Abstract
Background:
The importance of patient-reported outcome measures (PROMs) is becoming increasingly recognized in the orthopaedic field. The Banff Patellofemoral Instability Instrument 2.0 (BPII 2.0) is a disease-specific tool designed for patients with patellofemoral instability.
Purpose:
To adapt, translate, and validate the BPII 2.0 into Polish, to enable its use by Polish-speaking professionals for the evaluation of patients who present with patellofemoral instability.
Study Design:
Cohort study (Diagnosis); Level of evidence, 3.
Methods:
The final Polish version of the BPII 2.0 was investigated in patients with a confirmed diagnosis of recurrent patellofemoral joint (PFJ) instability. Patients completed 2 sets of PROMs, the BPII 2.0, Anterior Knee Pain Scale (Kujala), Norwich Patellar Instability (NPI) score, Lysholm knee score, and International Knee Documentation Committee [IKDC] Subjective Knee Form, at the clinic visit and 7 to 14 days later. The following psychometric properties were assessed: face validity, floor and ceiling effects, test-retest reliability, internal consistency, construct validity, standard error of measurement (SEM), and minimal clinically important difference (MCID). The reliability was evaluated by measuring the internal consistency (Cronbach alpha) and test-retest reliability (intraclass correlation coefficient). Pearson correlation coefficients were used to examine the validity.
Results:
The study population consisted of 80 postoperative patients (29 males, 51 females) aged 12 to 37 years with a confirmed diagnosis of recurrent PFJ instability. Face validity was assessed with an expert clinician group and an expert-patient group. No floor or ceiling effects were observed. The test-retest reliability of the BPII was 0.995 (P < .01). The BPII 2.0 score demonstrated excellent internal consistency, with a Cronbach alpha of 0.95 at baseline and 0.96 at the retest.
The correlations of the BPII with the other PROMs were statistically significant and classified as moderate to strong: IKDC Subjective Knee Form (Pearson rho = 0.725; P < .001), Lysholm score (rho = 0.58; P < .001), and Kujala score (rho = 0.533; P < .001). A significant negative correlation of moderate strength between the BPII and NPI score (rho = –0.615; P < .001) was calculated. The SEM was estimated at 1.329 and the MCID was 3.028.
Conclusion:
This study demonstrated that the Polish version of the BPII 2.0 is a reliable and valid instrument for Polish-speaking patients with recurrent patellofemoral instability. It will enable multicenter, international studies through uniform application of the functional assessment of patients with PFJ instability.
Keywords
Patellofemoral joint (PFJ) instability is one of the most common causes of limited physical activity and reduced quality of life in active sports patients, especially adolescents and young adults.5,11,17,18,27 For an objective assessment of patients’ experience of the intensity of PFJ instability and the effectiveness of treatment, it is necessary to use a repeatable, unambiguous, and reliable tool. A disease-specific patient-reported outcome measure (PROM) dedicated to the holistic assessment of the functional status and quality of life of patients with PFJ instability should be used to provide a reliable context of the severity of the condition.
