Abstract
Background:
Anterior cruciate ligament reconstruction (ACLR) is associated with a high incidence of posttraumatic osteoarthritis (PTOA). The incidence is even higher when ACLR is combined with meniscal surgery (ACLR+M). The mechanisms associated with the onset and progression of PTOA after injury and surgery are not fully understood, but altered joint biomechanics shortly after surgery are implicated.
Purpose:
To compare the side-to-side differences in joint kinematics (ie, osteokinematics) and articular cartilage arthrokinematics at 1 to 2 years after ACLR+M during 3 demanding activities that challenge the knee with high-impact loads, large rotations, and non–sagittal-plane movements.
Study Design:
Descriptive laboratory study.
Methods:
A total of 12 participants (8 male and 4 female) who had undergone ACLR+M at 1 to 2 years previously participated. Dynamic activities included single-leg landing, lunging, and pivoting. Tibiofemoral osteokinematic data of both knees were captured using dual fluoroscopy combined with model-based tracking. Osteokinematic analysis was conducted for 6 degrees of freedom kinematics. Cartilage arthrokinematic data, derived from magnetic resonance imaging–based cartilage models, were evaluated for tibiofemoral contact location, contact overlap, contact path, and excursion.
Results:
Compared with the contralateral knee, the ACLR+M knee underwent less flexion during landing (P < .001) and lunging (P = .04). It also exhibited increased anterior tibial translation during landing (P = .01) and pivoting (P = .002). We observed posterior shifts in contact location on the tibial cartilage surface and increased overlapping contact in both the medial and lateral compartments during landing and pivoting. The ACLR+M knee also exhibited increased anteroposterior excursion of the tibial contact path during lunging.
Conclusion:
ACLR+M was associated with alterations in osteokinematics and arthrokinematics in the surgical knee, suggesting that the surgical knee does not return to normal function during demanding dynamic activities at 1 to 2 years. This study provides novel in vivo evidence of joint abnormalities under functional loading conditions experienced during everyday activities and sports. These findings enhance our understanding of early biomechanical side-to-side deviations that may contribute to PTOA.
Clinical Relevance:
Identifying abnormal knee joint mechanics at 1 to 2 years after ACLR+M during high-demand tasks emphasizes the need for targeted rehabilitation and monitoring strategies aimed at mitigating altered loading patterns that may accelerate PTOA development.
An anterior cruciate ligament (ACL) injury is one of the most prevalent musculoskeletal injuries among young athletes such as soccer players and skiers.13,52 ACL reconstruction (ACLR) is a procedure that offers active patients the potential to resume their high-intensity athletic activities within a time frame of 6 to 12 months after surgery. 8 However, estimates as high as 50% to 80% of patients who undergo ACLR develop posttraumatic osteoarthritis (PTOA) within 10 to 15 years after surgery, with a 2.8 times greater risk in their surgical knee compared with the contralateral healthy knee.12,20,24,28,35,39,49 The risk of PTOA is consistent, regardless of the graft type used, but increases further when ACLR is combined with meniscal surgery (ACLR+M).28,45,48,51 The pathogenesis of osteoarthritis after ACLR and ACLR+M remains incompletely understood. However, alterations in joint biomechanics have been implicated as an important contributing factor to the degenerative pathway associated with PTOA.6,7,14,15,53
The ACL stabilizes tibial translation and rotation, and ACL deficiency leads to abnormal knee kinematics that often persist even after clinically successful ACLR.5,11,15,18,34,54 Previous research has indicated that the kinematic abnormalities are likely exacerbated during more intense and demanding activities, particularly those involving rapid deceleration and pivoting maneuvers.15,17,31,53
Abnormal osteokinematics alter the cartilage loading pattern, exposing cartilage regions that typically bear little or no loads to elevated shear and compressive stress, which may contribute to osteoarthritic changes over time.15,21,53,55 Furthermore, meniscal tears and surgery also contribute to changes in the loading pattern of articular cartilage 55 because of the critical contact stress role that menisci have for the knee joint through shock absorption and load distribution. 55 In addition, repair and/or resection of a damaged meniscus may not fully restore the meniscus’ capacity to absorb shock and distribute contact stress, which may be observed as increased overlapping contact of the articular cartilage surface. However, in vivo evidence of this phenomenon, particularly during high-demand activities in the ACLR+M population, remains limited.
