Abstract
Background:
Aging is associated with degenerative changes in both bone and tendon tissues, leading to impaired healing at the bone-tendon interface (BTI) during rotator cuff repair. The role of fibulin-7 (FBLN7) in enhancing BTI healing in aged populations warrants an investigation because of its potential regenerative properties.
Purpose:
To determine whether FBLN7 can enhance tendon-bone healing and regeneration in a model of rotator cuff repair in aging mice.
Study Design:
Controlled laboratory study.
Methods:
A total of 5 young (8 weeks old) and 5 aged (18 months old) C57BL/6 mice were used for a baseline supraspinatus tendon–humerus evaluation. Another 20 aged mice underwent rotator cuff repair and were allocated to either the FBLN7 (intervention) group (n = 10), receiving a fibrin sealant with FBLN7, or a control group (n = 10), receiving a standard fibrin sealant. Mice were euthanized at 4 (n = 5 per group) and 8 (n = 5 per group) weeks after surgery. The supraspinatus tendon–humerus complexes were harvested for micro–computed tomography, histological analysis, and biomechanical testing.
Results:
The expression of FBLN7 was significantly lower in aged mice at the BTI compared with young mice (P < .05). Histological and biomechanical assessments revealed that the FBLN7 group exhibited improved subchondral bone morphology (P < .05) and denser cortical bone (P < .05), along with significantly higher failure loads and stiffness (P < .05), compared with the control group. Additionally, the FBLN7 group demonstrated superior fibrocartilage formation (P < .05) and the upregulated expression of osteogenic (Runx2; P < .01 at week 4 and P < .05 at week 8), chondrogenic (Sox9; P < .01 at week 4 and P < .05 at week 8), and tenogenic (SCX; P < .05 at week 4) factors.
Conclusion:
FBLN7 significantly enhanced healing and regeneration at the BTI in aged mice, supporting its potential as a therapeutic intervention to improve rotator cuff repair outcomes.
Clinical Relevance:
These findings highlight FBLN7’s potential role in clinical sports medicine, suggesting that it could be utilized to promote better recovery and healing in aged patients undergoing rotator cuff repair, addressing a critical challenge in postoperative rehabilitation.
The bone-tendon interface (BTI) is a crucial connection point between bone and tendon tissues, encompassing various regions such as tendon, uncalcified fibrocartilage, calcified fibrocartilage, and bone.2,14 It plays a pivotal role in the transmission of mechanical forces, ensuring the proper functioning of musculoskeletal systems. 30 BTI injuries are common musculoskeletal problems that can significantly impair musculoskeletal function, resulting in pain, limited mobility, and diminished quality of life.7,11,21 In aging patients, these injuries can escalate, frequently resulting in rotator cuff tears due to degenerative changes in both bone and tendon tissues, which exhibit a noticeable reduction in their capacity for spontaneous healing. What is more, these patients experience prolonged recovery periods and an increased susceptibility to reinjuries. 32 Consequently, there is a need to address age-related BTI injuries and develop effective therapeutic strategies. Although surgical techniques are commonly employed for rotator cuff repair,9,12 the limited regenerative capacity of the BTI contributes to complications and high retear rates.13,29,33 Fibrocartilage regeneration is considered essential for durable healing,24,36 and the extracellular matrix (ECM) provides the structural and signaling cues necessary for this process. 23
Fibulin-7 (FBLN7), a matricellular protein, interacts with various ECM components, cell surface receptors, and growth factors, thereby influencing diverse cellular processes.20,22 It has been reported that FBLN7 is expressed in multiple vital tissues such as eyes, placenta, cartilage, bone, and teeth. 3 Currently, there is a limited understanding of the precise functions of FBLN7. Research has revealed that deletions in crucial regions of the FBLN7 gene, located on human chromosome 2q13, are associated with a variety of diseases and developmental abnormalities, known as 2q13 deletion syndrome. 18 Moreover, a previous study indicated that FBLN7 was implicated in inflammation and angiogenesis. 3 More remarkably, we found that FBLN7 plays a critical role in the assembly and organization of the ECM during embryogenesis. 3 However, its effect on BTI healing was not definitively proven.
