Abstract
Background:
Mucoid degeneration of the anterior cruciate ligament (ACL) is an uncommon clinical condition with a reported prevalence of <0.5%. There is a paucity of histopathological descriptions in the literature, which limits our understanding of the underlying pathophysiology.
Purpose:
To define the histopathology and gene expression patterns of ACL mucoid degeneration to guide future biological and surgical management options.
Study Design:
Case series; Level of evidence 4.
Methods:
Ten patients with ACL mucoid degeneration between 2002 and 2023 were included. Plain radiograph and magnetic resonance imaging were reviewed to obtain multiple anatomic and morphometric measurements. Tissue samples were obtained from all patients during arthroscopic partial debridement of the ACL, and ACL tissue from 3 normal cadaveric knees was sampled for comparison. Formalin-fixed and paraffin-embedded tissues were prepared for histopathologic examination of microstructure and composition and for multiplex gene expression analysis using the NanoString nCounter Human Fibrosis V2 Panel.
Results:
All 10 patients reported pain and limited knee flexion. Radiological evaluation exhibited a bulbous appearance, an increased posterior tibial slope, a narrow notch width index, and increased signal intensity of the ACL. Early degenerative changes were common in the medial compartment. Histopathological analysis revealed significant differences in tissue architecture compared with normal ACL controls, including a disorganized collagen matrix and increased glycosaminoglycan content. NanoString multiplex gene expression analysis revealed 155 differentially expressed genes (DEGs) between the mucoid degeneration and control groups. The 5 most upregulated DEGs identified were Fibronectin 1, COL5A1, COL6A3, COL3A1, and COL1A2. Significant differences were observed in the pathway scores for epithelial-to-mesenchymal transition, extracellular matrix degradation/synthesis, collagen biosynthesis, focal adhesion kinase, platelet-derived growth factor signaling, and PI3K–Akt.
Conclusion:
Histological findings demonstrated distinct abnormalities in ACL structure and composition. We hypothesized that repetitive microtrauma of the ACL would lead to cumulative damage that ultimately would result in mucoid degeneration. Also, we hypothesized that increased PTS and a narrow notch width index would contribute to cumulative ligament loading and subsequent mucoid degeneration.
Keywords
Mucoid degeneration of the anterior cruciate ligament (ACL) is an uncommon clinical condition with a reported 20 prevalence of <0.5%. This condition is characterized by an abnormal signal intensity and morphology of the ACL on magnetic resonance imaging (MRI). 21 Although the term intraligamentous ganglion has been used, ACL mucoid degeneration is a distinct and different condition, characterized by intrinsic changes in the extracellular matrix with deposition of glycosaminoglycan in the ligament matrix. 20 The terms mucoid or mucinous degeneration are now accepted to describe these changes. Patients typically have vague knee pain that is often exacerbated during terminal knee flexion and extension. Pain is often described as posterior pain and is exacerbated by knee flexion, as previously described. 23 Diagnosis is made using MRI, which demonstrates enlargement of the native ACL with increased signal intensity throughout the tissue.19,20 Some patients may also have limitations in terminal knee extension owing to mechanical impingement of the enlarged ACL in the intercondylar notch. Mucoid hypertrophy of the anteromedial bundle can lead to pain during flexion or cause flexion restriction by impinging against the lateral compartment or the posterior cruciate ligament (PCL). 5 Although uncommon, this condition represents a challenging clinical problem with varying treatment options based mostly on pain symptoms.
The cause of this condition is not well understood. However, a steeper posterior slope (PTS) and a decreased notch width index (NWI) have been reported to be associated with ACL mucoid degeneration. 14 There is a paucity of histopathological descriptions in the literature,20,37 limiting our understanding of the underlying cellular and molecular mechanisms. This lack of insight into the etiopathology of mucoid degeneration limits our ability to identify effective treatment approaches, 1 such as biological interventions, 29 to prevent its development or progression.
This study aimed to describe the histopathologic and transcriptional features of ACL mucoid degeneration to better define its underlying pathology and identify therapeutic targets for treatment and prevention.
Methods
Patient Characteristics
This study was approved by the institutional review board from the Hospital for Special Surgery (2022-2009). A consecutive series of 10 patients who met the inclusion criteria for ACL mucoid degeneration and who underwent arthroscopic debridement surgery between 2002 and 2023 were retrospectively identified. The patients were evaluated and treated by 2 senior attending orthopaedic sports medicine surgeons (S.A.R. and R.F.W.). Preoperative MRI, pathology reports, and arthroscopic images were available for all patients.
Control tissue consisting of normal ACL was obtained from the Joint Restoration Foundation Inc (JRF Ortho). Knees with a history of surgery or with arthritis, such as rheumatoid arthritis or collagen disease, were excluded from the study. Three knees from the mid–femur to the mid–tibia/fibula were analyzed (Table 1).
Descriptive Data of All Controls a

None of the controls had any history of knee surgery. BMI, body mass index.
Imaging
We excluded patients who had a distinctive cystic lesion within the ACL or those in whom the lesion extended into the bone at the insertion site without ligament hypertrophy. We also excluded patients with a cyclops lesion. All patients were evaluated using standard knee radiographs and MRI. The diagnosis was confirmed using MRI according to the diagnostic criteria defined by Mclntyre et al. 21 This included an ill-defined ACL, increased signal intensity of the ACL on both T1- and T2-weighted pulse sequences, increased overall ACL volume, and a celery stalk appearance with well-oriented and intact ligament fibers continuous from the tibial to femoral insertion.
