Abstract
Background:
Despite successful mechanical rotator cuff repair, the intrinsic quality of the tendon often remains poor, thereby increasing the risk of retear.
Purpose:
To investigate a proper differentiation of tenocytes from induced pluripotent stem cells (iPSCs) and a regenerative approach using iPSC-derived tenocytes (iPSC-TCs) combined with atelocollagen gel to enhance tendon healing after rotator cuff repair.
Study Design:
Controlled laboratory study.
Methods:
Tenogenic differentiation was induced in vitro from the human iPSC line 1231A3, with differentiation confirmed by cell morphology, flow cytometry, and immunofluorescence staining. The viability of iPSC-TC within 3% atelocollagen gel was analyzed. The Sprague Dawley rats were divided into 4 groups (n = 16 per group). Group 1 acted as the control without damage; group 2 with surgical repair of the fully torn supraspinatus; group 3 with additional injection of atelocollagen after the repair; and group 4 with additional injection of iPSC-TC embedded on atelocollagen after the repair. After tissue harvest at postoperative 6 weeks, quantitative evaluation of gene expression (messenger ribonucleic acid levels), immunohistochemistry (percentage of stained area), and Western blot (relative protein expression) was performed. All experimental results are reported as mean and error, and analyzed using the Student t test, the Mann-Whitney U test, and analysis of variance.
Results:
Differentiation to iPSC-TC was confirmed on day 19 of passage 3 with positive expression of cell surface markers and positive expression of tenogenic markers on immunofluorescence staining. Differentiated iPSC-TC showed significantly higher expression of tendon-related genes when compared with nondifferentiated iPSC. After the tissue harvest, PKH16-labeled iPSC-TC was identified only in group 4 under confocal microscopy. The Masson trichrome stain showed significantly greater collagen intensity in group 4 compared with the other repair groups (P < .05). Higher expression of type I collagen in group 4 was identified in both immunohistochemistry (P < .05) and Western blot analysis (group 1: 0.39 ± 0.07 vs group 4: 0.96 ± 0.27; P < .05).
Conclusion:
Our study demonstrates that an injection of atelocollagen containing iPSC-TCs into the lesion after rotator cuff repair produced excellent residual effects. The addition of iPSC-TCs embedded in atelocollagen to a surgically repaired rotator cuff could improve tendon quality by increasing type I collagen production.
Clinical Relevance:
The combination of atelocollagen with induced pluripotent stem cell-derived tenocytes could help tendon healing and thus potentially reduce retear. This approach may have the potential to improve the biological quality of repaired rotator cuffs, reduce the risk of retear, and ultimately enhance long-term surgical outcomes in patients with tendon injuries.
Unlike most tendon injuries that result from acute trauma, rotator cuff tears are primarily influenced by chronic degenerative changes within the tendon.5,6,14,32,34 This high prevalence, combined with the degenerative nature of these injuries, creates substantial treatment challenges. Because of the chronicity, existing treatment modalities for rotator cuff tears face practical limitations in achieving true tendon regeneration. Despite the constant development of surgical apparatus and skills, high retear rates are still reported from relatively large cuff tears, affecting up to 20% to 94% of patients depending on tear size, not only due to mechanical problems caused by loaded tension on the repaired tendon, but also due to incomplete restoration of the tendon-to-bone interface and the lasting and unchanged damaged condition of the repaired tendon itself.9,12 This problem is also seen in the treatment of partial-thickness tears, which represent approximately 25% to 50% of all rotator cuff pathology and are not indications for surgical repair, as most nonoperative treatment modalities focus on symptom relief while fundamental treatments that may recover or regenerate the tendon are lacking. 4
Among the various modalities aimed at enhancing the healing potential of torn tendons, atelocollagen therapy has recently gained attention and is increasingly used in the treatment of rotator cuff tears.2,8,11,15-17 Atelocollagen is a modified collagen of which terminal telopeptides are removed to lower the immunogenicity.23,24 Even though type I collagen is the main component of the extracellular matrix (ECM) in the tendon, simply providing a collagen supply cannot overcome the disadvantages of an acellular treatment and thus has a clear limitation in promoting actual regeneration. Tenocytes are known to be the main sources of collagen production within the tendons, and the application of collagen in combination with tenocytes could hold enormous potential for tissue regeneration. 7 However, tendon tissues are generally hypocellular, and tenocytes are unlikely to be present as a uniform population, making it a challenge to acquire as many native tenocytes as are needed in clinical practice. 30
In this study, the induced pluripotent stem cell (iPSC) technique was used to generate tenocytes.18,33 This study aimed to evaluate whether iPSC-derived tenocytes (iPSC-TCs), delivered within a commercially available injectable atelocollagen gel during surgical repair of full-thickness rotator cuff tears in a rat model, could survive the tendon-to-bone healing process, function as native tenocytes, and promote superior type I collagen production and tendon regeneration compared with atelocollagen alone or repair alone. We hypothesized that iPSC-TCs within the injected atelocollagen will promote superior type I collagen production and tendon regeneration over atelocollagen alone or repair alone.