The aim of this study was to investigate the effects of FBLN7 on BTI healing in a murine rotator cuff repair model. The effects of FBLN7 were assessed by micro–computed tomography (micro-CT), histological analysis, and biomechanical testing, with the hypothesis that it enhances BTI regeneration.
Methods
Animals and Study Design
A total of 5 young (8 weeks old) and 25 aged (18 months old) C57BL/6 mice were housed in a specific pathogen-free environment with unrestricted access to food and water. Initially, 5 young and 5 aged mice were euthanized to harvest bilateral supraspinatus tendon–humerus (SSTH) complexes for a baseline experimental evaluation. The remaining 20 aged mice were specifically chosen to establish a rotator cuff repair model and were randomly divided into 2 groups: the control group and the FBLN7 group, with 10 mice in each group. Fibrinogen solution (Sigma-Aldrich) and thrombin solution (Sigma-Aldrich) were prepared as per the manufacturer's instructions. In the FBLN7 group, fibrinogen and thrombin were utilized as carriers for the controlled release of recombinant FBLN7 at the interface of the rotator cuff repair site, as confirmed by in vitro release kinetics reported previously, 4 while mice of the control group were treated with a fibrin sealant without FBLN7. Subsequently, the mice were euthanized at 4 weeks (n = 5 per group) to evaluate early healing events such as tissue fusion and bone morphology or at 8 weeks (n = 5 per group) to assess long-term remodeling and maturation, and the bilateral SSTH complexes were harvested for subsequent evaluations. Ethical approval for this study was obtained from the Institute of Biological and Medical Engineering, Guangdong Academy of Sciences (K2022-01-167). The animal experiments adhered to the ARRIVE guidelines.
Animal Model and FBLN7 Treatment
All mice were intraperitoneally injected with 0.3% pentobarbital sodium (0.6 mL/20 g; Merck), and surgical procedures were performed under aseptic conditions using a stereo microscope. Mice were positioned in the right lateral decubitus position, and a longitudinal skin incision was made at the left shoulder. Subsequently, the deltoid muscle was incised to expose the supraspinatus tendon (SST). The SST was then secured in a figure-of-8 fashion using 6-0 polydioxanone (PDS) suture at the insertion point on the humeral head. The cartilaginous layer at the insertion point was gently removed, exposing cancellous bone. A 1 mm–diameter bone tunnel was created within the humeral head, and PDS suture was passed through the bone tunnel. Next, 12 μL of a fibrin sealant (10 μL fibrinogen saline solution, 10-30 mg/mL; 2 μL thrombin saline solution, 50-500 IU/mL) was applied at the repair site. In the FBLN7 group, the sealant was supplemented with 0.5 μg of recombinant FBLN7 (Abmart), corresponding to a final concentration of 0.5 μg/12 μL; controls received a sealant without FBLN7. The tightened limbs were then sutured to secure the SST at its original insertion site. Finally, the deltoid muscle and skin were sutured.
Micro-CT Scanning
The formation of subchondral bone at the attachment site of the SST was evaluated using micro-CT. After the collection of SSTH complexes from each group, the samples were fixed in 4% paraformaldehyde at 4°C for 24 hours and then washed with 0.9% saline to remove residual paraformaldehyde. Subsequently, the samples were placed in a vertical position in a test tube and subjected to micro-CT under the following conditions: 10.4-μm voxel size, 55 kVp, 0.36° rotation step (180° angular range), and 400 milliseconds per exposure. The scanning data for each specimen were subjected to 3-dimensional Gaussian filtering and global thresholding to distinguish bone voxels from surrounding soft tissue and bone marrow. A region of interest measuring 200 μm × 400 μm was selected for the evaluation of subchondral bone at the BTI healing site. The assessment parameters for bone quality within the region of interest included bone volume fraction (BV/TV) and bone mineral density (BMD).
