Abstract
Background:
There is growing interest in understanding the correlates of revision anterior cruciate ligament reconstruction (ACLR) failure, and conflicting evidence exists regarding body mass index (BMI) as a risk factor.
Purpose:
To evaluate the relationship between BMI and revision ACLR failure.
Study Design:
Case-control study; Level of evidence, 3.
Methods:
Patients aged 16 years or older who underwent revision ACLR between 2012 and 2021 with a minimum 2-year follow-up were included. Patients with inadequate follow-up, incomplete medical records, or concomitant lateral extra-articular tenodesis, anterolateral ligament reconstruction, osteotomy, osteochondral transplant, or multiligamentous injury were excluded. BMI was classified as normal (18.5 ≤ BMI < 25.0 kg/m2), overweight (25.0 ≤ BMI < 30.0 kg/m2), and obese 30.0 ≤ BMI < 35.0 kg/m2). Intraoperative data, concomitant injuries, complications, graft failure, and second revision ACLR data were collected.
Results:
A total of 174 patients who underwent revision ACLR were included; 37 (21.3%; 26 male, 11 female) experienced revision ACLR failure while 137 (78.7%; 83 male, 54 female) did not. The mean BMI did not differ between patients with and without revision ACLR failure. There was no difference in revision ACLR failure when stratifying patients by normal (n = 81), overweight (n = 57), or obese (n = 36) BMI.
Conclusion:
There is no significant relationship demonstrated between BMI and the risk of revision ACLR failure.
Anterior cruciate ligament (ACL) tears are one of the most common sports-related injuries, 20 with ACL reconstruction (ACLR) being the gold standard for patients to return to their preinjury sport level.14,15,22 However, there is a 5.2% to 34.2% graft failure rate,17,28 and failure rates of 3.5% to 33% for revision ACLR. 8 Revision ACLR outcomes are not as successful as primary ACLR regarding functional scores, patient satisfaction, and risk of developing knee osteoarthritis.11,21 In fact, each subsequent revision ACLR results in poorer patient satisfaction, functional scores, and lower return to sport (RTS) and activity levels.3,5-7,12,29 Thus, there is growing interest in the risk factors for revision ACLR failure.1,12,26,30,32
Common causes of ACLR revision failure include technical errors such as femoral tunnel malposition including increased graft angle, increased posterior tibial slope, biological factors such as hyperlaxity and narrow notch width, untreated secondary instabilities such as those sustained from trauma, and knee infection.9,19,24 Conflicting evidence exists on whether BMI plays a role in the risk of second revision ACLR. One retrospective study found that patients with a BMI >30 (n = 16) had 9 times the risk for repeat revision ACLR. 1 Vap et al 26 did not find an association between BMI and an increase in ACLR re-revision (n = 40); however, they did not control for other risk factors such as posterior tibial slope, tunnel position, graft type, and femoral tunnel drilling technique. Additionally, another study found no significant differences between the BMI of patients with stable revisions and patients requiring repeat revision ACLR (n = 20). 30 Given the small sample sizes and the lack of consensus in the limited literature that examines the association between BMI and re-revision ACLR, further studies are needed to assess this relationship.
Obese individuals may be at a greater risk for ultra-low-speed ACL injuries through noncontact mechanisms,10,25,27 possibly leaving patients with a high BMI at increased odds of revision failure. Importantly, patients with a BMI >28 have worse Lysholm knee scores than patients with a BMI <28 after repeat revision ACLR (fair or poor; P = .03). 11 It is, therefore, prudent to understand the association between BMI and revision ACLR failure to allow for improved surgical planning and patient counseling. We hypothesized that increased BMI would result in a greater risk of revision ACLR failure. The purpose of this study was to evaluate the relationship between a patient's BMI and the risk of ACLR revision failure.
Methods
Study Design
A retrospective case-control study assessed the relationship between BMI and the risk of revision ACLR failure.
Patient Identification
After obtaining approval from the institutional review board (IRB00103848), we performed a query of the administrative database at a single academic institution from 2012 to 2021 using the Current Procedural Terminology code 29888 (ACLR). Extensive chart review was performed to distinguish between successful primary revision ACLR and patients with revision ACLR failure. ACLR revision failure was defined as magnetic resonance imaging–documented retear of the first revision or return to the operating room for re-revision ACLR. Patients 16 years and older at the time of revision ACLR with at least a 2-year follow-up were identified for study inclusion. Patients with multiligamentous injury, concomitant osteotomy or osteochondral transplant, inadequate follow-up, or incomplete medical records were excluded from the study.
