Abstract
Background:
Posterior humeral avulsion of glenohumeral ligament (PHAGL) lesions involves detachment of the posterior band of the inferior glenohumeral ligament from the humeral neck. Although uncommon, these lesions have been associated with recurrent instability.
Purpose:
To compare clinical outcomes and recurrence rates of patients with PHAGL lesions undergoing surgical repair for posterior shoulder instability versus a pair-matched control group without PHAGL lesions undergoing isolated arthroscopic posterior labral repair.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
This was a retrospective study of patients diagnosed with PHAGL lesions and posterior shoulder instability who underwent primary arthroscopic repair between 2010 and 2019. These patients were matched 1:3 with a cohort of patients with isolated posterior labral tears (PLTs). Demographics, surgical data, and pre- and postoperative clinical outcomes scores (American Shoulder and Elbow Surgeons, Single Assessment Numeric Evaluation, 12-Item Short Form Survey Physical Component Summary, and short version of the Disabilities of the Arm, Shoulder and Hand questionnaire) were collected. Return-to-sport rates and satisfaction were also evaluated. Failure was defined as recurrent instability or progression to arthroplasty.
Results:
Nine patients diagnosed with PHAGL lesions who underwent primary arthroscopic repair were included. These 9 patients (PHAGL group) were age-matched to a cohort of 26 patients with PLTs who underwent repair (PLT group). The mean follow-up was 5.8 years (range, 2.0-14.2). No significant differences were found between the PHAGL and PLT groups regarding shoulder function, physical health, or patient satisfaction. The median satisfaction score for the PHAGL group was 8.5 of 10, compared to 10 of 10 for the PLT group (P = .144). One patient with PHAGL lesions experienced recurrent instability compared with 0 in the PLT group.
Conclusion:
This study shows that patients with PHAGL lesions who undergo arthroscopic stabilization can attain comparable mid-term functional outcomes to those with isolated PLTs. Failure rates are similar between the PLT group and those with PHAGL lesions, and surgical management remains an effective treatment option for both groups. Further studies are needed to optimize surgical techniques and refine patient selection criteria for improved long-term success.
Keywords
Posterior shoulder instability (PSI) has an incidence in the general population of 4.64 per 100,000 person-years, with a significantly greater incidence of PSI in male than female patients (8.86 vs 1.07 per 100,000 person-years). 14 It accounts for about 2% to 10% of all shoulder instability.11,12 The stability of the shoulder results from a combination of both static and dynamic constraints, and instability can be caused by a failure of 1 or more of the involved structures. 2 In 1949, Nicola 10 first described an avulsion of the anterior band of the inferior glenohumeral ligament. Later in 1995, Wolf et al 13 introduced the term HAGL to describe the humeral avulsion of the glenohumeral ligaments. The anterior HAGL is more common, but posterior injuries do occur and are referred to as reverse or posterior HAGL (PHAGL) lesions.
The incidence of these injuries is unknown, but in reported cases of HAGL lesions, anterior HAGL lesions represent 93% of reported cases, whereas PHAGL lesions represent only 7% of cases. 4 While PHAGL lesions are rare, they have been shown to contribute to recurrent instability.1,4 Patients who sustain a PHAGL lesion mechanism are typically younger and sustain a more violent injury to the shoulder, often involving a forced cross-body adduction on a forward flexed arm. 3
In a systematic review of 42 patients with anterior or posterior HAGL lesions, superior clinical outcomes and lower recurrence rates were reported in patients who were managed surgically, with a 90% recurrence rate in those treated nonoperatively. 8 Additionally, the surgical treatment of anterior HAGL lesions has demonstrated satisfactory clinical outcomes with low residual pain levels after stabilization, high rates of return to play, and low recurrence rates, with either arthroscopic or open stabilization.
The literature on PHAGL lesions is predominantly limited to case reports and case series, and there is still a lack of literature on patients with PHAGL lesions undergoing stabilization procedures compared with a control group without PHAGL lesions. Therefore, the purpose of this study was to compare clinical outcomes and recurrence rates of patients with PHAGL lesions undergoing stabilization versus a pair-matched control group with isolated posterior labral tears (PLTs) undergoing stabilization. The hypothesis was that patients with PHAGL lesions undergoing shoulder stabilization would have comparable patient-reported outcomes (PROs), functional outcomes, and failure rates at final follow-up compared with a pair-matched control group without PHAGL lesions who underwent stabilization of a PLT.