Most studies have focused on level walking gait because they follow the hypothesis that, in the long term, frequent cyclical abnormal loading will degenerate cartilage. However, a significant proportion of patients undergoing ACLR+M return to sports that involve high-demand activities, but the literature lacks experimental studies that quantify the effects of relevant higher demand activities on osteokinematics and arthrokinematics in the ACLR+M population, which should highlight larger mechanical changes. 47 Single-leg landing is particularly relevant to return-to-sports activities, producing high ground-reaction forces (GRFs) that stress cartilage in weightbearing regions under impact loading.2,19,50 Lunging allows evaluations at deep flexion angles in which quadriceps-hamstring coordination is essential for stability and in which deficits are common after ACLR.16,22,26 Pivoting challenges the reconstructed knee through rapid axial rotation, a movement strongly linked to early degenerative cartilage changes, and directly tests the ACL's role in rotational restraint.15,21,23,46 Given that even minor alterations in osteokinematics (joint angles and translation) after ACLR+M can significantly affect cartilage contact mechanics, gaining a deeper understanding of cartilage arthrokinematics (the relative position of articulating surfaces) in vivo in the early phase of healing is crucial.15,53 Previous dual fluoroscopy (DF) studies have primarily examined tasks with low flexion angles such as downhill running, but investigations of high-impact, large-rotation activities on the reconstructed knee may identify activity-specific deviations in joint mechanics with a direct application to physical activities after rehabilitation.1,32
Therefore, the objective of this study was to measure the side-to-side differences in osteokinematics and articular cartilage arthrokinematics at 1 to 2 years after surgery during 3 demanding dynamic activities: single-leg landing, lunging, and pivoting. We hypothesized that the ACLR+M knee would not fully restore osteokinematics or cartilage arthrokinematics compared with the contralateral healthy knee. Specifically, based on our previous work in walking and jogging as well as previous reports in the literature, we expected to observe reduced knee flexion, increased anterior tibial translation, posterior shifts in cartilage contact locations on the tibia, and increased contact overlap in the ACLR+M knee.21,23,25,31
Methods
Participants
This controlled laboratory study included 12 participants who were recruited between 1 and 2 years after ACLR accompanied by meniscal surgery. In addition to ACLR, participants had undergone either meniscal repair or partial meniscectomy, as detailed in Table 1. The inclusion of only patients with contralateral healthy knees was confirmed through magnetic resonance imaging (MRI) scans that were reviewed by a board-certified radiologist specializing in musculoskeletal conditions. Participants were excluded if quadriceps strength was <70% of the contralateral limb, as determined by isometric strength testing. This criterion ensured adequate strength recovery before participation. Of the initial 15 participants enrolled, 3 were withdrawn: 2 had an injury to their contralateral meniscus, and 1 faced scheduling conflicts that prevented participation. The final cohort for data collection consisted of 12 participants (8 male, 4 female), all of whom provided written informed consent in adherence to the study protocol approved by our university's institutional review board. Among them, 9 had undergone ACLR with an ipsilateral bone–patellar tendon–bone autograft, 1 had a quadriceps tendon autograft, 1 underwent ACLR with a patellar tendon allograft, and 1 received an ipsilateral bone–patellar tendon–bone graft with lateral extra-articular tenodesis (LET). The participants’ mean age was 26.2 ± 4.9 years, and their mean body mass index was 24.4 ± 3.0 kg/m2. The same cohort and data acquisition protocol described in our previous publication 25 were used in this study.
Characteristics of Participants and Meniscal Injuries
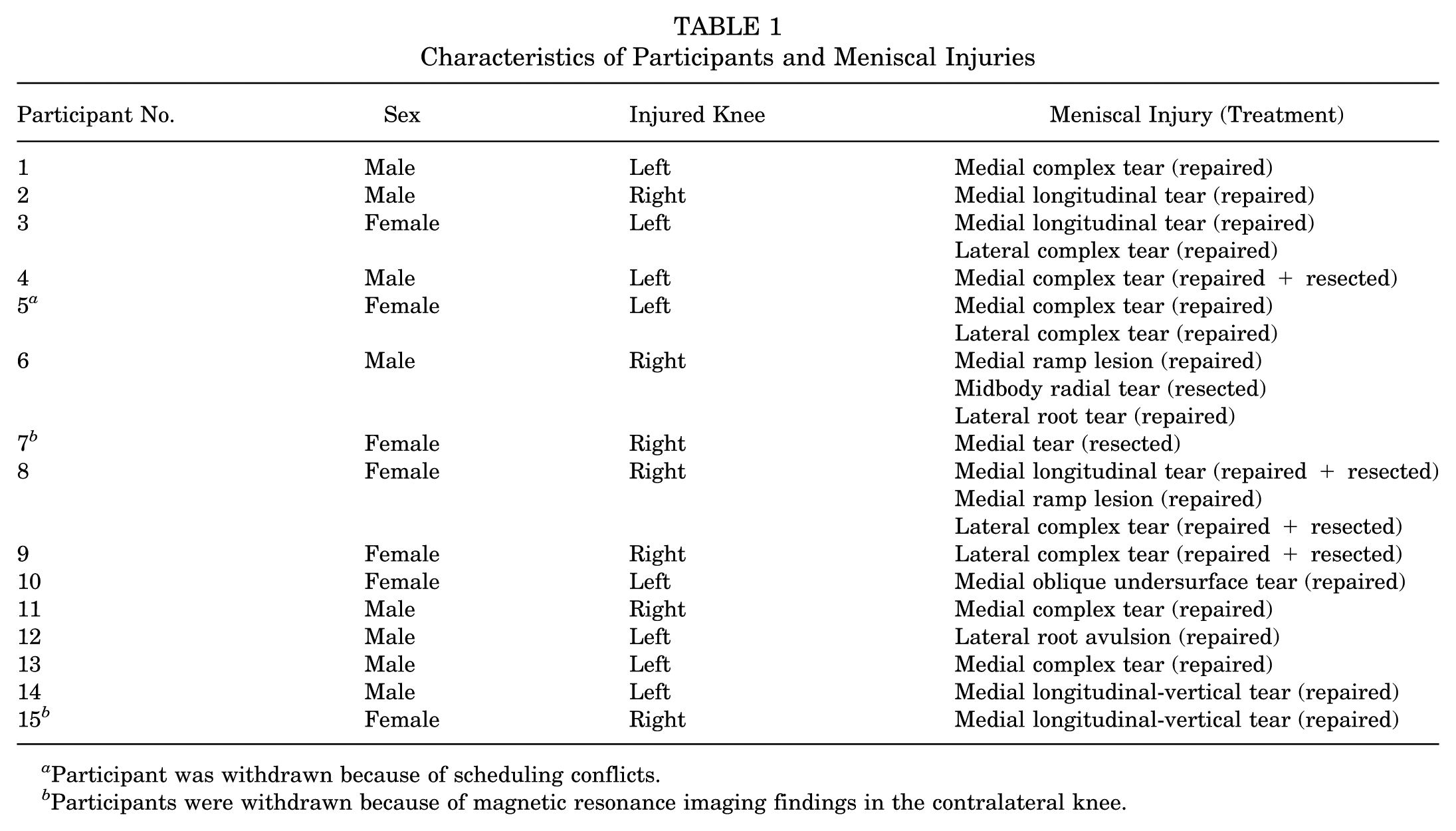
Participant was withdrawn because of scheduling conflicts.