Methods
Differentiation of Tenocytes From iPSCs
The human iPSC line 1231A3 was established at YipCell Corporation. 25 The iPSCs were maintained under feeder-free and xeno-free conditions. Briefly, the cells were cultivated on iMartix-511 (Nippi; 0.5 μg/cm2)-coated dishes with StemFit AK03N (AK03) (Ajinomoto) medium and passaged into single cells by dissociation using TrypLE Express (Life Technologies Corp). Before tenocyte induction, the iPSCs were seeded onto iMartix-511-coated dishes (1.1 × 103 cells/cm2) and cultured in AK03 medium for 3 days. The iPSCs were then differentiated into presomatic mesoderm in AK03 medium supplemented with 10 μM SB431542 (Sigma; a TGF-β/Activin inhibitor), 10 μM CHIR99021 (Sigma; a glycogen synthase kinase 3β inhibitor activating Wnt signaling), 2 μM DMH1(Sigma; bone morphogenic protein inhibitor), and 20 ng/mL fibroblast growth factor 2 (FGF2) (PeproTech; a fibroblast growth factor promoting cell proliferation and differentiation) for 4 days. For somite differentiation, the 4-day presomatic mesodermal cells were reseeded onto iMartix-511-coated dishes (1.8 × 104 cells/cm2) and cultured in AK03 medium supplemented with 10 μM SB431542 and 5 μM CHIR99021 for four days. On day 8 of differentiation, the somites were cultured in AK03 containing 100 nM smoothened agonist (SAG) (Sigma; smoothened agonist activating Hedgehog signaling) and 0.6 μM LDN193189 (Sigma; a BMP type I receptor inhibitor) for sclerotome differentiation. The 11-day sclerotome cells were detached from the dish using Accutase, seeded onto iMatrix-511-coated dishes (1.8 × 104 cells/ cm2), and subsequently differentiated toward syndetome in AK03 medium supplemented with 10 ng/mL FGF8 (PeproTech; a fibroblast growth factor involved in somite development) and 10 ng/mL transforming growth factor β3 (TGFβ3) (PeproTech; a transforming growth factor promoting tenogenic differentiation) for the first 2 days and in AK03 medium supplemented with 10 ng/mL TGFβ3 and 10 ng/mL bone morphogenic protein 7 (BMP7) (PeproTech; a bone morphogenetic protein supporting tendon matrix formation) for the next 6 days.
Flow Cytometric Analysis
To confirm the surface antigen markers of the cells, the iPSC-TCs were washed with phosphate-buffered saline (PBS) containing 1% fetal bovine serum (FBS) and then adjusted to a volume of 100 μL. After 5 μL of fluorescent antibodies (CD34, CD45, CD73, and CD105; eBioscience) were added and mixed, the cells were rewashed with PBS and incubated in the dark for 45 minutes at 2° to 8°C. Then, 1 × 106 cells were added to 1 mL of PBS containing 1% to 2% FBS, mixed, put into a fluorescence-activated cell sorter (FACS) tube, and analyzed on a FACS Aria Fusion (BD Bioscience).
In Vitro Immunofluorescence Staining
iPSC-TCs were seeded in a 4-well culture slide (2 × 104 cells per well), cultured for 3 days, fixed with 4% paraformaldehyde for 10 minutes, washed with PBS, and treated with 0.1% Triton X-100 for 8 minutes at 4°C. To prevent nonspecific antigen-antibody binding after the wash with PBS, a reaction with 10% normal horse serum (s-1000, Vector Laboratories, Inc) was conducted for 1 hour, followed by overnight treatment with antibodies for scleraxis (ab58655, 1:100; Abcam), tenomodulin (ab203676, 1:100; Abcam), type I collagen (NB600-450, 1:100; Novus Biologics), vimentin (1:100 Sigma-Aldrich), and octamer-binding transcription factor 4 (OCT4, 1:100; Sigma-Aldrich; Merck Millipore) at 4°C.
The cells were treated with a biotin-attached secondary antibody, biotinylated antimouse rabbit IgG (1:100, Vector Laboratories, Inc), for 1 hour at room temperature, then washed. Next, they were treated with streptavidin Alexa 488/595 (1:1000, Life Technologies, Invitrogen) for 1 hour at room temperature, then washed again. A confocal laser scanning microscope (LSM800; Carl Zeiss) was used for the evaluation.
Assay for Cell Viability
iPSC-TCs were cultured in 24-well plates (1.6 × 104 cells per well) for 24 and 48 hours. Cell viability was measured using the RealTime-Glo cell viability assay (Promega; G9712), which quantifies cell reducing power via light emission. After the mitochondrial cell-viability substrate and NanoLuc enzyme were dissolved in a 37°C water bath, the substrate (1000×) and enzyme (1000×) were diluted 1:500 and mixed in the cell medium. Then 1.6 × 104 iPSC-TCs were dispensed. After 24 and 48 hours, 2X RealTime-Glo reagent was added, and after 1 hour of reaction at 37°C, luminescence was measured with a fluorometer (Synergy MX; BioTek). The assay was performed in 3 independent experiments, each conducted in triplicate, to confirm reproducibility.
In Vitro RT-PCR
Ribonucleic acid (RNA) extraction from the iPSCs and iPSC-TCs was performed using Trizol (Ambion; Life Sciences). Also, 1 μg of RNA was synthesized into complementary deoxyribonucleic acid (cDNA) using an iScript cDNA synthesis kit (Bio-Rad Lab). Real-time polymerase chain reaction (RT-PCR) was performed using a CFX96 Touch Real-Time PCR Detection System (Bio-Rad Laboratories). Relative messenger RNA (mRNA) levels were calculated using CFX manager software and the 2–ΔΔCT method. Gene-specific oligonucleotide sequences are listed in Table 1. Gene expression levels were normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH), and relative expression was calculated as fold change compared with the control group. All experiments were performed in 3 independent replicates, each conducted in triplicate.
Primer Sequences for the In Vitro RT-PCR a

RT-PCR, reverse transcription polymerase chain reaction.
Octamer-binding transcription factor 4.
Fibromodulin.
Mohawk homeobox.
Glyceraldehyde 3-phosphate dehydrogenase.
Surgical Procedures and Tissue Harvesting
The Institutional Animal Care and Use Committee of our institution approved this study. (CUMC-2022-0133-07). Twelve-week-old Sprague Dawley rats (n = 64), with a mean weight of 400 to 500 g, were used. The rats were randomly assigned to 1 of 4 groups (n = 16 per group): group 1 received no treatment and acted as the control; group 2 received full-thickness rotator cuff tears with consequent surgical repair only; group 3 received the same rotator cuff injuries and repair plus an injection of atelocollagen; and group 4 received the same rotator cuff injuries and repair plus an injection of iPSC-TCs cultured in atelocollagen (Figure 1).

