Abstract
Background:
Rotator cuff tears occur with increased prevalence and worse outcomes in patients with diabetes mellitus. Glucagon-like peptide-1 receptor agonists (GLP-1RA) have demonstrated efficacy in improving glycemic control and reducing body weight, and early studies have examined their perioperative use in total shoulder arthroplasty.
Purpose:
To investigate the association between GLP-1RA use and postoperative outcomes following arthroscopic rotator cuff repair (RCR).
Study Design:
Cohort study; Level of evidence, 3.
Methods:
A retrospective cohort study utilizing the TriNetX database identified patients undergoing arthroscopic RCR from 2000 to 2024. Patients prescribed GLP-1RA within 6 months preoperatively were compared with a propensity-matched cohort without GLP-1RA exposure. Primary outcomes included 90-day postoperative medical complications and 2-year surgical outcomes. Outcomes between cohorts were compared using odds ratios (ORs) with 95% CIs.
Results:
After propensity score matching, 3066 patients were included in each cohort. The GLP-1RA cohort had a significantly lower risk of hospital readmission within 90 days (2.7% vs 3.6%; OR, 0.741; 95% CI, 0.555-0.989; P = .04) and reduced likelihood of subsequent RCR within 2 years (4.5% vs. 5.7%; OR, 0.768; 95% CI, 0.611-0.966; P = .02). No significant differences were observed in surgical-site infection, pneumonia, thromboembolic events, or emergency department visits within 90 days postoperatively. Likewise, no differences were found in other surgical outcomes, including shoulder arthroplasty, lysis of adhesions, or adhesive capsulitis at the 2-year follow-up.
Conclusion:
Preoperative GLP-1RA use for patients undergoing arthroscopic RCR was associated with a decreased risk of both hospital readmission at 90 days and subsequent RCR at 2 years postoperatively. There was no difference in the risk of shoulder arthroplasty, adhesive capsulitis, lysis of adhesions, or 90-day medical complications between groups. These findings suggest that GLP-1RA use appears safe in the perioperative period for patients undergoing arthroscopic RCR, providing reassurance for clinicians managing patients already prescribed these medications.
Keywords
Rotator cuff tears are among the most common injuries affecting the upper extremity, with an occurrence rate of approximately 20.7% in the general population and a significantly increased incidence among older individuals.26,28 Meanwhile, DM affects nearly 10% of the American population, a figure expected to rise to over 15% by 2030.18,20 It is well-established that DM and obesity are associated with an increased risk of rotator cuff tears, along with a greater likelihood of requiring surgical repair.9,13,23,24 Additionally, many studies have demonstrated that individuals with DM undergoing arthroscopic RCR may face a higher rate of postoperative complications, such as increased risk of retear, pain, infection, and extended hospital stays.1,3,4,17,21
Controlling blood sugar levels is critical for patients with DM, as studies have shown that maintaining stable levels significantly reduces the risk of postoperative complications during surgeries, including RCRs and major joint procedures such as hip, knee, and shoulder arthroplasties. 15 Several randomized controlled trials have demonstrated that patients using GLP-1RA, such as semaglutide, experience substantial reductions in both blood glucose levels and body weight.16,19,25 Due to their effectiveness in treating diabetes and obesity, GLP-1RAs have become increasingly popular, with semaglutide emerging as the leading drug on the market in 2023. 27
As the prevalence of diabetes continues to rise and the use of GLP-1RA increases, it is important to understand how GLP-1RAs affect surgical outcomes. Recent studies have examined the impact of GLP-1RA on perioperative complications following total shoulder arthroplasty (TSA)7,12; however, the impact of these drugs after arthroscopic RCR is not well understood. Therefore, this study aims to investigate the impact of GLP-1RA use on postoperative outcomes and complications following arthroscopic RCR through a matched cohort analysis.