Plain Radiograph
The posterior tibial slope (PTS) and NWI were measured. The PTS was measured using a lateral plain radiograph as previously described. 3 Circles were used instead of a line (used in the original study) to determine the midpoint of the tibial shaft in this study. First, 2 circles were placed tangential to the anterior and posterior cortices at the tibial diaphysis and metaphysis (and even to the tibial tubercle) or at 50 and 150 mm from the tibial subchondral bone. The tibial axis was defined as the line bisecting the centers of the 2 circles. The PTS was defined as the angle formed by the tibial axis and a line parallel to the tibial plateau. The NWI was measured using a flexed posteroanterior or standard anteroposterior view. The NWI is the ratio of the width of the intercondylar notch to that of the distal femur at the popliteal groove level. A ruler was placed parallel to the joint line. The narrowest portion of the notch was measured at the ruler level. In the previous report, the mean PTS in the control group was 8.6° in men and 8.4° in women, respectively. 3 An NWI of <0.20 in men and <0.18 in women was considered notch stenoses in this study, as previously described. 33
Magnetic Resonance Imaging
All MRI scans analyzed in this study were performed using a 1.5-T General Electric Sigma MRI scanner. Each patient had their knee placed in a GE Sigma Knee Phased Array coil in 10° flexion, which is the standard positioning at our institution. The measurement was performed using CD–viewer software (CD–viewer 2.0; General Electric) on a computer workstation for more accurate measurements. MRI was used to measure PTS, NWI (axial and coronal views), anterior tibial translation (ATT, sagittal view), and ACL signal intensity relative to the PCL. All radiological measurements were conducted by a single observer (L.A.).
Anterior Tibial Translation
We used a proton-density sagittal view of the mid-lateral compartment. A 90° angle perpendicular to the lateral tibial plateau extending to the posterior tibia was identified. A parallel line was then drawn from this point to the posterior edge of the subchondral bone of the lateral femoral condyle (the cartilage edge was not used as a measurement point to avoid differences in cartilage thickness in cases of osteoarthritis or cartilage lesions). An ATT of >7 mm was considered an ACL rupture or anterior tibial subluxation as previously described. 5
NWI on Axial Image
The proton-density axial image was used for measurement. According to the method reported by Souryal et al, 32 the femoral NWI was calculated by dividing the transcondylar or intercondylar width (ICW) by the femoral notch width (NW). The NW was measured as the length between the medial projection of the lateral condyle and the lateral projection of the medial condyle of the femur. The ICW was determined by measuring the line passing through the popliteal groove and running parallel to the line drawn between the condylar ends across the most distal aspect of the femur. Both measurements were performed on axial images. Employing the criteria of Domzalski et al, 8 a ratio >0.270 for the NWI was considered normal, whereas values ≤0.269 were considered low and indicative of notch stenosis.
NWI on Coronal Image
The proton-density coronal image was used to measure the femoral bicondylar and intercondylar notch widths that form the NWI. The image chosen for measurement was at the point of decussation of the ACL and PCL, as close as possible to the midsubstance of the ACL. 7 First, the bicondylar width was measured at the level of the popliteal groove in the lateral condyle of the femur, parallel to the joint line formed by the distal femoral condyles. Then, on the same line, we identified the most interior margins of the femoral condyles at the borders of the intercondylar notch. The distance between these points represented the intercondylar NW. The ratio of the intercondylar NW to the bicondylar width represented the NWI. Stenosis was assessed using the same ratio as for the axial images (>0.270 was considered normal, whereas ≤0.269 was considered low).
Signal Intensity of ACL/PCL
The signal intensity of the ACL was compared with that of the PCL on a single-slice sagittal image. Proton-density and T2-weighted fat-suppressed images were used to measure signal intensity using the free-line region-of-interest (ROI) tool in a picture archiving and communication system (PACS). The ROI was defined by drawing a freehand line along the edges of the ACL and PCL.
Surgical Treatment
We began by examining knee ligament stability in patients under anesthesia. All patients had intact knee stability with negative Lachman, anterior drawer, and pivot-shift test results. Standard arthroscopic evaluation of all intra-articular structures was performed. The diagnosis of ACL mucoid degeneration was verified based on the typical abnormality in the appearance of the ACL. Direct arthroscopic probing revealed that the ACL was intact with no tears. The ACL appeared abnormally enlarged in all patients, and there was often some splaying of the ligament fibers. There was apparent stenosis of the intercondylar notch and direct contact between the anterior aspect of the ACL and the roof of the intercondylar notch with the knee in full extension. In all patients, the notch was first widened using a 4.5-mm motorized shaver or bur, and arthroscopic biopsy specimens (3 × 3 mm) of the hypertrophic portion of the ACL were then obtained. The ACL was then debrided to reduce excess bulk, taking care not to remove >50% of the native ACL volume. The goal of debulking was to preserve as many ACL fibers as possible and remove conflicts with the intercondylar notch with knee motion. A stability examination after debridement verified the maintenance of the functioning ligament with no changes in the stability examination (Lachman, anterior drawer, and pivot shift). Postoperatively, patients were allowed weightbearing as tolerated with crutches and immediate full range of motion as tolerated.
Histopathology
A biopsy was performed to confirm the diagnosis of ACL mucinous degeneration and to rule out other conditions. Archived patient tissue samples were obtained from the Department of Pathology.
