Abstract
Background:
Lumbar spondylolysis (LS) has distinct pathologic stages, and worsening LS can cause poor bone union and low back pain. However, the risk factors involved in LS pathogenesis remain unclear.
Purpose:
To identify risk factors associated with the pathologic progression of LS at the L5 in adolescent male soccer players.
Study Design:
Case-control study; Level of evidence, 3.
Methods:
A total of 189 male adolescent soccer players aged 12 to 14 years were recruited. We used lumbar magnetic resonance imaging (MRI) to examine adolescent soccer players’ lumbar lordosis (LL), sacral slope (SS), sacral rotation angle, maturity stage of the lumbar vertebral bodies, and bone marrow edema (BMO) at L5. In addition, whole-body dual-energy x-ray scans measured bone mineral density and content as well as trunk lean body mass. Moreover, we tested the flexibility of the iliopsoas, hamstrings, and quadriceps, as well as evaluated the range of motion (ROM) of the hip in both external and internal rotation. LS stages were determined using computed tomography (CT)–like and short-tau inversion recovery (STIR) modes. The change over the 6-month period was the main outcome; participants were classified as “worsened,”“improved,” or “no change.” The worsened group was defined by a pathological progression on CT-like mode or new BMO appearance on STIR mode without CT-like change. Multiple logistic regression analysis was then performed to identify risk factors for the pathologic progression of LS.
Results:
Of the 163 players analyzed, 47 (28.5%) showed LS progression. BMO (odds ratio [OR], 5.54; 95% CI, 2.23-13.76), SS relative to LL (OR, 5.58; 95% CI, 2.32-13.51), sacral rotation angle ≤−1.5 or ≥1.5 (OR, 5.74; 95% CI, 2.36-13.96), and hip external rotation ROM (OR, 4.30; 95% CI, 1.67-11.06) were significantly associated with the pathologic progression of LS.
Conclusion:
Our study demonstrated that the risk factors for LS progression in male adolescent soccer players include BMO, sacral anteversion relative to LL, sacral rotation, and restricted hip external rotation ROM. Early BMO detection and sacral malalignment improvement may prevent pathologic LS progression in male adolescent soccer players.
Keywords
Lumbar spondylolysis (LS), characterized by a defect of the pars articularis, is caused by repetitive extension and rotation of the lumbar spine, 16 which is common in many sports activities. 28 Approximately 70% to 90% of LS cases occur at the L5 level, making this segment the most clinically relevant site of injury in adolescent athletes.6,29 The prevalence of LS is reported to be between 3% and 10% in the general population, whereas estimates around ~25% have been observed in athletes.2,3,14,19,22,28,33,37 Additionally, LS is classified according to distinct stages based on magnetic resonance imaging (MRI) and computed tomography (CT) evaluations.15,27 These range from a very early stage with only bone marrow edema (BMO) of the pars to a terminal stage resulting in pseudoarthrosis. Moreover, the latter stages of LS feature significant reductions in the rate of bone union.5,15,30 When aiming for bone union of LS, the first treatment involves immobilizing the lumbar spine while resting and wearing a corset for approximately 2 to 3 months. 30 Therefore, identifying factors associated with the onset and progression of LS and establishing prevention programs are crucial to avoid worsening of LS, and it may also be important to protect adolescent athletes from restrictions on participation in sports activities during adolescence.
High-quality prospective cohort studies are needed to prevent LS; however, to date, few such studies have been reported due to the difficulty of performing mandatory MRI and CT analyses. Previous studies of lumbar bone stress injuries (BSIs), corresponding to the very early stages of LS, have identified the following risk factors: long training hours, spinal malalignment, poor muscle flexibility in lower extremities, and an immature lumbar spine.13,14,37 However, these reports only evaluated BMO in the pars articularis of the lumbar spine; therefore, it is unclear whether bony structural failure (ie, defects in the pars articularis) actually developed. Given that low back pain (LBP) occurs in approximately 80% of high school and college athletes with LS, 10 identifying risk factors associated with the progression of LS, including structural failure of the bone, is required. Recent advances in MRI technology permit the use of a CT-like mode to evaluate both BMO and bone structure without radiation exposure.1,42 Therefore, conducting MRI scans using the CT-like mode could clarify how the LS stages worsen or improve.