Data Collection
Clinical and operative notes of all eligible patients were reviewed, and demographic, operative, and postoperative data were obtained. BMI was calculated at the time of revision ACLR and at the time of revision ACLR failure for those in the failure group. BMI was classified as normal (18.5 ≤ BMI < 25.0 kg/m2), overweight (25.0 ≤ BMI < 30.0 kg/m2), and obese (BMI 30.0 ≤ BMI < 35.0 kg/m2). Surgical variables included graft type, tunnel drilling method, and graft fixation type. Graft type included autograft, allograft, and allograft augmentation. Augmented allograft was an autograft hamstring tendon with either a semitendinosus, tibialis anterior, or tibialis posterior allograft added. All data were extracted from postoperative clinic notes as documented by the treating surgeon. Concomitant injuries and procedures for each reconstruction were collected. Level of activity before revision ACLR and RTS data were collected. Level of activity included recreational, high school, college, professional, or former elite sports. Study data were collected and managed using the Research Electronic Data Capture System (Vanderbilt University), a secure web-based software platform hosted at OrthoCarolina. 13
Surgical Technique
All patients were treated by fellowship-trained sports medicine orthopaedic surgeons (8 total) at our single academic institution. Overall patient health was assessed individually and used to determine patient appropriateness for surgery. Surgical approaches were not standardized for this study, but operative data were collected to control for this variable in the analysis. Lateral extra-articular procedures were not performed in any of the patients in the study.
Postoperative Rehabilitation and Follow-up
Postoperative rehabilitation varied per surgeon and for concomitant injuries but followed best practice guidelines. All patients received postoperative rehabilitation according to standard ACL rehabilitation protocols. Generally, this includes weightbearing as tolerated and range of motion as tolerated with patellar mobilization and closed-chain quadricep strengthening for the first 6 weeks. During this period, a hinged-knee brace locked in extension was used for walking and sleeping. RTS decisions were made with surgeon and physical therapist input, and standard physical therapy RTS testing was performed before clearance 6 to 9 months postoperatively. Patients could return to pivoting and cutting at the surgeon's discretion. Routine follow-up was scheduled at 1 to 2 weeks, 6 weeks, 3 months, 6 months, and 9 months. Two-year follow-up was recorded, as this was an inclusion criterion for our study. Equipment availability did not differ across rehabilitation facilities.
Femoral Tunnel Angle
Postoperative plain radiographs taken within at least 6 months of the revision ACLR were used to measure femoral tunnel angles on anteroposterior (AP) and lateral images using methods described by Sohn et al 23 (Figure 1). The angle between the femoral tunnel and a line parallel to the articular surface was measured on the AP radiograph. On the lateral radiograph, the angle between the femoral tunnel and the extension line of the posterior cortex of the femoral shaft was measured. Angle measurement was performed by 3 different authors (G.G., A.M.I., A.B.W.), who were taught and supervised in this measurement by the senior author (B.M.S.). Each measurement was performed once by each examiner. Given the simplistic nature of measuring tunnel angles, we did not observe any major differences in angles between reviewers.

Postoperative anteroposterior (AP) and lateral images obtained in a patient who underwent revision anterior cruciate ligament reconstruction demonstrating femoral tunnel angle measurements. On the AP radiograph, the angle between the femoral tunnel and a line parallel to the articular surface was measured. On the lateral radiograph, the angle between the femoral tunnel and a line parallel to the posterior cortex was measured.
Statistical Analysis
Descriptive statistics were calculated for all continuous and categorical variables. Continuous variables are reported as mean with standard deviation, and categorical variables as frequency with percentage. BMI was a continuous variable in our analysis. Bivariate analyses of patients with and without a revision ACLR failure were performed using the Wald chi-square test to analyze categorical variables, and a t test was used to test the difference in means for continuous variables. Binary logistic regression models were used to generate reported odds ratios and 95% confidence intervals, modeling failure as the outcome as predicted by different demographic and surgical variables. Because no significant relationships were found, no further modeling was done.
All statistical analyses were conducted using SAS/STAT software Version 9.4 of the SAS System for Windows (SAS Institute Inc). An a priori power analysis was not performed because all eligible patients were included in the study.
Results
Patient Characteristics
The cohort consisted of 395 patients, and after inclusion and exclusion criteria were applied, 174 patients were eligible (Figure 2).

CONSORT (Consolidated Standards of Reporting Trials) diagram for included patients. ACL, anterior cruciate ligament; CPT, Current Procedural Terminology.