There are many PROMs used in studies for the assessment of patients with patellofemoral instability. 11 However, most of them have not been specifically validated for patients with patellar instability.11,12,24 Only the Banff Patellofemoral Instability Instrument 2.0 (BPII 2.0) and Norwich Patellar Instability (NPI) score have been specifically validated in a patellofemoral instability population. 11 Standardized PROMs are essential for assessing the effectiveness of treatment and clinical response 11 and for comparative studies. In their systematic review, Høj et al 12 emphasized the necessity of high-quality comparative studies with consistent patient assessment measures. Furthermore, each PROM should undergo language and cultural adaptation to fulfill methodological requirements. 8
Currently, the most frequently used PROMs in clinical practice to assess patients with PFJ problems either do not concern patellar instability (Kujala score, Lysholm-Tegner score, and Knee injury and Osteoarthritis Outcome Score) or are not specific for PFJ (International Knee Documentation Committee [IKDC] Subjective Knee Form and Pediatric IKDC Subjective Knee Form). 11 Moreover, most of the tools currently used have not been adapted (validated) into Polish. This significantly limits the possibility of assessing both the initial functional status of the PFJ and the therapeutic effects of orthopaedists and physical therapists.11,22,23 One disease-specific PROM currently used to assess patellar instability is the BPII 2.0. 9 It was originally published in 2013 9 as a validated adaptation of the 1998 scale 21 to assess knee joint function in patients with anterior cruciate ligament insufficiency. In 2016, the BPII underwent factor analysis and item reduction to the current BPII 2.0 version. 16 The questionnaire contains 23 questions and collects information directly from the patient regarding the impact of patellar instability on their quality of life in 5 categories: (1) symptoms/physical complaints, (2) limitations related to work/school, (3) recreation/sports/activity, (4) daily activities/quality of life, and (5) social/emotional problems. Patients mark their answers on a visual analog scale measuring 100 mm. Each question has the same value, and the overall assessment result is the arithmetic mean of the individual questions. 16 Its validity, reliability, and responsiveness for the assessment of patients with patellofemoral instability, both surgically and nonsurgically treated, were successfully demonstrated in recent publications.9,16 Currently, the BPII 2.0 has been translated and validated into the German, 3 Dutch, 33 Portuguese, 7 French, 11 Swedish, 34 Indonesian, 26 Norwegian, 13 Spanish, 20 Turkish, 35 and Arabic 1 languages.
The BPII 2.0 has been endorsed by the European Society of Sports Traumatology, Knee Surgery and Arthroscopy Committee as a valid, reliable, and disease-specific tool for patients with patellar instability. 4 The consensus committee recommends including the BPII 2.0 and/or NPI scores as a minimum in studies concerning patellofemoral instability, while acknowledging that these PROMs are not validated for all languages. 4 Therefore, the aim of the study was to adapt and validate the BPII 2.0 into the Polish language. We hypothesized that a Polish version of the BPII 2.0 would be valid and reliable in patients with PFJ instability.
The aim of the study was to translate, adapt, and validate the BPII 2.0 into the Polish language. The linguistic adaptation allows for a correct and comprehensive assessment of Polish-speaking patients with recurrent PFJ instability. Validation provides Polish-speaking specialists with a disease-specific tool for assessing patients with PFJ instability.
Methods
Institutional review board approval (ID 1072.6120.213.2020) was obtained from the ethics committee. The main author (A.F.) received official recommendation for validation and Polish language adaptation of the BPII 2.0 from the Banff Sport Medicine Foundation and the Polish Arthroscopic Society.
Translation Procedure
According to Beaton et al, 2 the following steps were conducted:
Translation from English. Two independent, qualified translators transcribed the English version of the BPII 2.0 into Polish.
Synthesis of both translations. Both translations were merged into 1 document. The research team reviewed and discussed the 2 independent translations and agreed on a new Polish version of the BPII 2.0.
Back-translation into English. Two independent, qualified English translators fluent in Polish independently translated the new Polish version back into English.
Establishment of the preliminary Polish version of the BPII 2.0 (face validity from the professionals). The research team, consisting of translators, an orthopaedic surgeon, and 2 physical therapists, discussed and agreed on the translational agreement of both language versions of the research tool in Polish and the back-translated version into English. In addition, the research team contacted the authors of the original scale to confirm the English back-translation.9,16 These steps enabled the use of the BPII 2.0 to assess PFJ instability in Polish-speaking patients (see the Supplementary Materials for the Polish version of the BPII 2.0).
Testing of the preliminary Polish version of the BPII 2.0 (face validity from the patients). The research team randomly selected 32 patients with PFJ instability to ensure that all questions were easily understood. This group of patients was also asked to provide suggestions for modifying the questionnaire. None of the patients provided any suggestions for changes to the questionnaire. Therefore, the research team finalized the Polish version of the BPII 2.0 for testing in the study.
