Abstract
Background:
Groin pain syndrome (GPS) is a complex pathology of the pubic joint resulting from excessive torque on the pubic symphysis and surrounding structures. Although broadly recognized as a sports-related injury, GPS also affects nonathletes due to physical demands common in occupational labor or routine physical activity. Surgical management has demonstrated favorable outcomes in athletes, but its role in the general population remains uncertain.
Purpose:
(1) To compare surgical and nonsurgical outcomes in athletes versus nonathletes with GPS and (2) to compare outcomes between different surgical techniques for GPS.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
Patients treated for GPS between December 2013 and August 2024 were included. All patients were diagnosed clinically by physical examination and treated using a standardized diagnostic-therapeutic algorithm. Complete symptom resolution and residual pain on physical examination were compared between athletes and nonathletes for surgical and nonsurgical treatment. All patients were followed until complete resolution or treatment completion and assessed for long-term complications.
Results:
A total of 250 patients with GPS were evaluated, including 75 athletes and 175 nonathletes, with a median symptom duration of 13 months. Overall, 202 patients (80.8%) were treated surgically, and 247 patients (98.8%) achieved either complete (n = 154, 61.6%) or partial (n = 93, 37.2%) resolution of symptoms. Athletes demonstrated lower odds of residual pain than nonathletes (odds ratio [OR], 0.276; 95% CI, 0.098-0.778; P = .015). Surgery demonstrated lower odds of residual pain than no surgery (OR, 0.248; 95% CI, 0.100-0.614; P = .003), which remained significant only in the nonathlete subgroup analysis (OR, 0.164; 95% CI, 0.058-0.464; P < .001). Furthermore, the effect of surgery on residual pain was significantly modified by athletic status (P = .034). Robotic transabdominal preperitoneal (TAPP) mesh repair demonstrated higher odds of complete resolution (OR, 5.413; 95% CI, 1.837-15.949; P = .002) and lower odds of residual pain (OR, 0.152; 95% CI, 0.053-0.439; P < .001) than no surgery. Combined mesh repair and adductor tenotomy demonstrated higher odds of complete resolution (OR, 8.435; 95% CI, 1.648-43.181; P = .010) than no surgery.
Conclusion:
Under a standardized diagnostic-therapeutic algorithm, both athletes and nonathletes with GPS demonstrated significant improvement. Surgery, particularly robotic TAPP mesh repair, significantly improved outcomes in nonathletes. These findings extend GPS management beyond the elite, young, male athlete.
Keywords
Groin pain syndrome (GPS), traditionally known as “sports hernia,” is a complex pathology of the pubic joint affecting both athletes and nonathletes.49,74,84 Most cases develop chronically from excessive torque on the pubic symphysis and surrounding structures, leading to weakness of the posterior wall of the inguinal canal at the level of the transversalis fascia, without evidence of a true hernia.20,48,49 There may also be concomitant adductor longus or rectus abdominis tendinopathies or conjoint tendon tears. 37 Diagnosing GPS is complicated by significant symptom overlap with hip, thigh, and other groin-related pathologies.4,6,7,20,21,35,66
Among athletes, GPS is common in sports involving sudden changes in direction, including soccer, hockey, and football.15,32,33,35,57,81,82 In the National Football League, the number of surgeries for GPS has increased significantly since 1995. 32 In soccer, groin pain represents up to 19% of injuries, with GPS accounting for 4% of all groin injuries in professional soccer from 2001 to 2016.50,51,67,70,74,80-82 However, the prevalence of GPS in nonathletes, including manual laborers and other active individuals, is unknown. Nonathletes may similarly experience this injury through repetitive lifting, carrying, bending, or twisting movements during daily occupational or routine activities.
GPS is a topic of growing interest among general surgeons, orthopaedic surgeons, sports medicine physicians, and radiologists, as it is increasingly recognized as a challenging diagnosis.20,71 Many nondescriptive terms have been used to describe this entity, including sports hernia, athletic pubalgia, core muscle injury, sportsman’s hernia, sportsman’s groin, hockey groin, and Gilmore’s groin, with athletic pubalgia and sports hernia being the most commonly used.30,40,51,58 However, experts in groin-related injuries recommend avoiding the term sports hernia not only because the term is nonanatomic and nondiagnostic but also because true hernias are rarely present in athletes.10,30,84,85 Instead, the terms inguinal-related groin pain and inguinal disruption, or GPS more broadly, are considered more accurate, as they are based on clearly defined clinical findings and anatomy.7,30,69,80,84 In this study, GPS was used as an umbrella term to describe the clinical entity in question because it is broad, inclusive, and descriptive.
There has been a significant effort to define this entity and develop a structured approach to its categorization.6,7,12,28,70,80 Yet there remains significant confusion related to its definition and management, primarily due to significant heterogeneity in the underlying pathoetiology.4,7,20,68 According to the Doha 2014 meeting of a wide range of specialists, GPS has 3 major categories: (1) defined clinical entities for groin pain, including (a) adductor-related, (b) iliopsoas-related, (c) inguinal-related, and (d) pubic-related; (2) hip-related groin pain; and (3) other causes. According to the 2014 Manchester consensus meeting of a group of hernia specialists, a diagnosis of the inguinal-related subtype, which involves weakness of the posterior wall of the inguinal canal at the level of the transversalis fascia and the conjoint tendon, can be made if at least 3 of the following 5 criteria are met:
Pinpoint tenderness over the pubic tubercle at the point of insertion of the conjoint tendon
Palpable tenderness over the deep inguinal ring
Pain and/or dilation of the external ring, with no obvious hernia evident
Pain at the origin of the adductor longus tendon
Dull, diffuse pain in the groin, often radiating to the perineum and inner thigh or across the midline 70
Furthermore, there are no accepted standards for history taking, physical examination, or imaging in diagnosis, with significant variability in the reliability of clinical tests and magnetic resonance imaging (MRI) findings.14,27,40,54 Thus, GPS remains a clinical diagnosis, requiring a multidisciplinary approach that incorporates a range of diagnostic techniques, including imaging, to help corroborate the diagnosis.5,8,20,39,62,72,75,84 An important indicator is activity-related pain in the inguinal region that is intensified during resisted abdominal testing or Valsalva maneuvers. 41 Current recommendations include performing thorough palpation of the abdomen and groin and incorporating scrotal invagination and resistance testing to provoke pain and aid in classification.27,43
Existing studies on GPS focus almost exclusively on young male athletes, a majority of whom are at the top level of their sport. 83 These patients likely have greater access to higher-quality and consistent rehabilitation programs, as well as a stronger motivation to return to baseline function. Furthermore, outcomes are often assessed via return to sport, with minimal use and validation of patient-reported outcome measures (PROMs).52,84 Thus, the existing literature is hardly applicable to the general population, which is exacerbated by the continued use of the terms sports hernia and athletic pubalgia.59,79 Few studies have assessed outcomes of GPS in the general population,31,48,52 and only 1 study has directly compared outcomes between athletes and nonathletes. 52 This seminal study on GPS by Muschaweck and Berger 52 demonstrated that athletes treated surgically returned to sport faster and at a higher rate than nonathletes. However, all participants received the same open, suture-only “minimal repair” technique, and the analysis did not adjust for covariates, making it difficult to draw firm conclusions regarding the true differences in outcomes between athletes and nonathletes. As clinical awareness of GPS increases, nonathletes, who may have a unique recovery profile due to differences in rehabilitation patterns, make up a growing percentage of referrals to surgery clinics.42,48 Nonathletes, especially those in physically demanding occupations, can be significantly affected by GPS in their daily lives without definitive management. This makes it critical to study this condition in the general population more broadly.