Participants were withdrawn because of magnetic resonance imaging findings in the contralateral knee.
Computed Tomography (CT)
Bilateral CT images were obtained using an iCT SP imaging system (Philips). The CT acquisition parameters were configured to include a slice thickness of 0.67 mm, 120 kVp, and an in-plane resolution of 0.195 mm/pixel. The superior-inferior field of view (FOV) was set at 300 mm.
Motion Capture and DF
All patients performed 3 dynamic activities—(1) single-leg landing, (2) lunging, and (3) pivoting—in a motion capture laboratory, while DF images of the knee and retroreflective marker positions of the lower limb and torso were acquired simultaneously. High-speed DF images were acquired at 250 Hz using a custom system with emitters and image intensifiers on separate bases surrounding the laboratory walkway. 40 Marker locations were acquired using a 10-camera motion capture system (Vicon) operating at 125 Hz. GRFs were measured using a force plate (AMTI) embedded beneath the FOV of the DF imaging system. The GRFs were sampled at a rate of 1000 Hz. The order of testing the knee (left or right) and activity (landing, lunging, or pivoting) was randomized. Dynamic activities were repeated 3 times for both ACLR+M and contralateral healthy knees.
Dynamic movements were selected to represent common movements required for various sports. For the single-leg landing task, participants stood on one leg on a 10 cm–high box, performed a forward jump, and landed on the same single leg approximately 50 cm in front of their take-off point on a force plate (Figure 1A). A trial was considered successful if the participants maintained their landing position without their contralateral foot touching the ground and their knee was located within the DF imaging system's FOV. For lunging, each participant was required to perform a lunging activity with the knee of interest within the view of the DF system. Before beginning the activity, the participants’ feet were placed in a split stance (staggered position with the foot of the knee to be imaged in front), and they were instructed to achieve a lunge with their front knee in roughly 90° of flexion (Figure 1B). The participant was also instructed to perform the lunge without touching the rear knee to the ground. For the pivoting activity, participants completed a 3-stride approach toward the DF system, stepped onto the force plate with their foot facing forward, performed a 180° turn, and completed a forward stride in the opposite direction, leading with their contralateral foot (Figure 1C). Each participant was instructed to keep the foot stationary on the force plate during the 180° turn. A trial was considered successful if the participants held their knee in the FOV during rotation of the knee, hip, and torso while avoiding their opposite limb coming into the DF system's FOV.
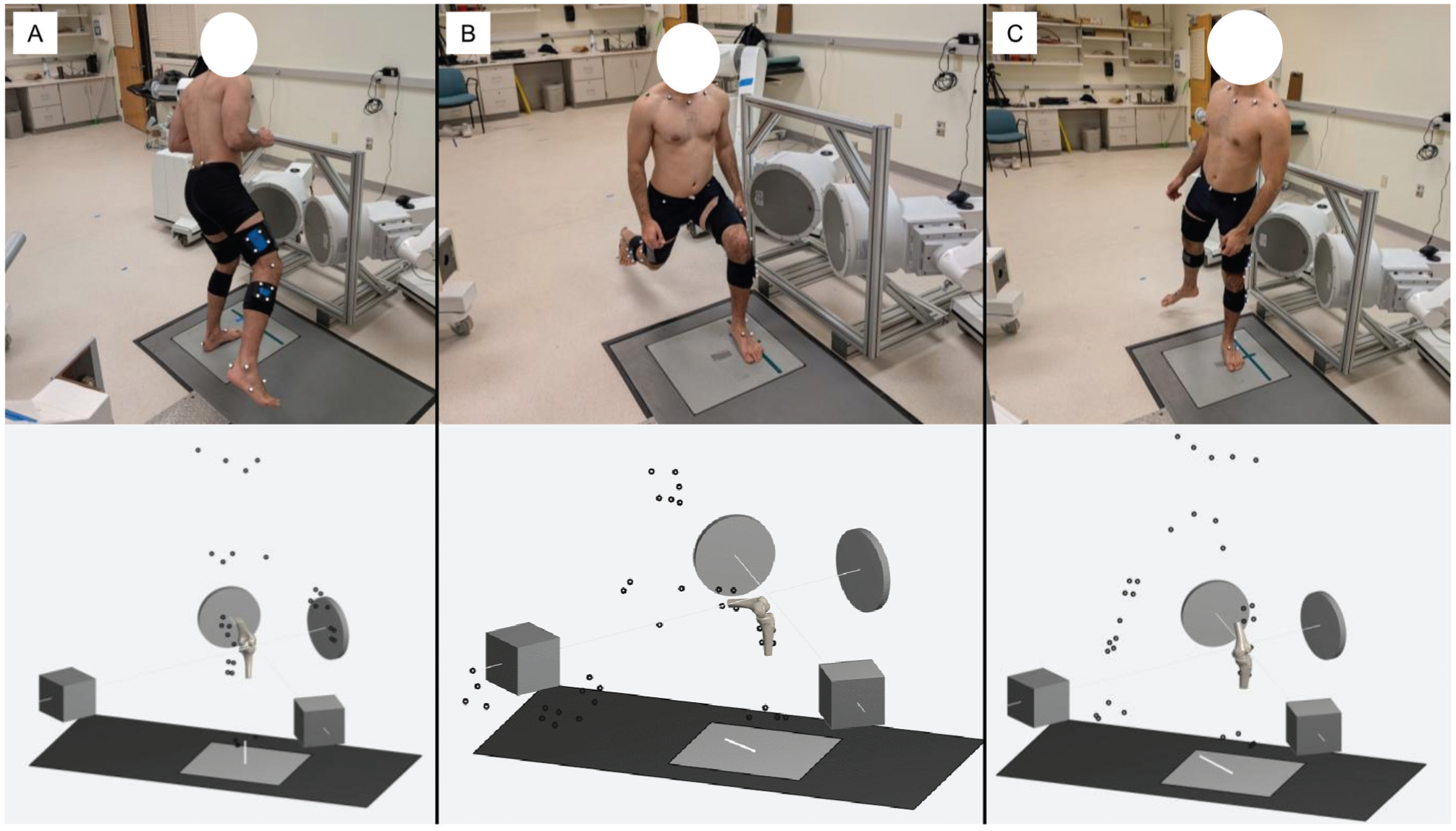
Experimental setup and activities completed with the simultaneous capture of dynamic dual fluoroscopy (DF) images and reflective skin marker positions. Each panel shows (top) a photograph of a participant performing the dynamic activity and (bottom) a schematic of the DF system and reflective marker set. The calibrated locations of the DF emitters are represented by cubes, and the image intensifiers are represented by short cylinders. Reflective markers (dots) were acquired by the motion capture system, and bone poses were derived using DF with model-based tracking. (A) Single-leg landing on the left knee, with the right knee outside the field of view (FOV). (B) Lunging with the left knee within the DF system's FOV. (C) 180° pivoting on the left knee, with the right knee outside the FOV.
