Abstract
Background:
Perioperative nerve blocks are commonly used for regional analgesia with anterior cruciate ligament (ACL) reconstruction (ACLR). Liposomal bupivacaine (LB) is a long-acting anesthetic agent providing up to 72 hours of nerve blockade. It is theorized that the addition of dexamethasone to LB (LB+dex) may prolong the analgesic duration.
Purpose:
To characterize pain control and opioid consumption with adductor canal block (ACB), interspace between the popliteal artery and capsule of the posterior knee (iPACK), and suprasartorial infiltration (SSI) regional anesthetic techniques utilizing LB after ACLR and to compare LB versus LB+dex.
Study Design:
Randomized controlled trial; Level of evidence, 1.
Methods:
Patients undergoing primary ACLR using bone–patellar tendon–bone or quadriceps tendon autograft were included. All patients received an ACB, iPACK, and SSI preoperatively. Patients were randomized to receive either LB or LB+dex for their anesthetic agent. Opioid consumption, visual analog scale (VAS) scores, pain control satisfaction, and duration of block effect were recorded postoperatively.
Results:
A total of 131 patients were included in the analysis. The mean opioid consumption for all patients was 1.20 ± 2.42 five-mg oxycodone tablets (8.97 ± 18.12 morphine milliequivalents [MME]). We did not observe a significant difference in oxycodone consumption between cohorts during the study period (1.42 ± 2.92 vs 0.97 ± 1.91; 10.65 ± 21.90 MME vs 7.28 ± 14.33 MME; P = .76). An estimated 95.2% of opioids prescribed went unused and 77.1% (101/131) of patients consumed no opioids. There was no significant difference in VAS score, patient satisfaction, or duration of block effect between cohorts.
Conclusion:
A single shot of LB with or without dexamethasone via ACB, SSI, and iPACK block provided excellent pain control and minimized opioid consumption after autograft ACLR. Nearly 80% of patients did not require opioids postoperatively, making opioid-free ACLR a realistic possibility for many patients. When narcotics were required, the dose of opioids was minimal; patients required 1.2 tablets of oxycodone on average, and >95% of the opioids prescribed went unused. Despite previous reports suggesting a prolonged duration of anesthetic effect when LB is combined with dexamethasone, we found no difference in opioid consumption, VAS score, patient satisfaction, or duration of block effect when patients received dexamethasone with their regional block. The utilization of regional anesthetic techniques such as ACB, SSI, and iPACK blocks in conjunction with LB-based anesthetics could allow providers to curtail or potentially eliminate opioid prescriptions after ACL surgery.
Registration:
NCT06006624 (ClinicalTrials.gov identifier).
Keywords
Femoral nerve blockade (FNB), adductor canal blocks (ACB), and the interspace between the popliteal artery and capsule of the posterior knee (iPACK) blocks are common techniques for regional analgesia with anterior cruciate ligament (ACL) reconstruction (ACLR). While there is a significant body of literature regarding the efficacy and utility of specific regional block techniques, there are fewer high-quality clinical data informing the use of specific anesthetic agents. 1
Liposomal bupivacaine (LB) is a long-lasting nonopioid analgesic that was initially utilized as an infiltrative agent but was recently approved by the US Food and Drug Administration for use in interscalene brachial plexus blocks and ACBs. 28 The combination of bupivacaine with a multivesicular-based lipid delivery platform is theorized to prolong the release of the anesthetic agent, which has been reported to deliver anesthetic effects for up to 72 hours after administration.5,6,17,26,30,32,38 Over 70% of opioid consumption occurs within the first 72 hours postoperatively after ACLR,13,14 making LB an enticing block agent. Thus, the use of long-acting LB either in the form of FNB, ACB, or as a local infiltrative block has become increasingly common for perioperative pain management after ACLR to address this critical early postoperative pain period to curtail opioid consumption.1,5,26,36
The addition of dexamethasone to LB has been theorized to further enhance drug delivery and prolong its anesthetic activity.1,33 It has been our experience that the addition of dexamethasone to LB, when administered via ACB, iPACK, and suprasartorial infiltration (SSI) has resulted in sustained pain control and patient satisfaction after ACL surgery. Therefore, the purpose of this study was to compare LB and LB combined with dexamethasone (LB+dex) to determine if the addition of dexamethasone significantly affects postoperative narcotic use when administered in an ACB, SSI, and iPACK regional anesthetic technique prior to ACLR. Secondarily, we aimed to determine the effect that these differing formulations would have on postoperative pain and perceived duration of neural blockade postoperatively. It was our hypothesis that the addition of dexamethasone to LB formulations would prolong the length of analgesic effects and reduce the total use of postoperative opioids.