Growing evidence supports the need for surgery to improve outcomes for patients with GPS, with the goal of addressing the underlying defect involving the posterior wall of the inguinal canal, distal insertion of the rectus abdominis, proximal adductor tendon attachment, and/or the genitofemoral nerve.21,26,48,65 Several surgical techniques in the literature have demonstrated success in athletes, particularly in comparison to nonsurgical treatment.* However, it is unclear whether these findings are applicable to the general population, as most of these patients are elite, young, male athletes assessed via return to sport. Thus, the primary aim of this study was to compare surgical and nonsurgical outcomes between athletes and nonathletes with GPS. The secondary aim was to compare outcomes between different surgical techniques. The following were hypothesized:
Nonathletes would have worse outcomes than athletes.
Surgical treatment would be more effective than nonsurgical treatment in both athletes and nonathletes.
There would be no difference in outcomes between different surgical techniques.
Methods
This was a single-center, single-surgeon retrospective cohort study using a GPS data registry. Inclusion criteria were patients who received treatment for clinically diagnosed GPS between December 2013 and August 2024. The institutional review board approved this investigation under IRB#HHC-2024-0141. Reporting was conducted in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology guidelines, as well as the minimum reporting standard for clinical research on groin pain in athletes.12,77 The strengths of this single-center, single-surgeon study include high internal consistency, with standardized diagnosis and treatment both surgically and nonsurgically; standardized postoperative plans and rehabilitation protocols; and detailed clinical data available to the research team. The limitations most notably involve surgeon bias in treatment selection. While a detailed treatment algorithm outlining multiple nonsurgical interventions helps to mitigate a bias toward operative management, a study involving multiple surgeons could reduce the potential impact of unconscious treatment bias.
Data Collection
In the construction of the data registry, an electronic medical record report was run searching for diagnosis International Classification of Diseases, 10th Revision codes referring to right and left lower quadrant pain (R10.31 and R10.32, respectively), which yielded 1436 patients (Figure 1). Charts were reviewed individually for a GPS diagnosis, which does not yet exist as a searchable code. Sixty-two variables pertaining to clinical, demographic, imaging, pathologic, and procedural characteristics of patients were retrieved from the electronic medical record. Of 389 patients with confirmed GPS, 139 were excluded because they were lost to follow-up (n = 71), in progress (n = 48), denied by insurance (n = 12), or not treated (n = 8). This left 250 patients with confirmed GPS who were included for further analysis. As such, the final sample size was determined based on the patients remaining in the database who met the inclusion criteria. Of the 250 patients included in the analysis, 75 were classified as athletes and 175 as nonathletes. For the purposes of this study, “athlete” was defined as a patient currently and routinely participating in competitive athletic activity on a weekly basis as a livelihood or an integral way of life, as derived from Meyers et al. 48 Additionally, their GPS symptoms must have led to a significant decline in their ability to participate in their sport.

Flowchart of patient inclusion. Following chart review of 1436 patients based on International Classification of Diseases, 10th Revision diagnosis codes for right and left lower quadrant pain, 389 patients with confirmed groin pain syndrome were assessed for inclusion. Of these, 139 were excluded because they were lost to follow-up (n = 71), in progress (n = 48), denied by insurance (n = 12), or not treated (n = 8). This left 175 nonathletes and 75 athletes for the final study cohort.
Treatment Algorithm
Diagnosis and Classification
All patients were evaluated and treated by a single surgeon (J.A.B.) using a standardized diagnostic-therapeutic algorithm throughout the duration of the study. Our universal management algorithm for GPS begins with a systematic history and physical examination, which includes sit-up sign (SUS), straight leg raise (SLR), clamshell, figure 4, and hip flexor test, assessing for positive hip flexor, adductor, testicular, inguinal floor, pubic symphysis, and/or pubic tubercle pain (Figure 2). While GPS is a clinical diagnosis, our practice routinely obtains a noncontrast MRI of the pelvis to corroborate the diagnosis and guide surgical decision-making, as MRI has been demonstrated to improve diagnostic post-test probability (Figure 3). 17

