Abstract
Background:
Quantifying glenoid bone loss (GBL) in patients with shoulder instability is essential for guiding surgical management and determining the need for bone augmentation procedures.
Purpose/Hypothesis:
The purpose of this study was to evaluate the diagnostic agreement and cost-effectiveness of 3-dimensional (3D) zero echo time (ZTE) magnetic resonance imaging (MRI) compared with 3D computed tomography (CT) for assessing GBL. It was hypothesized that ZTE MRI would demonstrate strong concordance with CT and serve as a cost-effective, radiation-free alternative.
Study Design:
Cohort study (Diagnosis); Level of evidence, 2.
Methods:
Patients undergoing MRI with both 3D ZTE and 2-dimensional proton density fat-saturated (PD FS) sequences, as well as 3D CT of the ipsilateral shoulder, were retrospectively reviewed. Patients with recurrent instability between imaging studies were excluded. GBL was measured independently by 2 raters using the perfect-circle linear method. Interrater and intrarater reliability were assessed using intraclass correlation coefficients (ICCs). Concordance with CT was evaluated using concordance correlation coefficients (CCCs). Cost-effectiveness was preliminarily assessed using institutional imaging charges and diagnostic agreement.
Results:
Eleven patients were included; 81.8% had ≥3 instability episodes. Mean GBL was 13.2% ± 9.3% (PD FS), 12.8% ± 8.9% (ZTE), and 12.7% ± 8.9% (CT). Inter- and intrarater reliability were excellent (ICC > 0.9). ZTE (CCC, 0.999; 95% CI, 0.997-0.999) and PD FS (CCC, 0.988; 95% CI, 0.974-0.994) demonstrated excellent agreement with CT. Estimated patient-billed costs were $1652 for CT + MRI, versus $1019 for either ZTE MRI or PD FS MRI alone. Corresponding institutional costs were $593 for CT + MRI and $329 for either ZTE or PD FS MRI.
Conclusion:
3D ZTE MRI is a reliable and reproducible alternative to CT for quantifying GBL in shoulder instability. With excellent diagnostic agreement, elimination of ionizing radiation, and reduced costs, ZTE MRI may serve as a single-modality solution for preoperative evaluation in select patients.
The glenohumeral joint is the most frequently dislocated joint in the human body, accounting for approximately 50% of all major dislocations. 40 In patients with traumatic anterior shoulder instability, osseous morphology, particularly glenoid bone loss (GBL), significantly affects clinical outcomes and the risk of recurrence.4,41 Accurate quantification of glenoid bone defects is therefore essential, as the extent of bone loss directly influences surgical planning and the potential need for bone augmentation procedures such as coracoid transfer or distal tibial allograft.5,25
Three-dimensional (3D) computed tomography (CT) remains the gold standard for evaluating GBL because of its high correlation with intraoperative findings.2,8 However, magnetic resonance (MR) imaging (MRI) is routinely obtained first to assess soft tissue pathology, including labral and capsular injuries, which also guide surgical decision making.12,31 As a result, many patients undergo both CT and MRI, increasing health care costs, patient burden, and exposure to ionizing radiation.
To reduce redundant imaging, several MRI techniques have been developed to improve on MRI's ability to capture osseous structures.22,24,32 Conventional MRI sequences are prone to inaccuracy and limited in cortical bone visualization due to rapid signal decay, particularly those with longer echo times (TEs). 6 Cortical bone appears dark on these sequences, requiring image readers to infer its borders based on surrounding bone marrow or soft tissue signal. 18 Proton density fat-suppressed (PD FS) sequences can improve contrast between bone marrow and adjacent soft tissues, which may indirectly aid in assessing bone loss, although they remain limited in directly visualizing cortical margins. Moreover, 3D reconstructions are generally not feasible from PD FS sequences because their slice thickness and resolution are not optimized for volumetric rendering.
In the context of shoulder imaging, the application of zero TE (ZTE) MRI is particularly valuable because of its ability to directly visualize cortical bone, which is crucial for assessing complex bony pathologies.3,11,38 Previous investigations have begun to explore ZTE's utility for detailed evaluation of glenoid morphology, identification of bone defects, and quantification of bone loss, which are critical factors in the diagnosis and surgical planning for shoulder instability. For instance, studies have leveraged ZTE's unique contrast to provide high-resolution images of the glenoid rim and quantify bone defects with accuracy comparable with CT, thereby reducing the need for ionizing radiation.10,23 Despite promising technical performance in recent studies, 3D ZTE MRI is not widely available in the United States, and its use is currently limited to certain scanner platforms (eg, GE Medical Systems Signa; Philips Ingenia Elition) that support specialized implementations. Moreover, few investigations have comprehensively evaluated ZTE MRI in clinically relevant domains, including measurement reliability, cost-effectiveness, and comparison with routinely used non-ZTE MRI sequences such as PD FS imaging.