Methods
Study Design
This was a prospective, blinded, randomized controlled trial designed in accordance with the CONSORT (Consolidated Standards of Reporting Trials) statement. 35 The study was registered on clinicaltrials.gov and approved by our institutional review board.
Patients undergoing primary ACLR using bone–patellar tendon–bone (BTB) or quadriceps tendon (QT) autograft from September 2023 to November 2024 performed by 5 fellowship-trained sports medicine surgeons (C.C.D., S.B.C., J.S., M.G.C., S.H.) at a single hospital-owned outpatient surgical center were screened for study eligibility. All regional blocks were performed by a single anesthesiologist (A.V.). Inclusion criteria were patients with ACL rupture undergoing primary reconstruction with BTB or QT autograft who were ≥18 years of age, English speaking, and able to provide informed consent with the ability to complete surveys via email or cellular phone. Patients were included if they underwent meniscal repair, partial meniscectomy, and lateral extra-articular tenodesis (LET) or anterolateral ligament (ALL) reconstruction. Exclusion criteria included revision procedures, multiligamentous knee injuries, cartilage or meniscal procedures that prevented adherence to immediate weightbearing and range of motion rehabilitation protocol, ACLR utilizing non-BTB or -QT autografts, allergies to study medications, known alcohol or narcotic abuse history, existing contract with a pain specialist due to underlying preoperative pain syndrome, and preoperative opioid use within the 3 months prior to surgery. All patients were instructed on the purpose of the study by the same single anesthesiologist and member of the research staff and signed an informed consent form prior to study inclusion.
Randomization and Blinding
Patients were randomized by a member of the research staff in a 1:1 ratio into 1 of the following 2 arms using a random number generator:
Study Group No. 1: 30 mL liposomal bupivacaine + 5 ml of 0.5% bupivacaine evenly distributed and administered in adductor canal, iPACK, and suprasartorial infiltration (35 mL total)
Study Group No. 2: 30 mL liposomal bupivacaine + 10 mg preservative-free dexamethasone + 5 ml of 0.5% bupivacaine evenly distributed for both adductor canal, iPACK, and suprasartorial infiltration (35 mL total)
Patients and their treating surgeon were blinded to their allocated study group. The treating anesthesiologist was notified of randomization status by research personnel just prior to administration of the regional anesthesia blocks. Careful consideration was given toward inclusion of a plain bupivacaine- or ropivacaine-based treatment arm at the time of study inception. However, given the success of liposomal bupivacaine–based regional anesthesia techniques at our center and the desire to explore the potential effects of dexamethasone highlighted in the basic science literature and limited clinical study,1,33 the authors made the decision to include the 2 treatment arms in an attempt to optimize postoperative pain management while avoiding any compromise in patient comfort or satisfaction.
Perioperative Multimodal Pain Control Regimen
All patients received the same perioperative multimodal pain control regimen as indicated in Table 1.
Perioperative Multimodal Pain Regimen

Patients were instructed to take acetaminophen, ketorolac, and metaxalone prescriptions on a scheduled basis and to take oxycodone on an as-needed basis.
Interventions
All patients received a preoperative block targeting the saphenous nerve via ACB (Figure 1), the intermediate femoral cutaneous and genicular nerves via SSI (Figure 2), and the nonmotor branches of the tibial nerve and branches of the obturator nerve via iPACK block (Figure 3). Patients, research personnel, and surgeons were all blinded from the treatment arm. Only the anesthesiologist was aware of block composition. ACLR began with the harvest of the BTB or QT autograft. Diagnostic arthroscopy was performed using standard anterolateral and anteromedial portals and all relevant meniscal pathology was addressed, including all-inside techniques when meniscal repair was performed. The femoral tunnels were drilled anatomically either through the use of an accessory medial portal or transtibial using an over-the-top guide or in an outside-in technique using a retrograde reamer depending on surgeon preference and graft option. The decision to proceed with ALL reconstruction or LET was made preoperatively based on patient age, presence of preoperative pivot shift, ligamentous laxity, primary sport, and surgeon preference.