Biomechanical Testing
Biomechanical testing was performed using a testing system (Model 5942; Instron) to evaluate failure load and stiffness. Before testing, the samples were thawed to reach room temperature. Subsequently, the humerus was clamped using a vise, and the SST was firmly fixed in the vise by sandpaper. The SSTH was positioned in the uniaxial testing machine according to the approximate anatomic location of the SSTH (Appendix Figure S1). A preload of 0.1 N was applied to the specimen for preconditioning, followed by loading the specimen at a rate of 0.03 mm/s until failure and recording the specimen's ultimate failure load and failure distance at maximum loading. Stiffness was calculated from the load-deformation curve.
Histological Evaluation
The dissected samples of the SST and attachment cartilage were fixed in buffered formalin, decalcified with 5% EDTA solution, dehydrated in ethanol, and embedded in paraffin for histological analysis. Subsequently, 5 μm–thick paraffin-embedded sections were obtained along the longitudinal axis of the SST and then subjected to staining with hematoxylin and eosin (H&E) and toluidine blue. Stained sections were scanned at 40× magnification, and ImageJ software (National Institutes of Health) was utilized for semiquantitative analysis of 3 random areas for each image. Based on the established criteria of a modified scoring system for tendon-to-bone maturation, this study assessed cellularity, vascularity, continuity, cell alignment of fibrocartilage, and tidemark on a scale of 0 to 4 points (Table 1).
Modified Tendon Maturing Score a

The percentage was a relative value compared with the uninjured bone-to-tendon insertion.
Immunohistochemistry Staining
The tissue sections were initially deparaffinized and rehydrated. Antigen retrieval was then performed using Proteinase K (20 mg/mL). The primary antibodies anti-FBLN7 antibody (1:300; PK42060S [Abmart]), anti-Runx2 antibody (1:400; PA5-87299 [Invitrogen]), anti-Sox9 antibody (1:300; ab185966 [Abcam]), and anti-SCX antibody (1:400; ab307722 [Abcam]) were incubated with the tissue sections overnight at 4°C. Subsequently, the tissue sections were incubated with a universal immunohistochemical secondary antibody reagent (GK500710; Gene Tech) for 20 minutes at room temperature. The staining signal was then detected using a DAB chromogen kit (Gene Tech) and counterstained with hematoxylin.
Statistical Analysis
All data were analyzed using GraphPad software (GraphPad Software), and the results are presented as mean ± standard deviation. To assess the effects of multiple factors, 2-way analysis of variance with Bonferroni post hoc testing was performed. The statistical significance level was set at P < .05.
Results
Expression of FBLN7 in BTI of Mice
Immunohistochemical analysis of FBLN7 was performed to evaluate the expression difference between old and young BTIs. The results revealed a significant decrease in FBLN7 expression at the BTI in 18-month-old aged mice compared with 8-week-old young mice (Figure 1, A and B).
