Abstract
Background:
Posttraumatic osteoarthritis occurs at an alarming rate after anterior cruciate ligament (ACL) injury and reconstruction (ACLR).
Purpose/Hypothesis:
The purpose of this pilot randomized controlled trial was to evaluate the preoperative effect of a 2-injection series of platelet-rich plasma (PRP) on the progression of inflammatory and chondrodegenerative biomarkers before ACLR. It was hypothesized that preoperative PRP injections would decrease the chondrotoxicity of the presurgical knee fluid.
Study Design:
Randomized controlled trial; Level of evidence, 1.
Methods:
A total of 24 participants were randomized to either a control or an intervention group. Both groups received joint aspirations within the first 10 days after ACL injury (visit 1). In addition to joint aspiration at visit 1, the PRP group received a leukocyte-poor, nonactivated PRP injection at visit 1 and an additional PRP injection 5 to 12 days after the initial visit (visit 2). Joint fluid samples were harvested in both groups at the time of ACLR (visit 3). The joint fluid and PRP samples were subject to inflammatory and chondrodegenerative biomarker analysis for the following: interleukin (IL)-1ß, IL-10, IL-6, granulocyte–macrophage colony–stimulating factor, IL-5, interferon (IFN)-γ, tumor necrosis factor–α, IL-2, IL-4, and IL-8.
Results:
The final analysis was completed on 8 participants in the control group and 8 in the intervention group. At visit 1, no significant differences were observed in cytokine concentrations between the control and intervention groups. Between visits 1 and 3, the control group demonstrated significant decreases in IL-8 (P = .007) and IFN-γ (P = .007), whereas the intervention group demonstrated significant decreases in all cytokine concentrations (P < .05) except for IL-8 (P = .08). At visit 3, similar to visit 1, no significant group differences were observed in cytokine concentrations. The PRP products in the intervention group were analyzed using complete blood counts and were consistent with defined concentrations found in leukocyte-poor PRP product.
Conclusion:
Intervention with aspiration combined with a 2-injection series of leukocyte-poor PRP in the acute ACL-injured knee resulted in a significant reduction in effusion inflammatory markers, whereas the control aspiration (without PRP injections) did not show such marker reduction.
Registration:
NCT04088227 (ClinicalTrials.gov identifier).
Keywords
In the United States, 80,000 anterior cruciate ligament (ACL) ruptures are estimated to occur annually, with an increased likelihood in individuals participating in sports and aged 15 to 25 years. 14 A potential long-term consequence of ACL injuries is the development of posttraumatic osteoarthritis (PTOA) in the years following injury, and roughly half of ACL-reconstructed patients show signs of PTOA within the first 5 to 15 years after surgery.22,38 There are no curative treatments for osteoarthritis, increasing the importance of minimizing the occurrence of PTOA following ACL injuries. Orthopaedic research has focused on methods to prevent PTOA after ACL injury, with studies indicating that biochemical changes in the knee joint cartilage, such as chondrocyte death, following injury can contribute to the development of PTOA.5,7,22,23,29,32
It has been theorized that chemical factors, other than or in addition to mechanical factors, may be responsible for early development of cartilage destruction.2,25 A chronic inflammatory reaction is likely to persist after resolution of the acute effusion and could be the cause of the cartilage degeneration seen in patients after ACL reconstruction.24,29 It is conceivable that inflammatory cytokines can promote cartilage catabolism by stimulating the synthesis of free radicals and matrix metalloproteases (MMPs), whose activities could result in the development of osteoarthritis. 16
A nonsurgical treatment option for the management of osteoarthritis includes injectables such as corticosteroids and platelet-rich plasma (PRP). These injectables work by positively affecting cartilage cells, also known as chondrocytes, and the cells of the joint lining tissue, also known as synoviocytes. PRP is an autologous-derived blood product—that is, a joint injectable made from the patient’s own blood at the time and location of injection with simple blood centrifugation. Studies in the bench-top laboratory setting have provided in vitro evidence that PRP decreases synoviocyte production of MMPs, an inflammatory protein with negative effects on cartilage, decreases the effects of inflammatory proteins such as interleukin 1 beta (IL-1ß) on chondrocytes and has an anti-inflammatory effect upon the gene expression of chondrocytes and synoviocytes.30,43,45 While these are mechanisms that can help manage osteoarthritis after it arises, they also are mechanisms that could help prevent osteoarthritis before it forms. Recent comparative studies of PRP to other injectables in the management of osteoarthritis suggest a greater, long-lasting effect of PRP than other injectables, including hyaluronic acid, corticosteroids, and saline.1,9,12,18,21,31,40,46 While different PRP formulations (including leukocyte rich and leukocyte poor) and preparations exist, there is no current consensus on optimal formulations nor preparations for a given indication. 9 The use of corticosteroid injections has recently been questioned since clinical studies involving rotator cuff repair have suggested a negative impact on tendon health. 33 Theoretical benefits of opting for PRP include the potential deleterious effects of steroid injections or increased infection risk at the time of surgical intervention; barriers for the use of PRP include the additional financial burden to medical cost and time/personnel burden to clinical practice.
