapy should be considered. 63 Adequate intake of vitamins should be promoted, especially in cases of hyperhomocysteinemia. 64 Evaluation of comorbid prothrombotic risk factors may help to identify those with higher stroke risk (eg, patients with FD and the concurrent presence of factor V Leiden). 65 Although there are no evidence-based data, feasibility of thrombolysis in acute ischemic stroke in FD was recently demonstrated, as reported in 2 patients. 66,67
Conclusions
The CNS is extensively involved in FD. Cerebrovascular findings include strokes, chronic WMLs, and brain vessel abnormalities. These clinical and neuroimaging findings are similar to those detected in other patients with cerebrovascular disease. Therefore, for diagnosing FD, an appropriate neurological workup that include detailed clinical history, neurological examination, and brain MRI investigations (search for WMLs, basilar artery dolichoectasia, and pulvinar sign) is necessary.
Until today, treatment studies did not address the specific issue of stroke prevention and recurrence. We hope that in the near future new studies on therapy will be designed, with the specific goal of neurological outcome for patients with FD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