Methods
Patient Selection
This was a retrospective review of prospectively collected data with approval by the Vail Health Hospital Institutional Review Board (VHH 2023-221). All patients with a PLT with and without PHAGL lesions who underwent arthroscopic repair from December 2008 to August 2021 by the senior surgeon (P.J.M.) were eligible for inclusion (minimum follow-up: 2 years). Posterior shoulder instability was diagnosed via physical examination, and PLTs with and without PHAGL lesions were identified and confirmed via noncontrast magnetic resonance imaging or computed tomography. PHAGL lesions were classified based on the classification system set forth by Ames and Millett 1 (Figure 1). Surgery was indicated for patients with instability that did not resolve through nonoperative treatment. Patients who underwent combined anterior and posterior labral repair or who did not have the minimum required follow-up were excluded.
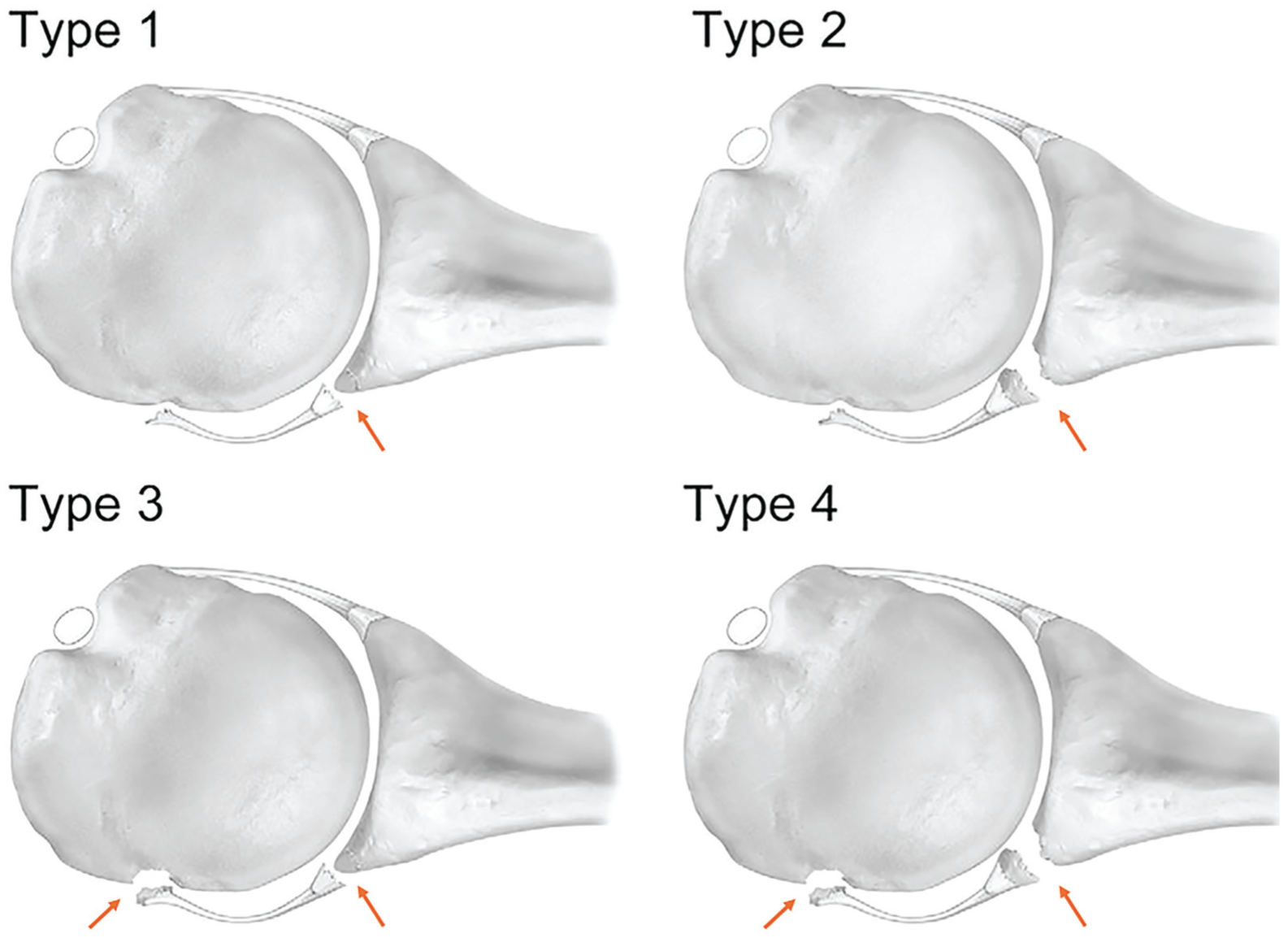
PHAGL subtypes as described by Ames and Millett. 1 Type 1: PHAGL with a posterior Bankart lesion. Type 2: PHAGL with a posterior osseous Bankart lesion. Type 3: PBHAGL with a posterior Bankart lesion. Type 4: PBHAGL with a posterior osseous Bankart lesion. PBHAGL, posterior bony humeral avulsion of the glenohumeral ligament; PHAGL, posterior humeral avulsion of the glenohumeral ligament.