Of the included patients, 137 (78.7%; 83 male, 54 female) did not have a revision ACLR failure, and 37 (21.3%; 26 male, 11 female) had a revision ACLR failure. A second revision ACLR was performed in 29 of the 37 patients with an identified failure of revision ACLR. The 8 patients who did not undergo a repeat revision ACLR were either lost to follow-up or elected not to have surgery. No patients underwent a third ACLR. The mean body mass index (BMI) was 26.6 ± 5.1 kg/m2 in patients without revision ACLR failure and 26.3 ± 4.1 kg/m2 for those with revision ACLR failure (P = .769) (Table 1). For patients without revision ACLR failure, there were 65 (47.4%) with a normal BMI, 41 (29.9%) with an overweight BMI, and 31 (22.6%) with an obese BMI. For those with revision ACLR failure, there were 16 (43.2%) with a normal BMI, 16 (43.2%) with an overweight BMI, and 5 (13.5%) with an obese BMI. There were no significant differences in sex, age, and BMI between the patients with and without revision ACLR failure (Table 1). Sports were the most common mechanism for rupture and rerupture of grafts.
Patient Characteristics a

Data are presented as mean [SD] or n (%). ACLR, anterior cruciate ligament reconstruction; BMI, body mass index; MD, mean difference; NA, not applicable.
Revision ACLR Technique
For the primary revision ACLR without failure group, 67.9% of patients utilized an autograft compared with 48.6% of those experiencing revision ACLR failure (P = .101) (Table 2). There was no difference between using autograft or autograft with allograft augmentation in patients with and without a revision ACLR failure (P = .101 and .674, respectively). Autograft type was not statistically different between patients without primary revision ACLR failure and those with primary ACLR revision failure. The sagittal femoral tunnel angle was 37.6°± 11.1° for patients without revision ACLR failure compared with 41.8°± 11.5° for patients with revision ACLR failure (MD, 4.16; 95% CI, –0.36 to 8.67; P = .071). The graft fixation type and femoral tunnel drilling technique did not significantly differ between patients with and without a revision ACLR failure. A breakdown of revision indications, mechanism, and tunnel positioning can be found in the Appendix (Appendix Table A1.)
Operative Variables and Concomitant Injuries and Procedures a
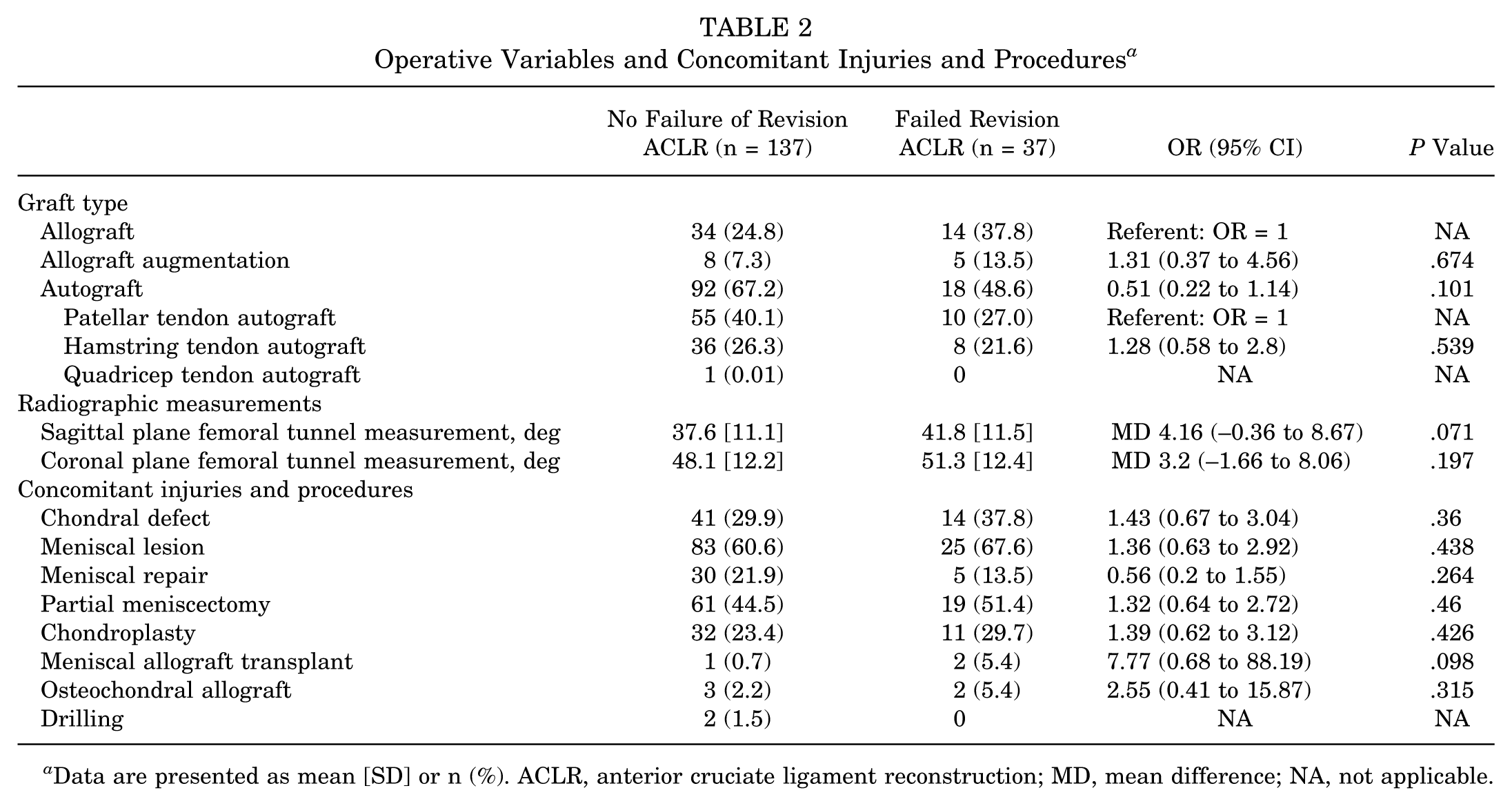
Data are presented as mean [SD] or n (%). ACLR, anterior cruciate ligament reconstruction; MD, mean difference; NA, not applicable.