Universal diagnostic-therapeutic algorithm for groin pain syndrome (GPS). With the exception of the posterior wall deficiency subtype, which is treated operatively right away, the GPS management algorithm follows a conservative approach with corticosteroid injections followed by physical therapy if symptoms persist. The adductor-dominant subtype of GPS commands additional management strategies, including platelet-rich plasma injections and adductor tenotomy prior to surgical mesh repair. Patients follow a standardized follow-up protocol of 4 weeks for injection-based treatment and 2 and 6 weeks for surgical treatment. Patients receiving steroid injections who demonstrate improvement within 4 weeks are referred to physical therapy for 6 weeks at a cadence of 2 to 3 visits per week, with a follow-up visit at 4 weeks to elicit progress. Patients receiving surgery who demonstrate improvement at 2 weeks are referred to physical therapy. Adductor-dominant subtype corresponds to adductor-related groin pain as described within the Doha agreement’s “defined clinical entities for groin pain,” and the posterior-wall deficiency subtype corresponds to both the pubic-related and inguinal-related groin pain entities. 80
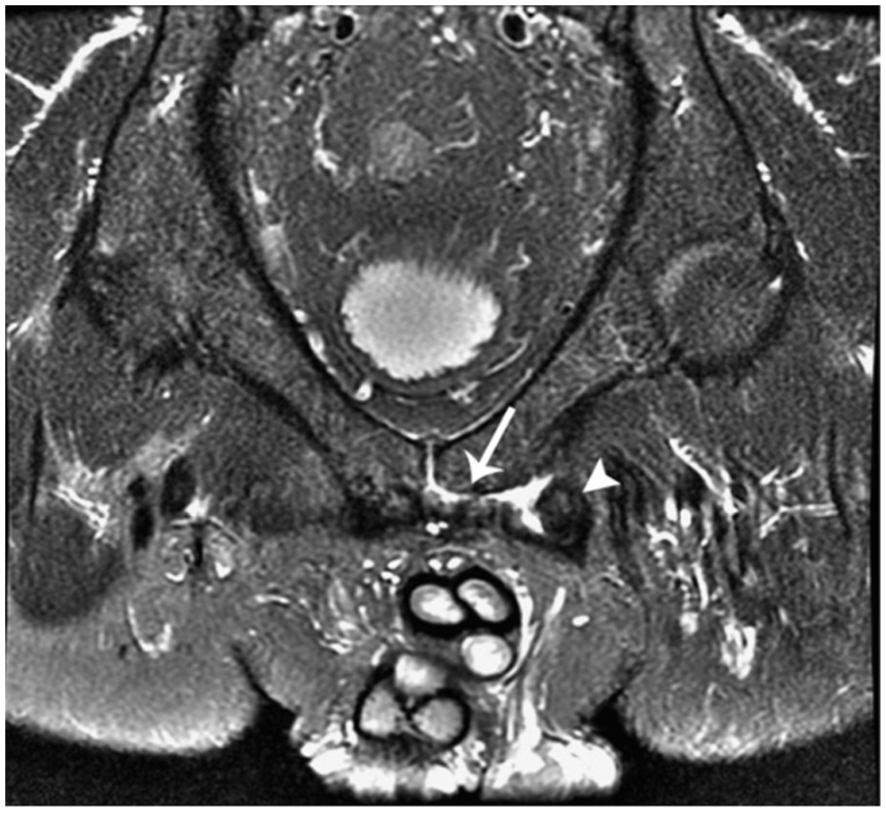
A positive coronal proton-density (PD) magnetic resonance imaging (MRI) demonstrating the secondary cleft sign (adductor brevis attachment, white arrow) with associated edema of the adductor muscle (white arrowhead). This MRI is consistent with the adductor-dominant subtype. The MRI protocol includes noncontrast T1, T2 fast spin echo (FSE), and PD imaging techniques with axial, sagittal, and oblique views. Positive MRI findings in groin pain syndrome include marrow edema, osteitis pubis, the superior/secondary cleft sign, and edema of the adductor or rectus aponeurosis. 13 Confirmation of the adductor-dominant injury pattern with the presence or absence of concomitant suprainguinal physical examination findings helps guide clinical decision-making per the treatment algorithm.
Following diagnosis, patients are classified into 1 of 3 GPS subtypes, including posterior wall deficiency, adductor-dominant, and combined type, using a combination of history and physical examination, which is derived from a prior methodology described in the literature.34,80 Posterior wall deficient subtype was defined as any suprainguinal complaints combined with positive suprainguinal pain with SLR or SUS. Adductor-dominant type was defined as any infrainguinal complaints combined with positive infrainguinal pain during figure 4, adductor squeeze, or clamshell. Combined type was defined as both supra- and infrainguinal complaints combined with positive supra- and infrainguinal pain during any of the physical examination maneuvers. MRI results are used to corroborate the clinical diagnosis and classification in positive cases based on the anatomic location and intensity of a given signal. Within this framework, the adductor-dominant subtype corresponds to adductor-related groin pain as proposed by the Doha agreement, and the posterior-wall deficiency subtype encompasses both pubic-related and inguinal-related groin pain, as proposed by the Doha agreement. 80 While the Doha agreement also describes the subtype of iliopsoas-related groin pain, our diagnostic criteria do not isolate this subtype, as patients with identifiable iliopsoas-related pathology are classified as having an “alternative diagnosis” and managed accordingly. This methodology is consistent with prior studies that incorporate the Doha classification system. 26

Robotic transabdominal preperitoneal mesh repair of right-sided groin pain syndrome of the posterior wall deficiency subtype in a 17-year-old male soccer player. Note the translucent, hypermobile transversalis fascia within the direct space (Hesselbach’s triangle), as well as the retracted musculotendinous portion of the distal rectus abdominis.
Nonsurgical Management
Nonsurgical treatment, which is initiated immediately following diagnosis, includes a combination of physical therapy (PT), corticosteroid injections, and/or platelet-rich plasma (PRP) injections, depending on the GPS subtype. Patients with combined or adductor-dominant subtypes receive 1 ultrasound-guided corticosteroid injection into the adductor longus tendon origin with a 4-week rest period, followed by PT if symptoms improve. Timing of PT after injection is resolution-dependent but typically begins at 4 weeks postinjection. If symptoms do not improve, a second injection is offered. Patients may receive a maximum of 2 corticosteroid injections before advancing through the treatment algorithm. The prescribed PT program is 6 weeks long at a cadence of 2 to 3 sessions per week, with an emphasis on improving postural stability, core strength, and adductor flexibility. Intensity is gradually increased in parallel to symptom improvement. All patients are followed up at 4 weeks to assess progress. Patients with persistent symptoms despite 2 corticosteroid injections and PT are offered ultrasound-guided PRP injection into the adductor tendon origin, which is followed by 4 weeks of strict rest without PT.