Surgical Technique
Surgery was performed as previously described.5,6,9 Local and general anesthesia were administered, and the patient was positioned in the beach-chair position or lateral decubitus position for standard sterile preparation and draping. Following an examination under anesthesia, a standard posterior portal was created, and the arthroscope was introduced into the glenohumeral joint. A high anterior superior portal was made in the rotator interval under direct visualization, and a diagnostic arthroscopy was performed. Patients with associated pathology of the long head of the biceps tendon underwent subpectoral biceps tenodesis. The presence of a PHAGL lesion was confirmed by viewing from an anterior superior portal. Posterior labral repair was then completed arthroscopically in standard fashion. An accessory posterolateral portal was established for improved anchor placement and repair of the PHAGL lesion. The mobility and reparability of the ligament were assessed using a grasper. The avulsion site on the humerus was prepared using an arthroscopic shaver to promote healing. Anchors loaded with sutures were inserted onto the humerus at the avulsion site (3.0-mm biocomposite SutureTak anchor, 3.5-mm biocomposite SwiveLock anchors, or 2.6-mm knotless FiberTak anchors; Arthrex). The number of anchors and the choice of anchor were based on surgeon preference. Sutures loaded in the anchors were then passed through the ligament using a suture-passing device in a mattress configuration. Arthroscopic knots were then tied extra-articularly, or a knotless mechanism was used, securing the posterior band of the inferior glenohumeral ligament back to its anatomic footprint. In cases of significant capsular laxity, additional plication sutures were placed to close the posterior capsular defect and/or reinforce posterior stability. After confirming adequate tension and stability, fluid was drained, and standard wound closure was performed.
Postoperative Rehabilitation
Rehabilitation was planned as previously described. 7 The patient was immobilized in a padded abduction sling with elbow, wrist, and hand range of motion allowed. Physical therapy could commence immediately postoperatively with passive range of motion or be delayed up to 2 weeks based on surgeon preference and patient-specific factors such as tissue quality and size of the surgical repair. Once passive range of motion was adequate, patients could progress to assisted active or active range of motion at 5 to 6 weeks postoperatively. After at least 8 weeks, resistance training could be introduced with advanced strengthening beginning at 3 months postoperatively. Return to full activities was anticipated after 4 to 5 months with shared decision-making by the surgeon and physical therapist.
Patient Characteristics and Operative Data
All patient demographic and surgical data were obtained prospectively but confirmed on the patients’ charts. This data included patient age at the time of surgery, sex, laterality of the injury, time from injury to surgery, and any further surgeries or revisions.
Outcome Measures
PRO measures were collected preoperatively, at regular postoperative time increments, and at a minimum 2-year follow-up. Outcome measures included the American Shoulder and Elbow Surgeons (ASES) score; Single Assessment Numeric Evaluation (SANE); short version of the Disabilities of the Arm, Shoulder and Hand questionnaire (QuickDASH); 12-Item Short Form Survey Physical Component Summary (SF-12 PCS); and subjective satisfaction (1-10 scale, with 10 = very satisfied). Pain levels were evaluated using the visual analog scale. Patients also received optional questions to determine postoperative return to sport, modifications of activity, and reasons for doing so at a minimum 2-year follow-up. Failure was defined as recurrent instability events or progression to arthroplasty.
Statistical Analysis
Data were analyzed using SPSS version 28.0 (SPSS). Patients were matched 1-to-3 using 5-factor match criteria (age, sex, year of surgery, biceps tenodesis, and isolated PLT) to PLT patients without PHAGLs. Categorical variables are reported as numbers and percentages. Nonnormally distributed continuous variables are reported as medians. A Kolmogorov-Smirnov test was used for continuous variables to assess normal distribution. Paired data were compared using a Wilcoxon signed-rank test. Comparison data across groups was done using a Mann-Whitney U test. Statistical significance was set at P < .05.
