Abstract
Background:
Platelet-rich plasma (PRP) has gained widespread use an adjunct to orthopaedic procedures, yet clinical studies have reported conflicting efficacy. The Minimum Information for Studies Evaluating Biologics in Orthopaedics (MIBO) guidelines were published in 2017 to standardize reporting of PRP preparation, characterization, and delivery, which may explain the heterogeneity of clinical outcomes in studies evaluating PRP for anterior cruciate ligament reconstruction (ACLR).
Purpose:
To evaluate adherence to MIBO guidelines in studies evaluating PRP-augmented ACLR and to assess whether reporting adherence improved after the publication of the 2017 guidelines.
Study Design:
Systematic review; Level of evidence, 3.
Methods:
A systematic literature search was conducted using the PubMed/MEDLINE, Embase, and Web of Science databases in March 2025 to identify studies evaluating PRP-augmented ACLR. Included studies were randomized controlled trials (RCTs), prospective and retrospective cohort studies, and case-control studies published in the English language. Two independent reviewers extracted data on study characteristics and assessed adherence to all 23 MIBO checklist items.
Results:
A total of 26 studies (14 RCTs, 12 observational studies) involving 1319 patients were included. The mean MIBO adherence was 40.1% (9.2/23 items) for pre-MIBO studies and 50.1% (11.5/23 items) for post-MIBO studies, with no statistically significant improvement (P = .115). The range of adherence was 17.4% to 82.6%. The most poorly reported items included CONSORT (Consolidated Standards of Reporting Trials)/STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) compliance, anti-inflammatory medication use, platelet recovery rate, PRP storage conditions, and final PRP analysis. No significant differences in MIBO adherence were found between RCTs and observational studies (P = .578). Half of the included studies reported positive outcomes with PRP augmentation, while half reported neutral outcomes.
Conclusion:
Adherence to the 2017 MIBO guidelines in studies evaluating PRP-augmented ACLR remains poor, with no significant improvement after guideline publication. Critical information regarding PRP preparation, characterization, and platelet recovery rate continues to be inadequately reported, limiting study reproducibility and the ability to determine true PRP efficacy.
Keywords
Anterior cruciate ligament (ACL) injury is one of the most common orthopaedic sports injuries, with an incidence of 68.6 per 100,000 person-years. 33 ACL reconstruction (ACLR) is considered the preferred surgical approach and is recommended as the primary treatment for ACL injuries by the American Academy of Orthopaedic Surgeons (AAOS) guidelines.6,43 Despite the promising results of ACLR and advancements in surgical techniques, complications after ACLR remain relatively common, including anterior knee pain, stiffness due to graft malpositioning, secondary meniscal lesions, and graft rerupture. 30
Platelet-rich plasma (PRP) has gained attention in orthopaedics as a primary or adjunctive treatment to enhance both operative and nonoperative outcomes for various pathologies. PRP is thought to exhibit anti-inflammatory and potentially regenerative properties through the mediation of numerous biochemical pathways, including but not limited to vascular endothelial growth factor, transforming growth factor-β, platelet-derived growth factor, and other cytokines and growth factors, as well as through modulation of inflammatory pathways such as nuclear factor-κB. 37 Preclinical studies have suggested that PRP has the potential to improve the healing of tendinous ACL grafts both histologically and biomechanically.3,12,51 However, recent systematic reviews evaluating the efficacy of PRP-augmented ACLR in human patients have reported conflicting findings. Some studies demonstrate only short-term benefits of PRP augmentation, while others show no improvement at all.7,36,53
One possible challenge and explanation for the heterogeneity of findings in PRP-augmented ACLR studies is the variability in preparation methods, composition, and application of PRP. To address these inconsistencies, the AAOS released the Minimum Information for Studies Evaluating Biologics in Orthopaedics (MIBO) guidelines in May 2017. 25 These guidelines provide a structured 23-item framework to improve transparency and reproducibility in research involving biological materials, including PRP. Despite the release of these guidelines, several studies have found continued poor adherence to reporting guidelines in other orthopaedic applications, including knee osteoarthritis, lateral epicondylitis, and rotator cuff repair.11,15,26
The purpose of the present systematic review was to evaluate the adherence to MIBO guidelines in the literature evaluating PRP-augmented ACLR. By identifying gaps in literature reporting, we hope to evaluate the reliability of existing evidence and guide improvements in the future design and publication of PRP-augmented ACLR studies. Clarifying these methodological shortcomings may help resolve ongoing uncertainty regarding the true efficacy of PRP in ACLR.