Diagram showing group allocation and study design. iPSC-TC, induced pluripotent stem cell-derived tenocytes.
The rats allocated to groups 2, 3, and 4 were anesthetized with an intraperitoneal injection of ketamine (75-95 mg/kg) and xylazine hydrochloride (5 mg/kg). All operations were performed using a sterile technique. After the skin was shaved and sterilized, an incision was made at the shoulder along with deltoid splitting. The anterior margin of the supraspinatus was identified adjacent to the biceps tendon, and the posterior margin was determined by the junction with the infraspinatus tendon fibers (Figure 2A). The tendon was sharply dissected from the greater tuberosity to establish a full-thickness tear that exposed the humeral head and the cut end of the torn tendon. Subsequent repair was done with 5-0 Ethibond and the Mason-Allen technique (Figure 2B). Group 3 received an injection of 0.5 mL of pure 3% atelocollagen at the repaired tendon-to-bone interface, and group 4 received an injection of iPSC-TCs embedded in atelocollagen. The tendons were harvested 6 weeks after surgery, which was considered enough time for the manipulated rotator cuff injury in a rat model to have healed. 20 Through the same incision, the remaining supraspinatus was exposed and harvested from the muscle origin, along with an osteotomy of the humerus mid-shaft without damaging the tendon insertion. All surgical procedures and tissue harvesting were performed by a single surgeon (H.J.L.).
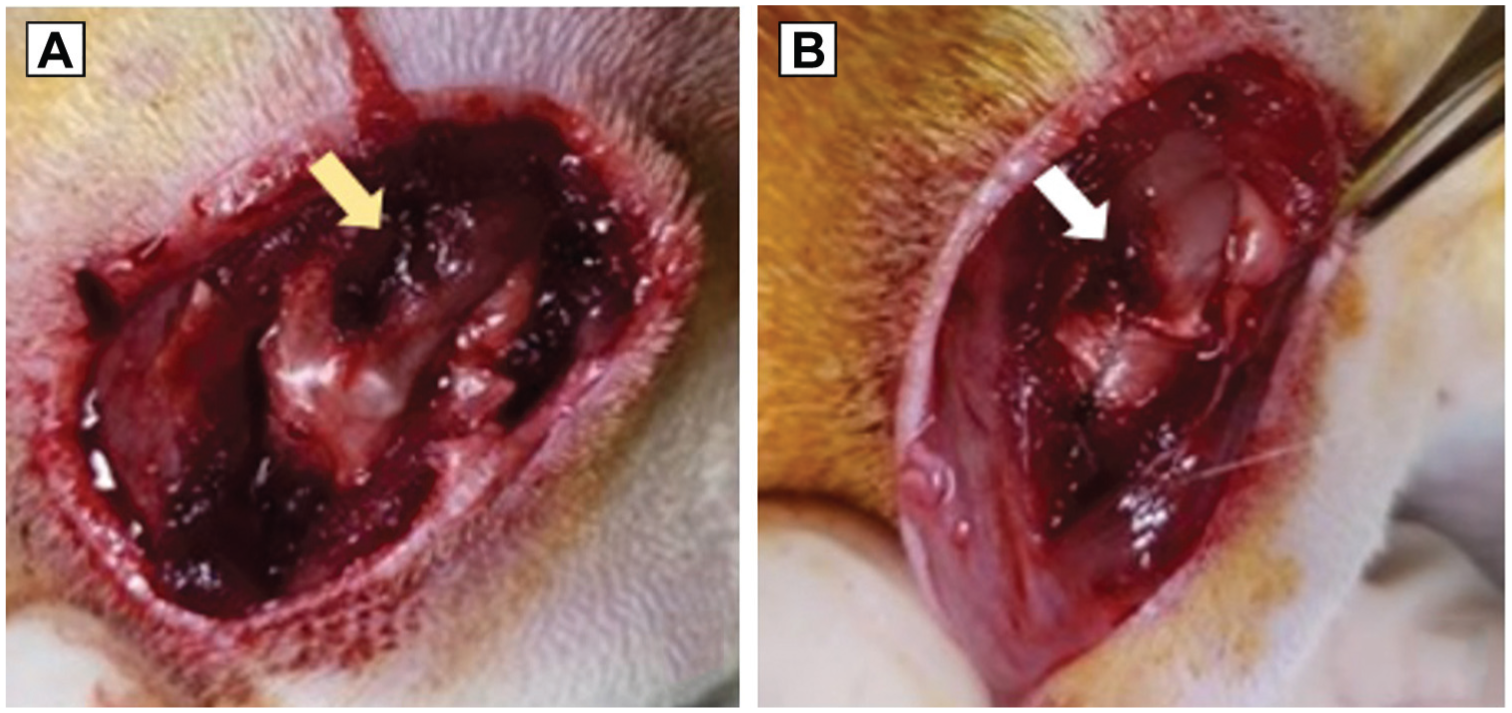
Rotator cuff injury in a 12-week-old Sprague Dawley rat. (A) Full-thickness tear (yellow arrow indicates the tear site). (B) Repair of the torn tendon using the Mason-Allen suture technique (with arrow indicating the repair site).
In Vivo Fluorescence Spectrometry
PKH26-labeled iPSC-TCs mixed with 50 μL of atelocollagen were injected into the rotator cuffs of 16-week-old male Sprague-Dawley rats. Samples were collected 6 weeks after the injection. Fluorescence expression (fluorescence excitation range: 400 to 750 nm; emission range: 500-875 nm) was measured on an in vivo imaging system (IVIS) (Lumina XRMS; PerkinElmer Inc). In vivo fluorescence was analyzed using Living Image software, Version 4.4 (Revvity), with an IVIS imaging system. Regions of interest (ROIs) were defined at the experimental sites, and the mean fluorescence intensity within each ROI was measured. The raw intensity values were used for quantitative comparison between groups.