This study aimed to provide a comprehensive evaluation of 3D ZTE MRI in the assessment of GBL in shoulder instability. Specifically, we (1) compared the accuracy of 3D ZTE and 2-dimensional (2D) PD FS MRI sequences with 3D CT, (2) assessed inter- and intrarater reliability of GBL measurements, and (3) conducted a preliminary cost comparison analysis to evaluate the feasibility of adopting ZTE as a standalone imaging modality. It was hypothesized that ZTE MRI would demonstrate strong agreement with CT, outperform PD FS in reliability and accuracy, and reduce the economic burden associated with dual imaging.
Methods
Study Design and Patient Selection
This retrospective study was approved by the institutional review board at INOVA Health System. We queried the institutional surgical database from 2010 to 2024 to identify patients who underwent surgical stabilization for anterior shoulder instability, using Current Procedural Terminology codes 23455 (open Bankart repair), 23462 (coracoid transfer/Latarjet), and 29806 (arthroscopic capsulorrhaphy). Advanced imaging was ordered at the discretion of the treating surgeon for diagnostic purposes and preoperative planning. At our institution, all shoulder MRIs were performed with arthrograms in accordance with radiology department policy.
Inclusion criteria were
Imaging of the operative shoulder with both 3D CT (with humeral head subtraction) and MR arthrogram on a 3-T GE scanner using standard protocols at our institution
MR arthrograms that included both PD FS and optimized ZTE sequences at our institution
Exclusion criteria included
MRI lacking either PD FS or ZTE sequences
Imaging performed for posterior instability
Previous shoulder surgery
Interval between CT and MRI >90 days
Documented instability events between the CT and MRI studies
The sequence of imaging (CT or MRI first) was not an exclusion criterion. Demographic variables (age, sex, body mass index [BMI]) and the number of preoperative instability events (subluxations, dislocations) were recorded. To mitigate concerns regarding interval changes, we reviewed patient records to confirm that no new instability events, such as dislocations or subluxations, occurred between the CT and MRI scans for any enrolled participant.
Imaging Protocol
For CT, all patients for this study were scanned using multidetector CT scanners (GE Medical Systems Lightspeed VCT 64-slice scanner; GE Medical Systems Optima CT660 64-slice CT scanner; Philips Brilliance 64-slice CT scanner; or Siemens Somatom Dual Source CT scanner). CT shoulder protocols used 0.625-mm standard collimation, axial thin-slice interval ranging from 0.625 to 1.25 mm, standard bone reconstruction algorithm in the sagittal and coronal planes, field-of-view ranging from 25 to 30 cm, tube voltage of 120 or 140 kVp, auto mAs tube current, rotation speed of ≤1 second, pitch <1, and table speed of ~30 mm/rotation. The GE Advantage Workstation was used at the 3D labs at all sites to create 3D models of the shoulder.
For MRI, all patients for this study were scanned using a 3-Tesla (3-T) scanner (GE Medical Systems Signa; Philips Ingenia Elition). In addition to a standard MRI arthrogram protocol, a ZTE sequence was performed with a field of view of ~20 cm, a slick thickness of 1 mm, 160 slices per slab, a TE ranging from 0.01 to 0.02 ms, a repetition time ranging from of 400 to 550 ms, a flip angle of 1° or 2°, 384 spokes per segment, number of excitations ranging from 5 to 7, and a bandwidth ranging from 50 to 65 kHz. The acquisition time for the ZTE sequence was typically 4 to 5 minutes.
For patients scanned on a GE Medical Systems Signa 3-T scanner, the standard MRI arthrogram protocol included PD FS sequences performed in axial, coronal, and sagittal planes. The sequences were performed with a field of view of 13 to 14 cm, a slice thickness between 3 and 4 mm, number of images ranging from 15 to 30 images per plane, a TE ranging from 25 to 55 ms, a repetition time ranging from 2000 to 5000 ms, a flip angle of 111°, number of excitations of 1 or 2, a bandwidth ranging from 60 to 90 kHz, and automated shimming. The acquisition time for each plane was typically 2 to 4 minutes.