Validation Process
The analyzed group included patients >12 years of age who were primarily surgically treated for patellar instability (at least 2 objective patellar dislocation episodes in the past or a first-time acute patellar dislocation with the presence of a noncomminuted osteochondral fracture >1 cm2). Medial patellofemoral ligament reconstruction (double-bundle, ipsilateral gracilis tendon autograft) was performed in all. Osteochondral fractures were fixed with multiple absorbable transosseous sutures (PDS No. 0). Additional bony corrections were performed concomitantly based on radiological criteria from preoperative magnetic resonance imaging examination with the following thresholds: tibial tubercle osteotomy (TTO) with medialization (Elmslie-Trillat) for Tibial Tubercle to Trochlear Groove Distance (TTTG) >16 mm, TTO with distalization (Bayoumi) for a Caton-Deschamps index >1.2, deepening trochleoplasty (Bereiter) for high-grade trochlear dysplasia (Lateral Trochlear Inclination [LTI] <11° and Ventral Trochlear Prominence [VTP] >7 mm), and femoral derotational osteotomy (supracondylar, lateral distal subvastus approach) for femoral antetorsion >30° (as measured by the Murphy method 23 ). See Table 2 for details of surgical treatment.
The postoperative time frame for the BPII 2.0 completion ranged from 24 to 36 months after surgery. Patients were excluded from participating if they were unable to understand or complete the questionnaires or refused to give their written informed consent, or if additional injuries were present.
After providing written consent to participate in the study, each patient was assessed according to the developed study protocol. The study protocol consisted of (1) demographic data, (2) preoperative data, (3) data regarding the procedure, (4) postoperative data, and (5) PROMs, including the BPII 2.0, NPI score, Anterior Knee Pain Scale (Kujala), Lysholm knee score, and IKDC Subjective Knee Evaluation Form. The study patients completed the PROMs during the clinic visit.
Then, 7 to 14 days after the clinic visit, the patients received a second set of PROMs along with a question about whether there were any events (eg, trauma) after the clinic visit that could affect the result of the repeat PROMs. In the event of a negative answer, the patients completed the questionnaires again and emailed them to the main researcher.
The study enrollment is shown in Figure 1.

CONSORT (Consolidated Standards of Reporting Trials) flow diagram of patients eligible, recruited, followed up, and included in the analysis.
All patients enrolled in the study completed both sets of PROMs (test and retest).
Inclusion criteria to participate in the present study were recurrent patellofemoral instability, age ≥12 years, and formal consent provided. Exclusion criteria were a lack of formal consent to participate in the study and additional injuries of the lower extremities.
Outcome assessment instruments used in the present study are shown in Table 1.
Characteristics of Questionnaires Used in Validation of Polish BPII 2.0 a

PFJ, patellofemoral joint.
Psychometric Properties
The study sample size was determined according to recommendations from Terwee et al 32 suggesting a minimum of 50 patients for assessing construct validity. The initial score validation from Hiemstra et al 9 was successfully conducted with subsamples between 17 and 62.
The following psychometric properties were assessed: face validity, floor and ceiling effect, test-retest reliability, internal consistency, construct validity, the minimal clinically important difference (MCID), and the standard error of measurement (SEM).
Face validity assesses the degree to which experts agree that a questionnaire accurately assesses the construct.22,31 Face validity can also evaluate whether the individual questions of a PROM adequately reflect the construct being measured. 8
Floor and ceiling effects were evaluated as an indicator of whether the content of the BPII demonstrated limitations at either the top end (ie, the ceiling) or the bottom end (ie, the floor) of the scale. 22
Reliability presents the extent to which scores for patients who have not changed are the same for repeated measurement under several conditions: for example, using different sets of items from the same PROMs (internal consistency) over time (test-retest) by different persons on the same occasion (interrater) or by the same persons (ie, raters or responders) on different occasions (intrarater).8,25
Construct validity shows the degree to which the scores of a PROM instrument are consistent with hypotheses (eg, regarding internal relationships, relationships to scores of other instruments, or differences between relevant groups) based on the assumption that the PROM instrument validly measures the construct being measured.8,25
Statistical Analysis
The initial validity analysis included visual inspection of histograms, checking for normality of the distribution of key variables using the Shapiro-Wilk test, and testing for the homogeneity of variance with the Levene test. Descriptive statistics, that is, ranges, means with standard deviations, and medians with interquartile ranges, were obtained. It was assumed that if <15% of the patients scored minimum or maximum, there was no floor or ceiling effect. Possible outliers were defined as observations with scores either less than the first quartile minus 1.5 interquartile range (negative outliers) or more than the third quartile plus 1.5 interquartile range (positive outliers).