Model-Based Tracking and Arthrokinematic Analysis
A previously validated DF with model-based tracking system combined with CT bone models was used for acquiring the anatomic coordinate systems located in the tibia and femur, bone poses, and 6 degrees of freedom kinematics. 40 CT images were used for patient-specific 3-dimensional bone model reconstruction by a semiautomatic segmentation process (Seg3D Version 2.4.4; University of Utah). Model-based tracking was conducted using the X4D application within the DSX Suite (C-Motion), which generated digitally reconstructed radiographs from CT-based bone models to align with processed radiographic images. Joint angles and translation, or osteokinematics, were determined from transformation of the tibia to the femur in successive rotations about the flexion-extension axis, abduction-adduction axis, and internal-external rotation axis and the position of the femur's origin relative to the tibia's origin, respectively. 29
MRI segmentations were used for reconstruction of the bone and cartilage surface models, and the surface models were smoothed and meshed with an edge length of 0.75 mm (MeshLab Version 2012.12; Visual Computing Laboratory). The MRI-based bone models were aligned with the corresponding CT models to determine the positions of articular cartilage in each frame. This process involved registering the MRI bones to the CT bones to obtain the MRI-to-CT transformation matrix. The MRI-to-CT transformation matrix aligned the MRI bone-cartilage models with the CT bones. The MRI-based articular cartilage model poses in each time frame were then found by combining the MRI-to-CT transformation matrix with the model-based tracking solution that determined the transformation from the CT model to the motion capture laboratory. The center of contact was determined using a distance-weighted centroid algorithm. 10 Additionally, contact overlap was defined as the percentage of tibial cartilage surface mesh elements in contact with femoral cartilage surface mesh elements relative to the total tibial cartilage surface mesh elements within the corresponding compartment.
Each activity was normalized to key biomechanical events to ensure comparable data across participants, despite differences in task execution timing. Time points for analysis were selected to maximize the number of valid trials across all participants. Landing trials were time normalized relative to the peak vertical GRF (vGRF), spanning the loading phase from 50% of the peak vGRF to the peak (100%) and back to 50% of the peak vGRF. Outcomes for landing were evaluated at 5 time points: 50%, 75%, 100%, 75%, and 50% of peak vGRF. Lunging movements were angle normalized to the maximum knee flexion angle, covering the range from 85% of maximum flexion to the peak (100%) and returning to 85% of maximum flexion. Outcomes for lunging were assessed at 7 time points: 85%, 90%, 95%, 100%, 95%, 90%, and 85% of maximum flexion. Finally, the pivoting activity was normalized to the maximum anteroposterior (AP) position of the lateral knee marker, ranging from 90% to 100% and returning to 90%, to evaluate the pivoting movement relative to AP knee displacement. Pivoting outcomes were analyzed at 5 time points: 90%, 95%, 100%, 95%, and 90% of the AP lateral knee marker (ALKM) position. One participant was excluded from the pivoting analysis because the contralateral knee was not fully captured within the region of interest (90% of the ALKM position). Consequently, 44 trials (from both reconstructed and contralateral healthy knees) were analyzed from 12 participants for single-leg landing and lunging, and 44 trials were analyzed from 11 participants for pivoting.