This prospective study aimed to identify risk factors associated with the pathologic progression of LS at the fifth lumbar vertebra (L5) in adolescent male soccer players. We hypothesized that malalignment of the sacrum (ie, increased sacral slope relative to lumbar lordosis [LL], defined as a difference ≥5°), 37 which is located inferior to L5, is associated with stage progression, considering LS is a fracture of the inferior articular process.
Methods
Study Design
A 6-month prospective cohort design was adopted to identify LS stage progression factors in a sample of male adolescent soccer players. This period was set based on evidence that BMO persists for 12 to 16 weeks in adolescents 27 and that approximately 40% of adolescent soccer players with lumbar BMO developed lumbar bone stress injury after 6 months of play. 35
Participants
Overall, we aimed to recruit 150 participants, divided between 2 groups, each comprising 60+ participants; around 20% were expected to be excluded because of missing follow-up appointments or the existence of terminal LS or other lumbar disorders at baseline.
In total, we recruited 189 soccer players from the same club for this study. Recruitment took place during the competitive seasons, from May to November in 2023, 2024, and 2025. The club was at the competitive level, and the players practiced for 2 to 3 hours 5 times a week after school and on weekends. Additionally, the 6-month follow-up coincided with a period within the competitive season, during which regular training sessions and games were held. Additional inclusion criteria required that male soccer players be aged 12 to 14 years at the time of baseline measurements. Exclusion criteria comprised lack of participation in soccer games or practices for a ≥2-week continuous cessation during the 6-month follow-up from the baseline measurement, as well as those with an injury or illness affecting athletic performance.
All measurements were taken at Waseda University, and all experimental procedures conducted during this study were approved by the ethics committee of Waseda University (Approval No. 2021-218), and the participants, with their parents, signed informed consent to approve their participation.
Baseline Testing Parameters
At baseline, we measured body height, weight, lumbar MRI, dual-energy x-ray (DXA) scan, and muscle flexibility test for all participants. All measurements were conducted under assessor-blinded conditions, ensuring that examiners were unaware of each player's clinical status or imaging findings.
We used a stadiometer (YG200DN; Yagami) calibrated to the nearest 0.1 cm, as well as a digital scale (BC622; TANITA) calibrated to the nearest 0.1 kg, to measure participants’ height and weight, respectively. Next, we measured bone mineral density (BMD), bone mineral content, and regional lean body mass (LBM) of the trunk using a DXA platform (Hologic). The trunk region was defined as the body section between the first cervical vertebra and the femoral neck, excluding the region of the arm from the axillary fossa to the end of the fingers.34,37 DXA and region analyses were then performed by an experienced orthopaedic surgeon (S.T.).
Next, to determine LS and assess lumbar alignment and maturity of the lumbar vertebral body, we used a 3.0-Tesla whole-body MRI system (Signa 3.0T; GE Healthcare). Using this setup, we collected data using 2 imaging modes: short-tau inversion recovery (STIR) and 3-dimensional liver acceleration volume acquisition (ie, CT-like). STIR and CT-like mode scanning parameters were as follows:
STIR: repetition time/echo time = 1800/minimum (60) ms; slice thickness = 2 mm; field of view = 24 cm; matrix = 320 × 224 pixels; TI = 150 ms.
CT-like: repetition time/echo time = 4.5/2.1 ms; slice thickness = 0.80 mm; field of view = 24 cm; matrix = 384 × 384 pixels.