Surgical Management
When nonsurgical options fail, surgical repair of the underlying anatomic defect is considered. Surgical management is indicated when the patient has received a minimum of 1 corticosteroid injection (maximum of 2), followed by 4 weeks of rest without improvement. For the posterior wall deficiency subtype, however, surgery is recommended immediately once careful examination has excluded alternative causes of groin pain. 80 This is because tearing of the direct (Hesselbach) space musculature, composed of the medial fibers of the internal oblique aponeurosis and deeper fibers of the transversus abdominis aponeurosis fusing as the conjoint tendon, indicates a need for surgical reinforcement, as this musculotendinous injury will not adequately heal on its own. 52 Imaging findings are sometimes used to support surgical decision-making. 54 This is particularly useful for patients with adductor-dominant symptomatology, as osteitis pubis on MRI lowers our threshold for surgical treatment in these patients. The surgical approach depends on the underlying anatomic defect and GPS classification and may include a robotic transabdominal preperitoneal (TAPP) mesh repair, laparoscopic totally extraperitoneal (TEP) mesh repair, combined mesh repair and adductor tenotomy, or standalone adductor tenotomy. A standalone adductor tenotomy is indicated for patients with only adductor (infrainguinal) symptoms, whereas the combined mesh repair and adductor tenotomy is indicated for patients with combined suprainguinal and infranguinal symptoms. For the mesh repair, the decision to use a robotic TAPP versus a laparoscopic TEP approach is based primarily on logistical factors, including facility availability of the robot platform, operating room scheduling, and geographic proximity to the patient’s residence. For the purposes of this study, there were no distinct clinical indications favoring one approach over the other, as both techniques are considered equivalent and are routinely used in the surgical management of GPS. 37
The postoperative recovery protocol, which is nearly identical to the nonsurgical treatment algorithm, involves an initial 2-week rest period with no PT, exercise, or lifting more than 10 pounds, with the first follow-up appointment occurring at 2 weeks postoperatively. For nonathletes without access to an athletic training program who have demonstrated improvement of symptoms, a standardized handout with recommended at-home rehabilitation exercises is provided. The postoperative PT protocol is identical to that of nonsurgical treatment, with 6 weeks of progressive core and adductor strengthening, stretching, and postural stability exercises. For athletes on a sports team with access to an athletic training program, the training staff takes over with the same PT protocol provided to nonathletes. All patients are seen for follow-up at 6 weeks postoperatively. For patients with persistent symptoms at 2 weeks postoperatively, the rest period is extended an additional 2 weeks, after which they are referred to PT. Corticosteroid and PRP injections are offered to patients with refractory adductor symptoms following surgical repair.
Surgical Technique
Robotic TAPP Mesh Repair
Following standard docking of the Da Vinci robot (Intuitive Surgical), a peritoneal flap is created, exposing the inguinal floor from the linea alba to the lateral aspect of the internal ring. The entire myopectineal orifice is exposed, taking care to eliminate preperitoneal fat from Hesselbach’s triangle (Figure 4). A Bard 3D Max (Becton Dickinson) medium standard-weight mesh is positioned to overlap the pubic symphysis medially and cover the internal ring laterally, secured at the rectus muscle superomedially, Cooper’s ligament inferiorly, and the transversalis fascia laterally. The peritoneal flap is closed with a running 2-0 V-Lok suture.
Laparoscopic TEP Mesh Repair
A periumbilical incision is used to access the inguinal space on the deep surface of the posterior rectus sheath, through which a dissecting balloon is deployed and subsequently replaced by a structural balloon and two 5-mm trocars placed infraumbilical. Dissection, mesh selection, and placement are identical to the TAPP mesh repair from this point forward, although optimal fat dissection using this technique is comparatively limited.
Adductor Tenotomy
The patient is placed in a bilateral figure four position to expose the adductor tendon origin. A curvilinear incision at the groin crease over the palpable tendon is made and carried down to the tendon sheath. The tendon sheath is opened longitudinally, and the tendon origin is dissected bluntly with right-angle forceps 1.5 cm off the pubis and divided with cautery.
Outcome Measures
Two primary outcome measures were operationalized to assess treatment outcomes, including (1) complete resolution of all symptoms and physical examination findings (“complete resolution”) and (2) positive findings on any physical examination maneuver at the final follow-up visit (“residual pain”). This dual approach was implemented with the understanding that patients with chronic GPS may continue to experience symptoms despite showing no signs on physical examination. Thus, complete resolution of symptoms was defined as the absence of self-reported symptoms during the final follow-up visit combined with the absence of positive findings on physical examination. Residual pain was defined as having at least 1 positive physical examination finding at the final follow-up visit and is therefore independent of the patient’s historical report of pain in other contexts. To minimize time-related confounding and estimate treatment-proximal effects, outcomes were censored at the time of complete resolution or at the completion of the standardized management algorithm. Time-based outcome measures (ie, time to resolution/return to sport) were not used but were adjusted for in the multivariable analysis.
Due to the stringency of the primary outcome measures, complete or partial resolution of symptoms was also assessed as a secondary outcome measure to provide additional insight into the broader efficacy of the management algorithm. In this case, complete or partial resolution was defined as the absence of at least 1 physical examination finding that was present at the initial evaluation visit.
In addition to physical examination–based outcome measures, the Numeric Rating Scale (NRS) was also used, although only 175 of 250 (70.0%) of included patients had complete data for this outcome measure. The minimal clinically important difference (MCID) for the change in NRS from the initial evaluation to the final follow-up visit was assessed according to guidelines proposed by Farrar et al 18 for chronic pain syndromes, which use a raw change of −1.74 as being clinically important. All included patients were also evaluated for long-term complications, including deep vein thrombosis, hematoma, neuropathy, adductor tendon reattachment, and reinjury.
Statistical Analysis
Data processing and preliminary analyses were performed in Microsoft Excel. Advanced statistical analyses were performed using IBM SPSS Statistics Version 31.0.0.0. Categorical variables were analyzed using contingency tables, and statistical significance was assessed with χ2 tests. When ≥20% of cells had a count of <5, the Fisher exact test was applied. Continuous variables were assessed for normality using the Shapiro-Wilk test. Parametric data were analyzed using the independent samples t test, while nonparametric data were analyzed using the Mann-Whitney U test. Statistical significance was set at an α of <0.05.
Odds ratios (ORs) with 95% confidence intervals were calculated using binary multivariable logistic regression. The covariates included in the model along with athletic status and surgery were time from initial evaluation to completion of the treatment algorithm, age, mechanism of injury, pain location, PRP injection, cortisone injection, PT, and positive imaging findings for GPS. Time spent in the treatment algorithm was included to account for temporal differences in the timing of treatment due to variations in treatment response times, as well as scheduling or insurance difficulties; age was included because nonathletes were significantly older than athletes; mechanism of injury (MOI) was included because it differed significantly between groups; pain location was included to account for anatomic differences in injury sites (ie, adductor longus origin vs rectus abdominis origin); the rates of cortisone and PRP injections differed significantly between groups and were therefore included; PT was also included despite not differing significantly between groups to control for presumed differences in rehabilitation patterns between athletes and nonathletes. Interaction effects between athlete and surgery were tested by including a multiplicative interaction term in the regression model and assessing for significance. Subgroups including only athletes or only nonathletes were analyzed using the same model. Potential overfitting was addressed individually for each outcome variable by maintaining an events-per-variable ratio of ≥5 and variance inflation factors of ≤5. Nonparametric bootstrap resampling (1000 samples with replacement) was performed to assess coefficient stability in each model (athlete vs nonathlete and surgical technique). E-values were also calculated to assess the minimum strength of association that an unmeasured confounder would need with both exposure and outcome to explain away observed associations. 76
Patients were included in the study only if complete data were available for the variables in the model. Cases with substantial missing data were classified as “in progress” (n = 48) and excluded from the final analysis. The only variables with missing data included race and ethnicity data, which were missing for 10 patients in total, and NRS, which was missing for 75 patients, but these variables were not included in the regression model. Multiple imputation was not used. Finally, no formal sample size calculation was performed because all available patient records that met the predefined inclusion/exclusion criteria were included in the analysis.