The center of contact on the tibia was tracked at each time point to measure the length of the tibial contact path for each activity. Furthermore, AP and mediolateral excursions of the contact path were calculated as the difference between the maximum and minimum contact locations for each respective outcome across the entire trial for both compartments.
Statistical Analysis
All comparisons were performed using linear mixed models with repeated measures. For outcomes with multiple time points, the models included limb (ACLR+M vs contralateral), time point, and their interaction as fixed effects, with participant as a random effect. Including the limb × time point interaction allowed for pairwise comparisons between limbs at each time point. For outcomes defined by a single value across the entire trial (eg, excursion and contact path), models included limb as a repeated factor and participant as a random effect. All available trials for each participant were included in analyses to maximize statistical power rather than averaging trials within participants. Results are reported as least squares means and standard errors (SEs) from the mixed models. Statistical significance was set a priori at an alpha of .05. All analyses were performed using SAS (Version 9.4; SAS Institute).
Results
Single-Leg Landing
During single-leg landing, the ACLR+M knee showed increased anterior tibial translation relative to the femur at all time points, reaching a mean value of 3.6 mm (SE, 0.7 mm; P = .01) at peak vGRF (Figure 2A) compared with the contralateral healthy knee. The ACLR+M knee also showed greater abduction (1.8° [SE, 0.7°]; P = .02 at peak vGRF) compared with the contralateral healthy knee at 75% and 50% after peak vGRF (Figure 2A). At the last time point of 50% after peak vGRF, ACLR+M knees demonstrated less flexion (8° [SE, 0.7°]; P < .001), and the tibia was located more lateral (0.6 mm [SE, 0.3 mm]; P = .06) and further inferior (2.1 mm [SE, 0.9 mm]; P = .04), compared with the contralateral healthy knees (Figure 2A). Only internal/external rotation of the tibia relative to the femur did not exhibit a significant difference between ACLR+M and contralateral knees during landing (Figure 2A).
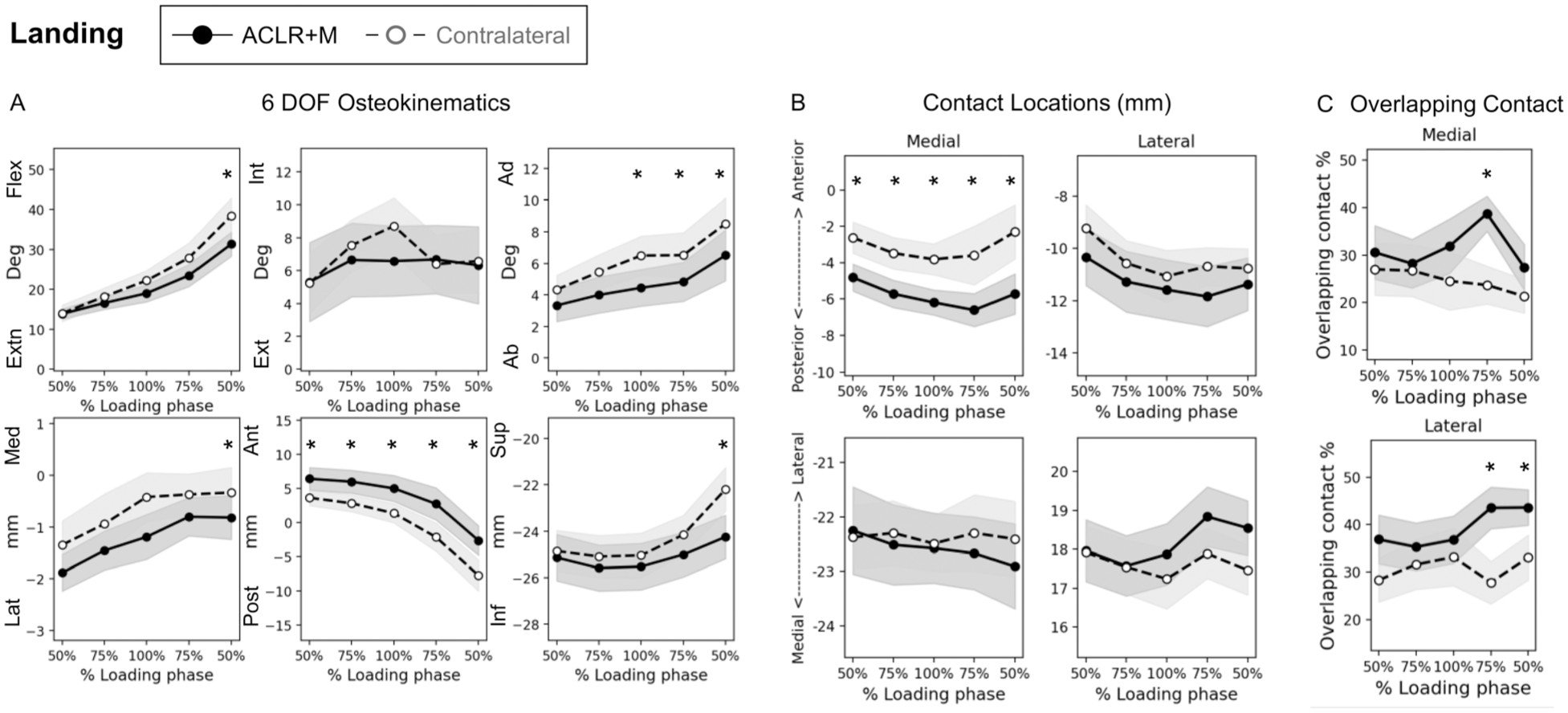
Single-leg landing. (A) Six degrees of freedom osteokinematics (tibia relative to femur), (B) anteroposterior (top) and mediolateral (bottom) contact locations on the tibia, and (C) contact overlap calculated as a percentage of the corresponding tibial compartment. Shaded areas represent standard errors. Significant differences between ACLR+M and contralateral healthy knees are indicated with an asterisk (P < .05). Ab, abduction; Ad, adduction; Ant, anterior; Ext, external; Extn, extension; Flex, flexion; Inf, inferior; Int, internal; Lat, lateral; Med, medial; Post, posterior; Sup, superior.