Imaging Analysis
Two independent raters measured GBL twice, 3 weeks apart, to ensure temporal blinding. The senior rater (U.R., rater 1) was a board-certified, fellowship-trained musculoskeletal radiologist; the junior rater (S.S.T., rater 2) was a trained medical student under supervision by the senior author (E.S.C.).
All measurements were performed on Visage 7 software (Visage Imaging, Inc). The primary outcome was percentage GBL, calculated using the perfect circle method through overlay of a circle on the inferior glenoid matching the curvature of the posteroinferior glenoid.14,36 Secondary outcomes included glenoid diameter and linear width of the anteroinferior glenoid defect. GBL was calculated as the ratio of the glenoid defect divided by the perfect circle diameter (Figure 2).
ZTE sequences were performed with the ZTE scanner sequence (GE Healthcare). For 3D ZTE and PD FS MRI, flat multiplanar reformations were used to generate axial and sagittal realignments, respectively, for an en face glenoid view matching native glenoid version (Figures 1 and 2). Critical GBL was defined as >15%.28,29,35
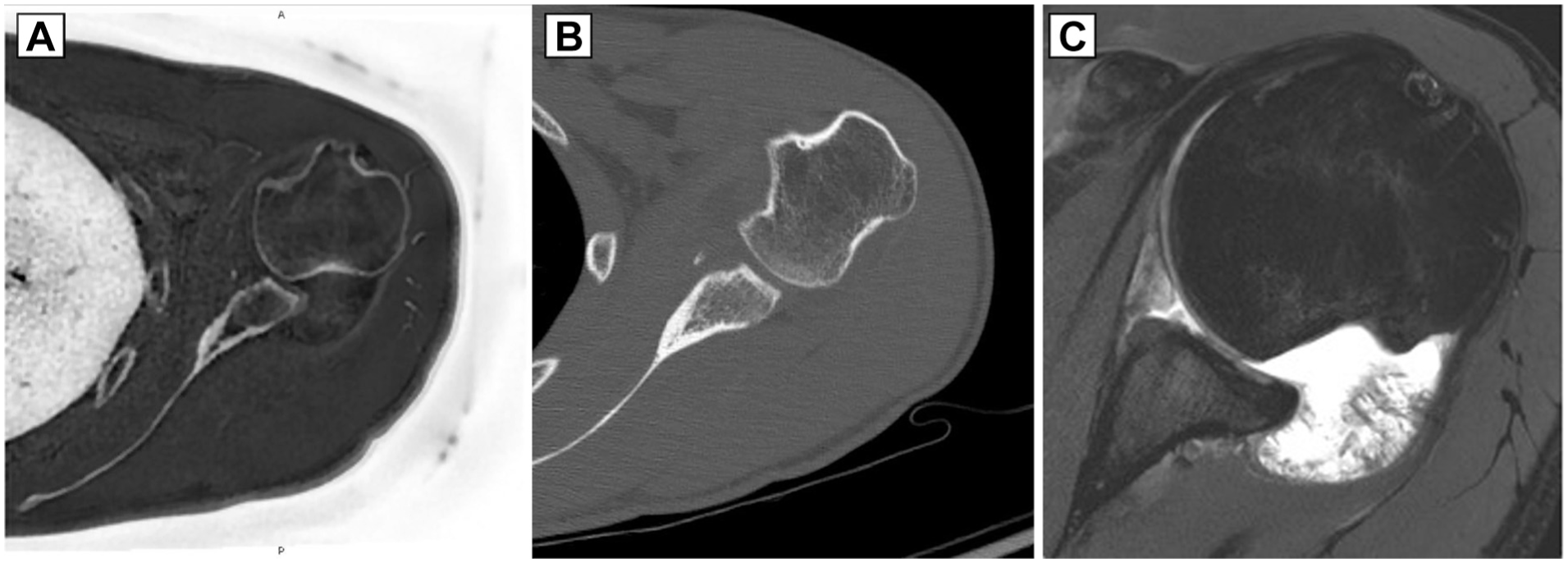
Axial images of a patient with critical glenoid bone loss, Hill-Sachs, and reverse Hill-Sachs lesions on (A) zero echo time magnetic resonance sequence, (B) computed tomography, and (C) proton density fat suppression magnetic resonance sequence.