Methods
Protocol
The methodology for this systematic review followed the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses). 28 The review protocol was registered in the PROSPERO international systematic review database (registration No.: CRD420251031511).
Literature Search
A systematic literature search was conducted to identify studies that examined PRP-augmented ACLR. The review included peer-reviewed research articles published in the English language. No date limit was applied. No artificial intelligence–generated content filters were applied during the search. One reviewer (D.M.) performed a literature search on March 15, 2025, using the following research databases: PubMed/MEDLINE, Embase, and Web of Science. The following search strategy was used: (“Platelet-Rich Plasma”[MeSH] OR “Blood platelets”[MeSH] OR “Platelet-Rich Plasma” OR “PRP” OR “Platelet”)AND (“Anterior Cruciate Ligament”[MeSH] OR “Anterior Cruciate Ligament Reconstruction”[MeSH] OR “Anterior Cruciate Ligament/pathology”[MeSH] OR “Anterior Cruciate ligament” OR “ACL Reconstruction” OR “Anterior Cruciate ligament reconstruction.”
Inclusion and Exclusion Criteria
This systematic review included randomized controlled trials (RCTs), prospective and retrospective cohort studies, and case-control studies investigating PRP-augmented ACLR in humans. Included studies were published in peer-reviewed journals in the English language. Animal studies, cadaveric studies, review articles, expert opinions, case reports, and conference abstracts were excluded. Studies were also excluded if they had a low sample size (n ≤ 10), studied PRP for non-ACLR procedures, or used other orthobiological therapies.
Study Selection and Data Extraction
The final search retrieved 1319 references. This set was uploaded to Covidence (https://www.covidence.org/) for screening, which identified 484 duplicates, leaving 835 studies eligible for initial screening. Two authors (D.M. and T.W.) independently screened the titles and abstracts of the identified articles. Any conflicts were discussed with a third party (M.W.) until a consensus was reached. Abstracts that did not include sufficient information to determine eligibility for inclusion were retrieved for full-text evaluation. A total of 37 articles met the inclusion criteria for full-text review, while 798 studies were excluded from the analysis. Full-text review was performed independently by 2 reviewers (T.W. and M.W.). Any conflicts were discussed with a third party (D.M.) until a consensus was reached. After the full-text review, 26 studies remained eligible for the data extraction process. A PRISMA flow diagram illustrating the data selection process can be found in Figure 1. Data from eligible studies were extracted by 2 reviewers (T.W. and M.W.). A standardized data extraction sheet was used to collect study characteristics (authors, year of publication, journal, and study design), patient characteristics (age and sex), and adherence to the MIBO guidelines, which was assessed according to the 12 MIBO categories and 23 statements for PRP (Figure 1).

PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram.
Statistical Analysis
Descriptive statistics were presented in tabular form. Studies were classified as having a positive outcome if they reported statistically significant improvements histologically or radiographically (graft healing or decreased tunnel widening on magnetic resonance imaging), decreased rates of arthrofibrosis (defined by decreased range of motion 3 months postoperatively), early graft failure/revision, decreased inflammatory markers, or improved patient-reported outcome measures (Lysholm score, International Knee Documentation Committee score, Knee injury and Osteoarthritis Outcome Score, visual analog scale score, or Tegner activity scale score). The overall MIBO item percentage normality was evaluated using the Shapiro-Wilk test. It was then determined that parametric testing was appropriate and 2-sided Student t tests were performed to compare overall item percentage adherence to MIBO guidelines between studies published 2018 and earlier (pre-MIBO) and after 2018 (post-MIBO), as well as across study design types, using SPSS Statistics (Version 29.0.2.0; IBM). As the MIBO guidelines were published in mid-2017, studies through 2018 were classified as pre-MIBO to allow adequate time for adoption. Individual MIBO items for pre-MIBO and post-MIBO were evaluated using chi-square tests. Significance was reported using Fisher exact (exact 2-sided) tests, where a P value <.05 was considered significant.