Results
Patient Characteristics
Twelve patients diagnosed with PHAGL lesions underwent primary arthroscopic repair by the senior surgeon between August 2010 and June 2019. In the posterior instability group, 162 patients underwent primary arthroscopic repair from December 2008 to August 2021. Of the 12 patients with PHAGL lesions, 3 were excluded because of multidirectional instability, os acromion fixation, or follow-up of <2 years from surgery (Figure 2). The 9 patients with PHAGL lesions were age-matched to a cohort of 26 patients from the posterior instability group. Of the 9 patients with PHAGL lesions, 7 had type 1 lesions and 2 had type 2 lesions. 1 All 9 of the patients with PHAGL lesions were eligible for a minimum 2-year follow-up, with 6 patients (67%) having preoperative PROs and 7 patients (78%) having postoperative PROs. The posterior instability group had all 26 patients eligible for a minimum 2-year follow-up, with 25 having preoperative PROs (93%) and 20 having postoperative PROs (80%). The mean follow-up for both groups was 5.8 years (range, 2.0-14.2). The mean time from injury to surgery was 412 days (range, 3.0-3278 days; P = .559), and mean age at surgery was 30.0 years (range, 16.3-66.4 years; P = .624). All patients were male, with 1 worker's compensation case. For the PHAGL group, 6 patients (66.7%) were operated on in the beach-chair position, while 3 patients (33.3%) were in the lateral decubitus position. In the posterior instability group, 18 patients (69.2%) were operated on in the beach-chair position while 8 patients (30.8%) were operated on in the lateral decubitus position (Table 2). The most common mechanism of injury in the PLT group was either through an insidious onset or by skiing or snowboarding. In the PHAGL group, the most common injury was either by skiing or snowboarding or through a team sport (Table 1).

Flowchart of patient inclusion. MDI, multidirectional instability; PHAGL, posterior humeral avulsion of glenohumeral ligament; PLT, posterior labral tear; PRO, patient-reported outcome; TSA, total shoulder arthroplasty.
Mechanism of Injury by Lesion Type a

PHAGL, posterior humeral avulsion of glenohumeral ligament; PLT, posterior labral tear.
Patient Demographics a
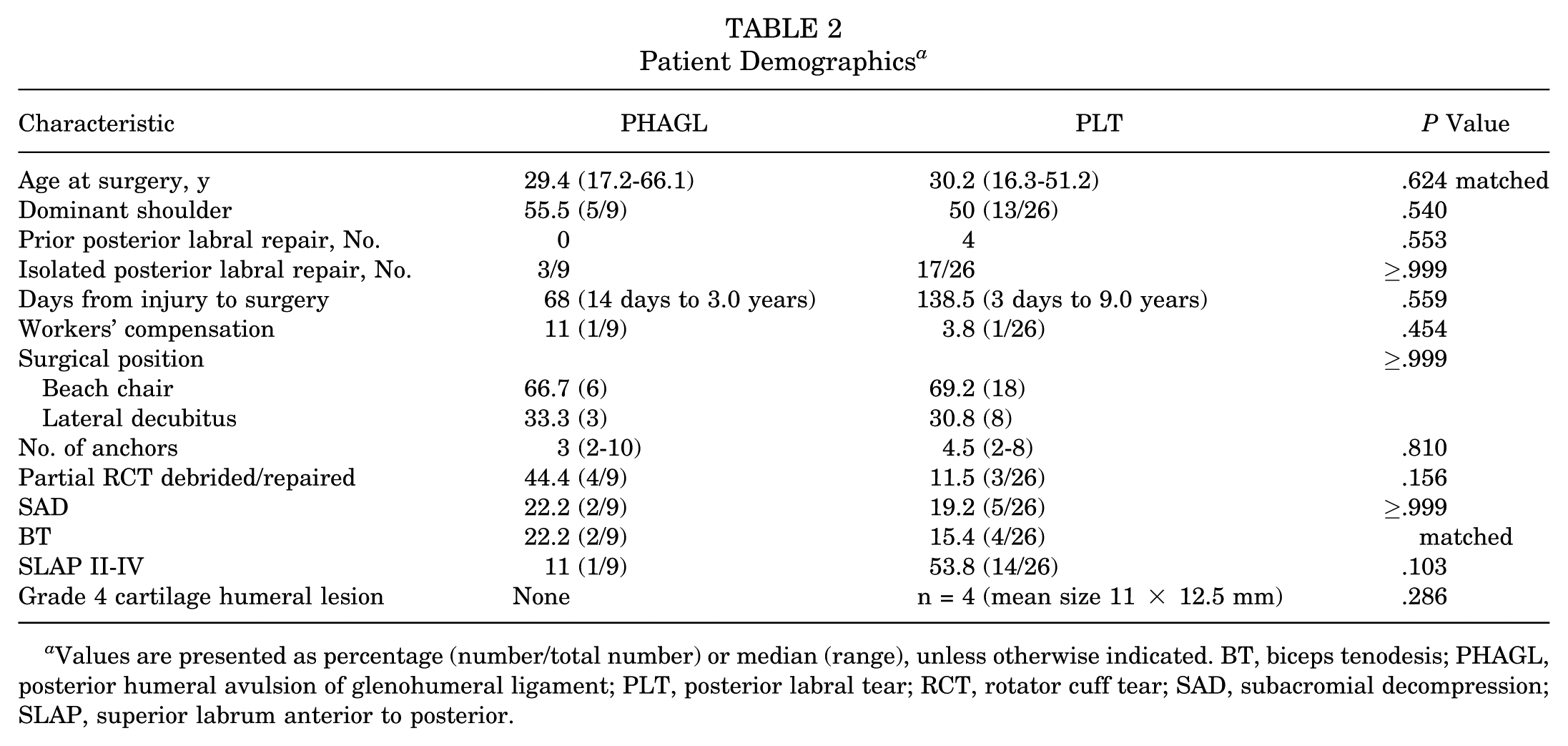
Values are presented as percentage (number/total number) or median (range), unless otherwise indicated. BT, biceps tenodesis; PHAGL, posterior humeral avulsion of glenohumeral ligament; PLT, posterior labral tear; RCT, rotator cuff tear; SAD, subacromial decompression; SLAP, superior labrum anterior to posterior.
Outcome Assessment