In the medial compartment, the contact location was shifted posteriorly on the tibia in the ACLR+M knee compared with the contralateral healthy knee at all time points, reaching a mean value of 2.5 mm (SE, 1 mm; P = .02) at peak vGRF (Figure 2B). Increased tibiofemoral overlapping contact was observed in the ACLR+M knee in the medial compartment (16% of tibial compartment; P = .01) at 75% after peak vGRF, the lateral compartment (15% of tibial compartment; P = .01) at 75% after peak vGRF, and the lateral compartment (12% of tibial compartment; P = .04) at 50% after peak vGRF (Figure 2C).
In the lateral compartment, the ACLR+M knee also demonstrated a shorter tibial contact path (2.4 mm [SE, 1.1 mm]; P = .05) and smaller mediolateral excursion (1.3 mm [SE, 0.5 mm]; P = .03) compared with the contralateral healthy knee. A trend toward a smaller AP excursion was observed in both the medial and lateral compartments (1.5 mm [P = .08] and 1.6 mm [P = .06], respectively).
Lunging
During lunging, the ACLR+M knee demonstrated significantly less flexion (4.5° [SE, 2°]; P = .04) and greater internal rotation (2° [SE, 0.9°]; P = .05) of the tibia at peak flexion compared with the contralateral healthy knee (Figure 3A). No other significant osteokinematic differences were observed during lunging. In the medial compartment, a lateral shift in the tibial contact location was observed at 90% after peak flexion (1.7 mm [SE, 0.8 mm]; P = .05) (Figure 3B). Larger AP excursion of the tibial contact path in the lateral compartment was observed during lunging (0.36 mm [SE, 0.13 mm]; P = .02). No other significant differences in arthrokinematics and overlapping contact were found during lunging.

Lunging. (A) Six degrees of freedom osteokinematics (tibia relative to femur), (B) anteroposterior (top) and mediolateral (bottom) contact locations on the tibia, and (C) contact overlap calculated as a percentage of the corresponding tibial compartment. Shaded areas represent standard errors. Significant differences between ACLR+M and contralateral healthy knees are indicated with an asterisk (P < .05). Ab, abduction; Ad, adduction; Ant, anterior; Ext, external; Extn, extension; Flex, flexion; Inf, inferior; Int, internal; Lat, lateral; Med, medial; Post, posterior; Sup, superior.
Pivoting
During pivoting, the ACLR+M knee demonstrated consistent anterior tibial translation at all time points, reaching a mean side-to-side difference of 3 mm (SE, 0.9 mm; P = .002) at the peak ALKM position (Figure 4A). Compared with the contralateral healthy knee, greater lateral tibial translation was observed in the ACLR+M knee at the peak ALKM position (0.9 mm [SE, 0.4 mm]; P = .02), at 95% after the peak ALKM position (1 mm [SE, 0.4 mm]; P = .009), and at 90% after the peak ALKM position (1.2 mm [SE, 0.04 mm]; P = .004) (Figure 4A). The reconstructed knee was more internally rotated (3.9° [SE, 1.6°]; P = .02) at 90% after the peak ALKM position. The contact location was located more posterior on the tibia in the ACLR+M knees compared with the contralateral healthy knees at 95% before the peak ALKM position in the medial compartment and at 90% after the peak ALKM position in the lateral compartment (Figure 4B). In the medial compartment, overlapping contact was greater in the ACLR+M knee (11.3% of tibial compartment; P = .02) at the peak ALKM position as well as at 90% and 95% before the peak ALKM position (Figure 4C). Overlapping contact was greater in the ACLR+M knee's lateral compartment at 90% after the peak ALKM position (Figure 4C). No side-to-side difference was found in contact path and excursion during pivoting.