Methods
Data Source
This retrospective cohort study utilized data from the TriNetX database, a global federated health research network, to identify patients aged ≥18 years who underwent arthroscopic RCR between 2000 and 2024. The TriNetX database comprises 108 participating health care organizations with >131 million patient electronic medical records with data regarding procedures, diagnoses, medications, and laboratory values. This study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology guidelines for consistent reporting of observational data. 6
Cohort Identification
The study population was established using International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM), and Current Procedural Terminology (CPT) coding to identify patients aged >18 years who underwent arthroscopic RCR. Two cohorts were established: patients who were taking GLP-1RA and patients who were not taking GLP-1RA. Patients were included in the GLP-1RA group if the database had recorded a prescription for a GLP-1RA within 6 months before their surgery. Patients were excluded from analysis if they underwent surgery prior to having a GLP-1RA listed in their medical record. Patients in the control group (no GLP-1RA) were identified as those who did not have a prescription in the database within 6 months prior to their surgery. Medications that qualified for inclusion in the GLP-1RA cohort were semaglutide, tirzepatide, exenatide, liraglutide, lixisenatide, dulaglutide, and albiglutide. The ICD-10-CM and CPT codes used for cohort identification, matching, and outcomes can be found in the Supplemental Material (Table S1; available separately). To minimize potential confounding, cohorts were matched by 1:1 propensity scoring based on age, sex, race, ethnicity, obesity, hemoglobin A1c (glycated hemoglobin), and medical comorbidities.
Outcomes and Statistical Analysis
Baseline patient demographics, GLP-1RA prescription status, comorbidity data, hemoglobin A1c, and nicotine dependence were collected. The primary outcomes of interest were the risk of 90-day postoperative medical complications and 2-year surgical outcomes between cohorts. Surgical outcomes included subsequent arthroscopic RCR, shoulder arthroplasty, lysis of adhesions, and adhesive capsulitis. Medical complications included surgical-site infection, pneumonia, renal failure, myocardial infarction (MI), stroke, deep vein thrombosis (DVT), pulmonary embolism (PE), hospital readmission, and emergency department (ED) visit.
Chi-square tests were used to determine differences in categorical comorbidity variables, and Student t tests were used to analyze differences in continuous comorbidity variables, as indicated. Outcomes between cohorts were compared using odds ratio (OR) with 95% CI, risk differences, and risk ratios. A P value of <.05 was used to determine statistical significance. All statistical analysis was performed using the TriNetX database analysis software.
Results
Cohort Demographics
There were 3079 patients taking GLP-1RA and 130,821 patients not taking GLP-1RA who underwent arthroscopic RCR meeting inclusion and exclusion criteria. After matching, 2 well-balanced cohorts of 3066 patients were obtained.
Before Propensity Score Matching
At time of index surgery, patients in the GLP-1RA cohort were more likely to be female (P < .001), Hispanic or Latino (P < .001), and Black or African American (P < .001). Patients in the GLP-1RA cohort were less likely to be White (P < .001) and Asian (P = .03). There were no differences between cohorts with respect to American Indian (P = .26) or Hawaiian or Pacific Islander race (P = .84). The GLP-1RA cohort had a higher prevalence of DM (66.2% vs 7.3%; P < .001), nicotine dependence (9.0% vs 4.4%; P < .001) and were more likely to be overweight or obese (38.3% vs 6.7%; P < .001) (Table 1). Regarding medical comorbidities, the GLP-1RA cohort had a higher prevalence of hyperlipidemia (P < .001), chronic kidney disease (P < .001), liver disease (P < .001), thyroid disorders (P < .001), and hypertension (P < .001).