Biopsies were performed during arthroscopy in all patients, and the specimens were immediately preserved in 4% buffered formalin, followed by standard processing for paraffin embedding. Similar processing was performed on control ACL specimens. Tissues were sectioned at 6 μm and were stained with hematoxylin and eosin. Alcian Blue solution, and Picrosirius red, and the slides were viewed using a DXM1200 microscope camera (Nikon Corp).
Each parameter was classified as abnormal when fiber alignment was disoriented, type I collagen was insufficient, or cellularity, glycosaminoglycan amount, angiogenesis, chondrogenesis, and lipid deposition were increased.
Immunohistochemistry
For Collagen III immunohistochemistry (IHC), sections were baked at 70°C for 1 hour. Antigen retrieval was performed using a citrate-based pH 6 epitope retrieval solution with heat-induced epitope retrieval (HIER) on the BOND automated system for 30 minutes at 100°C. Sections were then incubated with the primary antibody (Collagen III antibody, ab7778, polyclonal; Abcam) at a dilution of 1:500 in Bond Antibody Diluent (AR9352). A secondary polymer-based detection system (Leica Bond Refine Detection Kit, DS9800) and the Leica Mix DAB Refine Detection Kit were used for visualization after the manufacturer’s immunostaining procedures. The sections were counterstained with hematoxylin for 5 minutes. Sections stained with Collagen III IHC were examined using the same microscope camera as the other stained slides. Some specimens were unsuitable for IHC analysis due to poor tissue quality. All histological and immunohistochemical assessments were conducted qualitatively by a single observer (Y.O.).
NanoString
For gene expression analysis, ribonucleic acid (RNA) was isolated from the archived formalin-fixed paraffin-embedded (FFPE) sections. Three consecutive 6 μm sections were pooled to generate RNA for each sample. Total RNA was extracted using the High Pure FFPET RNA Isolation Kit (Roche Molecular Diagnostics) according to the manufacturer’s protocol. An additional ethanol precipitation step was performed to concentrate the RNA with a final elution of 15 μL in nuclease-free water, and the samples were stored at −80°C until further analysis. RNA concentration and integrity were evaluated using an Agilent BioAnalyzer 2100 (Agilent Technologies Inc). Smear analysis was performed, and the correct RNA input was calculated for each NanoString hybridization reaction by measuring the proportion of RNA fragments within 50 to 300 nucleotides, following the manufacturer’s instructions. A corrected input of 50 ng of total RNA was used for NanoString analyses using the NanoString nCounter Human Fibrosis V2 Panel for multiplex molecular analysis of 770 genes. Analysis was performed using the accompanying nSolver software (4.0) (NanoString Technologies), and only probes with counts above the background were included, following the manufacturer’s instructions.
Statistical Analyses
NanoString pathway score data are presented as mean ± standard deviation. Intergroup differences were evaluated using the Mann-Whitney U test. Statistical analysis was performed using GraphPad Prism 6 (GraphPad Software, Inc.). Statistical significance was defined as P < .05.
Results
Clinical Features
There were 4 women and 6 men, with a mean age of 53.7 years (range, 40-69 years). All cases were unilateral, with 3 left knees and 7 right knees. Three patients were current smokers at the time of surgery. Seven patients received intra-articular steroids with or without analgesic injections before surgery, with an initial improvement in symptoms, followed by gradual recurrence. Among the 7 patients, 6 had injection dates recorded in the electronic medical records, and the mean interval from injection to surgery was 31.9 ± 18.8 weeks. Four patients had previously undergone arthroscopic surgery—including 2 partial medial meniscectomies, 1 arthroscopic examination, and 1 arthroscopic debridement. Three patients were diagnosed with hyperlipidemia; 1 with hypertension; 1 with type 2 diabetes; 1 with pulmonary embolism and osteoporosis; 1 with ulcerative colitis, kidney stones, and hepatitis B; and 1 with Lyme disease.
Nine patients had a documented body mass index at the time of surgery, with a mean of 25.7 kg/m2. Three patients had traumatic injuries. Clinically, all 10 patients reported knee pain and flexion limitation. The pain was often described as deep within the joint or posterior, worsening with flexion. Some patients reported pain during both terminal flexion and extension. Symptoms started insidiously, except in 2 patients who reported a history of trauma, and the duration of symptoms ranged from 6 to 24 months, with a mean duration of 15.7 months. Six patients exhibited mild extension deficits. Patients undergoing debridement for mucoid degeneration of the ACL experienced pain relief and range-of-motion restoration soon after surgery, and none exhibited instability caused by ACL deficiency.
Imaging Findings
The image evaluation results are presented in Table 2. Standard radiographs of all the patients demonstrated early degenerative changes and mild intercondylar notch stenosis (Figure 1). MRI demonstrated bulbous enlargement of the ACL with increased signal intensity on all pulse sequences. There was a normally oriented ACL parallel to the Blumensaat line. The MRI appearance was consistent with the celery stalk appearance on T2-weighted pulse sequences (Figure 2). 21
Imaging Characteristics of All Patients a

ATT, anterior tibial translation; MRI, magnetic resonance imaging; NWI, notch width index; PD, proton density; PTS, posterior tibial slope; SIR A/P, signal intensity ratio of the anterior cruciate ligament/posterior cruciate ligament; T2FS, T2-weighted fat-suppressed.