Pivoting. (A) Six degrees of freedom osteokinematics (tibia relative to femur), (B) anteroposterior (top) and mediolateral (bottom) contact locations on the tibia, and (C) contact overlap calculated as a percentage of the corresponding tibial compartment. Shaded areas represent standard errors. Significant differences between ACLR+M and contralateral healthy knees are indicated with an asterisk (P < .05). Ab, abduction; Ad, adduction; Ant, anterior; Ext, external; Extn, extension; Flex, flexion; Inf, inferior; Int, internal; Lat, lateral; Med, medial; Post, posterior; Sup, superior.
Discussion
The objective of this study was to assess how ACLR+M affects 3-dimensional osteokinematics and articular cartilage arthrokinematics by comparing the reconstructed knee with the contralateral healthy knee during 3 dynamic activities that challenge the knee and are common during participation in everyday and sporting activities. We hypothesized that ACLR+M would not restore sagittal-, frontal-, and transverse-plane osteokinematics and cartilage arthrokinematics compared with the contralateral healthy knee.15,21,22,36,53 As expected, we found altered osteokinematics and cartilage arthrokinematics in the reconstructed knee compared with the contralateral healthy knee. The main findings of this study were as follows: (1) increased anterior translation of the tibia in the ACLR+M knee along with posterior shifts in contact locations on the tibia during landing and pivoting, (2) less flexion (ie, more extension) in the ACLR+M knee during landing and lunging, (3) more overlapping contact in the ACLR+M knee in both the medial and lateral compartments during landing and pivoting, and (4) larger AP excursion of the tibial contact path in the lateral compartment during lunging. Our findings build upon previous reports1,32 by demonstrating that osteokinematic and arthrokinematic deviations persist primarily during high-demand, low-flexion activities such as landing and pivoting, whereas restoration appears more complete during high-flexion movements such as lunging.
Posterior shifts in contact locations, along with increased anterior tibial translation, during landing and pivoting indicated that ACLR+M did not fully restore normal AP knee joint movements at 1 to 2 years after surgery. This finding aligns with previous studies that have focused on ACL-deficient and ACLR knees during walking, double-leg squatting, and lunging.9,21,27,37 Hosseini et al 21 reported a 4.8-mm posterior shift in the center of contact on the lateral tibia in ACLR knees compared with contralateral knees at 0° and 15° of flexion, with no differences observed between 30° and 90° of flexion during a single-leg quasistatic lunge at 6 months after surgery. Similarly, in a static MRI study, Amano et al 4 reported anterior tibial translation in reconstructed knees at 1-year follow-up. In our study, we observed a consistent 2.3- to 3.0-mm posterior shift in the tibial contact location in the medial compartment during single-leg landing and a 2.7-mm posterior shift in the medial compartment of the tibia at 95% before the peak ALKM position during pivoting. However, no differences were detected in contact locations during lunging. Integrating our findings with previous studies, it can be concluded that anterior tibial translation is more evident during activities with less knee flexion, such as landing and pivoting, in which patients do not exceed 30° to 40° of flexion. This may be attributed to reduced involvement of the ACL and the reconstructed graft at higher knee flexion angles in which no AP differences between reconstructed and healthy knees have been observed. 21 Anterior translation of the tibia in ACLR+M knees may be indicative of permanent elongation of the reconstructed graft at 1 to 2 years after surgery, 11 potentially changing the contact loading pattern in articular cartilage and exposing it to the onset and progression of degeneration and subsequently the clinical manifestation of PTOA.
We found that the reconstructed knee exhibited a lower flexion angle (ie, more extended) compared with the contralateral healthy knee during the last phase of single-leg landing, despite no such side-to-side differences being observed during earlier phases within the activity. This observation supports the findings of previous studies on knee mechanics during single-leg hopping in patients with ACL-deficient and ACLR knees.15,19,41,42 In 2010, Deneweth et al, 15 through a DF model-based tracking study, observed that the reconstructed knee consistently maintained a more extended position throughout single-leg hop landing, with a mean side-to-side difference of 8° to 10°. However, their study reported osteokinematics within the initial 0 to 250 milliseconds after landing, potentially overlooking variations in landing speed or differences in GRFs resulting from higher initial jump heights among patients. 15 In contrast, our study measured osteokinematics from 50% to 100% and back to 50% of the peak vGRF to determine single-leg landing mechanics with regard to the loading phase. We observed that there was no side-to-side difference during earlier phases within the activity; however, a significant difference was evident at the final time point (at 50% after peak vGRF). This finding suggests that while patients initially land with similar flexion angles in both knees, over the course of the activity, they progressively adopt a strategy of less flexion in their ACLR+M knee. A more extended landing posture may represent a compensatory mechanism to limit abnormal anterior tibial translation. 15 Specifically, by avoiding deeper flexion, patients may reduce the demand on the posterior femoral rollback mechanism, thereby enhancing joint stability.15,42,43 Moreover, we found lower maximum flexion during lunging in the reconstructed knee compared with the contralateral healthy knee. Reduced peak flexion observed during both single-leg landing and lunging might be attributable to a relatively weakened quadriceps muscle, leading to a diminished knee extensor moment, which is a frequent consequence of ACL injuries and reconstruction.3,22,26,36