Administration of adductor canal and suprasartorial infiltration in a left leg. Red arrow signifies the tip of the needle at site of anesthetic administration. FA, femoral artery; FV, femoral vein.

Administration of suprasartorial infiltration in a left leg. FA, femoral artery.

Administration of interspace between the Popliteal Artery and the capsule of the posterior knee (iPACK) Block. Left leg. PA, Popliteal Artery.
Upon completion of surgery prior to discharge from the surgical center, patients were given standardized opioid education by a multidisciplinary team consisting of the lead anesthesiologist as well as the attending surgeon. Specifically, patients were instructed to utilize opioid medications when their pain exceeded a 7 of 10 on the Numeric Rating Scale and were instructed on nonpharmacological pain control methods such as elevation and application of ice to the surgical site as needed. Patients were specifically instructed to utilize ketorolac postoperatively for 5 days. Patients were encouraged to discontinue the use of acetaminophen and metaxalone when their pain levels felt appropriately managed without pharmacologic interventions (including narcotics). Patients were also given a visual analog scale (VAS) as well as instructions for completion of a postoperative pain recording diary via RedCap survey. Patients were instructed to rate their VAS pain score on a linear sliding scale of 0 to 100 in which a 0 indicated “no pain” and 100 indicated the “worst imaginable pain.” Additionally, patients were instructed to indicate their satisfaction with pain control on a similar linear sliding scale of 0 to 100 in which 0 indicated “completely unsatisfied” and 100 indicated “completely satisfied.” Patients were asked to record their VAS scores, opioid and nonopioid medication consumption, satisfaction with pain control, and perception of regional block function daily beginning on the day of surgery and continuing until postoperative day 7. On postoperative day 7 and again on postoperative day 14, patients were surveyed to again verify their total opioid consumption, satisfaction with postoperative pain control and regional anesthesia, duration of block effect, and VAS pain score. Duration of block effect was established by asking patients each day if they still felt the effects of their regional block (yes/no). The block duration was considered the last day patients felt the effects of their block. Regional block efficacy for the respective day was indicated as a “1” if the response was “yes” and was indicated as a “0” if the response was “no”; therefore, if days 0 to 4 were indicated as “yes” and on day 5 the patient indicated “no,” then the duration of block effect was 4 days.
Postoperative Rehabilitation Protocol
Postoperatively, all patients were placed into a hinged knee brace. Patients were permitted to bear weight as tolerated immediately after surgery with the brace locked in extension. Range of motion to 90° of flexion without weightbearing was permitted with physical therapy guidance immediately after surgery. Progression of range of motion varied based on concomitant meniscal pathology; however, this did not substantively differ between patients during the study period. Patients returned to the clinic for the first postoperative visit and suture removal during the second postoperative week.
Outcome Assessment
The primary endpoints were postoperative opioid consumption and VAS pain scores after ACLR. Secondary endpoints included patient satisfaction and duration of nerve block efficacy (in days). The acute pain period was defined as the first 3 days after surgery. Primary and secondary endpoints were assessed daily, during the acute pain period, and during the first week postoperatively. All data collection and study communication after enrollment was performed by members of the research team, which was a separate entity from the treating surgeon. Opioid consumption was reported as the number of tablets consumed as well as morphine milliequivalents (MMEs).
Statistical Analysis
An a priori power analysis was conducted prior to the study based upon postoperative opioid consumption after ACLR using an effect size of 0.5, a power of 0.80, and an alpha level of .05. The analysis indicated that a sample size of 128 patients was required. To account for potential attrition, a total of 156 patients was deemed necessary for enrollment.
Following data collection, descriptive statistics were computed. Continuous data are presented as mean and standard deviation, and categorical data are presented as count and percentage. t tests or Mann-Whitney U tests were used to compare continuous data based on normality, while chi-square tests or Fisher exact tests were used to compare categorical data. For comparison of duration of nerve block efficacy, chi-square analyses between responses for both treatment groups were made based on daily responses. A P value of <.05 was considered statistically significant. All statistical analyses were performed using R Studio (Version 4.1.2).