In Vivo RT-PCR
RNA was extracted from iPSCs and iPSC-TCs using Trizol (Ambion; Life Sciences). Also, 1 μg of RNA was synthesized into cDNA using an iScript cDNA synthesis kit (Bio-Rad Lab). RT-PCR was performed using a CFX96 Touch (Bio-Rad Lab). Relative mRNA levels were calculated in CFX Manager software using the 2–ΔΔCT method. 26 The values obtained for each gene were normalized to the housekeeping gene, hypoxanthine guanine phosphoribosyl transferase, and the uninjured control. Gene-specific oligonucleotide sequences are listed in Table 2.
Primer Sequences for the In Vivo RT-PCR a

HPRT, Hypoxanthine-guanine phosphoribosyltransferase; RT-PCR, reverse transcription polymerase chain reaction.
Western Blot Analysis
Proteins were extracted using a protease inhibitor cocktail (Xpert Protease Inhibitor Cocktail Solution, GenDEPOT) combined with radioimmunoprecipitation assay analysis buffer (Cell Signaling Technology). The extracted protein in each sample was quantified using a Pierce Bicinchoninic Acid (BCA)Protein Assay Kit (Thermo Scientific) and subjected to 8% sodium dodecyl sulfate polyacrylamide gel electrophoresis. The proteins were transferred to a nitrocellulose membrane and reacted with Ponceau S (Amresco; OH44139) solution for about 5 minutes. The confirmed nitrocellulose membrane was then left in 5% bovine serum albumin for 1 hour. The primary antibodies for glyceraldehyde 3-phosphate dehydrogenase (GTX100118; 1:10000, GeneTex), scleraxis (ab5865, 1:500; Abcam, MA02453), tenomodulin (ab203676, 1:2000; Abcam, MA02453), type I collagen (NB600-450, 1:1000; Novus Biologicals, CO 80112), and type III collagen (NBP-05119, Novus Biologicals, CO 80112) were administered and left overnight at 4°C. Then, the samples were washed with peroxidase-labeled anti-mouse and anti-rabbit antibodies at room temperature for 1 hour. After detection with a SuperSignal West Pico chemical substrate kit, the results were quantified using a PXi4 image analyzer (Syngen) and GeneTools software. Full-length/uncropped lots are provided in Appendix Figure A1.
The Masson Trichrome Stain
Slides were deparaffinized with xylene and washed. Then, the Bouin solution at 60°C was used as a mordant for 1 hour. The samples were rewashed, Weigert’s hematoxylin solution was used to stain the nuclei, and the samples were washed with distilled water once again. Next, the samples were stained with Biebrich scarlet-acid fuchsin solution for 5 minutes, washed with distilled water, soaked in phosphotungstic/phosphomolybdic acid solution for 10 minutes, and shaken off. The slides were then left in aniline blue solution for an additional 10 minutes. They were washed with distilled water, treated with 1% acetic acid for 1 minute, dehydrated, and sealed. The slides were scanned with a slide scanner (Pannoramic SCAN II, 3D Histech), and the amounts of stained factors were expressed quantitatively using an automated method. Positively stained pixels were digitally selected on all slides, and the immunoreactivity positivity ratio for each slide was calculated as the total number of positively stained pixels divided by the total number of pixels.
Immunohistochemical Analysis
After the samples were fixed with 10% formalin, they were washed, dehydrated, and embedded with paraffin. The tissue block was cut into 4-μm-thick sections, which were then mounted on slides. After 1 hour on a 60°C slide warmer, each deparaffinized and rehydrated sample was heated twice for 5 minutes in a microwave with 10 mM citrate buffer, pH 6.0, to express the antigen in the tissue, then washed with PBS. To inhibit endogenous peroxidase activity, samples were incubated with Dako REAL peroxidase-blocking solution (Agilent Tech) for 15 minutes. To prevent nonspecific antigen-antibody binding, samples were reacted with 10% normal horse serum (s-1000, Vector Laboratories, Inc) for 1 hour. The primary antibodies, collagen types I and III (NB600-450, NBP-0519, Novus Biologics), were diluted 1:100 and incubated with the samples overnight at 4°C. The next day, the secondary antibody, EnVision + System-horseradish peroxidase–labeled polymer anti-mouse (Dako), was added and left for 30 minutes at room temperature. After reaction with diamino-benzidine (Vector Laboratories, Inc) for 5 to 10 minutes, the samples were washed with water and counterstained with hematoxylin QS (Vector Laboratories, Inc). Surgipath MM24 mounting medium (Leica Biosystems Richmond, Inc) was used to seal the slides, and they were observed with an established microscope (AX70, TR-62A02; Olympus) and scanned with a slide scanner (Pannoramic SCAN II, 3D Histech). The same method was used as the Masson trichrome stain for quantitative analysis.
Statistical Analysis
A power analysis was conducted to estimate the minimum sample size required to detect a statistically significant difference in type I collagen expression among 4 experimental groups in the rat rotator cuff tear model. Based on previous literature and pilot observations, a medium effect size (f = 0.4) was anticipated for the primary outcome measure (collagen type I protein expression as measured by densitometry from Western blot analysis). 12 The calculation assumed an alpha level (α) of .05 and a desired statistical power (1 –β) of 0.80. Under these assumptions, the required total sample size was 32 rats, with at least 8 per group, sufficient to detect statistically significant differences using 1-way analysis of variance (ANOVA). All experimental results are reported as mean ± error, and the data were statistically processed. Statistical analyses were performed using the Student t test, the Mann-Whitney U test, and ANOVA. P < .05 was considered significant. All statistical measurements were performed using SPSS Statistics 21 (IBM).