Results
Patient Characteristics
A total of 250 patients with GPS were evaluated, including 75 (30%) athletes and 175 (70%) nonathletes (Table 1). Most patients were male (n = 219, 87.6%), with a mean age of 42 ± 15 years and a mean body mass index of 27.8 ± 5.8 kg/m2. Among the athletes, the most common sports played were soccer (n = 17, 22.7%), hockey (n = 12, 16.0%), and football (n = 11, 13.3%). Overall, 50 patients (20.0%) were injured specifically at work, primarily in occupations that require heavy lifting and twisting. A total of 48 (19.2%) patients had a history of inguinal hernia, with a higher proportion in nonathletes (25.1% vs 5.3%, P < .001). In total, 38 (15.4%) patients were referred to the practice from orthopaedics, 14 (5.7%) from urology, 5 (2.0%) from gastroenterology, 3 (1.2%) from general surgery, 2 (0.8%) from gynecology, and 9 (3.6%) from multiple specialists. The remaining 178 patients (71.3%) presented directly to the clinic.
Univariable Analysis of Demographics, Treatments, and Outcomes Based on Athletic Status in Patients With Groin Pain Syndrome a

Data are presented as number (%) unless otherwise indicated. Categorical variables were analyzed using contingency tables, and statistical significance was assessed with χ2 tests. When ≥20% of cells had a count of <5, the Fisher exact test was applied. Continuous variables were assessed for normality using the Shapiro-Wilk test. Parametric data were analyzed using the independent samples t test, while nonparametric data were analyzed using the Mann-Whitney U test. Statistical significance was set at an α of 0.05, with significant P values depicted in bold. Complete resolution is defined as the absence of self-reported symptoms during the final follow-up visit, combined with the absence of positive findings on physical examination. Residual pain is defined as having at least 1 positive physical examination finding at the final follow-up visit. Complete or partial resolution is defined as the absence of at least 1 physical examination finding that was initially present at the first pretreatment visit. The MCID for the change in NRS from the initial evaluation to the final follow-up visit was assessed according to guidelines proposed by Farrar et al 18 for chronic pain syndromes (raw change of −1.74). BMI, body mass index; DVT, deep vein thrombosis; GI, gastrointestinal; GPS, groin pain syndrome; MCID, minimal clinically important difference; NRS, Numeric Rating Scale; OB/GYN, obstetrics/gynecology; PRP, platelet-rich plasma; TAPP, transabdominal preperitoneal; TEP, totally extraperitoneal.
Clinical Presentation
Overall, 137 patients (54.6%) had suprainguinal pain, and 40 patients (16.1%) had infrainguinal pain. Pain location distribution differed significantly between athletes and nonathletes (P = .010), as athletes more often presented with suprainguinal pain, whereas nonathletes more often presented with infrainguinal pain. In total, 35 patients (14.1%) had both supra- and infrainguinal pain, and 38 patients (15.3%) had pain directly at the inguinal crease. All patients were categorized into 1 of 3 GPS subtypes, including posterior wall deficient (n = 99, 39.6%), adductor-dominant (n = 50, 20.0%), and combined type (n = 101, 40.4%). At the initial physical examination evaluation, 170 (68.0%) patients had a positive SLR, and 167 (66.8%) had a positive SUS. There were 167 (66.8%) patients with positive adductor pain, 74 (29.6%) patients with positive testicular pain, and 33 (13.2%) patients with positive pubic symphysis pain. Nonathletes had a significantly higher rate of testicular pain (34.3% vs 18.7%, P= .013) and pubic symphysis pain (16.0% vs 6.7%, P = .046). The mean initial NRS was 4.83 ± 1.66 for the overall cohort (out of the n = 179 with initial NRS data) and did not differ significantly between groups (P = .613). A total of 49 (19.6%) patients had bilateral GPS.
Imaging
All included patients received a noncontrast MRI; 127 patients (50.8%) had positive GPS findings, with significantly greater positivity in athletes (70.7% vs 42.3%, P < .001). The median imaging delay was 6 months, with no significant difference between athletes and nonathletes. The main GPS-related imaging findings described in radiology reports included osteitis pubis, pubic symphysis bone marrow edema, pubic symphysis irregularity, subchondral cystic changes of the pubic symphysis, pubic symphysis degenerative arthrosis, rectus abdominis-adductor aponeurotic plate edema, partial-thickness tear of the rectus abdominis aponeurosis, secondary cleft formation, adductor aponeurosis tear, adductor origin edema, and adductor origin abnormality.
Treatment
The median time from initial evaluation to completion of the treatment algorithm was 5 months, and the median time from initial treatment to completion of the treatment algorithm was 3 months, with no significant differences between groups. The median duration of symptoms was 13 months, with a significantly longer duration in nonathletes (11 vs 15, P = .016). The median evaluation delay was 6 months, with no significant difference between groups. Overall, 202 patients (80.8%) had surgery. The most common surgical technique was a robotic TAPP mesh repair (n = 100, 40.0%), followed by laparoscopic TEP mesh repair (n = 68, 27.2%), combined mesh repair and adductor tenotomy (n = 20, 8.0%), and standalone adductor tenotomy (n = 14, 5.6%). In total, 100 patients (40.0%) had a cortisone injection, and 44 patients (17.6%) had a PRP injection, with a significantly higher rate in nonathletes for both (P= .024, P = .025). Most patients (n = 201, 80.4%) had at least 1 session of PT, with no significant difference between groups. Compliance with prescribed rehabilitation, which was assessed qualitatively at each scheduled follow-up visit based on patient self-report and clinician documentation of adherence to the prescribed PT protocol, was uniformly high across the cohort. All patients were documented as being at least moderately compliant with their postoperative or nonsurgical rehabilitation regimen, and no patients were noted to have poor adherence during follow-up.