A recent study involving the postinjury ACL joint milieu suggested that an early intervention of joint aspiration and corticosteroid injection has positive effects, including a decrease in biomarkers responsible for chondral degeneration. 20 This, combined with historical in vitro studies on the effects of PRP on chondrocyte and synoviocyte activity suggest that PRP has the potential to affect the ACL-injured joint.
The primary objective of this pilot randomized controlled trial (RCT) was to determine if an early intervention of joint aspiration and PRP injection would be safe and would positively affect the biomarkers representative of chondral degeneration in patients with ACL injuries. We hypothesized that the PRP interventions would be safe and reduce the concentration of inflammatory and chondrodegenerative biomarkers following ACL injury, thus decreasing the chondrotoxicity of the presurgery knee effusion fluid. We also aimed to quantify the cellular content of each PRP sample.
Methods
Participants
This was a prospective, pilot RCT with 2 groups conducted in accordance with CONSORT (Consolidated Standards of Reporting Trials) 2010 guidelines. Institutional review board approval was obtained for the study protocol, and the study was posted on clinicaltrials.gov (NCT04088227). Enrollment started in September 2019 and was completed in April 2022 and was executed in the outpatient setting at the primary institution. Follow-up was completed in April 2022, and final data analysis was completed by August 2022. No changes were made to methods, protocols, or outcome measures after trial commencement.
Patients with a recent ACL tear between the ages of 14 and 40 years who had closed growth plates (as visualized on plain radiograph), no history of previous traumatic ipsilateral knee injury, and no clinical evidence of posterior cruciate ligament injury (no more than grade 1 medial or lateral collateral ligament injury) were screened for participation in this study by members of the research team (A.W.A., E.A.B., S.E.J., R.V.O.). Exclusion criteria included open growth plates (as visualized on plain radiography), patients without a palpable knee effusion, an injury occurring >10 days before enrollment, previous ipsilateral knee surgery, intra-articular cortisone or PRP injection into either knee within 3 months of injury, participation in another clinical drug trial within the 4 weeks before injury, and a history of any inflammatory disease. Patients who had difficulty obtaining internet access, did not have an active email address, or were unable to comprehend study documents or give informed consent were also excluded. If eligible and interested in participating, the potential participants went through the informed consent process (enrollment) with a member of the research team. No specific advertising nor recruitment materials were utilized. No compensation was given to participants in this study. A power analysis was impossible to perform before the study due to the lack of literature evaluating the effect of PRP on inflammatory and chondrodegenerative biomarkers as they potentially relate to PTOA in patients undergoing ACL reconstruction.
The investigation consisted of 2 study arms: (1) a control group (aspiration only) and (2) an intervention group (aspiration followed by PRP injections). A final sample size of 24 participants (12 in each arm, accounting for 2 withdraws per arm) was enrolled in this study, which is consistent with early-phase pilot studies of this nature. Upon enrollment, patients were randomized at a ratio of 1 to 1 using a simple computer-generated randomization sequence and sequentially numbered containers. The randomization method was generated and accessible by the research director (H.A.P.) but not by members of the study team or the principal investigator. The participants were randomized into the 2 study groups and were subsequently scheduled by the research team to receive the assigned treatment. In the control group, 1 patient withdrew after the initial visit, and 1 patient had insufficient aspiration at time of surgery for analysis. In 1 patient, the surgeon forgot to aspirate at the time of surgery, and 1 patient’s surgery had to be postponed >5 weeks. This left 8 patients for analysis in the control group. In the intervention group, 4 patients had insufficient aspirate at time of surgery for analysis, leaving 8 patients for analysis in the intervention group. A flow diagram of the patient inclusion process is shown in Figure 1.

CONSORT (Consolidated Standards of Reporting Trials) flow diagram of patient inclusion in the study.
Aspiration and Injection Regimen
The Arthrex Autologous Conditioned Plasma (ACP) System (Arthrex) was used to prepare all PRP injections. All injections were prepared and delivered using the described regimens immediately at the point of care in the clinic (within approximately 20 minutes of the initial blood draw).
Both groups underwent a joint aspiration performed at their initial visit, within the first 10 days after injury (visit 1), and again at the time of ACL reconstruction (visit 3), within 5 weeks of injury. The control group did not receive an injection at the time of aspiration. The intervention group had an initial joint aspiration, followed by an intra-articular injection of PRP at the initial visit (visit 1). A second intra-articular injection of PRP was performed at a second visit 5 to 12 days after the initial visit (visit 2), followed by aspiration at the time of ACL reconstruction (visit 3). Joint aspirations and injections were performed through the superolateral, suprapatellar approach. Patients were instructed not to take any prescription or over-the-counter nonsteroidal anti-inflammatory medications. All patients were encouraged to rest, ice, and elevate their injured knee, and all were monitored for adverse events and complications throughout the duration of the study.