Between the PHAGL and PLT groups, no statistically significant differences were observed in shoulder function, physical health, or outcome scores. The median satisfaction scores were 8.5 of 10 (range, 1-10) for the PHAGL group and 10 of 10 (range, 6-10) for the PLT group (P > .144). There was also no significant difference in functional outcome or sports-specific preoperative and postoperative scores demonstrated between the groups (P > .05) (Table 3). Eighteen patients in the PLT group and 7 patients in the PHAGL group reported their postoperative return to sport. Of the patients, 61.1% (n = 11) in the PLT group and 57.1% (n = 4) in the PHAGL group were participating at or above their preinjury level, respectively. Pre- to postoperative comparisons of PROs in the PLT group showed statistically significant improvements in ASES, SANE, QuickDASH, and SF-12 PCS scores (P < .05). There was a statistically significant improvement in the QuickDASH (P = .028) and SF-12 PCS (P = .043) scores for the PHAGL group (Table 4). One patient in the PHAGL group developed recurrent posterior instability. Recurrent instability was confirmed by follow-up visits or, as indicated in postoperative surveys, with a subluxation or dislocation event. One patient had a traumatic dislocation of their contralateral shoulder noted on their postoperative survey, while the second patient reported a dislocation that required relocation by another person. In contrast, 1 patient in the PLT group required an arthroplasty 12 years after the index surgery.
Minimum 2-Year Postoperative Comparison of Patient-Reported Outcomes for PHAGL and PLT a

ASES, American Shoulder and Elbow Surgeons; PHAGL, posterior humeral avulsion of glenohumeral ligament; PLT, posterior labral tear; QuickDASH, short version of the Disabilities of the Arm, Shoulder and Hand questionnaire; SANE, Single Assessment Numeric Evaluation; SF-12 PCS, 12-Item Short Form Survey Physical Component Summary.
Pre- to Minimum 2-Year Postoperative Comparison of Patient-Reported Outcomes for PHAGL and PLT a

Bold values indicate statistical significance at P < .05. ASES, American Shoulder and Elbow Surgeons; PHAGL, posterior humeral avulsion of glenohumeral ligament; PLT, posterior labral tear; QuickDASH, short version of the Disabilities of the Arm, Shoulder and Hand questionnaire; SANE, Single Assessment Numeric Evaluation; SF-12 PCS, 12-Item Short Form Survey Physical Component Summary.
Discussion
The findings of this study indicate that patients undergoing arthroscopic stabilization for PSI with concomitant PHAGL lesions achieved comparable outcomes to those with isolated PLT repairs. While both groups demonstrated significant postoperative improvement in PROs, no statistically significant differences were observed between the groups in terms of postoperative shoulder function, physical health scores, and satisfaction levels. These results suggest that when addressed surgically, PHAGL lesions do not appear to negatively affect mid-term clinical outcomes when compared with isolated PLT repairs.