Participants remained in the supine position during scanning, with the knee in extension and the ankle fixed at a 90° angle of dorsiflexion. We then measured the angles of the LL (ie, between the superior endplate of L1 and the inferior endplate of L5) and the sacral slope (SS) (ie, between the superior endplate of S1 and the horizontal line) (Figure 1A). We used these values to quantify the balance of the lumbar curve by subtracting LL from SS.37,39 The sacral rotation angle is defined as the difference between the spinous process of L5 and the line that divides the intervertebral joint into halves by passing medially through the superior articular process of the sacrum (Figure 1B-1). A horizontal plane that transects the lumbosacral articular surface of the sacrum was also used to calculate the sacral rotation angle (Figure 1B-2). The landmarks (spinous process of L5 and superior articular process of the sacrum) were identified visually, and lines were drawn digitally using OsiriX measurement tools. LL, SS, and sacral rotation angle analysis were performed by a single examiner (T.T.) using image-processing software (Osirix MD; Pixmeo). Intraexaminer reliabilities, assessed by repeated measurements performed 1 day apart on 10 participants, resulted in intraclass correlation coefficient (ICC) (1,1) values of 0.92 (95% CI, 0.87-0.95), 0.92 (0.88-0.94), and 0.85 (0.81-0.90) for LL, SS, and sacral rotation, respectively, indicating excellent to good reliability. In addition, a same-day test-retest substudy (n = 8) yielded ICC (2,1) values of 0.97, 0.97, and 0.94, with corresponding minimal detectable change at the 95% confidence level values of 2.96°, 3.36°, and 0.63°.

Lumbosacral alignment analysis. (A) Mid-sagittal plane. Lumbar lordosis is the angle between the superior endplate of L1 and the inferior endplate of L5. Sacral slope is the angle between the superior endplate of S1 and a horizontal reference line. (B-1) Axial slice at L5-S1. The sacral rotation angle is the angle between (1) a black line drawn through the spinous process of L5 and (2) a white line that divides the intervertebral joint into halves by passing medially through the superior articular process of the sacrum. (B-2) Diagram showing the orientation of the plane used to calculate the sacral rotation angle. L1-L5, first to fifth lumbar vertebrae.
Based on previous studies,9,38 we assessed lumbar vertebral body maturity stage by determining the lesion degree of the epiphysis at L5. All observations were classified into 1 of 3 stages: cartilaginous, apophyseal, and epiphyseal.
Next, we used a standard goniometer to assess muscle flexibility and joint range of motion (ROM). Muscle flexibility was evaluated for the iliopsoas, hamstrings, and quadriceps. Iliopsoas flexibility was assessed using the Thomas test, 18 in which participants lay supine and actively flexed 1 hip, bringing the thigh toward the chest until the lumbar spine flattened and the pelvis stabilized; the examiner then maintained the position for measurement. Hamstring flexibility was measured as the knee extension angle with the hip fixed at 90° of flexion and quadriceps flexibility as knee flexion in the prone position. The reliability of these measurements has been reportedly high. 41 Hip internal rotation and external rotation (ER) tests were then performed with the angle of the hip flexed at 90° in the supine position. Muscle flexibility and ROM were measured by a physical therapist (N.I.) (Appendix Figure A1).
Determination and Classification of LS
LS stages were determined using both STIR and CT-like modes. In the first mode, BMO at the pars articularis was evaluated (Figure 2A). In the second mode, the degree of fracture at the pars articularis was evaluated. Consequently, LS stages were classified as follows: normal (Figure 2B), early with a hairline (Figure 2C), progressive with a clear gap (Figure 2D), and terminal with pseudoarthrosis (Figure 2E).

Classification of stages of lumbar spondylolysis. (A) Short-tau inversion recovery sequence showing bone marrow edema at the pars interarticularis (arrow). (B-E) Computed tomography–like magnetic resonance imaging showing each stage of fracture: (B) normal (no abnormal finding), (C) early (incomplete fracture), (D) progressive (complete fracture), and (E) terminal (pseudoarthrosis). Arrows indicate the site of pathologic findings (fracture line or bone marrow edema) used to determine the stage. BMO, bone marrow edema.