Outcomes
Overall, 154 patients (61.6%) achieved complete resolution of symptoms, with a significantly lower proportion in nonathletes (54.9% vs 77.3%, P < .001). In total, 85 patients (34.0%) had residual pain on physical examination at their final follow-up visit after completion of the treatment algorithm, with a significantly higher proportion in nonathletes (40.6% vs 18.7%, P < .001). Nearly all patients (247, 98.8%) achieved either complete (n = 154, 61.6%) or partial (n = 93, 37.2%) resolution of symptoms following completion of the treatment algorithm, with no significant difference between nonathletes and athletes (98.9% vs 98.7%, P= .899). Three patients (1.2%) had no improvement following treatment (1 athlete, 2 nonathletes). The mean NRS at the final follow-up visit for the overall cohort (out of the n = 175 with final NRS data) was 0.91 ± 1.58, with a significantly lower mean in athletes (0.54 ± 1.22 vs 1.11 ± 1.71, P = .021). Nearly all patients with available data achieved the MCID in NRS for chronic pain (165/175), with no significant difference between athletes and nonathletes (98.2% vs 92.5%, P = .250). The most common persistent finding on physical examination at the final follow-up visit was adductor pain (n = 58, 23.6%), with a significantly higher proportion in nonathletes (28.1% vs 13.3%, P = .012).
In the multivariable analysis, there was no significant effect of surgery or athletic status on complete resolution after adjustment for patient characteristics. However, patients treated surgically had a significantly lower odds of residual pain on physical examination than those treated nonsurgically (OR, 0.248; 95% CI, 0.100-0.614; P = .003). Athletes also had a significantly lower odds of residual pain than nonathletes (OR, 0.276; 95% CI, 0.098-0.778; P = .015). Furthermore, the effect of surgery on residual pain was significantly modified by athletic status (P = .034). In the subgroup analyses, only in the nonathlete group did surgical treatment demonstrate significantly lower odds of residual pain than nonsurgical treatment (OR, 0.164; 95% CI, 0.058-0.464; P < .001), with no significant effect in athletes (OR, 1.703; 95% CI, 0.047-61.993; P= .772) (Table 2). There was no significant difference in outcomes between the 3 GPS subtypes. Internal validation with bootstrap resampling demonstrated that the coefficients were stable in this model, with minimal bias and comparable significance levels across resamples.
Multivariable Analysis of Outcomes for Groin Pain Syndrome Based on Athletic Status and Surgical Versus Nonsurgical Treatment a

Complete resolution is defined as the absence of self-reported symptoms during the final follow-up visit combined with the absence of positive findings on physical examination. Residual pain is defined as having at least 1 positive physical examination finding at the final follow-up visit. Covariates included athletic status, surgical/nonsurgical treatment, time from initial evaluation to completion of treatment algorithm, age, mechanism of injury, pain location, platelet-rich plasma injection, cortisone injection, physical therapy, and positive imaging findings for groin pain syndrome. For OR <1, the inverse OR was used before applying the E-value formula. OR, odds ratio. Statistical significance was set at an α of 0.05, with significant P values depicted in bold.
Across the different surgical techniques, robotic TAPP mesh repair demonstrated significantly higher odds of complete resolution (OR, 5.413; 95% CI, 1.837-15.949; P = .002) and lower odds of residual pain (OR, 0.152; 95% CI, 0.053-0.439; P < .001) compared to nonsurgical treatment in the overall study cohort (Table 3). A combined mesh repair and adductor tenotomy also demonstrated higher odds of complete resolution than nonsurgical treatment (OR, 8.435, 95% CI, 1.648-43.181; P = .010). A laparoscopic TEP mesh repair and a stand-alone adductor tenotomy both failed to achieve significance for these outcomes. In the subgroup analyses, the nonathlete group demonstrated that a robotic TAPP mesh repair was associated with lower odds of residual pain than nonsurgical treatment (OR, 0.127; 95% CI, 0.040-0.401; P < .001). No other subgroup analyses demonstrated significant differences in outcomes between surgical techniques. Internal validation with bootstrap resampling demonstrated that the coefficients were stable in this model, with minimal bias and comparable significance levels across resamples.
Multivariable Analysis of Outcomes for Groin Pain Syndrome in Athletes and Nonathletes Based on Surgical Technique a
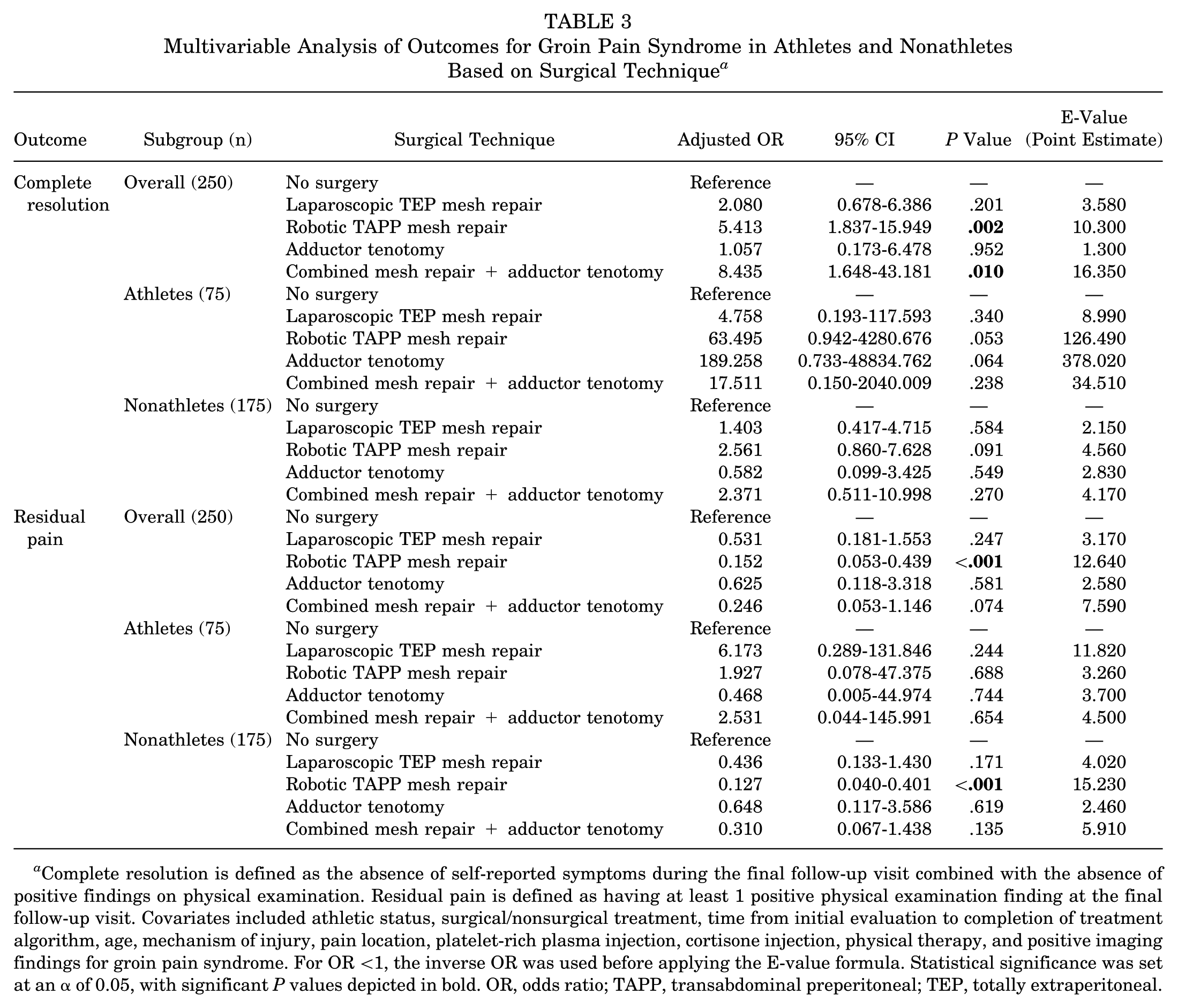
Complete resolution is defined as the absence of self-reported symptoms during the final follow-up visit combined with the absence of positive findings on physical examination. Residual pain is defined as having at least 1 positive physical examination finding at the final follow-up visit. Covariates included athletic status, surgical/nonsurgical treatment, time from initial evaluation to completion of treatment algorithm, age, mechanism of injury, pain location, platelet-rich plasma injection, cortisone injection, physical therapy, and positive imaging findings for groin pain syndrome. For OR <1, the inverse OR was used before applying the E-value formula. Statistical significance was set at an α of 0.05, with significant P values depicted in bold. OR, odds ratio; TAPP, transabdominal preperitoneal; TEP, totally extraperitoneal.