Concomitant Injuries and Procedures
In patients without revision ACLR failure, 21.9% had a meniscal repair during the first ACLR revision, and in those experiencing revision ACLR failure, 13.5% had a meniscal repair (P = .264). There were no differences in those with meniscal lesions (60.6% vs 67.6%; P = .438) or partial meniscectomy (44.5% vs 51.4%; P = .46) between the 2 groups (Table 2). There were no significant differences in chondral injuries or chondral procedures between patients with and without revision ACLR failure. Of the patients who went on to have revision ACLR failure, 2.7% were due to high velocity trauma and 62.2% were due to sports injury.
Return to Sport
Regarding pre-revision activity levels, the majority of patients in both groups participated at the recreational level (68.6% vs 67.6%), with smaller proportions engaged in high school (13.1% vs 5.4%), college (10.9% vs 16.2%), and professional (2.2% vs 0) sports, and none of these distributions differed significantly between groups (all P > .05) (Table 3). Overall RTS rates were numerically higher in the no-failure group compared with the failure group (51.8% vs 40.5
Return to Sport a

Data are presented as mean [SD] or n (%). Boldface P value indicates a statistically significant difference between groups (P≤ .05). ACLR, anterior cruciate ligament reconstruction; NA, not applicable; RTS, return to sport.
Discussion
This study found no significant association between BMI and the risk of revision ACLR failure, with comparable BMI distributions between patients who experienced revision ACLR failure and those who did not. These findings contribute important insights to the limited existing literature on risk factors for re-revision ACLR.
The lack of association between BMI and re-revision risk contrasts with some previous findings but aligns with others in the literature. While Alm et al 1 found that patients with BMI >30 had 9 times the risk for re-revision ACLR, their study had a notably smaller sample size (n = 16). The findings presented here align more closely with those of Vap et al 26 and Yoon et al, 30 who found no significant relationship between BMI and re-revision risk in their respective studies. The larger sample size in the present study (n = 174) provides greater statistical power to evaluate this relationship, although the retrospective nature has inherent limitations.