We used MRI data to perform stage classification of LS at baseline and repeated this after 6 months. The change over this period is the main outcome of this study, and participants could therefore be classified into 1 of 3 categories: worsened, improved, and no change. The worsened group is characterized by a pathologic stage progression in CT-like mode or the appearance of BMO in STIR mode combined with no change in CT-like mode. The improving group is explained as a pathologic stage regression in a CT-like mode or as a stage in which the absence of BMO occurred in STIR mode with no change in CT-like mode. To examine the differences in the side (eg, pivot or kicking) of the LS according to this subanalysis, the worsened group was considered the side with the worse LS pathogenesis for 6 months. When both sides presented the same stage of LS pathology, the one for which we had image findings at baseline was used for further experimental data acquisition. In addition, we excluded players who displayed any of the following: terminal stage (pseudoarthrosis) with no expected improvement, 15 imaging of multilevel LS, and disc herniation. Furthermore, an orthopaedic surgeon (S. Torii) with >40 years of diagnostic experience evaluated all lumbar injuries. In a blinded 2-session rereading conducted at least 1 week apart (n = 75) on MRI scans from a separate group of adolescent athletes not included in the main cohort, the examiner was blinded to all participant information and to the results of the first reading. Intrarater reliability for MRI-based LS assessments was high: STIR agreement, 94.7%; Cohen's κ, 0.84 (95% CI, 0.67-0.97); CT-like staging agreement, 92.0%, and weighted κ, 0.95 (95% CI, 0.87-0.99).
Statistical Analyses
For a sample size and power calculation, we expect the incidence rate of participants with high-risk factors to be approximately 3 times higher than the control group. 37 The power to detect a difference between the null and alternative hypotheses as proposed is 80% (ie, 0.05 on a 2-tailed test).
We compared the means of all response variables between the “worsened” group of LS pathogenesis and a control group that included both “no change” and “improved” players. All data were reported as mean and standard deviation for normally distributed data and percentage and number (N) for categorical data; to test mean differences for these 3 kinds of data, we used 2-sample t tests, Mann-Whitney U tests, and chi-square tests, respectively. Next, to identify the most predictive variables for worsened LS pathogenesis, we conducted a multivariate logistic regression using variables that showed P < .10 from the 2-group comparisons. Independent variables were selected from chronological age, body height, weight, bone mineral content, BMD, trunk LBM, LL, SS, SS relative to LL, lumbar vertebral body maturity stage, BMO (positive/negative), sacral rotation (presence/absence), hip ER and internal rotation ROM, and muscle flexibility of the iliopsoas, hamstrings, and quadriceps. Only variables with significance P < .05 remained in the model for each set. Models were developed using forward stepwise regression. Next, based on the mean and standard deviation, continuous variables identified by multivariate logistic regression were divided into high and low groups. A final multivariate stepwise logistic regression analysis was conducted for all categorized variables to determine the best overall predictive model. Next, we reported odds ratios (ORs) and profile likelihood 95% confidence intervals from the final model. A likelihood ratio test was also performed, and differences between the −2 logarithmic likelihood of the submodel and final model were calculated to determine the effect size of each predictor variable within the final model. In addition to the prespecified multivariate logistic regression, we fitted modified Poisson regression models with a log link and robust standard errors as sensitivity analyses to estimate risk ratios (RRs) for the binary outcome. To explore potential nonlinear associations between continuous exposures (SS relative to LL and sacral rotation) and outcomes, we employed restricted cubic splines with 4 degrees of freedom, adjusting for chronological age, height, and weight. We set the reference point at 0° (corresponding to RR = 1) and restricted both the inference and the visual representations to the 5th to 95th percentiles to avoid issues with extrapolation. We assessed overall associations and nonlinearity using Wald tests. For this subanalysis, we categorized the players into the worsened LS pathogenesis group and the worsened pivot and kicking sides group. Based on this distinction, we performed a 1-way analysis of variance to compare sacral rotation angles among 3 groups: the 2 specified above and a control group. Differences in pain frequency between groups were analyzed using the chi-square (χ2) test. Primary analyses were conducted in SPSS Statistics v27 (IBM), and supplementary analyses were performed in R (version 4.5.1) using standard packages.
Results
A total of 189 adolescent soccer players were recruited for this study. Of these, we excluded 5 who were in the terminal stage of LS, 1 player who showed multilevel LS at baseline, 17 players with inadequate data, 1 with disc herniation, 1 with sacral BSI, and 1 with multilevel LS. As a result, we included a total of 163 players in our analyses (Figure 3). Among these, 116 players were classified as having no abnormal MRI findings at follow-up, consisting of 88 controls (who remained free of abnormal findings throughout the study) and 28 players in the improved group. The relationship between positions and LS is presented in Appendix Table A1. The worsened group comprised 47 players (28.5%), of whom 31 (66.0%) reported pain during lumbar extension in the Kemp test; in contrast, 23 of the remaining 116 in the control group (19.8%) had pain. This difference was statistically significant (χ2 = 32.5, P < .001). Furthermore, at follow-up, pain was more frequently observed in the worsened group than in the improved group (31 of 47 [66.0%] vs 10 of 28 [35.7%]; χ2 = 6.16, P = .013).