Results
At passage 3 (day 19), confluent iPSC-TCs showed the longish, slender morphologic characteristics typical of tenocytes, unlike the iPSCs at passage 1 (Figure 3).

Tenogenic differentiation process from induced pluripotent stem cells. (A) 40× on day 3. (B) 100× on day 3. (C) 40× on day 7. (D) 100× on day 7. (E) 40× on day 11. (F) 100× on day 11. (G) 40× on day 19. (H) 100× on day 19. Scale bar = 100 μm.
Tenogenic differentiation of iPSCs was confirmed in various ways. In the flow cytometric analysis of cell-surface marker expression, the iPSC-TCs were positive for CD73 and CD105 and negative for CD34 and CD45 (Figure 4).
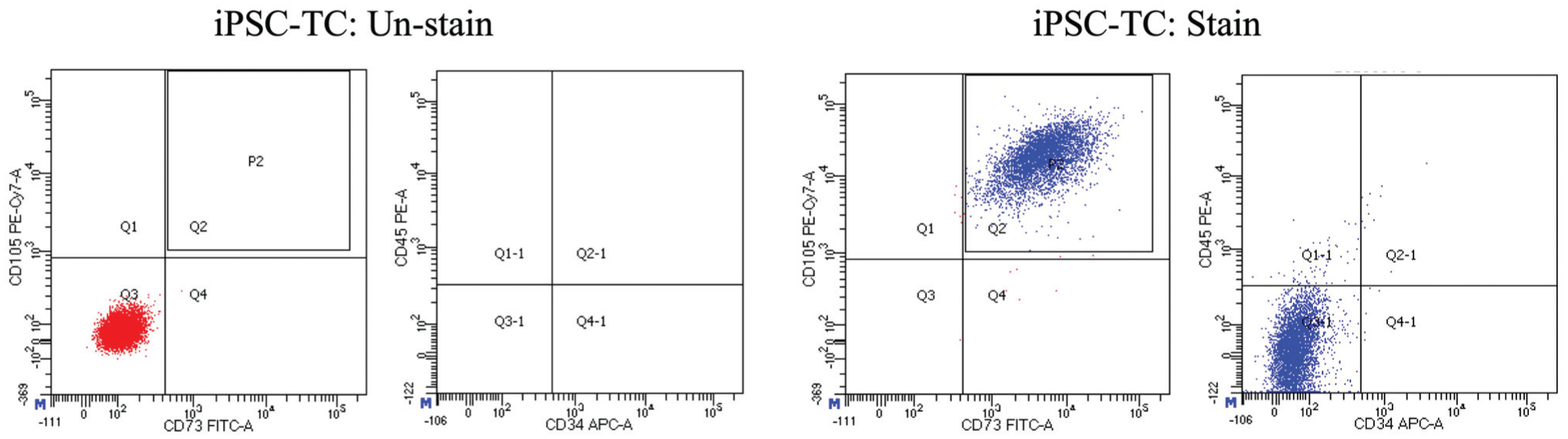
Flow cytometric analysis: The expression of cell surface markers on iPSC-TCs. APC, allophycocyanin; CD, cluster of differentiation; FITC, fluorescein isothiocyanate; iPSC, induced pluripotent stem cell; iPSC-TCs, iPSC-derived tenocytes. PE-Cy7, phycoerythrin-cyanine 7.
Differentiation from iPSCs to tenocytes was confirmed by in vitro immunofluorescence staining. The iPSC-TCs expressed collagen type I, vimentin, scleraxis, and tenomodulin and did not express OCT4. The opposite result was found with undifferentiated iPSCs (Figure 5).

In vitro immunofluorescence staining. Cells were treated with specific antibodies (red or green), counterstained with 4′,6-diamidino-2-phenylindole (DAPI) (blue), and visualized under a confocal laser scanning microscope. iPSC, induced pluripotent stem cell; iPSC-TC, iPSC-derived tenocyte; OCT4, Octamer-binding transcription factor 4. Scale bar = 20 μm.
In the analysis of the cell viability assay, the iPSC-TCs showed significantly higher luminescence levels in the collagen-added medium, which can be interpreted to indicate that collagen did not harm the activity of the iPSC-TCs because the levels of cell viability and luminescence are proportional (Figure 6).

Assay for cell viability. Cell viability was measured using the RealTime-Glo cell viability assay (Promega; G9712), which quantifies cell reducing power via light emission. Significantly higher luminescence was detected in the medium containing atelocollagen and iPSC-TCs. Error bars indicate standard error. Asterisks indicate statistically significant differences between groups (P < .05). iPSC, induced pluripotent stem cell; iPSC-TC, iPSC-derived tenocyte.
The expression of tendon-related genes (fibromodulin, Mohawk homeobox, type I alpha 1 and 2 collagen, scleraxis, and tenomodulin) was significantly higher in the differentiated iPSC-TCs than in the undifferentiated iPSCs. Meanwhile, the expression of OCT4, a marker of undifferentiated iPSCs, was significantly lower in the iPSC-TCs than in the iPSCs (Figure 7).

In vitro comparison of gene expression levels between iPSCs and differentiated iPSC-TCs. The expression of tendon-related genes was significantly higher in iPSC-TCs than in iPSCs. The expression of OCT4, a marker of undifferentiated iPSCs, was significantly lower in the iPSC-TCs than in the iPSCs. Error bars indicate standard error. Asterisks indicate statistically significant differences between groups (P < .05). FMOD, fibromodulin; iPSCs, induced pluripotent stem cells; iPSC-TCs, iPSC-derived tenocytes; MKX, Mohawk homeobox; OCT4, octamer-binding transcription factor 4.
The presence of iPSC-TCs in the rat tendons was confirmed using an in vivo fluorescence spectrometric analysis. An accumulation of PKH26-labeled iPSC-TCs was detected in the repaired rat tendons from group 4 after tissue harvest, whereas no such accumulation was observed in the other groups. Quantification of radiance efficiency among the groups also showed a significant difference only in group 4 (Figure 8).