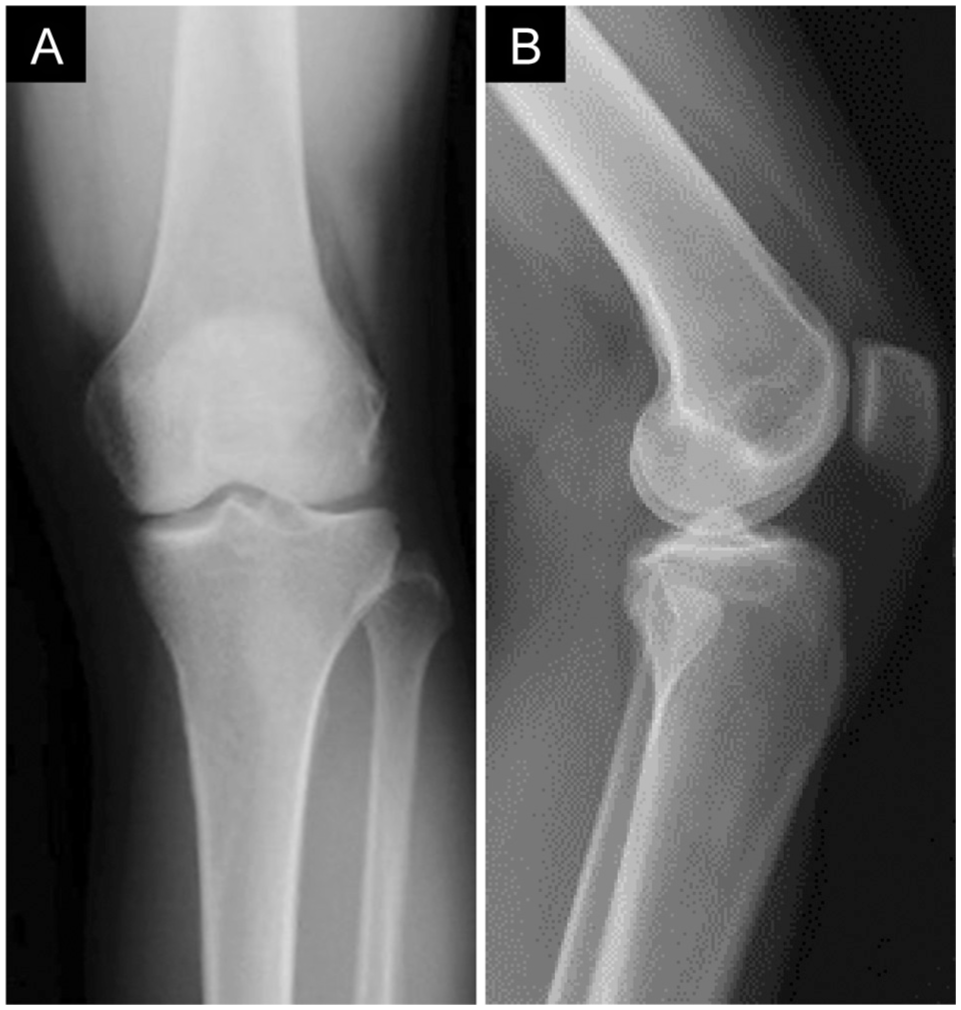
Representative plain radiographs. (A) Anteroposterior view. (B) Lateral view showing a large posterior tibial slope (19°).

Representative magnetic resonance imaging exhibiting a bulbous ACL. (A) Proton density image. The ACL (white arrow) is bulbous and shows iso- to high-signal intensity compared with the PCL (white arrowhead). (B) Inversion recovery image. The ACL (white arrow) is bulbous and shows iso- to high-signal intensity compared with the PCL (white arrowhead). ACL, anterior cruciate ligament; PCL, posterior cruciate ligament.
Arthroscopic Findings
Arthroscopic inspection revealed obvious enlargement of the ACL. There was often a poorly developed or absent synovial lining on the ACL and a longitudinal separation of the ligament fibers. Mucoid material was often expressed by probing the ligament tissue (Figure 3). Most patients had early degenerative changes—including articular cartilage lesions and meniscal tears. Medial meniscal tears were observed in patients No. 2, No. 3, No. 5, No. 9, and No. 10 (50%). There were no lateral meniscal tears. Medial chondral lesions were observed in patients No. 3 to 7 (50%). Lateral chondral lesions were observed in patients No. 3 and No. 7. Abnormalities in the medial compartment (meniscal tears, medial cartilage lesions, or both) were observed in eight patients.

Representative arthroscopic findings. (A) Abnormal ACL is confirmed. (B) The ACL exhibited a bulbous morphology (white ellipse). (C) Debridement was performed using an arthroscopic shaver (black arrow). (D) The debrided ACL did not show impingement with the femoral notch. ACL, anterior cruciate ligament.
Histopathology
Histopathological evaluation revealed multifocal myxomatous degeneration with increased metachromasia, indicative of elevated glycosaminoglycan (GAG) deposition compared with control. Nine samples from the mucoid degeneration group showed disorganized collagen fibers, reduced type I collagen, and hypercellularity. All samples exhibited an abnormally high abundance of glycosaminoglycan. Furthermore, angiogenesis, chondrogenesis, and occasional lipid deposition were confirmed in 6, 3, and 7 samples, respectively. Conversely, samples from the control group presented well-oriented fibers, abundant type I collagen, normal cellularity, and GAG. No metaplasia was identified in the control group (Table 3 and Figure 4).