PRP Preparation
The ACP system has a reported platelet recovery rate of 200% compared with whole blood. All preparations and injections were completed by the primary investigator (A.W.A.). Preparation involved harvesting 15 mL of whole blood from the patient’s forearm following standard venipuncture technique. The blood was immediately centrifuged at 1500 rpm for 5 minutes at room temperature within an Arthrex centrifuge. The top layer of the soft stack was carefully aspirated into the inner injection syringe as standard in the ACP preparation technique, stopping within 0.5 mL of the middle layer (buffy coat) before reaching the bottom layer (erythrocytes), which produced 4 to 5 mL of a leukocyte-poor PRP. The bottom layer (containing mostly red blood cells) was disposed of according to standard operating procedures. The inner syringe was then unscrewed and removed, making the ACP ready for use at the point of care. We set aside 100 μL in a microcentrifuge tube during injections for quantification of the PRP with an automated hematocytometer (XN-350; Sysmex). The remaining liquid PRP (approximately 4-5 mL) was used for the injection regimen. Due to the immediate use of the PRP at the point of care, anticoagulant citrate dextrose solution A was not used during preparation. Samples of joint fluid (from aspiration) and the PRP product were transferred to our institution for immediate analysis and subsequent storage. Figure 2 presents the overview of PRP preparation.

Overview of platelet-rich plasma (PRP) preparation.
PRP Quantification
A freshly collected sample of PRP product was kept at room temperature while a complete blood count (CBC) was performed using an automated hematocytometer. An amount of 50 μL of each tested PRP sample was loaded into the hematocytometer and internally mixed with Cellpack DCL Diluent, Lysercell WDF Reagent, Sulfolyser Hematology Reagent, and Fluorocell WDF Fluorescent Reagent (all Sysmex). This allowed for quantification of platelets, red blood cells, white blood cells, lymphocytes, monocytes, and neutrophils.
Inflammatory and Chondrodegenerative Biomarker Analysis
All joint fluid and PRP samples from both the control and the intervention groups were quickly thawed from a −80°C freezer using a hot water bath and subsequently centrifuged at 4000 rpm and 4°C for 10 minutes. Each sample was then analyzed for inflammatory biomarker (cytokine) content using the Luminex MAGPIX xMAP instrument (Luminex). Human Cytokine Magnetic 10-Plex Panels (Invitrogen) were used for analyzing each sample on 96-well plates. Each panel contained antibody beads for 10 cytokines for analysis, including IL-1β, IL-10, IL-6, granulocyte-macrophage colony-stimulating factor, IL-5, interferon (IFN)–γ, tumor necrosis factor (TNF)–α, IL-2, IL-4, and IL-8. Before analysis, the Luminex MAGPIX xMAP instrument was calibrated and verified using the Luminex MAGPIX Calibration/Verification Kit (Luminex) and the probe height was adjusted for each new kit.
Each 96-well plate was prepared for sample loading by adding 25 μL of 1× antibody beads to each well, washing twice with 200 μL of 1× wash solution, and then adding 50 μL of incubation buffer to each well. Using the provided standard stock of cytokines, 12 standard dilutions were prepared in a serial fashion, and 100 μL of each diluted standard was loaded into the wells in duplicate. Duplicate 100-μL blanks (wash solution) and 100-μL controls (human serum) were plated. The remaining wells were utilized for sample analysis by adding 50 μL of assay diluent and 50 μL of each sample (off the top) into the wells. All samples were also plated in duplicate onto the 96-well plate and allowed to agitate on an orbital plate shaker overnight at 2° to 8°C.
After incubation, the wells were decanted and washed twice with 200 μL of 1× wash solution before adding 100 μL of 1× biotinylated detector antibody to each well. The plate was then allowed to agitate on an orbital plate shaker for 1 hour at room temperature. After incubation, the wells were decanted and washed twice with 200 μL of 1× wash solution before adding 100 μL of 1× streptavidin-RPE to each well. The plate was then allowed to agitate on an orbital plate shaker for 30 minutes at room temperature. After the final incubation, the wells were decanted and washed 3 times with 200 μL of 1× wash solution.
After the final wash, 150 μL of 1× wash solution was added to each well and allowed to agitate on an orbital plate shaker for 2 to 3 minutes before analysis. The plate was then inserted into the Luminex MAGPIX xMAP for analysis. The standard concentrations were quantified to generate a standard curve for each cytokine, which was then used to quantify the concentration of each cytokine within the unknown samples. All data were exported from the instrument to an Excel sheet (Microsoft) for external statistical analysis.