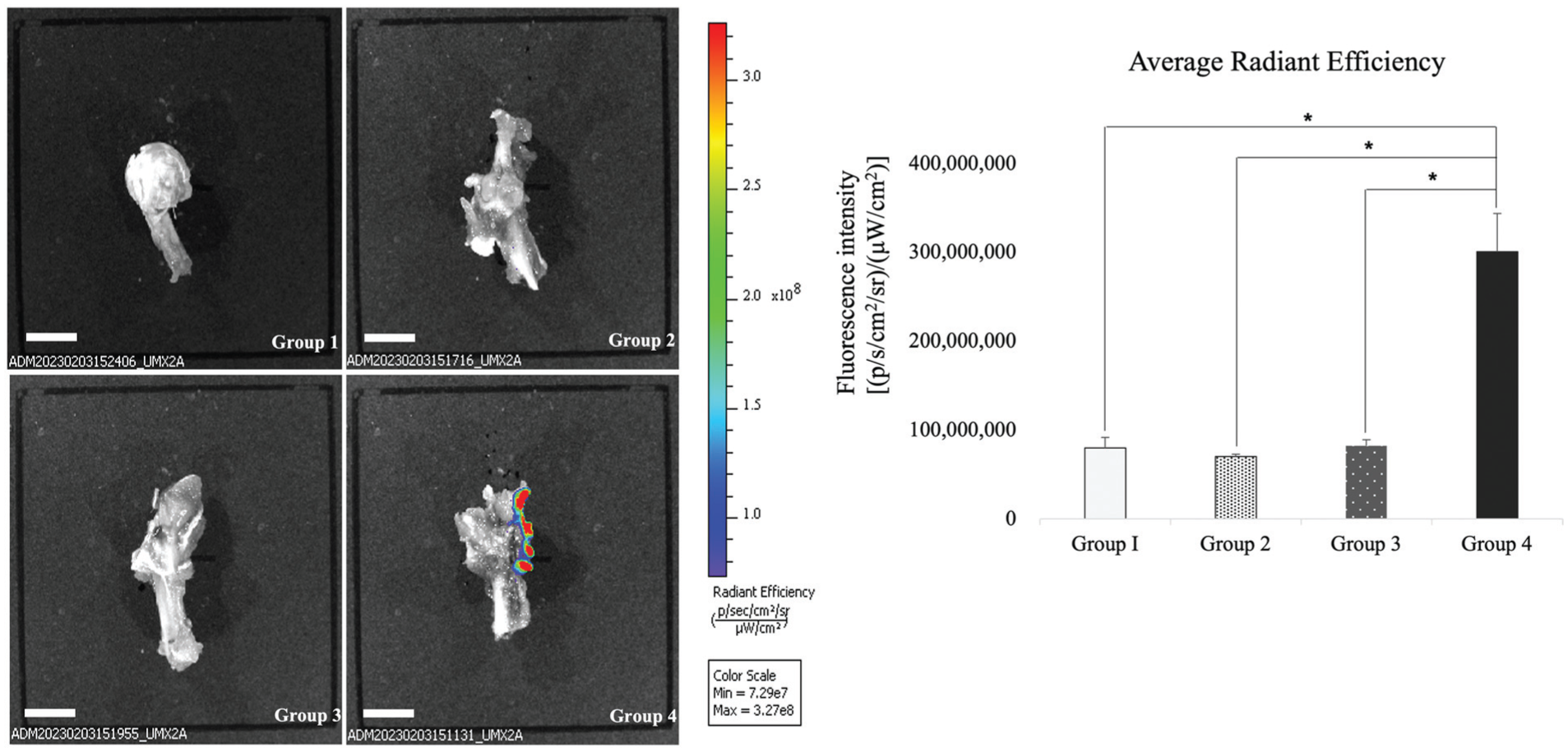
In vivo fluorescence spectrometry and the analysis of average radiant efficiency. Spectrometric analysis confirmed the presence of iPSC-TCs in rat tendons. An accumulation of PKH26-labeled iPSC-TCs (Pharmacia kit for hemocytometry) was detected in the repaired rat tendons of group 4 after tissue harvest, whereas no such accumulation was observed in the other groups. Quantification of radiance efficiency among the groups also showed a significant difference only in group 4. Error bars indicate standard error. Asterisks indicate statistically significant differences between groups (P < .05). Scale bar = 4 mm. iPSC-TC, induced pluripotent stem cell-derived tenocyte.
The gene expression analysis also showed differences among the groups. The expression of tenomodulin was highest in group 4, and the difference from group 2 was statistically significant. The expression of scleraxis, tenascin C, and type III collagen was not the highest in group 4. However, type I collagen expression was markedly and significantly higher in group 4 than in the other groups (Figure 9).

In vivo RT-PCR. Col1 expression was significantly higher in group 4 than in the other groups. The expression of Tnmd was highest in group 4, and the difference from group 2 was statistically significant. Error bars indicate standard error. Asterisks indicate statistically significant differences between groups (P < .05). Col1, type 1 collagen; Col3, type 3 collagen; RT-PCR, reverse transcription polymerase chain reaction; SCXA, scleraxis; TNC, tenascin C; Tnmd, tenomodulin.
Similar to the RT-PCR results, the presence of proteins related to tendon activity also differed among the groups. Western blotting for type I collagen showed that group 4, which received iPSC-TCs, had higher expression than the other groups that did not. The difference from the control group was statistically significant. Furthermore, the atelocollagen-treated groups (3 and 4) tended to have higher levels of type I collagen. However, type III collagen expression was highest in group 2, which received no treatment other than simple surgical repair of the torn cuff (Figure 10).