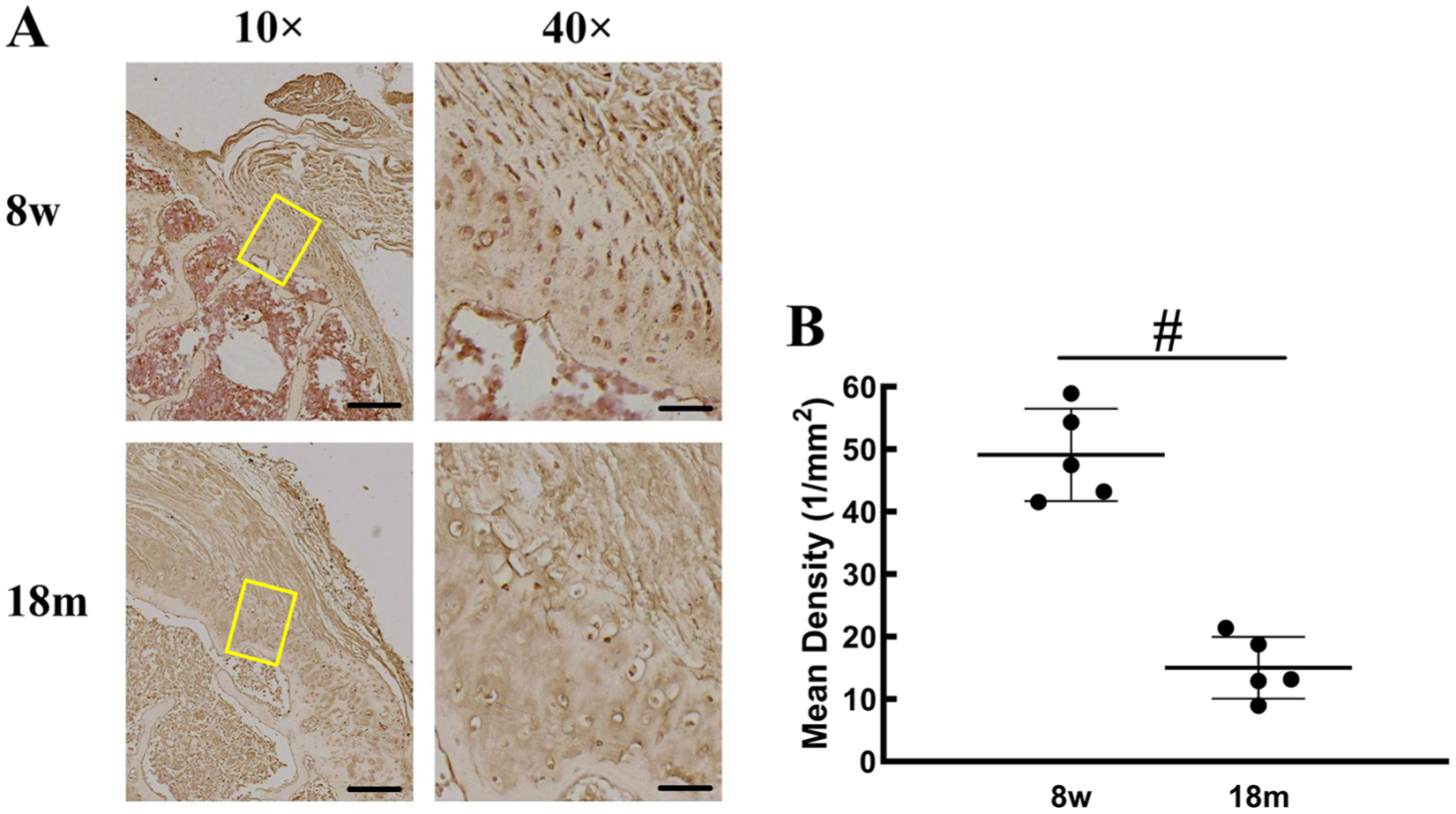
Immunohistochemical analysis of FBLN7 expression at the bone-tendon interface (BTI) of aged and young mice. (A) Representative images of FBLN7 staining. (B) Quantification of FBLN7 expression. Scale bars = 200 μm (10×) and 50 μm (40×). #P < .01.