Results
Baseline Demographics
A total of 187 patients were deemed eligible and approached for study participation, with 29 electing not to participate (15.5%). Thus, 158 patients were enrolled in the study; but a total of 131 of these patients (82.9%) completed the required surveys and were thus included in the final analysis (Figure 4). A total of 78 male and 53 female patients were included, with a mean age of 27.1 ± 7.4 years. There were no significant differences between cohorts with regard to age (P = .62), sex (P = .94), race (P = .95), BMI (P = .98), graft selection (P = .17), or concomitant procedures (P > .05) (Table 2).

Study flow diagram.
Baseline Patient Demographics and Characteristics for Each Group a
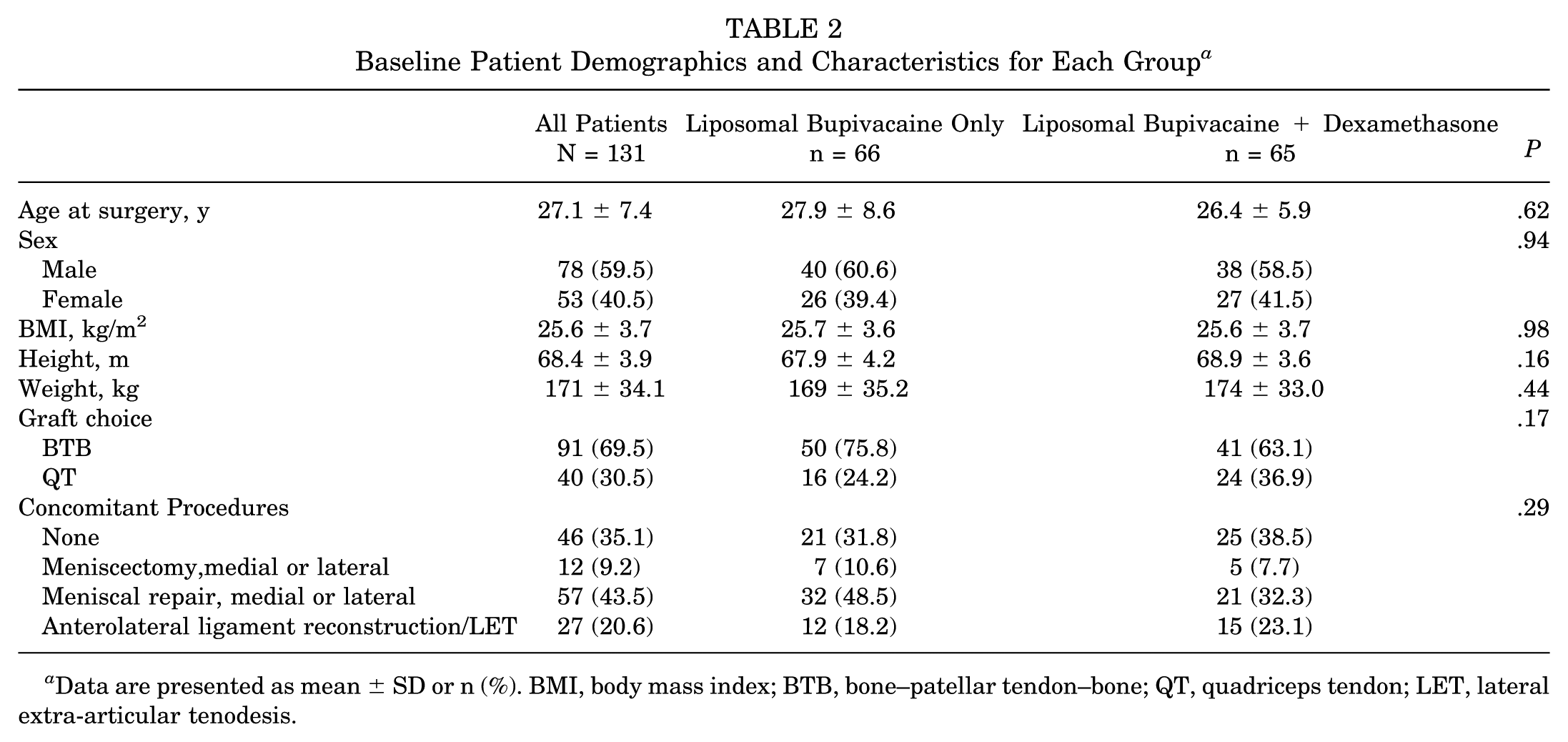
Data are presented as mean ± SD or n (%). BMI, body mass index; BTB, bone–patellar tendon–bone; QT, quadriceps tendon; LET, lateral extra-articular tenodesis.