Complications
In the long-term follow-up at an average of 5 ± 1 years, there was 1 deep vein thrombosis, 2 hematomas, 16 neuropathies, and 1 tendon reattachment. Seventeen patients (6.9%) who had either complete or partial resolution of symptoms had a reinjury following the completion of treatment. No significant differences were observed in complication rates between athletes and nonathletes, or between surgical and nonsurgical treatment.
Discussion
The most important findings of this study are as follows: nonathletes with GPS more frequently had residual pain than athletes, surgery reduced the rate of residual pain in nonathletes, and, overall, 98.8% of patients treated via the standardized diagnostic-therapeutic algorithm achieved either complete or partial resolution of symptoms. By characterizing GPS in the nonathlete population in comparison to athletes, these findings challenge the prevailing focus on athletes in GPS research. They also highlight the critical need for broader recognition of surgical treatment as an effective option for the general population. A more inclusive understanding of GPS is needed.
The observed proportions of complete resolution (61.6%) and residual pain (34.0%) should be interpreted in light of the stringency of these binary endpoints. Complete resolution was defined as global recovery across all symptoms and provocative physical examination maneuvers, and residual pain was defined as the persistence of pain with any single maneuver. In contrast, the complete or partial resolution endpoint captured objective improvement in nearly all patients in the study cohort across both athletes and nonathletes (98.8%), and 94.3% of patients achieved the MCID in chronic pain for NRS. These findings strongly support the overall efficacy of the diagnostic-therapeutic algorithm in providing immediate structural and functional benefit for patients with GPS.
Pathoanatomy of Groin Pain Syndrome
While several underlying mechanisms for GPS have been described, a predominant theory is related to strain transmission due to excessive shear forces across the anterior pubic symphysis from the strong adductors against the contralateral distal rectus sheath.2,3,11,14,51,53 Recent cadaveric studies describe a pyramidalis-anterior pubic ligament-adductor longus complex that may explain the structural and functional integration of these tissues.44,60 However, there is likely no single underlying pathology for GPS, which would explain why patients frequently have inconsistent pain patterns and often require multiple office visits and evaluations before a definitive diagnosis can be made.7,44,53,63,78 The current study emphasizes the role of surgical management of GPS as restoring the structural integrity of the inguinal floor. This approach was described in 2010 by Dr Ulrike Muschaweck with a “minimal repair,” a nonmesh reinforcement of the inguinal floor with excellent results in both athletes and nonathletes. 52 Progress with minimally invasive surgery techniques has led to the development of a mesh repair performed in the same manner with similarly positive results, as seen in the present study. The utility of this approach may also extend beyond the posterior wall deficiency subtype, as patients with adductor-dominant symptoms may also benefit from mesh reinforcement. While an adductor tenotomy alone appears to be effective at treating symptoms for this subtype, the use of this technique may just be a consequence of uncertainty regarding the underlying anatomy.24,26 This is supported by findings suggesting that surgery for inguinal-related groin pain is more effective than surgery for adductor-related groin pain, and a combined surgery of the adductor and inguinal regions is more effective than surgery of the adductor region alone. 26 Thus, the role of other surgical techniques, such as a mesh repair or an open adductor to rectus abdominis turn-up flap, in addressing adductor involvement needs to be further explored.55,59
Surgical Techniques for Groin Pain Syndrome
The current study demonstrated that, overall, a robotic TAPP mesh repair yields better outcomes compared to a laparoscopic TEP mesh repair. This is most likely explained by the TAPP approach enabling a more extensive dissection of preperitoneal fat off the inguinal floor, particularly at Hesselbach’s triangle, thereby resulting in a more secure mesh fixation. For a true inguinal hernia repair in nonathletes, TEP and TAPP approaches are considered equivalent in terms of recurrence rates. For GPS, however, there is no definitive evidence favoring one technique over the other. A meta-analysis by Kler et al 37 found no significant difference between TEP and TAPP repair for GPS. However, the meta-analysis was limited to athletes only. Thus, prospective studies including both athletes and nonathletes are needed to provide definitive evidence on the comparative efficacy of these similar surgical approaches for GPS in the general population.