Flow diagram of exclusion criteria and outcomes.
At baseline, 10 players (6.1%) were in the progressive stage, 34 (20.9%) were in the early stage, and 119 (73.0%) showed no abnormal findings on CT-like mode. After 6 months, progression was observed in 9 of 10 players (90.0%) from the progressive stage, 12 of 34 (35.3%) from the early stage, and 26 of 119 (21.8%) from those with no abnormal findings (Figure 4A). In addition, 50 players (30.7%) were positive on STIR mode at baseline, and half of these progressed during follow-up (25/50, 50.0%), whereas only 22 of 113 players (19.5%) without baseline findings worsened (Figure 4B).

Stage changes for pathologic LS status. (A) Computed tomography–like mode. (B) Short-tau inversion recovery mode.
Univariate analysis revealed that players in the worsened LS pathogenesis group had more positive BME and sacral rotation (Table 1), significantly lower hamstring flexibility (P = .040) and hip ER ROM (P = .002), and higher SS (P = .026) and SS relative to LL (P < .001) than the control group (Table 2).
Univariate Analysis of Categorical Variables a

BMO, bone marrow edema; OR, odds ratio.
Univariate Analysis of Continuous Variables a

Data are presented as mean ± SD. BMC, body mass content; BMD, bone mass density; ER, external rotation; IR, internal rotation; LBM, lean body mass; LL, lumbar lordosis; ROM, range of motion; SS, sacral slope.
Mann-Whitney U test.
Unpaired t test.
Next, variables for which univariate analysis identified differences between worsened LS pathogenesis and control groups were entered into a multivariate logistic regression model. The 5 variables were as follows: presence of BMO, sacral rotation angle ≤−1.5 or ≥1.5, SS relative to LL ≥5°, hip ER ROM ≤40°, and hamstring flexibility ≥40°. Multivariate logistic regression results showed that BMO (OR, 5.54; 95% CI, 2.23-13.76), SS relative to LL (OR, 5.58; 95% CI, 2.32-13.51), sacral rotation angle ≤−1.5 or ≥1.5 (OR, 5.74; 95% CI, 2.36-13.96), and hip ER ROM (OR, 4.30; 95% CI, 1.67-11.06) were significant predictors of worsened LS pathogenesis (Table 3). As a sensitivity analysis, modified Poisson models with a log link and robust standard errors yielded directionally consistent RRs, including BMO (RR, 2.20; 95% CI, 1.44-3.35), SS relative to LL >5° (RR, 2.44; 95% CI, 1.44-4.13), sacral rotation angle ≤−1.5 or ≥1.5 (RR, 2.39; 95% CI, 1.49-3.84), and hip ER ROM (RR, 1.48; 95% CI, 1.01-2.18) without changing inference (Table 3). In supplementary analyses modeling SS relative to LL and sacral rotation as continuous variables using restricted cubic splines, adjusted RR curves revealed a significant overall association with worsening and a nonlinear pattern (Wald tests, both P < .001) (Appendix Figure A2).