The Characteristics of the Mucoid Degeneration of the Anterior Cruciate Ligament Compared With the Control Group
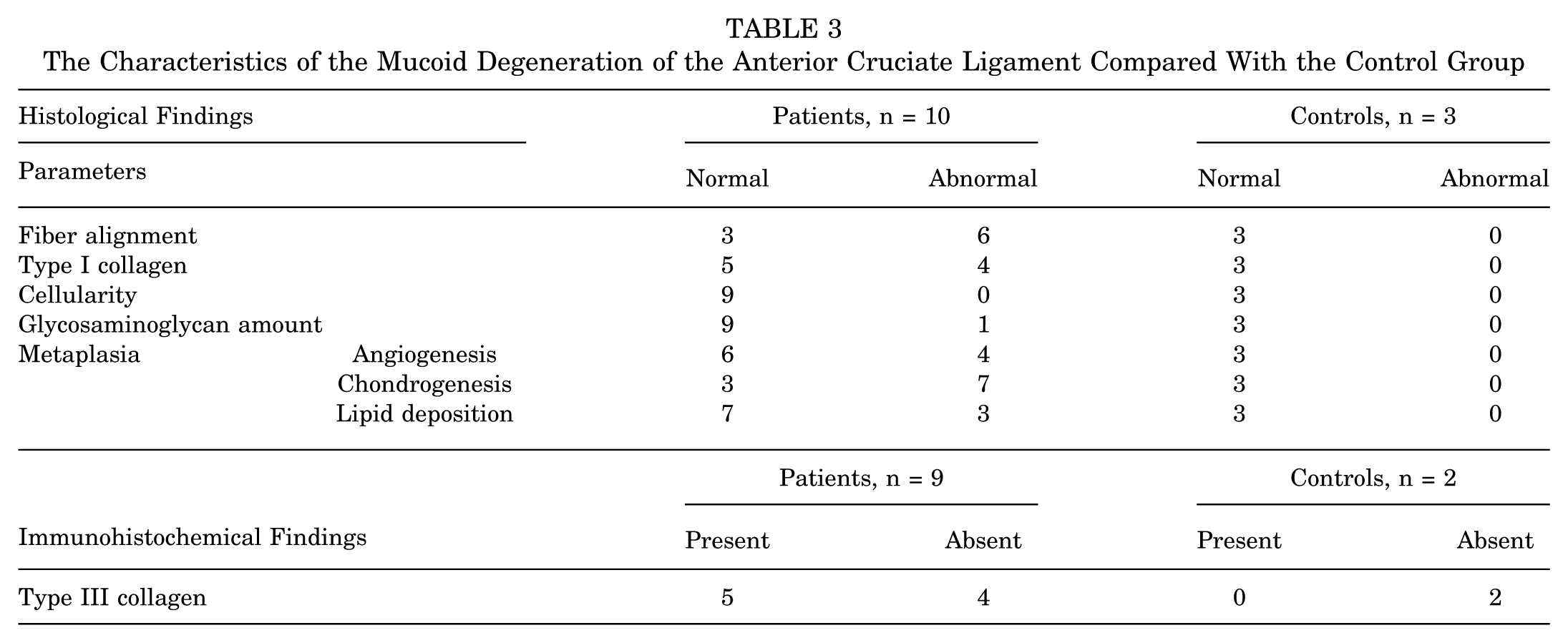

Representative histological findings. (A and B) Disorientation of the fibers. (B) An abundant glycosaminoglycan (the blue-stained area). (A and B) Angiogenesis (black arrow) and lipid deposition (region outlined in black) are observed in the mucoid degeneration group compared with the control group. (D and E) In contrast, the control group shows well-oriented fibers with less cellularity and glycosaminoglycan, as well as (F) abundant fibers (indicating type I collagen), compared with the mucoid degeneration group. Bars = 200μm. H&E, hematoxylin and eosin stain.
Immunohistochemistry
Type III collagen staining was not observed in the control group (Figure 5, A-C). In contrast, type III collagen immunostaining was detected in ACL samples with moderate mucinous degeneration (Figure 5, D-F) but was absent in samples with more advanced mucinous changes. (Figure 5, G-I). The extent of degeneration was evident from the presence of mucopolysaccharides (as indicated by the intensity of staining) using Alcian Blue staining (Table 3).

Representative immunohistochemical findings. (A-C) Type III collagen staining was not observed in the control group. In contrast, (D-F) it was detected in samples with moderate mucinous degeneration. However, (G-I) it was absent in samples with more severe mucinous changes. Bars = 100 μm in A-F. Bars = 250μm in G-I.
Transcriptional Profiling
NanoString multiplex gene expression analysis revealed 155 DEGs between the mucoid degeneration and control groups. The 20 most upregulated DEGs are presented in Table 4. The 5 most upregulated DEGs identified included Fibronectin 1 (FN1), COL5A1, COL6A3, COL3A1, and COL1A2. We also found significant differences in the pathway scores for epithelial-to-mesenchymal transition (EMT), extracellular matrix degradation/synthesis, collagen biosynthesis, focal adhesion kinase, platelet-derived growth factor (PDGF) signaling, and PI3K-Akt (Figure 6).