Risk of Bias Assessment
Two independent reviewers (D.M. and A.P.) assessed the risk of bias for each included study. RCTs were appraised using the Cochrane risk of bias tool, while observational studies were evaluated using the Newcastle-Ottawa Scale. Discrepancies were resolved through discussion or consultation with a third reviewer. Risk of bias analysis was performed to adhere to PRISMA guidelines; however, because the primary aim of this review was to evaluate MIBO compliance rather than study outcomes, risk of bias was not expected to directly influence the findings.
Results
Four studies included in this review were high risk of bias, mainly due to concerns with randomization and blinding of patients/investigators. The remaining 10 studies were deemed as “some concerns” (Figure 2).

Cochrane risk of bias assessment.
Of the 12 observational studies included in this review, 2 were determined to be moderate risk of bias, with the remaining 10 being considered low risk of bias (Table 1).
Newcastle-Ottawa Risk of Bias Assessment a
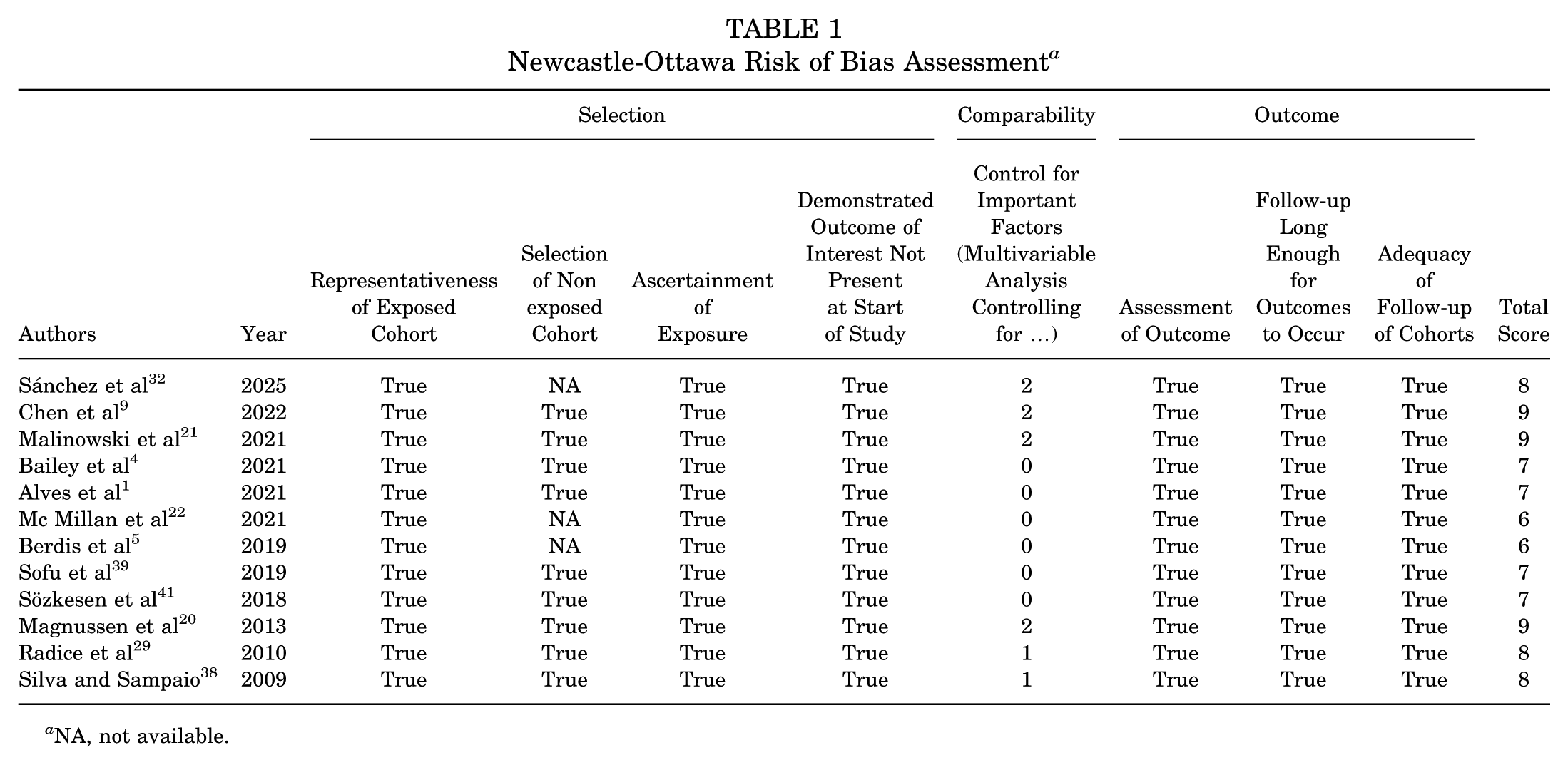
NA, not available.