Multivariate Logistic Regression Analysis of Factors Associated With Worsened Sacral Slope Pathology a
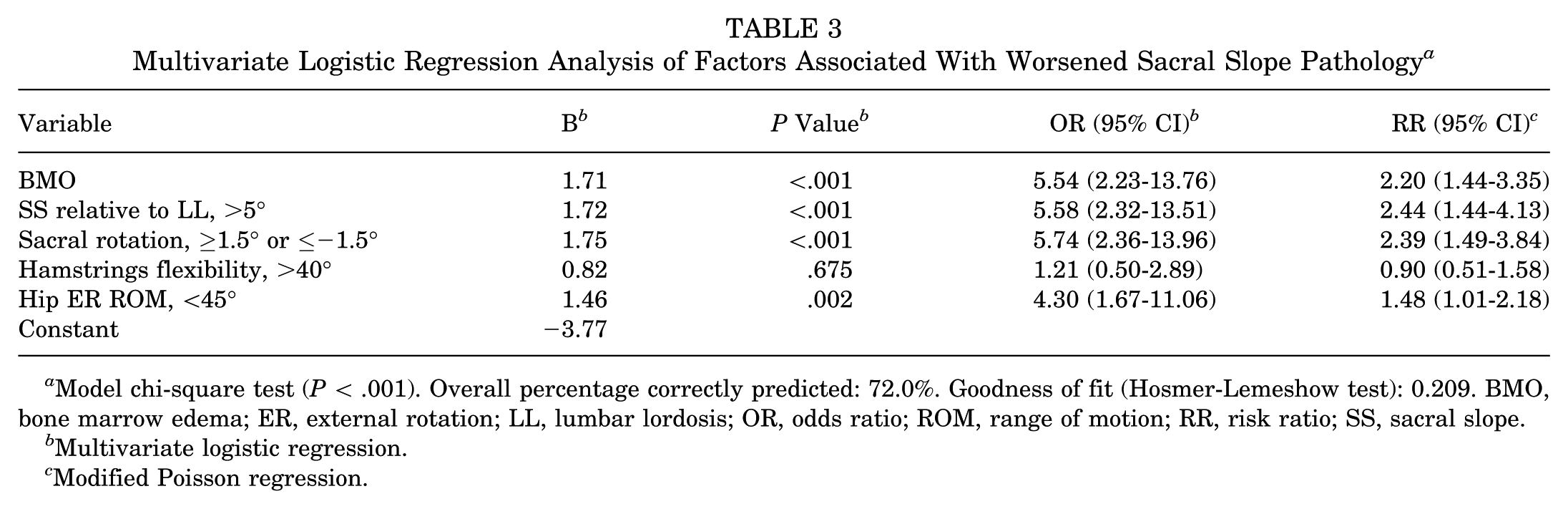
Model chi-square test (P < .001). Overall percentage correctly predicted: 72.0%. Goodness of fit (Hosmer-Lemeshow test): 0.209. BMO, bone marrow edema; ER, external rotation; LL, lumbar lordosis; OR, odds ratio; ROM, range of motion; RR, risk ratio; SS, sacral slope.
Multivariate logistic regression.
Modified Poisson regression.
Regarding the subanalysis, the worsened LS pathogenesis group (n = 47) was divided into 33 players who showed worsening on their pivot side and 14 players who showed worsening on the kicking side; these groups were compared with respect to the direction of sacral rotation. Figure 5 shows the sacral rotation for the worsened group of players with progressed LS pathogenesis on the pivot and kicking sides, as well as for a control group. The groups showing worsening on the pivot and kicking sides had a greater sacral rotation angle toward the pivot (1.60°± 0.87°, P < .001) and kicking sides (−1.61°± 1.16°, P < .001), respectively, relative to the control group (0.16°± 1.05°) (Figure 5).

Distribution of sacral rotation (○) in three groups. Negative values indicate sacral rotation toward the kicking side, and positive values toward the pivot side. Black, worsened on the kicking side (n = 14); gray, worsened on the pivot side (n = 33); open, control (n = 33).
Discussion
The major findings of this prospective study were that 28.5% of adolescent male soccer players showed pathologic progression of LS over 6 months and that this progression was significantly associated with the presence of BMO on STIR imaging, greater SS relative to LL, larger sacral rotation, and reduced hip ER ROM. These results highlight the key physical and morphologic factors that may predispose adolescent athletes to LS progression during active growth. In addition, the pathologic progression of LS was associated with BMO, increased SS relative to LL, greater sacral rotation, and lower hip ER ROM. Lumbar BSI, including LS during adolescence, is a high-risk injury 43 that requires a longer time before returning to their sports activities than other sports-related injuries. In addition, given that LS prevalence is a potential trigger for LBP, 10 LS risk management in adolescents may be influenced by their participation in sports activities.