Top 20 DEGs With Adjusted P < .05 and Fold Change >1.5 in Patients With Mucoid Degeneration and Controls a
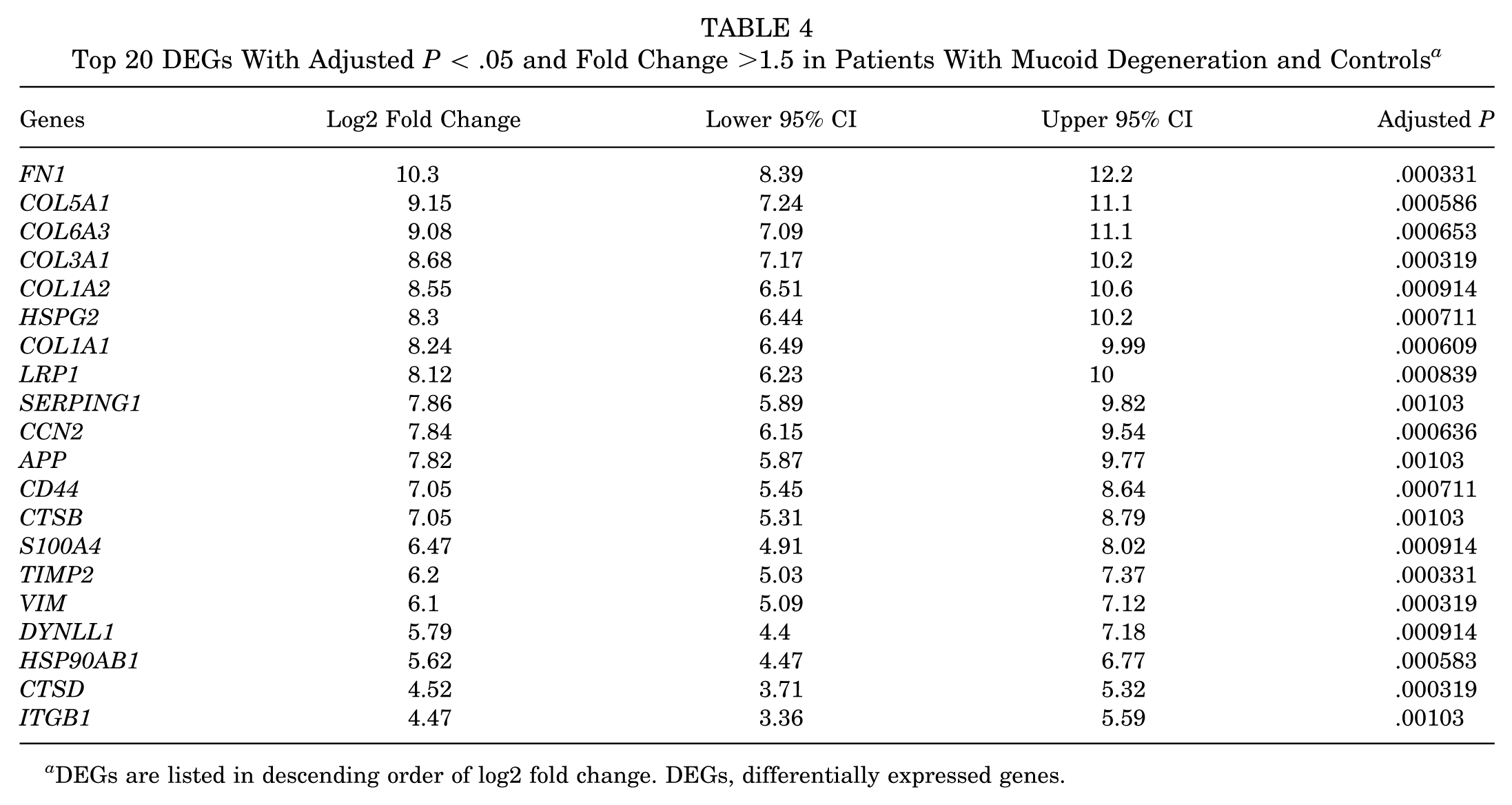
DEGs are listed in descending order of log2 fold change. DEGs, differentially expressed genes.
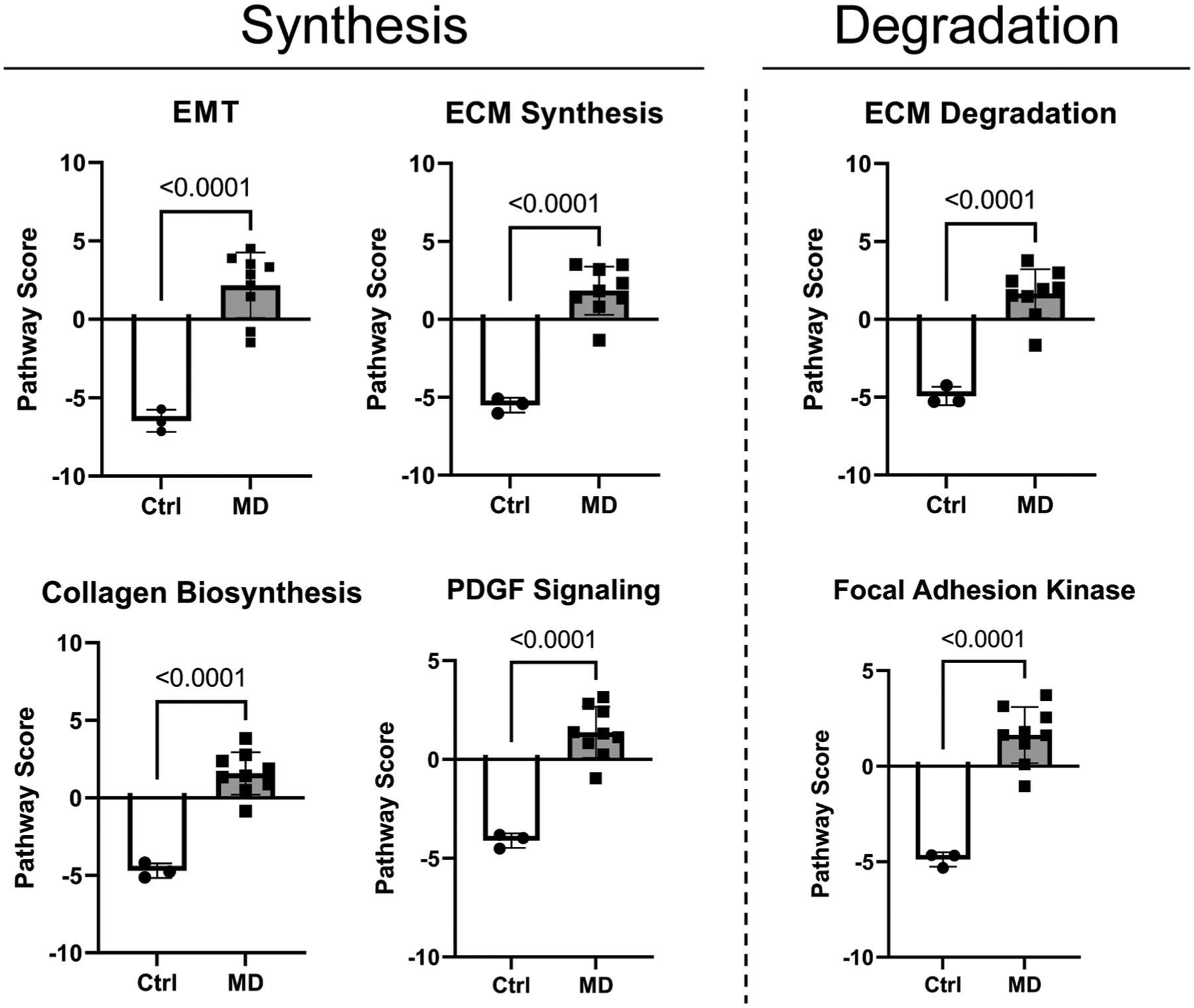
Representative pathway scores. Significant upregulation in both anabolic and catabolic pathways is observed in the mucoid degeneration group compared with those in the control group. ECM, extracellular matrix; EMT, epithelial-to-mesenchymal transition.
Discussion
The primary findings of this study are abnormalities in tissue microstructure and composition, with upregulation in both anabolic and catabolic gene expression pathways. These features were consistent with changes in signal and morphology of the ACL seen on gross inspection and MRI. Although previous studies have analyzed tissue histology, none have comprehensively analyzed gene expression patterns. The pathogenesis of mucoid degeneration is not well understood. Several theories have been proposed to account for its development—including the herniation of synovial fluid and/or synovial tissue into the ACL after injury to the protective synovial sheath over the ACL (synovial theory), repetitive trauma leading to disruption of ligament fibers (traumatic theory), congenital displacement of synovial tissues into the ACL (ectopic synovial theory), or age-related degenerative changes in the joint (degenerative theory). 31 The findings of this study demonstrate changes in the structure and composition of the ACL consistent with matrix turnover and remodeling. We found a disorganized collagen matrix and increased glycosaminoglycan content, consistent with adaptive remodeling in response to repetitive ACL microinjury.