The revision ACLR failure group had higher sagittal femoral tunnel angles compared with the no-failure group, although this was not statistically significant (41.8°± 11.5° vs 37.6°± 11.1°; P = .071). This aligns with previous literature emphasizing the critical importance of proper tunnel placement in revision ACLR.9,19,24 Recent biomechanical studies have provided new insights into optimal tunnel placement angles. Cheng et al 4 demonstrated that a femoral tunnel angle of 45° in both the coronal and sagittal planes provided the lowest peak stress and strain on both the bone tunnels and the ACL graft. Similarly, Zhang et al 31 found that angles between 43° and 45° resulted in optimal biomechanical properties. The patients who experienced revision failure had mean sagittal angles (42.2°) approaching this optimal range, while those without failure maintained significantly lower angles (37.3°). This may suggest that there is a range of tunnel angles that predispose patients to failure, and there are likely other more significant contributing factors. The relationship between overweight BMI and steeper sagittal femoral tunnel angles in failed revisions warrants further investigation and may reflect technical challenges in achieving optimal tunnel placement in patients with higher BMI, although the exact mechanical and surgical factors remain to be elucidated.
The RTS rates in this cohort were significantly different between groups, with 51.8% of the primary revision group returning to sport compared with 40.5% of the re-revision cohort (P = .038). Limited RTS rates in both revision groups have several contributing factors, including decreased subjective stability as well as early arthritis in the multiply injured knee. Griffith et al 12 and Engler et al, 7 have documented progressively worse functional outcomes and lower return to activity levels with multiple revisions. The lower RTS rates in the re-revision cohort could also be related to patient apprehension. In revision ACLR cohorts, fear and persistent symptoms are the leading self-reported reasons for not returning. 16 Additionally, higher-level athletes are more likely to RTS and recreation-level participation is associated with not returning after revision ACLR.2,18
Several limitations should be considered when interpreting these results. First, the retrospective nature of the study introduces potential selection and confounders. Thus, known factors associated with ACLR failure such as age, activity level, graft type, and RTS were examined. Posterior tibial slope was not examined in this cohort. Second, while the sample size is larger than those in previous studies examining re-revision risk factors,1,26,30 it may still be underpowered to detect subtle BMI-related effects. Third, the follow-up period was limited to 2 years, which may not capture all potential failures. Furthermore, this is a multisurgeon study, which may contribute to variability with regard to graft selection, surgical technique, and postoperative rehabilitation protocols, limiting the generalizability of the study findings. Factors such as hypermobility and notch width, stability as measured on the KT-1000 test, muscle strength, and specific sport played on return were also not collected in this study. All these factors could have played a role in graft failure.
The significant decline in RTS rates after re-revision emphasizes the importance of optimizing primary revision surgery and carefully counseling patients about realistic outcome expectations.3,5-7,12,29
Conclusion
The BMI values in patients who experience a successful revision ACLR and patients who experience a revision ACLR failure do not differ significantly. These results show no clear association between patient BMI and the risk of revision ACLR failure.
Footnotes
Appendix
Revision ACLR Indications, Mechanism of Injury, and Preoperative Tunnel Positioning a

| Primary Revision ACLR (n = 134) | Re-Revision ACLR (n = 44) | OR (95% CI) | P Value | |
|---|---|---|---|---|
| Indication for revision | ||||
| Traumatic | 76 (56.7) | 21 (47.7) | Referent: OR = 1 | NA |
| Surgical error | 40 (29.9) | 15 (34.1) | 1.36 (0.63 to 2.92) | .434 |
| Other | 15 (11.2) | 5 (11.4) | 1.21 (0.39 to 3.7) | .743 |
| Injury mechanism | ||||
| High energy (trauma) | 2 (1.5) | 1 (2.3) | Referent: OR = 1 | NA |
| Low energy (sports) | 97 (72.4) | 28 (63.6) | 0.58 (0.05 to 6.6) | .659 |
| Ultra-low energy (weight shift/trip) | 26 (19.4) | 10 (22.7) | 0.77 (0.06 to 9.45) | .838 |
| Preoperative tunnel positioning | ||||
| Femoral tunnel mispositioned | 61 (45.5) | 14 (31.8) | 0.68 (0.3 to 1.5) | .336 |
| Tibial tunnel mispositioned | 40 (29.9) | 10 (22.7) | 0.85 (0.36 to 2) | .709 |
Data are presented as mean [SD] or n (%). ACLR, anterior cruciate ligament reconstruction; NA, not applicable.
Final revision submitted October 1, 2025; accepted October 31, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: D.P.P. has received education payments from Arthrex and consulting fees from Zimmer Biomet. J.E.F. has received education payments from Peerless Surgical, nonconsulting fees from Smith & Nephew, and hospitality payments from Arthrex. B.M.S. has received consulting fees from Bioventus; hospitality payments from Arthrex; education payments from Peerless Surgical, Medwest Associates, Arthrex, and Smith & Nephew; and a grant from Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Wake Forest University School of Medicine IRB (IRB00103848).