(A) Western blots (cropped) showing type I and III collagen expression in the repair sites of each group. Densitometry of types I (B) and III (C) collagen from the Western blotting assay. Group 4 had the highest expression of type I collagen. The groups treated with atelocollagen (groups 3 and 4) had significantly higher type I collagen expression than group 1. Error bars indicate standard error. Asterisks indicate statistically significant differences between groups (P < .05). The expression of type III collagen was highest in group 2, which received no treatment except for simple surgical repair of the torn cuff.
The Masson trichrome staining showed collagen-fibril formation in the tendon-bone junction. The control group, with uninjured tendons, showed concentrated and orderly collagen fibrils with high density, and the groups that did not receive iPSC-TCs (groups 2 and 3) showed loose collagen fibrils within the tendon and junction. According to the calculation of stained areas (tendon area only), the expression of collagen fibrils within the tendon was significantly higher in group 4 than in groups 2 and 3, which did not receive additive cell therapy. Group 4, which received iPSC-TCs, had high collagen fibril expression and an orderly pattern comparable to that of the control group (Figure 11).

The Masson trichrome analysis. Group 4, which received iPSC-TCS, showed concentrated and orderly collagen fibrils with high density comparable to those of group I with the uninjured tendon. Whereas the groups that did not receive iPSC-TCs (groups 2 and 3) showed relatively loose collagen fibrils within the tendon and junction. Error bars indicate standard error. Asterisks indicate statistically significant differences between groups (P < .05). According to the calculation of stained areas (tendon area only), the expression of collagen fibrils was significantly higher in group 4 than in groups 2 and 3. Scale bar = 200 μm.
Immunohistochemical staining confirmed that group 4, which received iPSC-TCs and atelocollagen, had the highest type I collagen expression, and that the difference from group 2, which received no additional cell treatment, was significant. However, the expression of type III collagen did not differ significantly among the groups (Figure 12).

Immunohistochemical staining results. Group 4, which received iPSC-TCs and atelocollagen, had the highest type I collagen expression, and the difference from group 2, which received no additional cell treatment, was statistically significant. The expression of type III collagen did not differ significantly among the groups. Error bars indicate standard error. Asterisks indicate statistically significant differences between groups (P < .05). Scale bar = 200 μm.
Discussion
The major findings of this study are as follows: First, tenogenic differentiation of iPSCs produced cells that exhibited the morphological characteristics of native tenocytes and expressed appropriate tenocyte-specific markers. Multiple validation methods were employed to confirm proper differentiation into iPSC-TCs and minimize residual undifferentiated iPSCs. Second, cell survival assays demonstrated that a commercially available atelocollagen gel effectively served as both a medium and scaffold for the iPSC-TCs. Third, the differentiated iPSC-TCs functioned as native tenocytes, as evidenced by the de novo production of type I collagen, the principal component of the tendon extracellular matrix.
The principle of atelocollagen treatment for tendon damage focuses on defect filling, structural support of the tissue, and enhancement of regeneration by supporting existing cells.2,10,17 This theoretical background has been supported by numerous basic and clinical studies that have led to the actual application of atelocollagen in regenerative medicine. However, single-injection atelocollagen therapy has the clear disadvantage of lacking a cellular component. In fact, the therapeutic mechanism for an injured tendon mainly depends on ECM production, and tenocytes are the main source of collagen, which forms 95% of ECM.1,3
The difficulty in isolating and harvesting native tenocytes has limited their clinical application to damaged tendons. Despite advances in tendon tissue engineering, a donor-site defect is an inevitable consequence of harvesting autologous tenocytes. To solve the problem, stem cells have been used and observed in various tendon pathologies. Stem cell-based therapy has a long history and is a promising regenerative strategy for various musculoskeletal problems, but its ethical and safety issues remain a barrier to clinical applications. Many attempts have been made to address the fundamental problems with stem cell techniques, and the newly introduced iPSCs are completely free from the ethical issues associated with human embryonic stem cells. Another advantage of the iPSC technique is its ease of accessibility and expandability. 33 With the growing clinical adoption of the iPSC technique, several protocols for tenogenic differentiation from iPSCs have been reported; however, only a few studies have evaluated their in vivo validity in tendon disease models.19,25 In fact, our experiments are the first to combine iPSC-related therapy with an existing clinical treatment in a synergistic way to treat rotator cuff tears.
In this study, rather than directly applying iPSCs in vivo, we differentiated them into tenocytes in vitro to minimize the risk of unexpected ectopic transcription and subsequent neoplastic development. The in vitro differentiation and subculture of iPSCs provide a tractable environment for examining differentiation into the target cell. In considering the clinical application of stem cells, including iPSCs, we were acutely aware of potential safety risks, particularly the persistence of undifferentiated cells or improper lineage specification, which could lead to severe adverse events such as teratoma formation, ectopic tissue generation, or dysregulated extracellular matrix deposition. To address these concerns, we conducted multiple in vitro verification steps before transplantation. These included confirming the absence of pluripotency-associated markers, validating the expression of tendon-specific genes and proteins, and ensuring stable phenotypic characteristics under culture conditions. Only after these safety criteria were met did we proceed to in vivo experiments. In fact, according to Lui et al, 22 predifferentiating mesenchymal stem cells into tendon-forming cells before transplantation was proposed as a potential strategy to improve outcomes and reduce risks associated with undifferentiated mesenchymal stem cell transplantation.