The ACL possesses intrinsic potential for healing and metabolic activity.17,26,35 However, once the ACL is severely or repeatedly damaged, degradation, chondrogenesis, and degeneration begin.2,9,28 Transcriptional profiling revealed 155 DEGs between the mucoid degeneration and control groups, with the most upregulated genes being important matrix proteins—including FN1, COL5A1, COL6A3, COL3A1, and COL1A2. We also found significant differences in the pathway scores for EMT, extracellular matrix degradation/synthesis, collagen biosynthesis, focal adhesion kinase, PDGF signaling, and PI3K-Akt. These changes are consistent with cellular and molecular responses to repetitive injury and with both catabolic and anabolic matrix remodeling processes. Furthermore, deoxyribonucleic acid (DNA) replication occurred frequently (as confirmed by the upregulated cell cycle pathway score). Errors may occur during the synthesis of DNA, RNA, or proteins, leading to phenotypic mutations. 25 These mutations, along with upregulated catabolic pathways, ultimately result in tissue (ACL) degradation.
An important associated finding was increased PTS (the mean value was 14° in this study compared with the previous study 3 ) and narrow NWI (5 knees out of 10 in this study according to the previously defined value 8 ). It is well established that an increased PTS leads to increased ATT and an elevated strain in the ACL.10,30 A narrow NW also leads to impingement of the intercondylar notch. These morphological changes may cause pain in extension due to impingement and predispose patients to repetitive subclinical injury to the ACL. The patients included in the present study had a steep PTS and narrow NWI as previously described.6,12-14,16 While mechanical stress is beneficial for maintaining homeostasis in ACL cells, excessive loading can be detrimental.22,34 A previous study demonstrated that a steeper PTS can lead to ACL overload and subsequent degeneration after high tibial osteotomy. 13 Furthermore, in the anteromedial bundle transection model of the ACL, the thickness of the glycosaminoglycan layer in the posterolateral area gradually increased until 8 weeks, peaking at 4 weeks. 26 Narrow NW could result in impingement between the ACL and the femoral notch, causing repeated/cumulative damage to the ACL.