Statistical Analysis
All participant data were deidentified before being analyzed by a statistician, allowing them to remain blinded. Descriptive statistics (means, standard deviations, and ranges) were reported for demographic data, PRP product data, and inflammatory and chondrodegenerative biomarker data. Statistical significance was considered at P < .05. The Wilcoxon signed-rank test was conducted to compare paired data, and the Wilcoxon rank-sum test was performed to compare independent samples.
Results
The control group participants had a mean age of 19.4 ± 8.0 years, and a male-to-female ratio of 6 to 2. In the intervention group, the mean age was 19.9 ± 6.8 years and the male-to-female ratio was also 6 to 2. The patient characteristics according to study group are summarized in Table 1. None of the participants showed any indication of infection nor complication during the duration of the study. No adverse events or serious adverse events were found. No patients underwent additional procedures after ACL reconstruction.
Demographics of the Trial Participants a
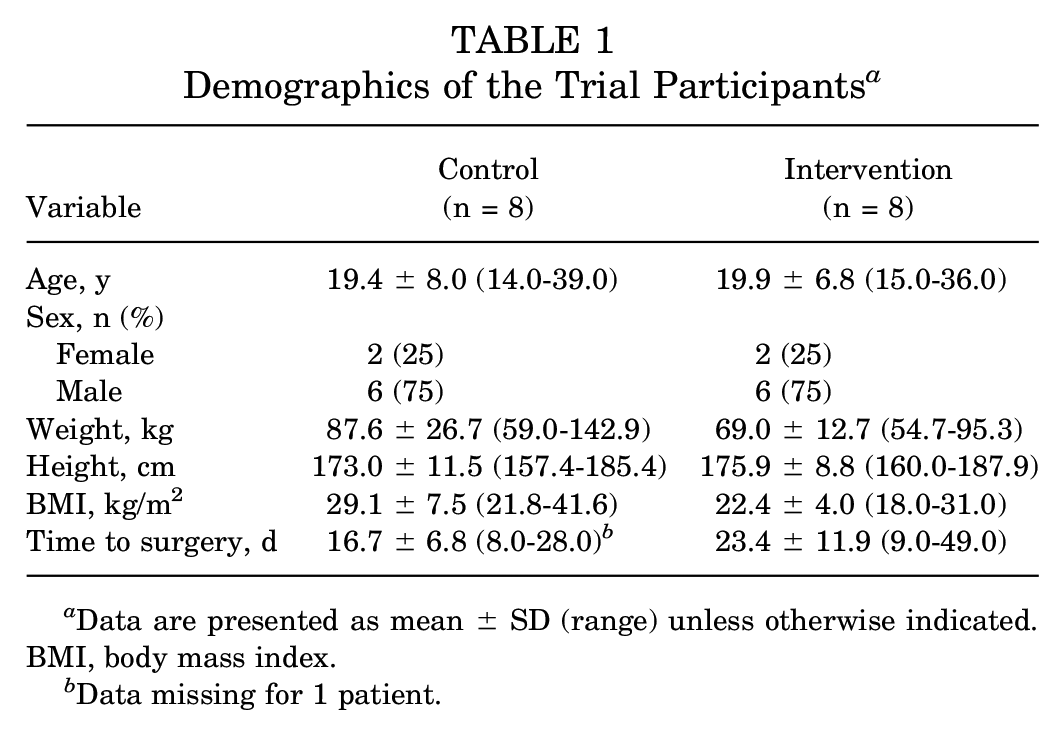
Data are presented as mean ± SD (range) unless otherwise indicated. BMI, body mass index.
Data missing for 1 patient.
All tested inflammatory and chondrodegenerative biomarker concentrations for the control group and intervention group at visit 1 and visit 3 are presented Table 2 and Figure 3. The control group demonstrated a decrease in all cytokine concentrations, which was not statistically significant from visit 1 to visit 3 except for IL-8 and IFN-γ; that is, 2 of the 10 markers studied showed a statistically significant decrease. The intervention group demonstrated a statistically significant decrease in all cytokine concentrations from visit 1 to visit 3, except for IL-8 (P = .08); that is, 9 of the 10 markers studied showed a statistically significant decrease. When comparing the control group with the intervention group at visit 1, no significant difference was seen between cytokine concentrations. Likewise, comparing the control group with the intervention group at visit 3 (time of ACL reconstruction), no significant difference was seen between cytokine concentrations.