There is also evidence suggesting that adding an adductor tenotomy to a mesh repair may enhance functional outcomes for athletes, which is supported by the current study’s findings.19,25,34,37 A systematic review by Hatem et al 26 found that, in athletes with GPS, outcomes were better following surgery for inguinal-related symptoms compared to surgery for adductor-related symptoms, and a combined surgery of the adductor and inguinal regions was also more effective than the adductor region alone. Thus, while a standalone adductor tenotomy may not directly address the underlying structural defect, its addition to a mesh repair may mitigate persistent pain due to adductor-related pathology and improve symptom resolution. This is particularly important because adductor pain was the most common residual physical examination finding in our cohort for both athletes and nonathletes. Thus, further investigation into the surgical management of adductor-related symptoms is warranted, specifically regarding the utilization of the adductor tenotomy. However, restoring the structural integrity of the inguinal floor remains critical and should therefore represent the primary goal of surgical treatment in both athletes and nonathletes at this time.
Nonsurgical Management of Groin Pain Syndrome
When comparing surgical to nonsurgical treatment outcomes, the literature is not only equivocal but also of low methodological quality.9,29,36,65,73 Some studies suggest that structured active rehabilitation programs yield significant improvements in pain reduction and return to sport, sometimes even more so than surgical treatment, while others suggest that surgical treatment yields earlier return to sport.1,9,36,65,73 In the current study, only the nonathlete subgroup analysis demonstrated a significantly greater benefit from surgical treatment compared to nonsurgical treatment. While our athlete cohort was relatively smaller, this finding is corroborated by the significant interaction effects between the athlete and surgery variables. Thus, while surgery is broadly effective for minimizing residual pain, nonsurgical treatment—specifically active rehabilitation—plays a critical role in the management of GPS, particularly in athletes. This may be caused by existing differences in rehabilitation adherence, access to high-quality rehabilitation resources, and overall conditioning levels between these populations.
A randomized trial by Hölmich et al 29 found that nonsurgical management with an active rehabilitation program may be especially effective for the adductor-dominant subtype in athletes, with specific exercises aimed at improving postural stability and the strength and coordination of the muscles acting on the pelvis. This is particularly important because adductor-related groin pain appears to be the most prevalent GPS subtype in the athlete population, particularly for sports that involve kicking and changes of direction. 72 However, in a systematic review, Serafim et al 65 found that individuals undergoing surgery for GPS may return to sport earlier than those receiving nonsurgical treatment. This is further complicated by the finding that surgery of the adductor origin alone appears to be less effective than surgery to the inguinal or pubic areas, as well as combined adductor and inguinal areas, in athletes with GPS. 26 Overall, while active rehabilitation is highly effective, surgery may also be immediately indicated for GPS, particularly a mesh repair of the inguinal floor for patients with a deficiency of the posterior wall. This may be particularly applicable to nonathletes, for whom a structured active rehabilitation program may not be equally accessible or beneficial due to social factors such as employment demands, out-of-pocket treatment cost, and transportation.
Future work on GPS should aim to clearly define the role of specific nonsurgical treatment protocols within a multidisciplinary approach by integrating surgical and nonsurgical options into a unified treatment algorithm based on the GPS subtype. Furthermore, a better understanding of the role of various active rehabilitation programs, techniques, and exercises in nonathletes is needed. Studies would benefit from including nonathletes to ensure that findings are broadly applicable and not limited to an athlete context when nonathletes are similarly affected by this condition.
Limitations
The authors appreciate the limitations of this study, beginning with its retrospective design and single-surgeon experience, which invites the possibility of unconscious treatment bias. However, this concern is alleviated bythe transparent presentation of a standardized diagnostic-therapeutic algorithm. Nevertheless, we recognize that there may also be other possible management algorithms. Second, while this represents one of the largest GPS cohorts reported to date, particularly in nonathletes, the sample size may have limited the statistical power for comparisons between surgical and nonsurgical treatment for both outcome measures. Subgroup analyses may have been particularly limited, especially to detect small differences between surgical and nonsurgical treatment outcomes in athletes. Third, the definition of “athlete” is difficult to define objectively and is a recognized limitation in the literature. The definition used in this study was derived from the literature and incorporated the individual’s subjective experience as well as quantitative elements (ie, weekly participation) to improve reproducibility. Fourth, while cortisone and PRP injections were adjusted for as binary variables, 20% of patients also received these treatments postoperatively for refractory symptoms, which may introduce temporal bias and cumulative treatment effects that were not directly accounted for in the model. Fifth, while complications were not the primary outcome of this study, the assessment of long-term complications was performed via retrospective chart review rather than through a routine follow-up visit. This was due to the retrospective design of the study and lack of a preestablished protocol mandating follow-up visits after symptom resolution or treatment course completion. Thus, there may have been additional patients unaccounted for who experienced complications, reinjury, or relapse but did not present to the clinic or their primary care provider. Sixth, the absence of workers’ compensation status as a covariate represents a potential limitation, as patients involved in these claims tend to report poorer pain and functional outcomes. 61 This may have disproportionately affected the nonathlete subgroup, potentially underestimating their degree of improvement. Seventh, besides the NRS, additional validated PROMs, such as Copenhagen Hip and Groin Outcome Score, were not reported in this study. Additionally, only 70% of patients had complete NRS data, and the NRS remains without a validated MCID for GPS, specifically. While this may limit the interpretation of treatment effect, our completion rate was relatively higher than other studies in the orthopaedic literature, and a validated MCID was used. Additionally, the use of objective outcome measures based on physical examination findings adds reliability despite lacking formal validation. Finally, return-to-sport/work outcomes were not assessed. Future studies on GPS outcomes would benefit from implementing multiple PROMs, physical examination findings, and return-to-sport/work outcomes in parallel.
Conclusion
Under a standardized diagnostic-therapeutic algorithm, 98.8% of patients with GPS achieved either complete or partial resolution of symptoms. Both athletes and nonathletes demonstrated clinically significant improvement in symptoms. Compared to athletes, nonathletes demonstrated lower rates of complete resolution and higher rates of residual pain. Surgery, particularly robotic TAPP mesh repair, significantly improved outcomes in nonathletes. These findings extend GPS management beyond the elite, young, male athlete.
Footnotes
Final revision submitted October 14, 2025; accepted November 19, 2025.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was waived by Hartford Healthcare IRB (#HHC-2024-0141).