Internal consistency was determined by calculating the Cronbach alpha coefficient. A Cronbach alpha ≥.90 was considered excellent. Test-retest reliability was evaluated using intraclass correlation coefficients (ICCs) and 95% confidence intervals. Calculations were based on a 2-way, random, single-measure model with absolute agreement. A value >0.4 was considered sufficient (poor, 0.00-0.20; fair, 0.21-0.40; good, 0.41-0.60; very good, 0.61-0.80; and excellent, 0.81-1.00).
To verify the construct validity, Pearson correlation coefficients were calculated.
Positive correlations were expected for the BPII 2.0, and negative correlations were expected for the NPI score. A Spearman rho coefficient of 0.5 to 0.69 indicated correlations of moderate strength, while a coefficient ≥0.7 was considered to show a strong correlation.
Finally, in the case of the BPII 2.0, the SEM and MCID were estimated based on the observed score distribution. The SEM and MCID were calculated from the standard deviation observed in the first application of the test and the ICC, using the following formulas:

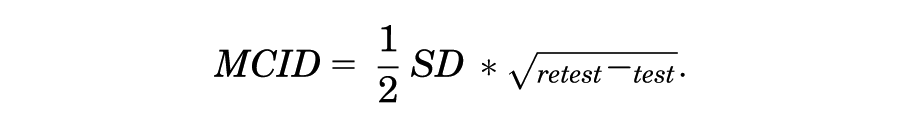
For the present analysis, the level of statistical significance was set at a P value of .05 and power of 80%.
In the case of a P value <.001, the notation “P < .001” was used. All calculations were performed in R (Version 4.0.2; R Foundation for Statistical Computing).
Results
The study population consisted of 80 patients (29 males, 51 females) aged 12 to 37 years at the time of assessment. The majority were adolescents with a median age of 16, with half of the patients in the interquartile range of 14 to 17 years of age. The demographic data, preoperative data, and procedures are given in Table 2.
Demographic Data, Preoperative Data, and Additional Procedures a

DFO, distal femoral osteotomy; MPFLr, medial patellofemoral ligament reconstruction; OCF, osteochondral fracture fixation; TTO, tibial tuberosity osteotomy.
Descriptive Statistics
BPII 2.0 scores ranged from 28.17 (28.52 on retest) to the maximum of 100 points (one person, who scored 99.78 on the retest). The mean scores were 68.44 (SD, 19.19) and 67.49 (SD, 18.93) on test and retest, respectively. The score distribution differed significantly from normal, being mildly left-skewed (first quartile [Q1] = 54.36, median [Me] = 69.76, third quartile [Q3] = 85.82 for test; Q1 = 53.79, Me = 68.33, Q3 = 84.01 for retest). Also, inspection of histograms revealed that a possible second peak could be observed, roughly between the score values of 85 and 90 (both on test and retest).
Face Validity
The team, consisting of an orthopaedic surgeon (J.F.) and 2 physical therapists (A.F. and K.Z.J.), discussed the relevance and comprehensiveness of the translated BPII 2.0. The second stage of assessing face validity was to investigate in the patient group if the questions in the Polish version of the BPII 2.0 were comprehensible.
The expert committee agreed that the Polish version of the BPII 2.0 had good face validity. Furthermore, support for good content validity was found, as interviewed patients reported (1) a high relevance of included items, (2) no missing key aspects of their knee-related quality of life, and (3) comprehensible instructions and questions in the BPII 2.0. Furthermore, the patients interpreted the instructions and questions as intended.
As no difficulties were encountered, no changes were made to the final version that was used in the validation.