Inflammatory and Chondrodegenerative Biomarker Concentrations a
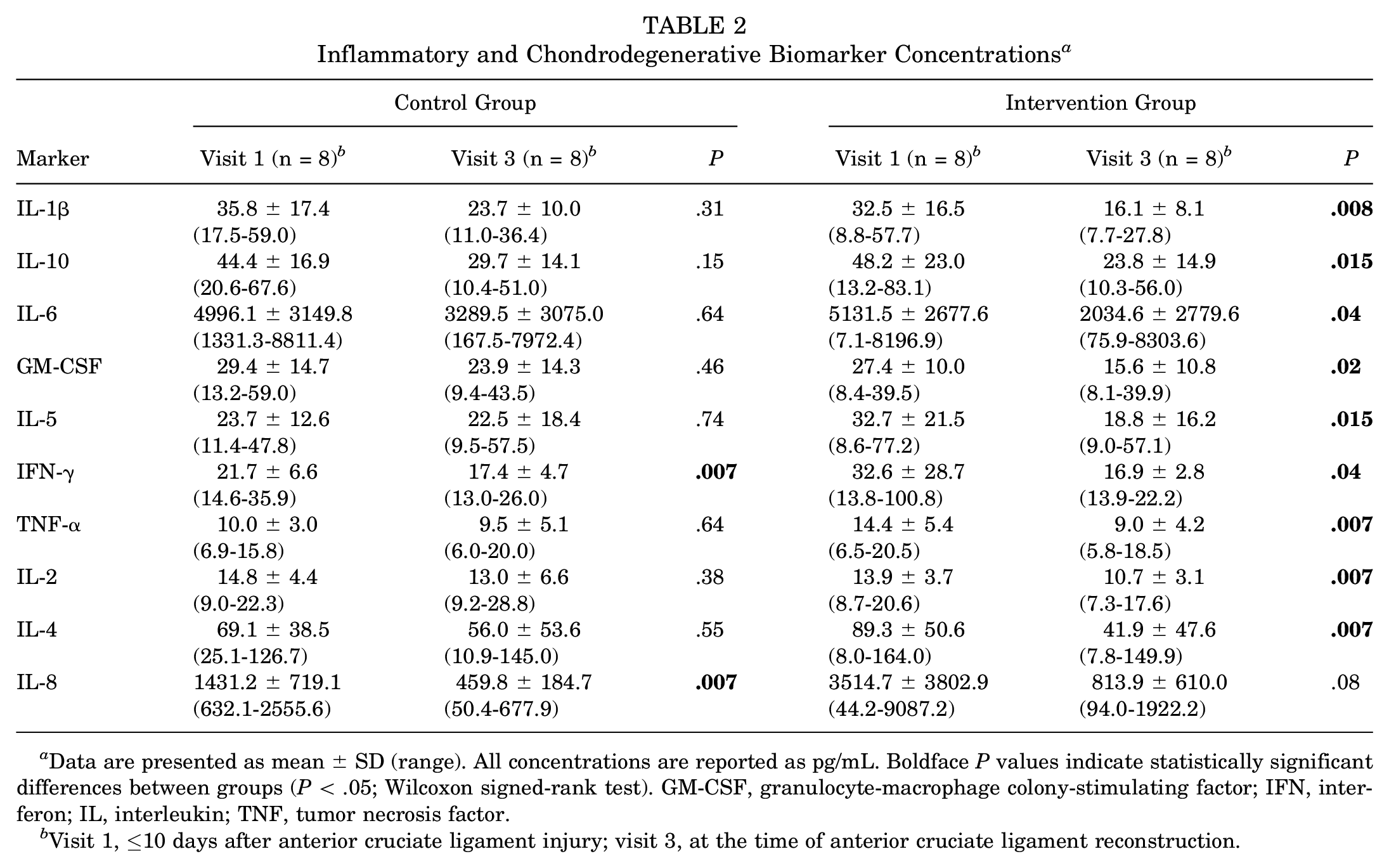
Data are presented as mean ± SD (range). All concentrations are reported as pg/mL. Boldface P values indicate statistically significant differences between groups (P < .05; Wilcoxon signed-rank test). GM-CSF, granulocyte-macrophage colony-stimulating factor; IFN, interferon; IL, interleukin; TNF, tumor necrosis factor.
Visit 1, ≤10 days after anterior cruciate ligament injury; visit 3, at the time of anterior cruciate ligament reconstruction.