Opioid and Analgesic Consumption
The mean opioid consumption for all patients was 1.20 ± 2.42 five-mg oxycodone tablets (8.97 ± 18.12 MME). Oxycodone consumption throughout the study period as well as the acute pain period is outlined in Table 3. An estimated 95.2% of opioids prescribed in the study period went unused (94.7% in the LB alone cohort and 95.7 LB+dex cohort) and 77.1% (101/131) of patients consumed no opioids. Only 7.6% (10/131) patients took >5 oxycodone tablets. Five patients in each group took >5 oxycodone tablets (X2 [1; n = 131] = 0.0006; P = .98). There were no significant differences in the amount of acetaminophen, metaxalone, or ketorolac taken between either group at any point in the study period (P > .05). Mean opioid consumption for patients undergoing reconstruction with BTB autograft was 0.72 ± 1.89 tablets (5.40 ± 14.18 MME) compared with 2.81 ± 5.04 tablets (21.08 ± 37.80 MME) (P = .02) for those receiving QT autograft. To assess potential data fragility, 3 outliers reporting increased postoperative pain and opioid consumption (10, 12, and 20 tablets consumed) in the QT cohort were excluded; however, the statistical differences between the BTB and QT cohorts persisted (1.88 ± 2.82 tablets in the updated QT cohort; P = .03). Patients undergoing meniscal repair consumed 1.45 ± 3.56 tablets (10.88 ± 26.70 MME), while those receiving concomitant meniscectomy consumed 0.92 ± 1.78 tablets (6.90 ± 13.35 MME) (P = .46). Patients undergoing concomitant ALL reconstruction or LET consumed 1.68 ± 4.16 opioid tablets (12.6 ± 31.2 MME) in contrast to 1.33 ± 3.19 (9.98 ± 23.93 MME) consumed by those who did not undergo anterolateral stabilization (P = .70).
Opioid Consumption, Satisfaction, and Duration of Nerve Block Effect a

Data are presented as mean ± SD. MME, morphine milliequivalent; VAS, visual analog scale.
Overall postoperative VAS pain score was calculated by generating the mean VAS pain score from all days during the study period.
Postoperative Pain
There was no difference in pain levels during either the acute pain period during postoperative days 0 to 3 (P = .28) or throughout the study period (P = .33). The mean pain score during the study period was 34.9 ± 11.7, including 36.7 ± 18.7 in the LB-alone group and 33.1 ± 16.7 in the LB+dex group (Table 3). When analyzed daily, there were no significant differences between cohorts with regard to daily VAS pain scores at any time point (P > .05). Mean VAS pain scores were highest on postoperative day 6 (38.0 ± 22.4) including 40.3 ± 22.5 in the LB cohort and 35.8 ± 22.2 in the LB+dex group (P = .26). Patients undergoing ACLR with BTB autograft experienced lower VAS pain scores (32.2 ± 17.2) throughout the postoperative period compared with those undergoing ACLR with QT autograft (41.0 ± 17.7) (P = .01).
Patient Satisfaction
Patient satisfaction with postoperative pain control was high throughout the study period (Table 3). There were no significant differences in patient satisfaction with pain control or with nerve block at any time point during the study (P > .05). Patient satisfaction with pain control was lowest on postoperative day 7 with a mean of 81.7 ± 20.9. Mean patient satisfaction was 85.3 ± 14.1 in patients undergoing ACLR with BTB autograft compared with 82.1 ± 15.1 with QT autograft (P = .25).