FBLN7 Improved BTI Healing
Next, we evaluated the effect of FBLN7 on BTI healing through imaging and biomechanical testing. At 4 weeks postoperatively, the control group still displayed notable defects in subchondral bone at the BTI, while the FBLN7 group showed enhanced shaping of subchondral bone (Figure 2A). Furthermore, at 8 weeks postoperatively, cortical bone at the BTI in the control group appeared thinner, with sparser trabeculae and evident osteoporosis, whereas the FBLN7 group showcased thicker and denser cortical bone at the BTI (Figure 2A). Quantitative analysis supported these observations, demonstrating significantly higher bone morphological parameters in the FBLN7 group. The BV/TV in the FBLN7 group at both 4 weeks (0.284 ± 0.045) and 8 weeks (0.464 ± 0.055) postoperatively was markedly greater than that in the control group (0.180 ± 0.039 and 0.358 ± 0.044, respectively) (P < .05 for all) (Figure 2B). Similarly, the BMD in the FBLN7 group at both 4 weeks (0.356 ± 0.037 g/cm3) and 8 weeks (0.382 ± 0.054 g/cm3) postoperatively was notably increased compared with that in the control group (0.254 ± 0.046 g/cm3 and 0.286 ± 0.036 g/cm3, respectively) (P < .05 for all) (Figure 2C). Biomechanical testing further elucidated superior healing in the FBLN7 group, as evidenced by significantly higher failure loads at 4 weeks (3.036 ± 0.336 N) and 8 weeks (4.298 ± 0.334 N) postoperatively compared with the control group (2.322 ± 0.214 N and 3.490 ± 0.249 N, respectively) (P < .05 for all) (Figure 2, D and E). Additionally, the FBLN7 group exhibited significantly higher stiffness values at 4 weeks (2.248 ± 0.253 N/mm) and 8 weeks (3.156 ± 0.327 N/mm) after surgery than the control group (1.506 ± 0.200 N/mm and 2.318 ± 0.330 N/mm, respectively) (P < .05 for all).

Imaging and biomechanical testing of the effect of FBLN7 on bone-tendon interface (BTI) healing. (A) Representative images showing subchondral bone shaping at 4 and 8 weeks postoperatively. (B) Quantitative analysis of bone volume fraction (BV/TV). (C) Quantitative analysis of bone mineral density (BMD). (D) Biomechanical testing results for failure load. (E) Biomechanical testing results for stiffness. Scale bars = 1000 μm. P < .05.
FBLN7 Improved Tissue Fusion and Formation at BTI
To investigate the effectiveness of FBLN7 in promoting tissue fusion at the BTI, we conducted a histological assessment. H&E staining revealed that tissue fusion at the BTI in the FBLN7 group was superior to that in the control group at 4 and 8 weeks after surgery (Figure 3A). Additionally, there was a substantial increase in fibrocartilage formation at the BTI in the FBLN7 group at 8 weeks after surgery (Figure 3A). Toluidine blue staining indicated a notably enhanced distribution of proteoglycans in cartilage in the FBLN7 group compared with the control group (Figure 3B). Furthermore, the histological scores of the FBLN7 group were consistently increased higher than those of the control group at both 4 and 8 weeks after surgery (Figure 3C).

Histological assessment of the effectiveness of FBLN7 at the bone-tendon interface (BTI). (A) Representative hematoxylin and eosin (H&E) staining images showing tissue fusion at the BTI at 4 and 8 weeks after surgery. (B) Representative toluidine blue staining images showing the proteoglycan distribution in cartilage at the BTI at 4 and 8 weeks after surgery. (C) Histological scores of tissue fusion and fibrocartilage formation at the BTI at 4 and 8 weeks after surgery. Scale bars = 200 μm (10×) and 50 μm (40×). *P < .05, #P < .01.
FBLN7 Upregulated Osteogenic, Chondrogenic, and Tenogenic Factors
Runx2, Sox9, and SCX are important transcription factors involved in the development and differentiation of skeletal tissues, including bone, cartilage, tendons, and ligaments.6,15 Here, we further investigated the healing process at the BTI by evaluating the expression of these factors. The results revealed that the expression of Runx2 at the BTI healing site in the FBLN7 group was notably higher at both 4 and 8 weeks postoperatively compared with the control group (Figure 4, A and B). Similarly, the expression of Sox9 at the BTI healing site in the FBLN7 group was markedly elevated at 4 and 8 weeks postoperatively, surpassing that in the control group (Figure 4, C and D). Additionally, the expression of SCX at the BTI healing site was significantly higher in the FBLN7 group at 4 weeks postoperatively, while no substantial statistical difference was observed between the 2 groups at 8 weeks postoperatively (Figure 4, E and F).