Of the 26 included studies, 13 were pre-MIBO ( 2018 or earlier publication date) and 13 were post-MIBO (publication date after 2018). Thirteen of the 26 studies found positive outcomes in PRP-augmented ACLR, while two studies found negative outcomes and the remaining eleven studies had neutral outcomes. Fourteen of the 26 included studies were RCTs, and 12 were observational studies. The range of MIBO item score percentages of the included studies was 17.4% to 82.6%, with both studies achieving scores of 82.6% being post-MIBO RCTs (Table 2).
Study Characteristics a

NR, not reported; MIBO, Minimum Information for Studies Evaluating Biologics in Orthopaedics; PRP, platelet-rich plasma; RCT, randomized controlled trial.
The most poorly reported MIBO items pre-MIBO were 1, 4, 5, 7, 8, 10, 11, 14, 15, and 18. While there were no statistically significant improvements in item scores post-MIBO, items 1, 4, 6, and 10 approached significant improvement (Table 3).
MIBO Scores by Item a

CONSORT, Consolidated Standards of Reporting Trials; MIBO, Minimum Information for Studies Evaluating Biologics in Orthopaedics; PRP, platelet-rich plasma.
Overall, there were no statistically significant differences in MIBO overall item scores pre- and post-MIBO guidelines in 2017, although improvement approached significance (P = .115) (Table 4). There were no differences between MIBO adherence by study type (RCT vs observational).
Statistical Analysis of MIBO Adherence by Publication Date and Study Design a
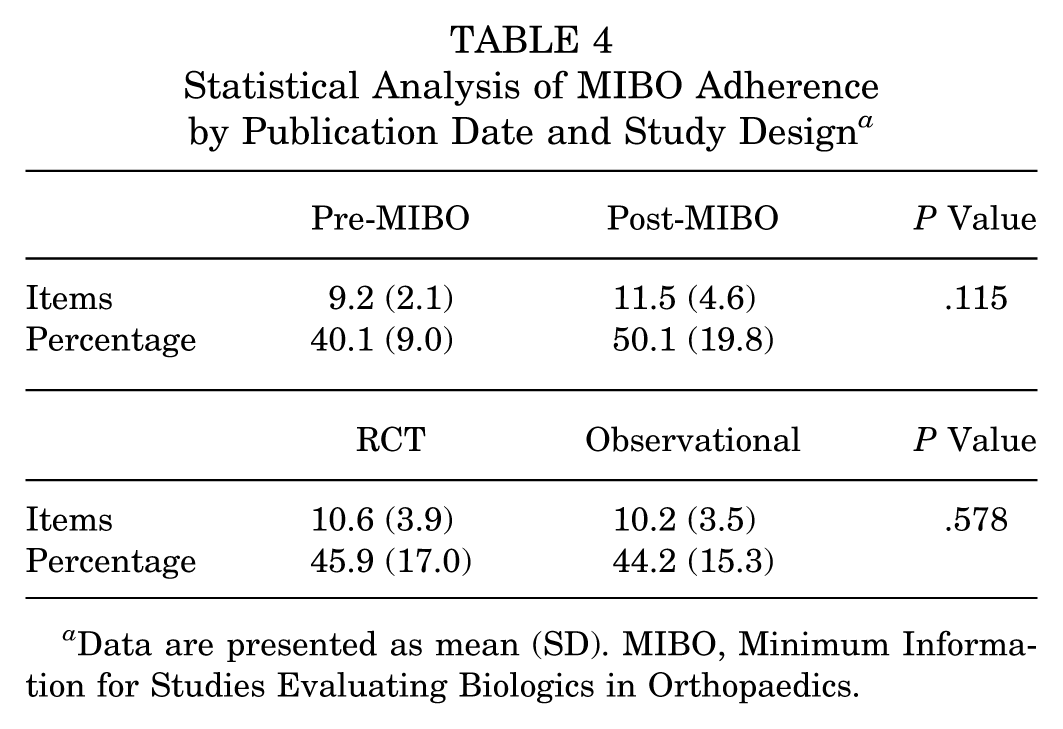
Data are presented as mean (SD). MIBO, Minimum Information for Studies Evaluating Biologics in Orthopaedics.