Comparison of Cohort Demographics and Comorbidities Before and After 1:1 Propensity Score Matching a
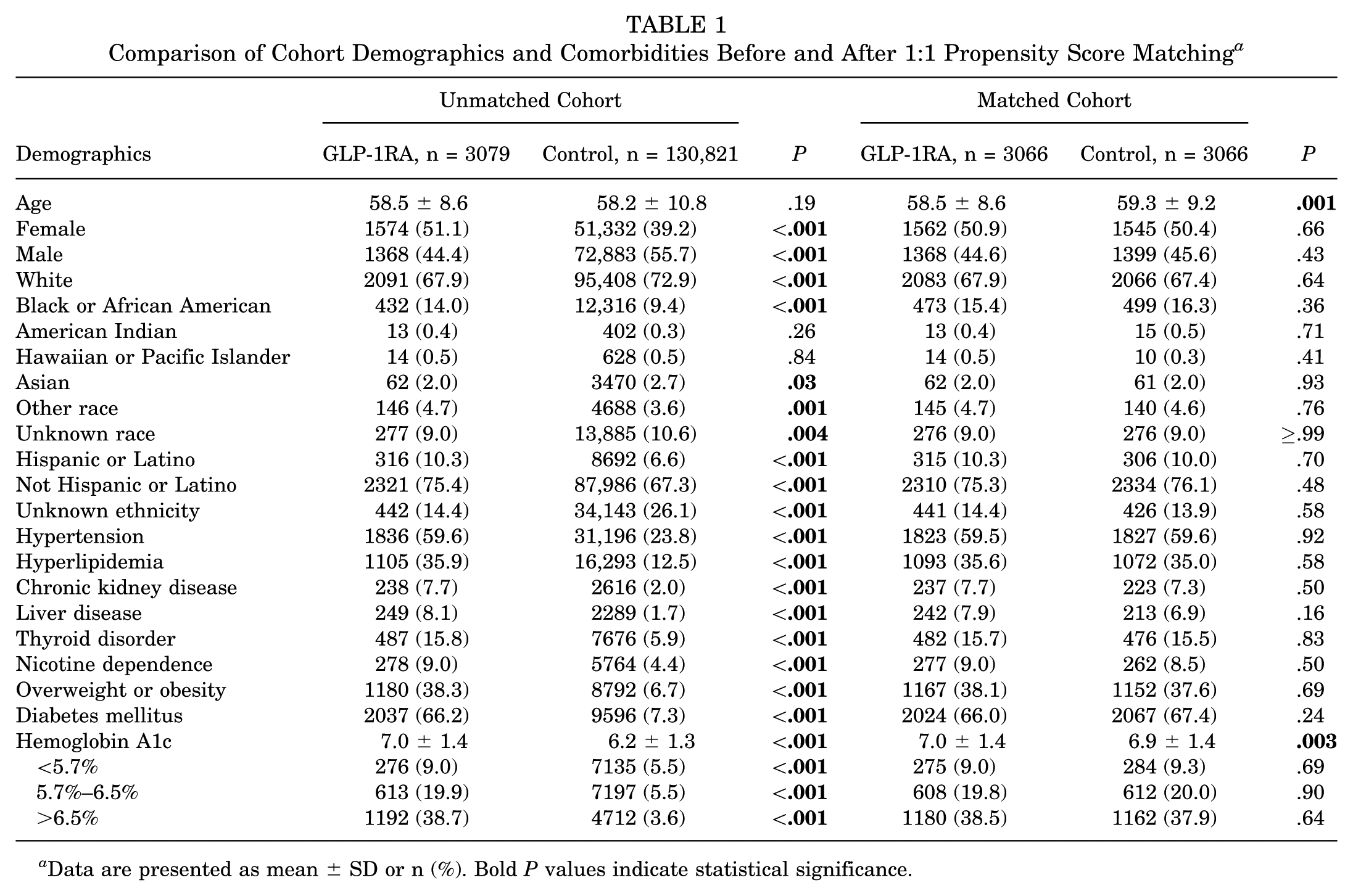
Data are presented as mean ± SD or n (%). Bold P values indicate statistical significance.
After Propensity Score Matching
Propensity score density plots indicate that 2 well-balanced cohorts were obtained (Figure 1).

Propensity score density plots representing differences in cohorts before (left) and after (right) matching. The purple line represents the GLP-1RA group while the green line represents the control group. Propensity score density plots demonstrate that 2 well-balanced cohorts were obtained.