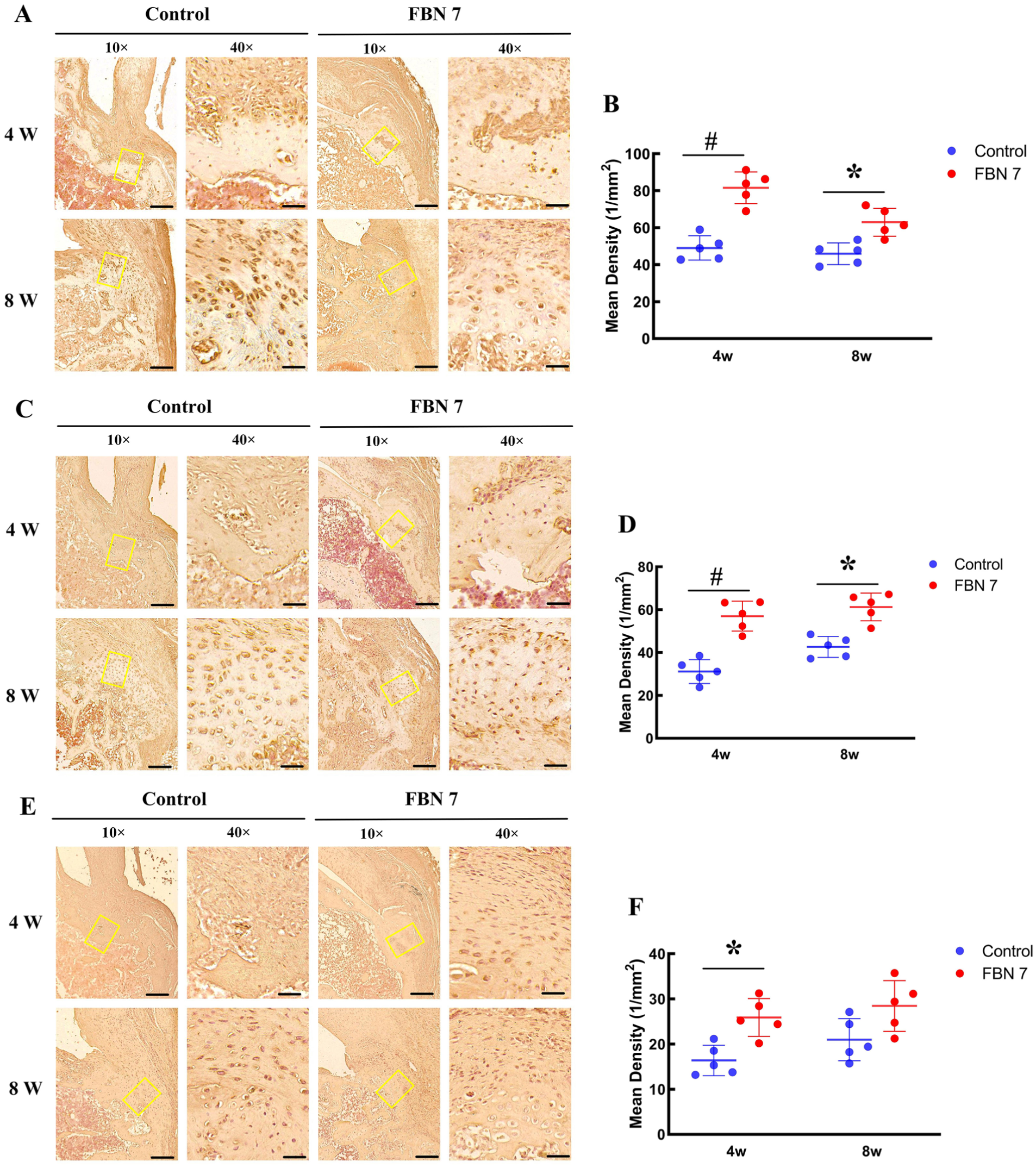
Evaluation of the expression of osteogenic, chondrogenic, and tenogenic factors in the healing process at the bone-tendon interface (BTI). (A, B) Expression of Runx2 at the BTI healing site at 4 and 8 weeks postoperatively. Representative images are shown on the left and quantification on the right. (C, D) Expression of Sox9 at the BTI healing site at 4 and 8 weeks postoperatively. Representative images are shown on the left and quantification on the right. (E, F) Expression of SCX at the BTI healing site at 4 and 8 weeks postoperatively. Representative images are shown on the left and quantification on the right. Scale bars = 200 μm (10×) and 50 μm (40×). *P < .05, P < .01.