Discussion
In this systematic review, we evaluated adherence to the MIBO guidelines for studies evaluating the use of PRP for ACLR augmentation. Among the 26 included studies, overall adherence to MIBO guidelines was poor, with a mean item score percentage of 40.1% pre-MIBO and 50.1% post-MIBO. Although there was a trend toward improved reporting after the release of the MIBO guidelines in 2017, the improvement did not reach statistical significance (P = .115). Similarly, there were no differences in reported item percentages between RCTs and observational studies. Certain checklist items, including CONSORT (Consolidated Standards of Reporting Trials) compliance, comorbidities, anti-inflammatory use, preoperative imaging, platelet recovery rate, PRP storage, and PRP analysis, remained poorly reported both before and after the release of the 2017 MIBO guidelines.
Comparison With Prior Literature
Previous systematic reviews have also evaluated the adherence to MIBO guidelines before and after their publication. DeClercq et al 11 evaluated MIBO adherence in 19 studies examining PRP for full-thickness rotator cuff repair published between 2008 and 2019. Similar to our findings, they noted reporting deficiencies in whole-blood and PRP processing, with no studies providing sufficient information to replicate PRP preparation protocols. Overall adherence across all studies was 58.5%, which included both pre-MIBO and post-MIBO studies, similar to the poor adherence rates observed in our study of 40.1% and 50.1%, respectively.
Similar findings have been reported in other PRP applications. Nakagawa et al 26 found that, in the context of PRP for knee osteoarthritis, the most poorly reported items included whole-blood analysis, PRP analysis, platelet recovery rate, and PRP activation. This study also found that adherence to MIBO guidelines paradoxically decreased in the post-MIBO group. While our study did not find a statistically significant improvement in MIBO item reporting in the post-MIBO group, our findings complement this previous work by identifying continued deficiencies in PRP analysis and platelet recovery rate reporting. This omission is clinically important, as prior studies have established that effective PRP should contain a platelet concentration of at least 3 to 5 times baseline whole-blood values, with a target of 1.4 to 1.9 × 106 platelets/μL, with an absolute dose of >3.5 billion platelets per treatment.2,10 It is therefore crucial to report PRP analysis and platelet recovery rate to ensure the preparation protocols are achieving adequate concentrations.
These findings are concerning because variability in PRP preparation and characterization is one of the principal explanations for heterogeneity in clinical outcomes across trials. Half of the included studies in our review demonstrated positive outcomes with PRP augmentation, while the other half found neutral or no impact. Previous systematic reviews evaluating the reporting of PRP preparation and composition in various musculoskeletal applications in studies from 2006 to 2022 found that only 10% to 12.1% of studies provided comprehensive reporting of preparation protocols that can be replicated by future investigators.8,18 Furthermore, only 16% to 26.6% of studies reported quantitative final PRP product composition.8,18 In our review, just 15.4% of post-MIBO studies reported final PRP analysis, highlighting the persistent gap between guideline recommendations and practice. Platelet recovery rate is likely critically underreported because commercial PRP systems do not routinely quantify this parameter. Additionally, there is a lack of a standardized method for calculating recovery rates. Limited reporting of platelet concentrations significantly inhibits meta-analysis of PRP studies and our ability to determine whether negative study outcomes result from suboptimal PRP preparation or true lack of efficacy.