After matching, each cohort consisted of 3066 patients, and there were no significant differences in demographics (Table 1). In the GLP-1RA cohort, the index surgery occurred at a mean age of 58.5 ± 8.6 years compared with 59.3 ± 9.2 in the control group. The GLP-1RA cohort was composed of 50.9% female patients and was predominantly White (67.9%), followed by Black or African American (15.4%). The control group was composed of 50.4% female patients and was predominantly White (67.4%), followed by Black or African American (16.3%) (Table 1).
Regarding medical comorbidities, mean hemoglobin A1c was the only variable that retained a statistically significant difference after matching (7.0% vs 6.9%; P = .003). Hemoglobin A1c was further matched by categorical range, which revealed no difference between cohorts (Table 1). The prevalence of DM in the GLP-1RA cohort was 66.0% compared with 67.4% in the control group (P = .24). Nicotine dependence was present in 9.0% of patients in the GLP-1RA cohort and 8.5% of patients in the control group (P = .50). The number of patients who were overweight or obese was 38.1% in the GLP-1RA cohort and 37.6% in the control group (P = .69). There were no significant differences in any other medical comorbidities between cohorts, including hyperlipidemia, chronic kidney disease, liver disease, thyroid disorders, or hypertension (Table 1).
Complications and Surgical Outcomes
Results of our primary analysis revealed that patients in the GLP-1RA cohort had a statistically significantly reduced risk of hospital readmission within 90 days postoperatively compared with the control group (2.7% vs 3.6%; OR, 0.741; 95% CI, 0.555-0.989; P = .04) (Table 2). There were no significant differences found in the risk of remaining medical complications at 90 days including surgical-site infection, pneumonia, renal failure, MI, stroke, DVT, PE, or ED visits between groups. The GLP-1RA cohort was less likely to undergo a subsequent arthroscopic RCR ≤2 years postoperatively compared with patients in the control group (4.5% vs 5.7%; OR, 0.768; 95% CI, 0.611-0.966; P = .02) (Table 3). No differences were found with the remaining surgical outcomes including shoulder arthroplasty, lysis of adhesions, or adhesive capsulitis.
90-Day Medical Complications After Rotator Cuff Repair With GLP-1RA Usage a

Bold P value indicates statistical significance. GLP-1RA, glucagon-like peptide-1 receptor agonist; OR, odds ratio.
2-Year Surgical Outcomes After Rotator Cuff Repair With GLP-1RA Usage a

GLP-1RA, glucagon-like peptide-1 receptor agonist; OR, odds ratio. Bold P value indicates statistical significance.
Discussion
This study revealed that patients receiving GLP-1RA were at a lower risk of undergoing subsequent RCR procedures at 2 years postoperatively when compared with the control group, as well as less likely to be readmitted to the hospital within 90 days postoperatively. There were no differences found in the risk of undergoing shoulder arthroplasty, lysis of adhesions, and adhesive capsulitis at 2 years, nor for risk of surgical-site infection, pneumonia, renal failure, MI, stroke, DVT, PE, or ED visits.
As the use of GLP-1RA continues to rise, it is important to understand the impact these medications have on postoperative outcomes. One recent study by Seddio et al 22 investigated the association between semaglutide use and complications following arthroscopic RCR in patients with type 2 DM. 22 The authors identified a greater retear rate at 2 years in the control group compared with patients using semaglutide (18.3% vs 12.5%, respectively). Furthermore, patients who were using semaglutide preoperatively had lower rates of minor and serious 90-day adverse events. Our study has similar findings and results, based on included patients using any of the commercially available GLP-1RAs at the time of surgery and regardless of DM diagnosis, further increasing the generalizability of these observances. 14
The influence of GLP-1RA on surgical outcomes and complications has also been previously explored in TSA patients with conflicting results.5,7,12 Lawand et al 12 demonstrated that patients receiving GLP-1RA exhibited higher rates of complication including DVT, MI, pneumonia, blood transfusion, and hospital readmission compared with the control group through a propensity-matched cohort. Additionally, there were no differences found with revision rate at 2 years between groups. Conversely, another study by Choudhury et al 5 identified a reduction in 90-day mortality in patients taking GLP-1RA prior to undergoing TSA, corroborating the protective effect that has been previously demonstrated in lower extremity arthroplasty literature.2,8,10,11 Even so, Elsabbagh et al 7 found no difference in 90-day postoperative complication rates in diabetic patients taking GLP-1RA after propensity score matching using the Mariner data set within the PearlDiver database, for those undergoing TSA. These mixed results in TSA literature highlight the need for continued investigation on the potential preoperative risks and benefits of GLP-1RA use in upper extremity orthopaedic procedures. Our study furthers this growing body of literature by demonstrating no significant increase in 90-day medical complications following arthroscopic RCR with a reduced risk of hospital readmission. Given the rising prevalence of GLP-1RA use in orthopaedic patients, future studies with large cohorts are warranted to better define its impact on postoperative outcomes.