En face views of a patient with critical glenoid bone loss (GBL) on (A) 3-dimensional (3D) zero echo time magnetic resonance imaging and (B) 3D computed tomography. (C) Best-fit circle linear method for measurement of native glenoid diameter and linear defect to calculate GBL (% GBL = defect width / glenoid diameter × 100). The red circle matches the contour of the posteroinferior glenoid, and the green line corresponds to the defect width.
Cost Comparison
We performed a cost comparison from the perspective of the health system to compare the downstream financial impact of using 3D CT, ZTE MRI, and PD FS MRI. We modeled the cost implications of 3 imaging strategies:
CT + MRI (standard approach): double-modality imaging with excellent accuracy, great bone and soft tissue visualization
MRI with ZTE only: single modality, excellent accuracy, and reduced radiation exposure, great bone and soft tissue visualization
MRI with PD FS only: low up-front cost, excellent accuracy, lower bone visualization
Direct imaging costs were estimated from institutional billing data; estimated professional interpretation fees and software analysis costs were incorporated using reimbursement rates and institutional data. In this study, we report total costs that are the sum of these values.
Statistical Analysis
A power analysis, with reference to Vopat et al, 34 determined that a sample size of 11 would yield 80% power to detect a strong correlation (r > 0.7) between MRI and CT for GBL. Descriptive statistics were reported for all measurements. Paired t tests and Shapiro-Wilk tests were used to evaluate between-rater agreement and normality of distributions. Intra- and interrater reliability was assessed using intraclass correlation coefficients (ICCs), with a 2-way mixed-effects model (ICC3 and ICC3,2 for single and mean measures, respectively), and classified as poor (<0.90), moderate (0.90-0.95), substantial (>0.95-0.99), and near-perfect (>0.99). 21
Agreement between imaging modalities was assessed using Bland-Altman plots. Linear regression models were created to evaluate correlation of GBL values between MRI sequences and CT. A 2-tailed alpha of .05 was set for statistical significance. All analyses were performed using jamovi Version 2.2.5.0.
Results
Study Cohort Characteristics
A total of 16 patients were identified to have 2D PD FS MRI, 3D ZTE MRI, and 3D CT reconstructions. Five were excluded: 4 for pending CT or MRI results and 1 for arthroplasty with glenoid retroversion; thus, 11 patients met inclusion and exclusion criteria and were included for analysis (Table 1). They were 90.9% male with a mean age 31.7 ± 15.9 years and BMI of 26.9 ± 4.6 kg/m2. They had sustained a median 2 subluxations (IQR, 2.3), 3 dislocations (IQR, 3.0), and 6 instability events (IQR, 3.5) before obtaining advanced imaging.
Patient Demographics and Clinical History a (N = 11)

BMI, body mass index; F, female; M, male.
Imaging-Based GBL Measurements
Overall mean percentage of GBL by imaging modality was 13.2% ± 9.3% on PD FS, 12.8% ± 8.9% on 3D ZTE, and 12.7% ± 8.9% on 3D CT. Descriptive statistics are in Table 2 for radiographic measurements by rater with inter- and intrarater reliability. All measurements (eg, linear defect, glenoid width) had excellent inter- and intrarater reliability.
Glenoid Bone Loss Measurements by Modality and Rater a

CT, computed tomography; FS, fat saturated; GBL, glenoid bone loss; ICC, intraclass correlation coefficient; PD, proton density; ZTE, zero echo time.
Agreement Between Modalities
There was good reliability for all measurements and excellent reliability for the primary outcome of % GBL. Concordance correlation coefficients (CCCs) between PD FS and CT were substantial, while CCCs between ZTE and CT demonstrated excellent agreement (Table 3). Bland-Altmann plots for modality types are exhibited in Figure 3.
Correlation of Imaging Modalities for Imaging Measurements a

CT, computed tomography; FS, fat saturation; GBL, glenoid bone loss; PD, proton density; ZTE, zero echo time.

Bland-Altman plots for comparing (A) 2-dimensional proton density fat-saturated magnetic resonance imaging (MRI) with 3-dimensional (3D) computed tomography (CT) for percentage glenoid bone loss (% GBL) and (B) 3D zero echo time MRI with 3D CT for % GBL.
Preliminary Cost-Effectiveness Comparison
The total patient cost for each strategy is as follows: CT + MRI: $1652; ZTE MRI only: $1019; and PD FS MRI only: $1019 (Table 4). The total institutional cost for each strategy is as follows: CT + MRI: $593, ZTE MRI only: $329, and PD FS MRI only: $329. The ZTE sequence added approximately 8 minutes to the MRI acquisition process, including both sequence acquisition and 3D reconstruction.