Our study also found poor compliance with CONSORT and STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) reporting guidelines for RCTs and observational studies, respectively, with 0% compliance pre-MIBO and 30.8% compliance post-MIBO. Our review includes studies published between 2008 and 2025, with the oldest observational study published in 2009. This is concerning given that CONSORT has been available since 1996 (with major updates in 2001, 2010, and 2025) and STROBE since 2007.16,49 Systematic reviews evaluating adherence to CONSORT guidelines have found generally poor adherence, although journals that specifically endorse CONSORT exhibited 1.5 to 2 times higher likelihood of reporting appropriate CONSORT items. 45 Similarly, STROBE checklist items are poorly reported across observational studies, with 1 study finding only 41.2% to 47.4% adherence across studies published in allergy and immunology fields. 50 Another study specifically evaluating STROBE compliance in the orthopaedic literature found an improvement in compliance from 38% in 2005 to 58% in 2011, but still poor adherence overall for specific items including study setting description, handling of missing data, potential bias, and power analysis. 40
Improving Reporting
Taken together, these findings highlight that poor adherence to reporting standards is not unique to PRP-augmented orthopaedic research but represents a broader issue across all medical research. The limited uptake of CONSORT and STROBE guidelines may help partially explain why the MIBO guidelines, despite being created specifically to improve reporting of orthobiological interventions, have similarly failed to demonstrate widespread adherence. Addressing these systemic issues will likely require stronger enforcement of guidelines by journals and peer reviewers, as well as increased awareness among investigators on the importance of transparent and standardized reporting of study design and methods. First, journals can play a central role by mandating the inclusion of completed reporting checklists on manuscript submission. Previous studies have shown that journal endorsement leads to improved reporting, suggesting that editorial policies can directly influence reporting quality.34,45 Interestingly, an RCT found that emailing investigators during the submission process to remind them to ensure CONSORT compliance did not improve reporting quality. 42 Taken together, these previous studies indicate that strict policies of adherence to published guidelines with submitted checklists may be the most effective way to improve reporting. Furthermore, funding bodies and orthopaedic societies like the AAOS could promote adherence by requiring MIBO compliance as a condition for grant funding or study endorsement. Ultimately, to improve the quality of orthobiological research, journals may need to require submission of the MIBO checklist with all applicable manuscripts and consider adherence in editorial decisions regarding rejection or progression to peer review.
Limitations
This systematic review has several limitations. First, the number of eligible studies was small, limiting our power to detect statistically significant differences between pre- and post-MIBO studies. Limiting our analysis to published, peer-reviewed studies in the English language introduces the possibility of publication bias. Adherence to MIBO criteria is based on reported information, which may not fully reflect whether items were performed and recorded but not included in the final paper. Additionally, we did not calculate interrater reliability statistics (eg, Cohen kappa) for MIBO scoring, which could strengthen the reproducibility of future systematic reviews evaluating the MIBO guideline adherence. Our study also primarily focused on reporting quality and did not evaluate methodological differences in studies, including type of graft used, application site of PRP, and reported outcomes (radiographic, clinical, histological, etc). Positive/neutral outcomes were reported with the sole intention of demonstrating the variability of PRP efficacy in ACLR, but not to assess the extent or validity of these outcomes. We did not perform sensitivity analyses for different outcome categories due to heterogeneity of outcome measures across studies.
Conclusion
The current study demonstrates that adherence to MIBO guidelines in studies evaluating PRP-augmented ACLR remains poor, with only slight nonsignificant improvements after the publication of the 2017 guidelines. From a clinical perspective, improving adherence to MIBO guidelines is essential to advancing the field of orthobiologics. Without standardized and transparent reporting, it is difficult to determine whether neutral/negative study outcomes reflect true lack of PRP efficacy, or methodological variability in PRP preparation and delivery. By enforcing guideline compliance, through journal requirements, peer reviewer oversight, and investigator education on the importance of transparent reporting, future research will be better positioned to determine which PRP applications offer meaningful improvements in tissue healing, patient-reported outcomes, and long-term musculoskeletal health. Ultimately, improved MIBO adherence is not just a methodological hoop to jump through, but rather a critical step toward clarifying the role of PRP in ACLR and other orthopaedic pathologies.
Footnotes
Final revision submitted December 6, 2025; accepted December 13, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: C.E. has received support for education from Smith & Nephew and hospitality payments from Arthrex; has ownership interest in JEASO; and has received research funding support from AANA, OREF, and the Weill Cornell Medicine CTSC Pilot Award (UL1TR002384), none of which are directly relevant to this work. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