Limitations
To our knowledge, this is the first large-scale, population-based investigation to assess the relationship between all commercially available GLP-1RA and their impact on postoperative outcomes and complications following arthroscopic RCR. This study is not without limitations, including those inherent to database studies; the most apparent of these are the potential for coding inaccuracies as well as the retrospective nature of the study, which has the potential to introduce biases for which we were unable to account—such as the reliance on CPT and ICD codes to identify patients undergoing RCR and to identify complications. We performed 1:1 propensity score matching for several comorbidities, including diabetes and obesity, to minimize confounding between groups. After matching, hemoglobin A1c remained higher in the GLP-1RA group (7.0% vs 6.9%; P = .003), although this difference was small. Another limitation is that the database does not record medication adherence or the exact timing of medication cessation prior to surgery, which may introduce bias. There were other potentially confounding variables we were unable to account for in our analysis, such as length of surgery, intraoperative complications, and daily activities that may have influenced outcomes. Because this study utilized the TriNetX database, which draws from predominantly US-based health care organizations, the findings may reflect the demographic and comorbidity profile of this population and may not be fully generalizable to other health care systems or international cohorts. Additionally, subgroup analyses by individual GLP-1RA agents or by specific patient characteristics (body mass index, glycemic control) could provide additional insight, but such analyses were limited by available sample sizes. Last, although GLP-1RAs are generally prescribed for patients with type 2 diabetes, the database does not reliably differentiate between insulin-dependent and non–insulin dependent diabetes, which may represent a residual confounder despite matching for diabetes diagnosis and hemoglobin A1c.
Conclusion
Preoperative GLP-1RA use for patients undergoing arthroscopic RCR was associated with a decreased risk of both hospital readmission at 90 days and subsequent RCR at 2 years postoperatively. There was no difference in the risk of shoulder arthroplasty, adhesive capsulitis, lysis of adhesions, or 90-day medical complications between groups. Importantly, these findings suggest that GLP-1RA use appears safe in the perioperative period for patients undergoing arthroscopic RCR, providing reassurance for clinicians managing patients already prescribed these medications.
Supplemental Material
sj-pdf-1-ojs-10.1177_23259671251412408 – Supplemental material for Effect of Glucagon-like Peptide-1 Receptor Agonists on Outcomes and Complications Following Arthroscopic Rotator Cuff Repair: A Matched-Cohort Analysis
Supplemental material, sj-pdf-1-ojs-10.1177_23259671251412408 for Effect of Glucagon-like Peptide-1 Receptor Agonists on Outcomes and Complications Following Arthroscopic Rotator Cuff Repair: A Matched-Cohort Analysis by Bradley J. Lauck, Charles B. Colson, Nicholas C. Bank, Nicholas A. Trasolini, Brian R. Waterman, Jessica Churchill and Alan W. Reynolds in Orthopaedic Journal of Sports Medicine
Footnotes
Final revision submitted October 24, 2025; accepted December 9, 2025.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto. This study was conducted in accordance with accepted ethical standards and relevant guidelines for research involving human data.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