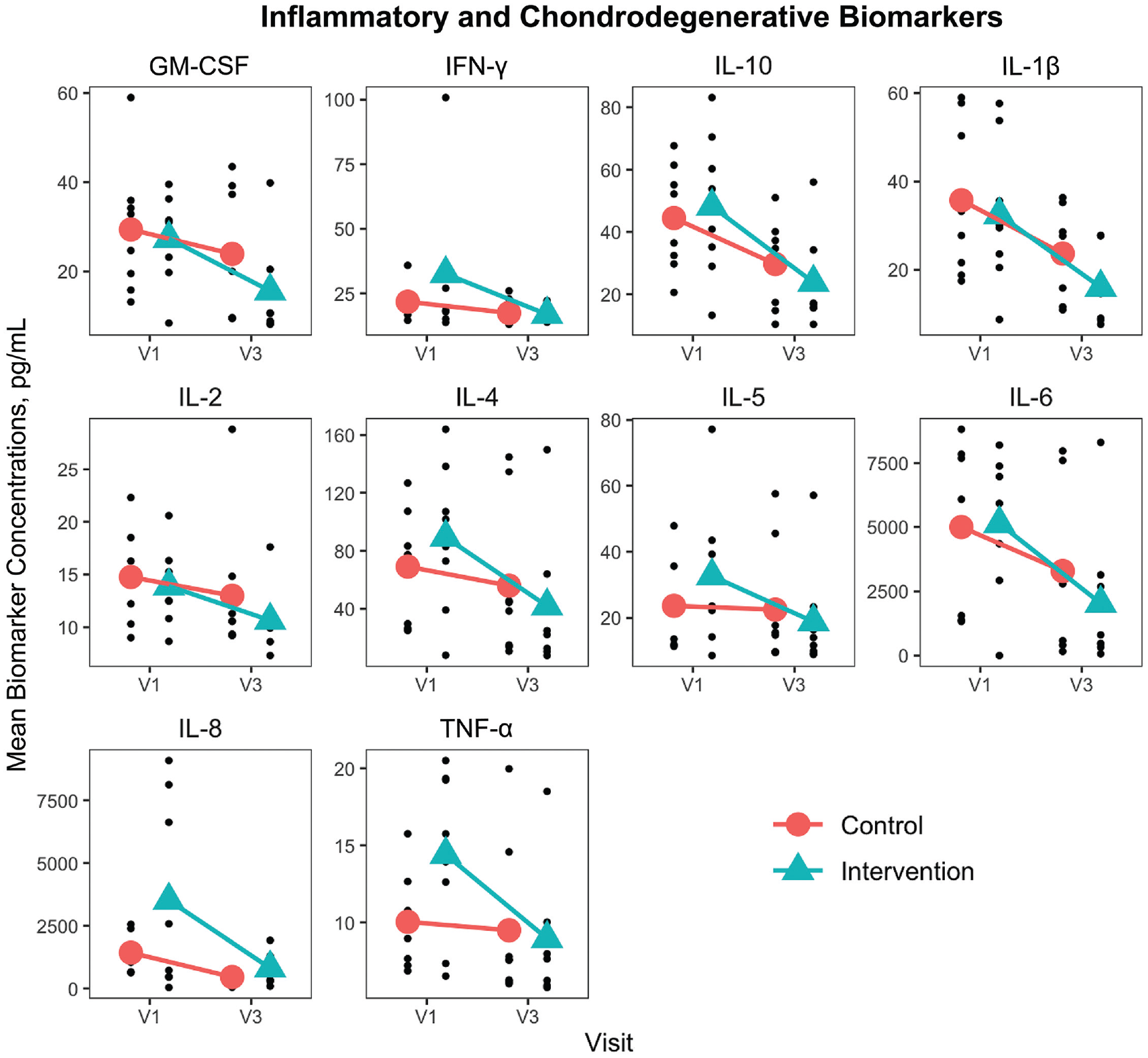
Inflammatory and chondrodegenerative biomarker concentrations. GM-CSF, granulocyte-macrophage colony-stimulating factor; IFN, interferon; IL, interleukin; TNF, tumor necrosis factor; V1, visit 1 (≤10 days after anterior cruciate ligament injury); V3, visit 3 (at anterior cruciate ligament reconstruction).
Samples of the intervention group PRP product were analyzed using CBC and were consistent with defined concentrations found in leukocyte-poor PRP product (Table 3).
Platelet-Rich Plasma Product Analysis a

CBC, complete blood count; PRP, platelet-rich plasma.
Discussion
The most important finding of this study was that preoperative injection of PRP before ACL reconstruction was safe (no adverse events) and the intervention group demonstrated a decrease in all tested marker concentrations from visit 1 to visit 3, which was statistically significant for all cytokines except for IL-8 (ie, 9 of the 10 markers studied). While the control group also demonstrated a reduction in mean concentration for most markers, this was not statistically significant for any marker except for IL-8 and IFN-γ (ie, 2 of the 10 markers studied). The magnitude of decrease was larger in the intervention group than the control group, but with comparison of the means between groups there was not a statistically significant difference. Another interesting finding was that 4 patients in the intervention group did not have sufficient effusions at the time of surgery for sample collection compared to only 1 patient in the control group not having sufficient effusions at the time of surgery. This implies an anti-inflammatory effect of the intervention upon the ACL-injury effusion.