We hypothesized that this cumulative loading would result in an aberrant cellular response and consequent mucoid degeneration. It is well-established that dense fibrous connective tissue responds to mechanical loads. This has been studied extensively in tendons, and it has been shown that physiological loads can maintain the tendon phenotype, whereas excessive mechanical loading can alter gene expression patterns and lead to adverse matrix changes—including chondrogenic differentiation. 40 Because mucoid degeneration is uncommon, we speculated that there may be underlying cellular and/or molecular abnormalities that lead to this response. Further insights into the pathophysiology of mucoid degeneration may be provided by single-cell transcriptional profiling.
Further support for the occurrence of cumulative microscopic matrix damage from repetitive loading comes from recent descriptions of fatigue damage of the ACL. Cumulative fatigue damage to the ACL may be an important factor in the occurrence of noncontact ACL injury. 39 Cadaveric studies using loading protocols that are known to strain the ACL submaximally result in the unraveling of the collagen molecular triple helix and the disruption of collagen fibrils. 4 Such microscopic changes are likely to gradually accumulate over time, as the ACL has a limited intrinsic ability to repair and remodel matrix damage. Although this concept has been more established in tendons and tendinopathy, it is a newer concept related to ligaments.
The deposition of glycosaminoglycans and mucinous material in these patients suggests a cellular response to an altered pericellular mechanical loading environment. Matrix changes alter the pericellular environment, likely affecting the mechanical signals cells experience. We hypothesized that such changes in mechanotransduction play an important role in abnormal cellular responses and subsequent mucoid changes. Previous studies on tendons demonstrated that tenocytes exposed to compression undergo metaplasia into a cartilaginous phenotype, and a similar response is likely to occur in ligament cells exposed to increased compressive loading.24,38
The presence of osteoarthritis may also contribute to mucoid degeneration due to intercondylar notch osteophytes, leading to repetitive microtrauma to the ACL. Also, 8 out of 10 patients presented medial lesions (either or both meniscal tears and cartilage lesions), suggesting an association between ACL mucoid degeneration and medial compartment pathology, as previously described. 15 Osteoarthritis is also associated with inflammatory mediators in the synovial fluid, which can potentially contribute to pathological changes in the ACL owing to the association between matrix-degrading enzymes (matrix metalloproteinases) 36 and a proinflammatory environment.11,27 Ultimately, the combination of biological (an inflammatory environment associated with early osteoarthritis) and mechanical (repetitive ligament impingement and cumulative matrix injury) factors may initiate aberrant matrix changes that result in ACL mucoid degeneration. Such a process is predicted to involve progression from an inflammatory phase to a phase of ligament remodeling and finally to established pathology. Each stage has distinct clinical, imaging, and histological characteristics. However, similar to many degenerative processes, surgical biopsy specimens are only available for late-stage disease, as in the present study. Therefore, this progression remains speculative. The lack of animal models makes it difficult to further study the pathophysiology of mucoid degeneration.
Limitations of this study include its retrospective design and the inclusion of a small number of patients and controls. The small size of the tissue samples also limited our ability to perform further analyses, such as immunohistochemical staining, to characterize the matrix proteins and specific cell types. Additional insights into the basic mechanisms of mucoid degeneration may be aided by analyses of its composition (proteomics), ultrastructure (electron microscopy), and molecular profile (single-cell RNA sequencing). Hypotheses regarding the roles of mechanical, cellular, molecular, and genetic factors require further study to confirm.
By defining the cellular and molecular mechanisms of this condition, we aimed to broaden treatment options to potentially identify biological interventions. The association between mucoid degeneration and medial compartment osteoarthritis suggests that patients may benefit from early intervention. Early treatment with debridement of the bulbous ACL, in addition to notchplasty, may prevent progressive accumulation of microdamage in the ACL, potentially halting the inflammatory and catabolic cycle. For persistent or recurrent cases, osteotomy to decrease PTS could be another treatment option.18,37 The surgeon should keep in mind that ACL debridement should be limited to the minimal amount necessary to relieve impingement, to maintain anterior-posterior stability of the knee.
Conclusion
Our study findings demonstrate changes in the structure and composition of the ACL consistent with matrix turnover and remodeling. Transcriptional profiling revealed the upregulation of both anabolic and catabolic pathways, which play important roles in matrix synthesis and degradation. These changes are consistent with adaptive remodeling in response to repetitive submaximal ACL injury caused by impingement. We found relative intercondylar notch stenosis and an increased PTS, both of which may contribute to cumulative mechanical stress on the ACL. The novelty of our study lies in the comprehensive analysis of gene expression patterns, which may pave the way for further studies of the underlying pathophysiology of this condition and ultimately help identify therapeutic targets.
Footnotes
Acknowledgements
The authors recognize the valuable support of Liang Ying (Orthopaedic Soft Tissue Research Program, Hospital for Special Surgery at that time) for technical support with histological analysis.
Final revision submitted October 1, 2025; accepted October 31, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was supported by The Uehara Memorial Foundation, Kodama Memorial Prize, Japan Orthopaedics Traumatology Foundation, International Travel Award from the Japanese Society for Matrix Biology and Medicine, and the Japan Society for the Promotion of Science KAKENHI (24K23295 and 25K19973). This study was funded in part by the Virginia Toulmin Foundation. S.A.R. has received consulting fees from Teladoc, Inc, Enovis-DJO, and Novartis Pharmaceuticals. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Hospital for Special Surgery (2022-2009).