To our knowledge, this is the first cohort study that follows adolescent soccer players and investigates changes in LS pathogenesis in that population. Most previous studies of LS have been conducted in hospitals, and to date, there is no clear understanding of how changes in LS pathogenesis can occur. Although the prevalence of LS in the general Japanese population is around 5.9%, 28 Rossi and Dragoni 22 reported that the incidence of LS in adolescent athletes is 2 to 5 times higher than in nonathletes. In addition, several prospective cohort studies discussing the incidence of LS revealed that 27.3% of cricket bowlers showed signs of a very early stage of LS, 13 as did 26.2% of adolescent soccer players. 37 Overall, these rates are consistent with the findings of this study. In particular, 90% (9/10) who were diagnosed as being in a progressive stage via CT-like MRI scanning, and 50% (25/50) who had BMO when examined using STIR mode at baseline showed worsening LS pathology. Kuroshima et al 15 reported that the rates of bone union in LS decreased as the LS stage worsened (ie, 92.6% to 40.5%). These findings suggest that LS reaching the progressive stage can make LS difficult to improve and may further result in disruption of the bony structure. In contrast, 17.6% of players showing LS pathology at baseline showed improved LS stages despite playing soccer for 6 months. Notably, 64.7% (22/34) of those identified as early-stage LS by CT-like MRI scans at baseline showed improvements over 6 months. Another previous study comparing the rates of return to sports of various physical therapy treatments, including pain management and trunk brace prescription (ie, the primary treatment of LS), found that both had a return to sports ≥95%. 12 These results are consistent with our study, and therefore, the early stages of LS may be crucial for determining whether the pathology worsens or improves. Furthermore, BMO has been reported to reflect remodeling processes in overloaded regions and may resolve or stabilize with appropriate symptom management and load modification. 40 Based on our findings, BMO may act as a trigger of pathologic progression; however, this also suggests that strategies aimed at reducing local stress could allow improvement or prevention of worsening in adolescent athletes. The implementation of a program to improve LS risk factors may help identify treatment options that do not prevent adolescent athletes from continuing to play soccer.
One physical risk factor for the pathologic progression of LS was increased SS relative to LL. Several studies of LS have reported an association between LL and SS in the sagittal plane. For example, Roussouly et al 23 and Yin et al 45 found significantly higher LL and SS values in LS patients compared with healthy control adults. Furthermore, Hanke et al 8 conducted a retrospective study in which athletes with symptomatic ischemic spondylolysis showed greater SS values when measured in high school. In a cross-sectional study, Tsutsui et al 39 identified that bilateral and unilateral LS patients had greater SS relative to LL than control subjects in a pool of adolescent athletes. It has therefore been suggested that an increased SS relative to LL may increase the mechanical stress 37 applied during lumbar extension25,26; this would include the compression stress between L5 and S1. Hence, a greater regional curve or tilt of L5/S—not necessarily than the entire lumbosacral spine—may serve to worsen LS pathology. In addition, this study also found that greater sacral rotation is a risk factor for the pathologic progression of LS. In general, the sacrum can move in any direction with respect to the ilium, although the magnitude of movement is slight. 4 A previous study found that the ROM of the sacroiliac joint exhibits a flexion-extension of approximately 3°, a rotation of approximately 1.58°, and a lateral flexion of approximately 0.88°. 21 The small sacral rotational ROM and the small axial rotation at L5-S1 44 suggest that a sacral rotation angle ≥1.5° or ≤−1.5° may increase stress on the pars articularis. In previous studies, LS has been shown to arise from the inferior articular process at the ventral side of the pars articularis. 36 Thus, direct compressive stress between the superior articular process of the sacrum and the inferior articular process of L5, as well as indirect shear stress via the articular process, can cause structural failure of the pars. Indeed, the subanalysis performed here showed that sacral rotation occurred ipsilateral to the side of the pathologic progression of LS; that is, the superior process of the sacrum and the inferior process of L5 were more closely rotated on the side of the pathologic progression of LS. Therefore, such sacral malalignments may result from physical characteristics that exacerbated mechanical factors such as lumbar extension and rotational loading, which have traditionally been regarded as the most common cause of pars stress in LS.25,26 Notably, to address sacral rotational malalignment, it may be important to improve the function of the multifidus muscle, which contributes to spinal stability and has direct attachments to the sacrum. 32 Although interventional studies are limited, a randomized trial of a 9-month Pilates program demonstrated reductions in LL and pelvic tilt in adolescents, 7 suggesting that core stability–focused approaches could represent a promising avenue for future investigation. In addition, a hip ER <45° was another risk factor for the pathologic progression of LS. McGovern et al 20 reported a greater length change in the deep rotators of the hip at 45° of ER and 90° of flexion position, while another study by Sadeghisani et al 24 identified greater lumbopelvic rotation ROM during passive hip ER in patients with LBP who were regularly involved in rotation-related sports. In addition, a study by Scholtes et al 31 suggested that patients with LBP who participated in rotation-related sports might exhibit greater and earlier lumbopelvic movement during active hip lateral rotation relative to patients without LBP. Thus, limited hip ER ROM may lead to compensatory lumbosacral motion. Given that the deep rotators of the hip also attach to the sacrum, improved gluteal muscle flexibility may help to modify malalignment of the sacrum and prevent pathologic progression of LS.