Duration of Nerve Block Effect
The mean duration of analgesic effects from the nerve block was 6.55 ± 2.50 days (Table 3). Of patients, 68% reported neural blockade on postoperative day 5, and there was a larger proportion of patients receiving LB+dex who experienced ongoing neural blockade (34/63; 54%) compared with those receiving LB alone (21/63; 33%) (P = .03) on postoperative day 6 (Figure 5). There were no significant differences in duration of nerve block effect at any other time point throughout the study (P > .05) or based on BTB (6.83 ± 2.56 days) or QT autograft (6.03 ± 2.34) (P = .11).

Comparison of duration of nerve block effect over the course of the study. LB, liposomal bupivacaine; LB+dex, liposomal bupivacaine with dexamethasone; POD, postoperative day.
Discussion
The primary finding of the present study was that the use of LB-based anesthetic formulations administered preoperatively via ACB, iPACK, and SSI in conjunction with a multimodal pain regimen resulted in a mean of 1.2 five-mg oxycodone tablets (9 MME) being consumed after ACLR with BTB or QT autograft and a perceived blockade duration >6 days. There appear to be no statistically significant differences between opioid consumption, VAS or patient satisfaction scores, or duration of regional anesthetic effects when dexamethasone is added to LB. These results contradict our initial hypothesis that the addition of dexamethasone to LB in adductor canal and iPACK blocks would result in lower postoperative opioid consumption, greater pain control, and longer duration of regional anesthetic effect after ACLR. Despite confirming our null hypothesis, the results of this study suggest that the addition of LB-based anesthetic formulations administered via ACB, iPACK, and SSI to a multimodal pain regimen may minimize narcotic requirements after ACLR and make opioid-free ACLR a realistic goal for nearly 80% of patients.
Recent studies of opioid consumption after ACLR estimate that between 2 and 15 five-mg oxycodone tablets are consumed after ACLR.13,14,18,23,34 However, studies reporting on the lower end of this range generally either do not specify or include heterogeneous graft sources including a high proportion of hamstring autograft as well as allograft, which are believed to offer lower morbidity and pain than BTB or QT autograft harvest.3,18,23 The potential reasons for the 1.2 five-mg oxycodone tablets (9 MME) consumed without significant differences between treatment groups in the present study are likely multifactorial. Most notably, previous studies utilizing a similar methodology have suggested that approximately 75% of all opioids consumed after ACLR occur within the first 3 days postoperatively—highlighting an “acute pain period” that must be addressed to curtail postoperative opioid consumption.13,14 It is therefore plausible that LB delivered via ACB and iPACK blocks, with a reported duration of analgesic effect most frequently reported at 72 hours,5,6,17,32,38 offers sustained analgesia necessary to bypass this period of heightened pain and opioid consumption. Following this logic, the addition of dexamethasone with the intention of further prolonging analgesic effect likely would not result in a statistically or clinically significant improvement in pain control or opioid consumption as was found in the present study. To this end, the majority of patients in both groups reported persistent sensory blockade beyond postoperative day 6. However, Liddy et al 18 and Moutzouros et al 23 have effectively highlighted the profound utility of a multimodal analgesic regimen in substantially reducing postoperative opioid consumption after ACL surgery. 24 In the present study, patients were strongly encouraged toward adherence to a multimodal approach including nonsteroidal anti-inflammatories and acetaminophen, as well as a muscle relaxer, with only a slight reduction in total opioid consumption compared with the aforementioned studies. Therefore, it is also plausible that strict adherence to such multimodal regimens supersedes other strategies such as regional nerve blocks as well as varying anesthetic agents and additives included in these blocks with regard to its importance in achieving acceptable postoperative pain control.