Cost Comparison Data for Different Imaging Strategies a

CT, computed tomography; FS, fat saturation; MRI, magnetic resonance imaging; PD, proton density; ZTE, zero echo time.
Required both scans.
Discussion
The primary finding of this study was that 3D ZTE MRI demonstrated substantial agreement with 3D CT for GBL measurements. Cost comparison analysis supported the imaging strategy of only ZTE, with its use reducing patient costs by $633 when compared with CT and MRI. ZTE also exhibited excellent intra- and interrater reliability across all measurements without underestimation of critical GBL, supporting its clinical consistency and reproducibility.
Importantly, 2D PD FS showed a similar correlation with 3D CT to 3D ZTE MRI for GBL measurements. However, the clinical risk of relying solely on conventional 2D MRI sequences as has been reported previously may favor the use of 3D ZTE.2,26 For example, mischaracterization of bone loss from lower bone cortical visualization and lack of 3D reconstruction capabilities can lead to inappropriate surgical decision-making: underestimation may result in failed Bankart repairs, carrying distinct risks, resource demands, and long-term implications. 31
Our preliminary cost comparison analysis favors ZTE as the most efficient and effective imaging strategy by minimizing radiation exposure and reducing overall costs. To our knowledge, this is one of the first clinical studies in the United States to evaluate 3D ZTE MRI in the context of shoulder instability. While ZTE offers significant advantages, it is important to acknowledge other 3D MRI techniques that share similar characteristics, such as 3D VIBE, 3D-Bone, and STAIR-UTE.7,9,13,16 These earlier 3D MRI techniques have achieved varying degrees of CT-like bone contrast. Moreover, each faces practical limitations that have restricted their routine use: resolution issues, susceptibility to motion artifacts, or scan time. ZTE seeks to address these constraints by delivering high-resolution isotropic images with improved bone–soft tissue contrast in a clinically feasible acquisition.
The relatively limited domestic adoption of ZTE technology in the United States, despite its promising technical performance, likely reflects a combination of factors, including initial costs associated with radiologist interpretation, advanced software analysis, and specialized expertise to integrate ZTE sequences into clinical workflows. Furthermore, the lack of widespread training and standardization may also contribute to slower adoption in routine musculoskeletal imaging. The limited adoption, despite promising technical performance, underscores the potential clinical value of broader implementation and justification for startup costs of this new technology.
Our findings are consistent with previous literature that validates the diagnostic utility of ZTE for bony shoulder assessment, as well as other uses such as evaluation of knee osteoarthritis and hip osseous morphology. 7 The data demonstrated substantial intermodality agreement between 3D ZTE MRI and 3D CT for quantifying GBL with a CCC of 0.999. The exceptional level of agreement observed in our study likely stems from several key methodological strengths. We used a standardized, single-institution imaging protocol with optimized ZTE sequences on uniform 3-T MRI scanners. Additionally, we employed volumetric 3D reconstructions with humeral head subtraction, which minimalized technical variability and operator-dependent differences. To mitigate bias, our raters were temporally blinded.
In contrast, previous studies evaluating ZTE MRI for glenoid assessment have reported lower correlations. Oishi et al 23 found a correlation coefficient of 0.89 (95% CI, 0.85-0.96) between ZTE and CT for GBL quantification, which may have been influenced by heterogeneous CT protocols, multiple imaging platforms, and manual postprocessing. Similarly, de Mello et al 10 reported moderate to substantial agreement between 3D MRI and 3D CT, with ICCs ≥0.94 and a bias of <1 mm across measurements, although their limits of agreement were wider at approximately 3 mm. This variability was likely due to use of multiple MRI resolutions, different patient and cadaveric cohorts, and operator-dependent postprocessing. By employing a single-center protocol with consistent 3D reconstructions and volumetric measurements, our study successfully minimized the technical and interobserver variability that might have impacted previous research, leading to the 0.999 CCC. These findings affirm ZTE's promise as a next-generation tool for comprehensive, bone-focused shoulder imaging.
Clinically, substituting CT with MRI for bone loss evaluation offers significant benefits, chief among them being the elimination of ionizing radiation to patients. This is particularly relevant in younger patients and those with recurrent instability who may require multiple imaging studies over time. A single shoulder CT exposes patients to a mean effective dose of approximately 10.83 mSv—equivalent to nearly 5700 chest radiographs.1,15,17,37 This corresponds to a lifetime attributable cancer risk of 0.1% to 0.2% and an increased relative risk of 16.1 to 17.5. 39 These figures underscore the long-term consequences of cumulative radiation exposure and support the growing demand for radiation-free diagnostic alternatives.