We also found that contact overlap was elevated in the reconstructed knee compared with the contralateral healthy knee at the fourth time point (75% after peak vGRF) in the medial compartment and at the last 2 time points (75% and 50% after peak vGRF) in the lateral compartment during single-leg landing. The participants who enrolled in our study (Table 1) underwent partial meniscectomy and/or meniscal repair. The increase in contact overlap observed in our study might be caused by meniscal injuries and surgery because the menisci play a critical role in shock absorption and contact stress distribution about the tibiofemoral joint.30,38 The observed increase in contact overlap in the reconstructed knee relative to the contralateral healthy knee during single-leg landing, in both compartments immediately after peak GRFs, suggests that the surgical knee may be less effective in transmitting high loads across the tibiofemoral joint, thereby leading to increased cartilage contact. In an in vitro study, McCann et al 30 demonstrated that meniscectomy results in elevated contact stress and an increased frictional coefficient at the articular interface, causing immediate surface fibrillation, biomechanical wear, and the permanent deformation of articular cartilage. Moreover, Peña et al 38 showed that maximal contact stress increases in response to axially directed femoral compressive loads at 0° of flexion after meniscectomy compared with a healthy joint using finite element analysis. With a similar methodology to our study, Zheng et al 55 reported an increase in contact overlap in patients who underwent isolated meniscectomy in the lateral compartment. The increase in overlapping contact observed in our study might serve as a biomarker for the development of PTOA after ACLR+M, as larger areas of articular cartilage are subjected to altered contact stress and potential fibrillation.30,38
The ACLR+M knee exhibited a reduced contact path length and diminished mediolateral excursion within the lateral compartment, alongside a trend toward a smaller AP excursion in both compartments, during single-leg landing. The reductions in the length of the contact path and excursion could be attributed to the decreased range of flexion-extension throughout the trial, with the reconstructed knee demonstrating less flexion at the final time point, despite no initial side-to-side differences. However, during lunging, an increase in AP excursion was observed in the ACLR+M knee relative to the contralateral healthy knee in the lateral compartment. This increase in AP excursion might reflect variations in muscle strength or the mechanical properties of the graft, such as differences in fiber orientation and stiffness compared with the native ACL, potentially leading to abnormal cartilage arthrokinematics. Consequently, during lunging, a broader area of cartilage is subjected to loading, particularly regions that are not typically loaded in this activity compared with the contralateral healthy knee. These altered loading patterns may serve as a biomarker for the initiation of PTOA after ACLR+M.
Several limitations are acknowledged in this study. First, the sample size of participants was relatively small, although it aligns with those in previous studies employing similar high-speed fluoroscopic methodologies. 44 Additionally, our use of nondeformable cartilage models tends to overestimate contact overlap. However, this approach introduces a systematic bias affecting both the ACLR+M and contralateral healthy knees, thereby minimizing the error in side-to-side comparisons. Furthermore, our emphasis on capturing natural movement patterns in patients using our DF system resulted in a small FOV, possibly limiting the range of motion collected and analyzed. This study included patients with varying graft types, tunnel placement, and meniscal procedures, and we did not control for this heterogeneity. These surgical differences may have contributed to a variability in postoperative kinematics and should be addressed in future studies with larger, more standardized cohorts. 38 Although previous DF work has found no significant differences in arthrokinematics between the ACLR and combined ACLR and LET groups, 33 the inclusion of a participant who had undergone LET remains a potential limitation, and further studies using standardized surgical techniques are warranted. We did not perform subgroup analyses comparing meniscal repair and partial meniscectomy because of the limited sample size. Although meniscal volume and procedure type may influence postoperative contact mechanics, all participants met the inclusion criterion of ACLR with a concomitant meniscal injury, a population with an elevated risk for PTOA and exposed to nonphysiological cartilage loading. Given our focus on shared biomechanical pathways rather than treatment-specific effects, participants were pooled. Future studies with larger cohorts should investigate the effect of meniscal procedure type on arthrokinematics. Moreover, the present study did not stratify results by the location of meniscal surgery (medial vs lateral) because of the small sample size and mixed injury patterns. Given the distinct biomechanical role of each meniscus, future studies with larger cohorts should examine compartment-specific effects on postoperative arthrokinematics.
Conclusion
This study provides in vivo evidence of altered knee joint kinematics and cartilage arthrokinematics at 1 to 2 years after ACLR+M. We highlight the presence of abnormal joint mechanics during high-demand activities—landing, lunging, and pivoting—that many patients resume within the first year after surgery. These activities challenge the reconstructed knee in ways that gait and low-impact tasks may not, exposing deficits in load dissipation and joint stability. Our findings suggest that the knee after ACLR+M may not effectively restore normal cartilage arthrokinematics, resulting in increased cartilage contact and altered loading patterns that could accelerate the degenerative process. This study is the first to provide in vivo evidence of joint dysfunction during such high-demand tasks in this population, offering a new framework for assessing postoperative joint health and motivating targeted rehabilitation strategies aimed at reducing the long-term PTOA risk. Combining the findings of this study with future research will enhance our understanding of PTOA initiation by tracking longitudinal changes after surgery.
Footnotes
Final revision submitted January 5, 2026; accepted January 12, 2026.
One or more of the authors has declared the following potential conflict of interest or source of funding: Research reported in this publication was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under award number R21AR077371 (N.M.F.). M.G. is an imaging consultant for Smith & Nephew. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the University of Vermont's Institutional Review Board (No. 1092).