Nearly 80% of patients in the present study required no opioids after ACLR, with dexamethasone providing no statistically significant benefit in opioid consumption, satisfaction, or postoperative pain control. This is comparable with the 81% of patients not requiring opioids reported by Walls et al 39 in a pediatric population of patients undergoing ACLR utilizing either LB or bupivacaine alone via single-shot ACB. However, the mean opioid consumption of 1.2 five-mg tablets in the present study is 3 times the 0.4 pills per patient reported in the same study. There are multiple reasons for such differences, including a study period of only 3 days, prescription of a hydrocodone/acetaminophen combination, and differing graft harvest sources (23/58 hamstring, 35/58 QT, 0/58 BTB) compared with the present study in which all patients underwent BTB or QT harvest, both of which are believed to have higher donor morbidity and resulting pain.3,42 In contrast, DeMaio et al 5 reported that only 21.2% (14/66) of patients receiving LB via ACB consumed no opioids with a median MME consumption of 28.6 (approximately 5.72 pills). Such differences in opioid consumption may stem from differing proportions of graft harvest, in which the DeMaio et al cohort utilized a larger proportion of QT autografts—a cohort who consumed significantly more opioids than patients with BTB autograft in the present study. However, it is also possible that the addition of iPACK and SSI in our study may have afforded patients greater pain control. While studies evaluating the efficacy of LB in a local infiltrative form have not clearly demonstrated clinical superiority to justify increased costs compared with nonliposomal formulations in both the knee and the shoulder,9,22,25,30 interestingly, both of these similarly designed studies in which LB was administered via ACB demonstrated reduced opioid consumption when compared with bupivacaine or ropivacaine alone.
Interestingly, patients undergoing ACLR with QT autograft consumed approximately 4 times more opioids and reported higher VAS pain scores throughout the study period compared with those receiving BTB autograft. This finding is somewhat counterintuitive, as it is generally believed that the bony preparation and donor-site morbidity of BTB harvest would be more painful than the soft tissue QT harvest utilized in the present study 16 and higher rates of postoperative anterior knee pain have led many to suggest that BTB autograft ACLR is more painful.10,19,20 However, this kneeling-associated anterior knee pain is typically reported months to years after the initial postoperative pain period analyzed in the present study19,29,31 and there are scarce data explicitly comparing acute postoperative pain and opioid consumption between BTB and QT autograft ACLR. 15 In our cohort, these differences persisted even after excluding 3 outliers reporting increased postoperative pain and opioid consumption in the QT cohort; therefore, it is unlikely that data fragility is the etiology of these findings. Furthermore, given the increased tendency for QT autograft ACLR to be performed in conjunction with LET/ALL procedures in our practice, it was considered that these additional procedures may lead to increased postoperative pain and opioid consumption in the QT cohort. However, subanalysis again demonstrated no significant difference in opioid consumption with or without additional LET/ALL procedure. While we believe that the suprasartorial component of the regional block used in the present study, which targets the intermediate femoral cutaneous nerve branch, provides adequate anesthetic coverage to the distal thigh to adequately cover patients undergoing QT autograft ACLR, it is possible that a different regional anesthetic strategy may be preferable for QT versus BTB autografts. Additionally, it may be possible that technical differences in bone tunnel preparation (outside-in retrograde drilling vs anteromedial vs transtibial), fixation strategies (suspensory fixation vs interference screw), or even utilization of suture augmentation (ie, Internal Brace; Arthrex) between BTB or QT graft choices could contribute to differences in postoperative pain. However, this is beyond the scope of the present study and is a topic that will require further careful research.
Despite adherence to recent recommendations, including from the American College of Surgeons opioids after surgery workgroup, for prescription of ≤20 opioid pain medication tablets after ACLR, >95% of opioids prescribed in the present study went unused. Such surplus is even greater than the 60% to 80% previously described after ACLR and other arthroscopic procedures13,14,21,37,41—highlighting the requisite modification in opioid prescription quantity in tandem with advances in nonopioid postoperative pain management strategies such as regional anesthesia blocks and strict adherence to multimodal regimens. Such surplus of unused opioids is particularly alarming considering reports as high as 75% of unused postoperative opioid prescriptions being handled improperly 2 and a recent meta-analysis implicating procurement of pharmaceutical drugs from friends and family as the primary source of nonmedical use of prescription drugs. 12 Ford et al. highlighted that opioids sourced from friends or relatives as being significantly associated with substance use disorders and other high-risk behaviors 7 and Hulme et al. posited that students, particularly those in fraternity or sorority environments, are those at a particularly high risk for pharmaceutical diversion. 12 Therefore, the typical demographic of those undergoing ACLR, largely consisting of the adolescent and young adult population as evidenced by a mean patient age of 27 years in the present study, is considered to be a group at “high risk” for opioid misuse and diversion.4,8,11,12,40 Thus, as postoperative pain management after ACLR becomes increasingly refined, providers must curtail their opioid prescription quantities to adequately combat misuse of prescription narcotics. In our study, we found that only 7.6% of patients required >5 tablets of oxycodone. In light of this, surgeons could curtail opioid prescriptions by 75% with very few patients requiring a refill of narcotics.