From a health systems perspective, cost is another critical factor in imaging selection. Despite increasing interest in optimizing imaging workflows, there remain limited data comparing the cost-effectiveness of different modalities for GBL assessment. A database study previously noted that the trend of obtaining both MRI and CT for preoperative planning could significantly increase costs and burden to both health care systems and patients. 27 In addition, Lander et al 16 found that the mean patient cost for an MRI with 3D reconstruction was $3940, while a combined 3D CT and MRI approach cost approximately $6610, roughly 1.7 times more expensive. Our study corroborated these findings: estimated patient billing in our institution was $1652 for CT + MRI and $1019 for either ZTE MRI or PD FS MRI alone.
At our institution, the total estimated institutional cost was $593 for CT + MRI and $329 for either ZTE MRI or PD FS MRI. Notably, there was no added cost to ordering the ZTE sequence or its 3D reconstruction, as this is incorporated into existing software licensing fees. Additionally, overall acquisition time increased by only 8 minutes with the addition of 3D ZTE. Despite similar baseline costs, PD FS imaging could carry a higher downstream cost because of its documented limitations in accurately classifying osseous morphology in shoulder instability. 30 Examples of downstream costs from misclassification are surgical failures, increased rates of instability, and the need for more complex and expensive revision surgeries. This can incur substantial long-term costs.19,20,33 Furthermore, patients with borderline findings often require additional CT imaging to clarify bone loss, increasing both cost and time to definitive care. These limitations highlight the risks associated with PD FS imaging and further support the integration of ZTE as a potentially more reliable and cost-effective imaging modality.
Overall, the data from this study for 3D ZTE MRI demonstrate its ability to reliably measure GBL comparable with CT. With studies showing that ZTE MRI provides measurements of glenoid and humeral bone loss with very high intermodality agreement and excellent ICCs to CT in various cohorts, ZTE MRI looks to be a singular imaging solution for shoulder instability that simultaneously evaluates soft tissues, accurately quantifies GBL, and minimizes radiation.10,23
Therefore, ZTE emerged as the most cost-effective strategy among the imaging modalities tested, providing high diagnostic accuracy, radiation-free imaging, and >$600 in cost savings to the payer. These findings support the integration of ZTE into institutional imaging algorithms and clinical decision-making pathways for anterior shoulder instability.
Limitations
This study has several limitations. First, the small sample size (N = 11), while adequately powered for modality comparison, limits the generalizability of the findings. However, this reflects the current limited adoption of ZTE MRI in the United States, where few institutions have incorporated this technology into clinical shoulder imaging. This study is intended to serve as foundational support for future investigations focused on this critical range.
Second, cost comparison values were derived from institutional estimates and should be validated through broader cost-utility analyses using real-world financial and clinical outcome data in larger cohorts. While the $633 reduction in patient costs with ZTE MRI was notable in our study, we acknowledge that the sustainability and generalizability of this difference may vary across institutions due to differing reimbursement models and payer systems. Our analysis was conducted within an institutional setting that serves a hybrid of Medicare and commercial payers, which may influence the specific cost reductions observed. In addition, certain costs, such as software licensing fees, were not able to be obtained. Future research, including multicenter studies and sensitivity analyses, would be valuable to provide broader context and assess the generalizability of these cost savings.
Furthermore, our study focused exclusively on GBL and did not evaluate the full clinical spectrum of injury, as it did not assess humeral-sided bone loss or the characterization of track lesions, including the increasingly relevant near-track and peripheral-track patterns. A comprehensive evaluation of these parameters would be critical for full surgical planning and should be a focus of future research. Last, imaging was not correlated with surgical or arthroscopic findings, which would further strengthen clinical validity.
Conclusion
3D ZTE MRI is a reliable and reproducible alternative to CT for quantifying GBL in shoulder instability. With excellent diagnostic agreement, elimination of ionizing radiation, and reduced costs, ZTE MRI may serve as a single-modality solution for preoperative evaluation in select patients.
Footnotes
Final revision submitted September 30, 2025; accepted November 9, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: S.M.F. has received support for education from Supreme Orthopedic Systems LLC. E.S.C. is a consultant for Avanos Medical Inc and has received support for education and research from Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto
Ethical approval for this study was obtained from INOVA Health System (reference No. U21-10-4593).