A similar study for comparison, Latterman et al 20 evaluated intra-articular, preoperative injections of saline and corticosteroids before ACL surgery in 49 patients to evaluate the natural progression of biomarkers after ACL injury and the possibility of steroid injection to alter this progression. Four groups of 12 patients were evaluated with 4 unique 2-injection regimens including a saline/saline group and a corticosteroid/corticosteroid group. They termed the saline group placebo, although it could be argued that saline dilution of a caustic synovial environment could have dilutional benefit. Naturally, chondrodegenerative markers worsened while pain scores improved for the first 5 weeks. The groups with a steroid injection at the earliest time points noted smaller increases of c-telopeptide fragments of type 2 collagen (CTX-II), a marker of cartilage breakdown, when compared with dramatic 250% increases in CTX-II demonstrated by the placebo group. 20 The authors concluded that PTOA began at the time of injury and that an early anti-inflammatory injection was safe and effective in lowering chondrodegenerative biomarkers.
Biomarkers within the synovial milieu have been studied in acute injuries, including ACL injury, and chronic osteoarthritis.3,41,47 Struglics et al 41 quantified the longitudinal concentrations of proinflammatory cytokines in the ACL injured knee, with baseline values taken between 0 and 6 weeks after injury. They demonstrated significantly increased local joint concentrations of TNF-α, IL-6, IL-8, and IFN-γ early after joint injury, with a 1050-fold elevation of median concentration for IL-6 compared with reference samples and other cytokines elevated between 6-fold (TNF-α) and 21-fold (IL-10). Furthermore, concentration half-life of cytokines in synovial fluid was found to vary widely from the shortest for IL-6 (0.9 years) and longest for IFN-γ (3.6 years). TNF-α elevation was found to persist up to 5 years after injury. The elevated cytokine levels observed in synovial fluid after injury were not reflected by an elevation in the serum cytokine concentrations, and there were no correlations between serum and synovial fluid cytokine levels. No correlation with age was seen for cytokines in synovial fluid. Their observations indicate an early postinjury joint cell activation and recruitment of neutrophils, macrophages, and monocytes, resulting in the release and activation of proteinases, degradation of matrix components, and inhibition of synthesis of matrix molecules.
Increased TNF-α levels in synovial fluid years after injury is concerning, as it indicates a continued increase in the local production of TNF-α in the joint similar to that found in osteoarthritic joints. 39 In patients with knee injury, expression levels of TNF-α in the synovial membrane correlated positively with chondral damage, 48 and TNF-α levels in synovial fluid from ACL-injured knees have been associated with chondral damage.25,44 Persisting local TNF-α levels in the injured joint, as observed in the present study, may provide continued stimulation of cartilage matrix degradation, and may eventually result in cartilage damage and the development of posttraumatic arthritis. These locally activated inflammatory pathways may provide a potential treatment target in future efforts to prevent postinjury osteoarthritis in humans.11,19 Not all cytokines are bad. For example, the 20-fold increase in synovial fluid of IL-10 after the acute phase of injury may signal the activation of tissue-protective processes to reverse the catabolic events induced by inflammatory cytokines and chemokines. Notably, IL-10 stimulates type 2 collagen and proteoglycan expression, inhibits expression of MMPs, IL-1ß, TNF-α, and IFN-γ, and protects against chondrocyte apoptosis. 35
Preclinical animal studies have evaluated the potential of intra-articular injections to alter PTOA after acute ACL trauma. In both rabbit and pig models, intra-articular corticosteroid injections have mitigated synovitis and chondrodegeneration associated with the development of PTOA after ACL injury.15,36 Sieker et al 36 evaluated triamcinolone in a porcine ACL transection model and found that early injection mitigated the injury-induced increase in synovial fluid collagen fragments. Heard et al, 15 in a rabbit model, induced trauma with cartilage drilling and found that a single postsurgical intra-articular administration of dexamethasone resulted in a reduction of the histological cartilage grade of PTOA within the cartilage and appears to be partially protected through a reduction in synovial inflammation as quantified by histological inflammation and expression of proinflammatory mediators. 15 However, human trials, such as the one performed by McAlindon et al 26 demonstrate the diminishment of intra-articular steroid injection benefits that occur within a 2-year period. Additionally, McAlindon et al observed the significant negative effects that have been associated with long-term injections, such as greater cartilage volume loss and overall joint degradation. 26
PRP has a growing evidence base for the treatment of osteoarthritis.4,8 -10,13,27,31,34,37 In vitro evidence of chondroprotection, chondrogenesis, and the ability to decrease chondrocyte apoptosis provides possible mechanisms of action.27,28 Most clinical trials on knee arthritis have used an injection series with multiple injections. 9 One basic science study 6 and 2 clinical studies13,42 have suggested superiority of a 3-injection series over a single injection. The manufacturer-specific instructions for the PRP used in this study did not indicate a need for thrombin activation during the preparation of the product before injection. There is no consensus on the necessity of activation PRP nor on which formulation is superior for a given indication. 45 In addition to a body of evidence around primary osteoarthritis, animal studies have looked at the use of PRP in posttraumatic models. Khatab et al 17 suggested that multiple intra-articular injections of PRP in a mouse model reduces synovial inflammation, prevents the early decrease in anti-inflammatory macrophages, and might have a protective effect on cartilage while at the same time reducing pain. The design of the current study considered the author’s (A.W.A.) clinical use of a 3-injection series for the treatment of osteoarthritis, the current body of evidence involving PRP, and the study by Latterman et al 20 involving the use of corticosteroids.