Floor and Ceiling Effects
There were no floor or ceiling effects, that is, no outliers. However, the score distribution was mildly left-skewed and differed significantly from the normal distribution, with roughly one-fourth of the patients scoring between 80 and 100 points.
Test-Retest Reliability
All patients enrolled in the study (N = 80) completed the second set of questionnaires 7 to 14 days after the clinic visit. Total scores and subscale scores of the first and second completions are given in Table 3.
Total Scores and Subscale Scores and Differences Over Time of Polish BPII 2.0 a
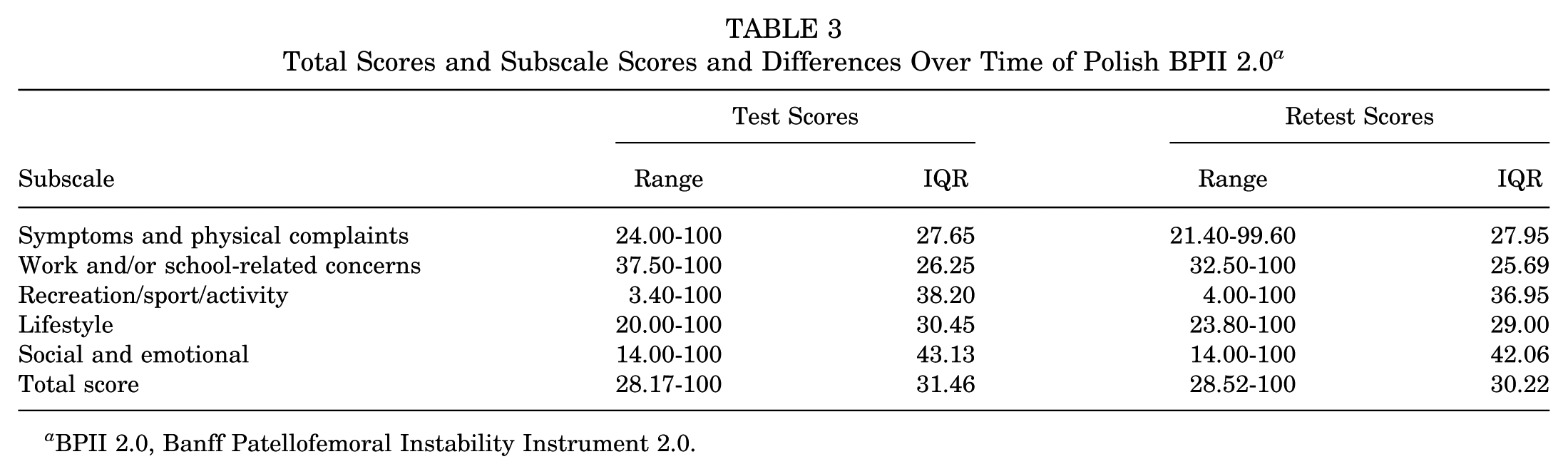
BPII 2.0, Banff Patellofemoral Instability Instrument 2.0.
The ICC for the first and second admission of the BPII 2.0 was 0.998 [F(79, 80) = 544.25; P < .01; 95% CI, 0.99-1.0].
Internal Consistency (Cronbach Alpha)
The BPII 2.0 demonstrated excellent internal consistency, with an Cronbach alpha of 0.95 at baseline (test) and 0.96 at the retest.
Construct Validity: Correlations With Other PROMS
The correlations of BPII 2.0 total scores with those of external measures were strong for the IKDC Subjective Knee Form (Pearson rho = 0.725; P < .001; 95% CI, 0.62 to 0.83) and moderate for the Lysholm knee score (rho = 0.58; P < .001; 95% CI, 0.40 to 0.70) and Kujala score (rho = 0.533; P < .001; 95% CI, 0.35 to 0.67) (Table 4). There was also an expected significant negative correlation of moderate strength between total scores in the BPII 2.0 and NPI (rho = –0.615; P < .001; 95% CI, –0.79 to –0.56).