Discussion
Effective healing at the BTI plays a critical role in the successful repair of rotator cuff injuries. 34 Unfortunately, aging patients frequently face challenges in this regard, as they often exhibit diminished healing capacity attributed to degenerative changes. 32 In this study, we aimed to investigate the effect of FBLN7 on BTI healing in a murine rotator cuff repair model. Initially, we provided compelling evidence that FBLN7 significantly improved tissue fusion and stimulated fibrocartilage formation at the BTI. Furthermore, our results confirmed that FBLN7 enhanced bone morphological parameters and the superior biomechanical properties of the BTI. Additionally, FBLN7 upregulated the expression of osteogenic, chondrogenic, and tenogenic factors.
It is well established that during the healing process at the BTI, regenerated tissue may not fully restore the structural organization and mechanical properties of the original fibrocartilage layer, leading to compromised functionality and resilience compared with the BTI before the injury. 8 In elderly patients, it is common to observe lower bone density, which increases the susceptibility to age-related injuries such as tendon ruptures, fractures, and degenerative changes. These conditions not only pose a higher risk of injuries but also make the repair process more challenging. 19 Therefore, it is crucial to strengthen the means of repair. In this study, we found a significant decrease in the expression of FBLN7 at the BTI in aged mice compared with young mice, indicating its potential role in age-related BTI injuries. Similar to a previous study, 1 our results showed subchondral bone beneath the tendon insertion of aged mice, which exhibited evident defects, characterized by thinner cortical bone, sparser trabecular bone, and significant osteoporosis. Intriguingly, FBLN7 improved the subchondral bone architecture, suggesting a potential role of FBLN7 in enhancing tissue regeneration and restoring biomechanical integrity.
From a mechanistic perspective, FBLN7 appears to regulate BTI healing through its interactions with ECM components, cell-surface receptors, and growth factors. As a matricellular protein, FBLN7 contributes to ECM assembly and structural organization, thereby influencing fibrocartilage formation and bone-tendon integration. Previous studies have implicated FBLN7 in tissue development, angiogenesis, and inflammatory modulation, and genetic deficiency has been linked to congenital malformations. 3 These functions are consistent with our findings of enhanced osteogenic (Runx2), chondrogenic (Sox9), and tenogenic (SCX) activities, suggesting that FBLN7 provides a favorable microenvironment for osteochondral regeneration at the BTI. A proposed schematic of these interactions is shown in Appendix Figure S2.