Sample Size Calculation for Future Studies
This was a pilot study focusing on multiple biomarkers as the primary outcomes. The data collected in this study are valuable for exploring and planning a pivotal study. Power analysis is useful for future sample size estimates. When we are interested in multiple primary outcomes, it is important to control the family-wise type 1 error. A common approach is to adjust the type 1 error of each statistical test using the Bonferroni correction. In this context, the power can be disjunctive, which is the probability of detecting ≥1 true difference (true effect if it exists) across all outcomes. Another option is to calculate the marginal power based on 1 selected outcome. We used the data from this pilot study to estimate the pooled standard deviation. Considering 80% power, with a type 1 error rate of α = .05 (.005 level of significance with Bonferroni correction), and 2 independent samples, Figure 4 displays a power analysis for all biomarkers, both with and without a Bonferroni correction. In a scenario where all biomarkers are considered as primary outcomes, Figure 4A can be used to determine the sample size needed to power future studies to detect the differences of interest. In a scenario where power analysis is used—for example, when considering only 1 biomarker (eg, IL-6) as the primary outcome of the study—we would need 92 patients in each group to detect a difference of 27 points between the 2 groups (see Figure 4B).

Sample size calculation for a power of 80% and type 1 error of .5% (A) n was inflated by 20% to account for loss to follow-up; power=0.8; α = 0.005 (Bonferroni correction) and (B) n was inflated by 20% to account for loss to follow-up; power = 0.8; α = 0.05.
Limitations
One limitation of this study was that the effect size for clinically significant change in inflammatory markers has not yet been defined. Biomarkers are subject to large variance, which limits their use as outcome measures. This study was underpowered. A larger sample size will be required in a follow-up, pivotal study to detect a true difference in means between the intervention and control group. Considering a natural decrease for these biomarkers after injury, it is possible that the control would decrease statistically if a larger sample size were tested. Preoperative intervention with PRP has the potential to consistently alter the concentration of chondrodestructive proteins. These data will be used to power a pivotal study with evaluation of radiographic and patient-reported outcomes for efficacy evaluation, with an attempt to reduced perioperative pain, improved rehabilitation efficacy, and the long-term goal of modulating the presence of posttraumatic arthritis. Patient withdrawal after allocation and initial visit was another limitation; in most cases, this was secondary to the patient-perceived disappointment in not receiving allocation to the intervention group. The withdrawal pattern likely contributed to the abnormal male-to-female ratio in the control group and difference in body mass index means, since one would expect these to be equal with appropriate randomization. These differences may have affected the findings of the study. An additional limitation was that there was no blinding of this study; however, since the outcome measures were biomarkers and not patient-reported outcome measures, this limitation should not affect the validity of results. Additional limitations include the lack of a control and the fact that no clinical effect variable such as patient-reported outcome measures or functional scoring was followed. It is important to consider that since there was an absence of intervention to the control group, the effects of PRP may be dilutional alone.
Conclusion
Intervention with aspiration combined with a 2-injection series of leukocyte-poor PRP in the acute ACL-injured knee resulted in a statistically significant reduction in effusion inflammatory markers, whereas the control aspiration (without PRP injections) did not show such an extent of marker reduction.
Footnotes
Acknowledgements
The authors acknowledge Jessi Truett, BCBA, Andrews Research & Education Foundation, who was responsible for manuscript proofreading, writing assistance, and technical editing.
Final revision submitted August 27, 2024; accepted September 4, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: Funding for this study was received from the State of Florida Department of Health. A.W.A. has received education payments from Arthrex, consulting fees from Arthrex and Bioventus, nonconsulting fees from Arthrex and Smith & Nephew, and royalties from Arthrex. E.A.B. has received grant support from Arthrex; education payments from Arthrex, Smith & Nephew, and MVP Orthopedics; and hospitality payments from Stryker. S.E.J. has received education payments from CGG Medical, nonconsulting fees from Arthrex, and royalties from Arthrex. R.V.O. has received hospitality payments from CGG Medical. B.J.K. has received consulting fees from Sonex Health. J.G.H. has received consulting fees and honoraria from Tenex Health. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Baptist Hospital–Pensacola (reference No. 1472599-1).