Although PHAGL lesions remain relatively rare, their contribution to posterior instability has been well documented.1,4 Previous literature has reported that HAGL lesions, whether anterior or posterior, frequently result in persistent instability when left untreated, with the overall rate of instability recurrence reaching 90%. 8 This current study supports previous evidence that surgical intervention leads to functional improvement and symptom resolution when appropriately repaired. In the PHAGL group, improvements were observed across all PROs at 2 years postoperatively, although not statistically significant overall. Notably, the QuickDASH and SF-12 PCS demonstrated a statistically significant improvement. While both groups had a similar failure rate with 1 patient each, the observed recurrence of posterior instability in the PHAGL cohort (1/9 patients) compared with none in the PLT group highlights the contribution of PHAGL lesions to recurrent instability when left untreated, and having an increased awareness to identify and repair them is critical to improve outcomes. Additionally, HAGL lesions are rare, and PHAGL lesions account for only a small subset of these. As a result, the overall incidence of PHAGL lesions is estimated to be <1% of all shoulder instability cases. PHAGL lesions can be visualized on noncontrast magnetic resonance imaging and confirmed intraoperatively. Future studies with larger sample sizes, likely including data from multiple centers, and longer follow-up durations would be valuable for identifying risk factors for failure and recurrence.
Both groups reported favorable rates of return to sport, with most patients resuming athletic activities at their preinjury levels. Additionally, no statistical significance was identified between those with isolated PLTs and those with combined PHAGL lesions. Given the younger demographic and the nature of injury mechanisms in the PHAGL cohort, this is an encouraging finding. However, further research is necessary to determine whether PHAGL repairs affect long-term return-to-play outcomes, particularly in contact athletes.
This study is not without limitations. Glenoid version was not assessed, which can contribute to the development of posterior instability and affect outcomes. Differences in surgical technique and rehabilitation protocols may have influenced outcomes. Potential confounders such as activity level and injury chronicity could not be fully controlled despite cohort matching using rigorous criteria. Time from injury to surgery was not included in matching criteria, and increased delay to operative treatment could contribute to decreased preoperative PROs. Additionally, the retrospective design and small cohort limit the generalizability of our findings. A formal power analysis was not performed due to the extreme rarity of PHAGL lesions. To optimize statistical power with the limited cases available, we used a 1:3 matched cohort design, pairing each patient with PHAGL lesions with 3 controls. This study is prone to type II error as a result of its small sample size, but given the infrequency of PHAGL, accruing a substantially larger sample size would likely require decades of data collection. Future multicenter studies with larger patient populations may offer more definitive conclusions regarding PHAGL lesion management.
Conclusion
This study shows that patients with PHAGL lesions who undergo arthroscopic stabilization can attain comparable mid-term functional outcomes to those with isolated PLT. Failure rates are similar between the PLT group compared with those with PHAGL lesions, and surgical management remains an effective treatment option for both groups. Further studies are needed to optimize surgical techniques and refine patient selection criteria for improved long-term success.
Footnotes
Final revision submitted September 30, 2025; accepted October 27, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: A.K. has received education payments from Smith & Nephew, grants from DJO, and reimbursement from Exactech. A.J. has received grants from AGA outside of the submitted work sponsored by Arthrex. M.T.P. receives royalties from Arthrex, Arthrosurface, Responsive Arthroscopy, and Anika Therapeutics and has received consulting fees from Arthrex, Joint Restoration Foundation, Zimmer Biomet Holdings, and Arthrosurface; grants from the Department of Defense, the National Institutes of Health, and DJO; and honoraria from Flexion Therapeutics. P.J.M. receives royalties and has received consulting fees and research support from Arthrex and holds stock options in VuMedi. The positions of C.J.H., M.P.H., C.S.D., M.H., and A.J. are supported by the Steadman Philippon Research Institute, which is a 501(c)(3) nonprofit institution supported financially by private donations and corporate support. The Steadman Philippon Research Institute (SPRI) exercises special care to identify any financial interests or relationships related to research conducted here. During the past calendar year, SPRI has received grant funding or in-kind donations from Arthrex, Canon, DJO, Icarus Medical, Medtronic, Ossur, Smith & Nephew, SubioMed, Stryker, and Wright Medical. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Vail Health Hospital Institutional Review Board (ref: 2023-221).