An important outcome of this study is that iPSC-derived tenocytes not only expressed specific tendon markers but also produced type I collagen, a major structural component of tendon tissue. This additional collagen formation was confirmed through morphological and quantitative comparisons with control groups that did not receive iPSC-TCs. Collagen expression in regenerated tissues was analyzed using RT-PCR, Western blotting, and immunohistochemistry. Among all groups, group 4, which received iPSC-TCs in combination with atelocollagen, consistently exhibited the highest levels of type I collagen expression across all analytical methods. These findings underscore both the therapeutic promise of iPSC-derived tenocytes and the necessity of rigorous preclinical safety screening to mitigate theoretical risks before clinical application. While not all results have reached statistical significance, the consistency of this finding across different analytical methods underscores its importance. The expression pattern of type III collagen, however, varied among other analyses. Notably, group II, which received only surgical repair without additional treatment, showed the highest levels of type III collagen—even exceeding its own type I collagen levels in Western blot analysis. This finding is particularly relevant because treatments aimed at improving tendon healing typically focus on promoting the transition from type III to type I collagen to enhance tendon strength and functionality. 21 Excessive or prolonged production of type III collagen can result in weaker scar tissue, increasing the risk of reinjury. 29 According to Wang, 31 an increased ratio of collagen type I to type III is considered a hallmark of tendon maturation and biomechanical integrity, as type I collagen fibers exhibit greater alignment and stiffness compared with the more randomly organized and extensible type III fibers. The results suggest that the combination of atelocollagen (primarily type I collagen) and iPSC-TC promotes tendon healing by modulating collagen expression patterns.
A few recent studies have reported the successful differentiation of tenocytes from iPSCs for the treatment of various tendon conditions. Komura et al 19 succeeded in tenogenic differentiation of iPSCs from mice, and the differentiated cells showed tenocyte-like properties and promoted tendon regeneration after transplantation into injured tendons of mice. Similar results were reported by Kaneda et al, 13 who used iPSC-derived tenocytes seeded on 3-dimensional printed scaffolds to treat an Achilles tendon defect in rats. Our study extends previous work on the treatment of rotator cuff tears and shows promising results.
Various in vitro and in vivo experiments were conducted to confirm proper differentiation of iPSC-TCs and demonstrate their competence as functional tenocytes. Undifferentiation was completely ruled out by the confirmation that OCT4 was absent in vitro, and proper differentiation to tenocytes was confirmed by detecting the expression of various tenogenic markers, specifically fibromodulin, Mohawk homeobox, collagen, scleraxis, and tenomodulin. After treatment, the iPSC-TCs’ function was assessed by comparing the expression of various tenocyte-related markers across groups. The RT-PCR, Western blot, Masson trichrome, and immunohistochemistry analyses consistently showed an increase in type I collagen in group 4, and it can be concluded that the iPSC-TCs were the sole factor responsible for this difference. The absence of OCT4 expression indicates the elimination of residual undifferentiated iPSCs, thereby reducing the risk of tumorigenicity and confirming cellular safety, while the expression of tenogenic markers and the enhanced production of type I collagen demonstrate functional efficacy. Together, these results support the use of iPSC-TCs combined with atelocollagen as a promising therapeutic option for rotator cuff tear repair.
Despite the novel findings of this study, it has a few limitations. First, only a biological evaluation was conducted to demonstrate the effect of the iPSC-TCs on surgically repaired tendon quality. Given the environmental similarities between the supraspinatus in rats and humans, sufficient objectivity regarding disease etiology and molecular biology is guaranteed; however, biomechanical testing on the relatively small rat tendons is bound to be inadequately objective when compared with the tendons of a larger animal.27,28 Still, an additional biomechanical evaluation would have added objectivity and confirmed the benefit of the collagen produced by the iPSC-TCs. Second, we designed this experiment to analyze data collected 6 weeks after surgery, as our original purpose was to elucidate the effects of iPSC-TCs on the early healing process of injured tendons. Although we have shown what happened as a result of the iPSC-TCs grafted within the tendon, the long-term effects of iPSC-TCs on tendon healing and whether the cells can survive within the atelocollagen and the repaired tendon for >6 weeks were not evaluated in this study. Also, to clearly reveal the mechanism and influence of one factor on another, periodic analysis is needed to compare differences across time points. We analyzed responses after tendon repair at a single time point and thus cannot explain the cascade of reactions in the torn cuff. However, this 1-time analysis was objective enough to verify the existence of certain reactions to the injury and treatment. Third, we used a rat model of acute rotator cuff injury in this study. Although it shares many anatomical features with human cuff tendons, it is not possible to reproduce the same chronic process that leads to rotator cuff tears in humans. These histological alterations and manipulations in rats cannot be precisely adapted to the differences from humans in movement and ambulation. Fourth, the conclusions about tendon regeneration may not apply to enthesis (tendon-to-bone interface) regeneration. Fifth, while type I collagen was analyzed as a key marker of extracellular matrix production and tendon regeneration, other collagen subtypes that play important roles in tendon healing were not examined, limiting the comprehensiveness of our evaluation of the healing process.
This work provides a novel insight: tenocytes derived from iPSCs can produce type 1 collagen, a key component of tendon healing and formation. Future work will elucidate the biomechanical properties acquired by the functioning of iPSC-TCs and potentially lead to practical and clinical applications of the combination of atelocollagen and iPSC-TCs to promote the healing of torn rotator cuffs. An injection of atelocollagen containing iPSC-TCs into the lesion after rotator cuff repair produced excellent residual effects. Adding iPSC-TCs in atelocollagen to a surgically repaired rotator cuff could contribute to tendon healing by increasing the production of type I collagen.
Conclusion
Our study demonstrates that an injection of atelocollagen containing iPSC-TCs into the lesion after rotator cuff repair produced excellent residual effects. The addition of iPSC-TCs embedded in atelocollagen to a surgically repaired rotator cuff could improve tendon quality by increasing type I collagen production.
Footnotes
Appendix
Final revision submitted October 1, 2025; accepted October 27, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: This work was supported by the National Research Foundation of Korea grant funded by the Korean government (No. 2021R1F1A1048010 and No. RS-2024-00358161, research grant to H.J.L.) and the Catholic Medical Center Research Foundation (program year, 2019). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Institutional Animal Care and Use Committee in the School of Medicine, The Catholic University of Korea (Receipt No. 2022-0127; Approval No. CUMC-2022-0133-07).