Limitations
This cohort study was conducted in only 1 soccer club; we were therefore unable to compare extrinsic risk factors, such as practice time, training load, or individual performance metrics. This is important because these have been previously reported to be associated with LS 14 ; however, given that training schedules were largely uniform across the team, individual differences in exposure were likely minimal. In addition, MRI scans were performed in the supine position, which is different from the positions used while patients play soccer. Although the ankle and knee were immobilized during MRI scans to eliminate the effect of lower extremity ROM (as much as possible), lumbosacral alignment may have been altered by this posture. Previous studies have shown that both LL and SS increase while pelvic tilt decreases when assessed in the upright position.11,17 Therefore, the application of this study to lumbar and sacral alignment may require clinicians and athletic trainers to evaluate athletes when they are placed in the supine position.
Conclusion
Our study demonstrated that the risk factors for LS progression in male adolescent soccer players include BMO, sacral anteversion relative to LL, sacral rotation, and restricted hip ER ROM. Early BMO detection and sacral malalignment improvement may prevent pathologic LS progression in male adolescent soccer players.
Supplemental Material
sj-docx-1-ojs-10.1177_23259671251401599 – Supplemental material for Physical Factors Associated With Stage Progression of Lumbar Spondylolysis: A Prospective Cohort Study in Male Adolescent Soccer Players
Supplemental material, sj-docx-1-ojs-10.1177_23259671251401599 for Physical Factors Associated With Stage Progression of Lumbar Spondylolysis: A Prospective Cohort Study in Male Adolescent Soccer Players by Toshiharu Tsutsui, Wataru Sakamaki, Seira Takei, Nodoka Ikegami and Suguru Torii in Orthopaedic Journal of Sports Medicine
Supplemental Material
sj-docx-2-ojs-10.1177_23259671251401599 – Supplemental material for Physical Factors Associated With Stage Progression of Lumbar Spondylolysis: A Prospective Cohort Study in Male Adolescent Soccer Players
Supplemental material, sj-docx-2-ojs-10.1177_23259671251401599 for Physical Factors Associated With Stage Progression of Lumbar Spondylolysis: A Prospective Cohort Study in Male Adolescent Soccer Players by Toshiharu Tsutsui, Wataru Sakamaki, Seira Takei, Nodoka Ikegami and Suguru Torii in Orthopaedic Journal of Sports Medicine
Supplemental Material
sj-docx-3-ojs-10.1177_23259671251401599 – Supplemental material for Physical Factors Associated With Stage Progression of Lumbar Spondylolysis: A Prospective Cohort Study in Male Adolescent Soccer Players
Supplemental material, sj-docx-3-ojs-10.1177_23259671251401599 for Physical Factors Associated With Stage Progression of Lumbar Spondylolysis: A Prospective Cohort Study in Male Adolescent Soccer Players by Toshiharu Tsutsui, Wataru Sakamaki, Seira Takei, Nodoka Ikegami and Suguru Torii in Orthopaedic Journal of Sports Medicine
Footnotes
Final revision submitted October 9, 2025; accepted October 26, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: This work was supported by JSPS KAKENHI (grant number JP24K20617). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the ethics committee of Waseda University (Approval No. 2021-218).
Data Availability
The data sets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