Limitations
This study must be considered with respect to its limitations. First, despite performing an a priori power analysis and reaching the recommended enrollment, this study was likely underpowered to detect a statistically significant difference in the primary study outcome of opioid consumption. Such finding likely stems from the reduced opioid consumption compared with expectation. Post hoc power analysis suggested that detection of a clinically meaningful difference between treatment groups would necessitate enrollment of nearly 1000 patients. However, with such limited opioid consumption between both treatment arms in the present study, the clinical utility to justify undertaking such a large randomized trial is limited. Second, LB can cost as much as 20 times that of bupivacaine at some centers. Numerous authors have asserted that the less costly bupivacaine-based formulations offer adequate postoperative pain control.9,30 The present study cannot offer conclusions or comparisons between LB or bupivacaine or regional anesthetic administration compared with local infiltration, although a trial in a pediatric population demonstrated superiority with regard to opioid consumption when LB was administered via ACB compared with bupivacaine alone. 39 Additionally, our center has begun analyzing the anesthetic effect of smaller doses of LB when administered via ACB, iPACK, and SSI in an attempt to offset the increased cost of LB. Furthermore, LB was approved for reimbursement by Medicare-associated surgery centers in 2019, and the novel NOPAIN Act mandating additional reimbursement for nonopioid perioperative pain management modalities could assuage some concerns about the additional costs of LB.6,27 Third, this study was conducted at a single, hospital-owned surgical center in which regional blocks were conducted by a single anesthesiologist. Therefore, our study design affords internal validity although these results may not be generalizable. Additionally, opioid usage rates were determined based on patient reports in their daily survey and opioid usage was not confirmed through the use of the opioid prescription monitoring program. However, we attempted to mitigate this limitation by surveying patients on their opioid usage daily in the initial postoperative period and verifying their reported use on surveys administered on postoperative day 7 and 14, with consistent agreement in reported use at all time points. Nonetheless, opioid consumption reports are subject to the Hawthorne effect characterized by patients potentially modifying their behavior and responses based on knowledge that their behavior is being observed, which could affect the results. However, it would be unlikely to affect the cohorts in a disparate manner. Finally, enrollment in the study was completely voluntary; therefore, the patients enrolled in this study were likely more motivated to curtail opioid use than an average patient potentially driving opioid consumption lower than could be expected for a more typical population.
Conclusion
A single shot of LB with or without dexamethasone via ACB, SSI, and iPACK block provided excellent pain control and minimized opioid consumption after autograft ACLR. Nearly 80% of patients did not require opioids postoperatively, making opioid-free ACLR a realistic possibility for many patients. When narcotics were required, the dose of opioids was minimal, with patients requiring 1.2 tablets of oxycodone on average and >95% of the opioids prescribed going unused. Despite previous reports suggesting a prolonged duration of anesthetic effect when LB is combined with dexamethasone, we found no difference in opioid consumption, VAS score, patient satisfaction, or duration of block effect when patients received dexamethasone with their regional block. The utilization of regional anesthetic techniques such as ACB, SSI, and iPACK blocks in conjunction with LB-based anesthetics could allow providers to curtail or potentially eliminate opioid prescriptions after ACL surgery.
Footnotes
Final revision submitted October 23, 2025; accepted October 28, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: A.V. has received speaking fees from Pacira Biosciences. S.H. has consulting fees from Arthrex Inc, speaking fees from Smith & Nephew, and support for education from Paladin Technology Solutions. S.B.C. has received royalties from Major League Baseball. M.G.C. is a member of the Major League Baseball Team Physicians Association. C.C.D. has received royalties and consulting fees from and owns stock or stock options in Aevumed. J.S. holds stock or stock options in Franklin BioScience and has received consulting fees from Stryker. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
This study received ethical approval by the Thomas Jefferson University Hospitals institutional review board (IRB No. 21D.369).