It is noteworthy that a low BMD in large nodules has been identified as a prognostic factor for postoperative recurrent tearing after rotator cuff repair. 17 To further evaluate the effect of FBLN7 on bone quality and density, we assessed BV/TV and BMD, which are key indicators of bone health.16,35 Our micro-CT analysis revealed a significant improvement in BV/TV and BMD in the FBLN7 group, indicating an enhancement in bone quality and density. Additionally, FBLN7 showed a notable effect on tensile load and stiffness, suggesting improved biomechanical properties. These findings suggest that FBLN7 may contribute to restoring tissue functionality and resilience after a BTI injury. However, it is necessary for future investigations to explore the underlying mechanisms through which FBLN7 influences tissue regeneration.
Our findings from H&E staining provided evidence that FBLN7 improved tissue fusion and formation at the BTI compared with the control group. These results were consistent with a previous study that demonstrated the role of FBLN7 in modulating tissue development and remodeling. 35 It has been previously shown that chondrocyte differentiation and hypertrophy could promote the formation and development of cartilage tissue, contributing to the repair of damaged rotator cuff tissue.5,28,31 Proteoglycans are considered as key components of normal cartilage tissue 10 and play a crucial role in promoting chondrogenic differentiation. 27 In our study, we observed an increase in the presence of fibrocartilage and proteoglycans at the BTI in the FBLN7 group. The transcription factors Runx2, Sox9, and SCX have been implicated in regulating chondrocyte and osteoblast biology.6,15 Sox9, through its regulation of cartilage matrix synthesis or cartilage-specific genes, promotes chondrocyte differentiation and cartilage tissue development.25,26 Our results indicated that FBLN7 upregulated the expression of key factors involved in osteogenesis, chondrogenesis, and tenogenesis at the BTI, including Runx2, Sox9, and SCX. These findings suggest that FBLN7 promotes healing at the BTI by facilitating tissue fusion, enhancing fibrocartilage formation, and upregulating the expression of osteogenic, chondrogenic, and tenogenic factors.
Our study employed 18-month-old mice in an aging model, which reflects certain physiological characteristics of elderly humans, such as reduced bone density and impaired tendon healing. However, most human rotator cuff tears occur in a chronic and degenerative environment, often with poorer tendon quality and substantial bone remodeling. While this model provides insight into age-related deficits, differences in tissue degeneration remain. Whether FBLN7 can reverse these chronic degenerative changes and modify the subacromial milieu warrants further studies.
From a translational perspective, FBLN7 could be delivered clinically in several ways. Potential approaches include a local injection at the repair site, incorporation into a fibrin-based sealant, or integration into bioengineered patches or scaffolds placed between tendon and bone. Each strategy carries specific advantages and challenges, particularly regarding stability, release kinetics, and sustained bioactivity. Future studies are required to determine the optimal delivery system and clinical applicability.
Limitations
This study has several limitations. First, the safety profile was not examined. Second, the molecular mechanisms by which FBLN7 promotes tissue fusion and regeneration at the BTI are still unknown. Further research is needed to understand the specific signaling pathways and cellular processes involved. Additionally, although structural healing at the BTI is generally considered desirable, clinical studies indicate that nonhealed repair sites on ultrasound may still yield functional improvements.37,38 This highlights that rotator cuff repair outcomes are multifactorial, with pain relief, neuromuscular compensation, and biological remodeling contributing to recovery. Lastly, the sample size was limited in this preliminary study, and future studies with larger cohorts will be conducted to validate our findings. Overall, more research is needed to explore the underlying mechanisms and potential clinical applications of FBLN7 in musculoskeletal disorders.
Conclusion
FBLN7 significantly improved BTI healing in aged mice, as evidenced by enhanced fibrocartilage formation, improved bone quality, and superior biomechanical strength. These findings highlight the therapeutic potential of FBLN7 in rotator cuff repair, particularly in an aging population.
Footnotes
Appendix
Final revision submitted October 23, 2025; accepted November 18, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was supported by the Guangdong Basic and Applied Basic Research Foundation (2022A1515110910), the Medical Scientific Research Foundation of Guangdong Province (A2023105), and the President Foundation of The Third Affiliated Hospital of Southern Medical University (YQ2021009). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