Polish BPII 2.0 Correlations With Other Scales a

Boldface type indicates statistical significance. BPII 2.0, Banff Patellofemoral Instability Instrument 2.0; IKDC, International Knee Documentation Committee; NPI, Norwich Patellar Instability.
The overall results are summarized in Table 5.
Summary of Measurement Properties of Polish BPII 2.0 a

Boldface type indicates statistical significance. BPII 2.0, Banff Patellofemoral Instability Instrument 2.0; ICC, intraclass correlation coefficient; IKDC, International Knee Documentation Committee; NPI, Norwich Patellar Instability.
SEM and MCID
The SEM was 1.329. Therefore, the MCID could be estimated at 3.028 based on the distribution observed in the current study.
Discussion
The most important finding of this study was that the BPII 2.0 was successfully adapted as the first validated, disease-specific, and reliable tool for use by Polish-speaking patients with recurrent PFJ instability. The Polish version of the BPII 2.0 can be used for primary and/or secondary endpoint evaluation in clinical conditions to understand how a patient feels, functions, or survives.
In the process of cross-cultural language adaptation, face validity is one of the most important properties to be investigated. Face validity identifies the degree to which the items of an instrument seem to be an adequate reflection of the construct to be measured. 31 The present study indicated that the Polish version of the BPII 2.0 is relevant and comprehensible, and overall, it holds acceptable standards when used to assess these patients. These findings are in line with a similar study by a Norwegian group. 13
When analyzing the data from the present study, no floor or ceiling effects were found. This aligns with findings from the original publication 9 and other language validation studies,1,3,7,13,20,25,32,35 indicating that the questionnaire can capture changes at both ends of the score range. However, the score distribution in our study was distinctly left-skewed and differed significantly from the normal distribution, with roughly one-fourth of the patients scoring between 80 and 100 points (Q1 = 54.36, Me = 69.76, Q3 = 85.82 for test; Q1 = 53.79, Me = 68.33, Q3 = 84.01 for retest). The mean scores were 68.44 (SD, 19.19) and 67.49 (SD, 18.93) on test and retest, respectively, close to the mean postoperative score of 65 (of 100) reported by the authors of the original questionnaire. 16
The research team assessed the test-retest reliability of the Polish version of the BPII 2.0. In the current study, there were 7 to 14 days between the 2 questionnaires, but most of the patients completed and returned the questionnaires 7 days after the first examination. The test-retest reliability was very high, with an ICC of 0.998 at baseline, which is comparable to that of the original English version. 9
The Cronbach alpha was calculated to assess internal consistency. The Cronbach alpha value was very good for the Polish BPII 2.0 at 0.95 on the initial administration. In the original study, the Cronbach alpha value for the BPII 2.0 was 0.91, which is slightly lower than that for the Polish version. 9 In the translation and reliability studies conducted for the BPII 2.0 to other languages, these values were found to be 0.93 and 0.95 for the German version, 3 0.903 for the Turkish adaptation, 32 0.97 for the Indonesian version, 25 0.97 for the Swedish version, 34 and 0.95 for the Norwegian version. 13
To assess the construct validity of the BPII 2.0 in Polish, correlation with other established scores commonly used to evaluate patellofemoral instability were calculated. The Kujala score, NPI score, Lysholm knee score, and IKDC Subjective Knee Evaluation Form were used in the study to assess the validity. A strong correlation (r = 0.725; P < .001) was observed between the BPII 2.0 and IKDC. A moderate correlation was obtained with the Lysholm score (r = 0.58; P < .001), Kujala score (r = 0.533; P < .001), and NPI score (r = –0.62; P < .001). These results are slightly lower than those of the Indonesian adaptation (r = 0.98) 25 or Norwegian translation (r = 0.83) 13 studies. To the best of our knowledge, the Norwegian 13 and Polish adaptations are the only studies in which the IKDC questionnaire was used to assess concurrent validity. The present study demonstrated a strong correlation between the BPII 2.0 and IKDC Subjective Knee Form (r = 0.725). In the Norwegian adaptation, a moderate correlation between the BPII 2.0 and IKDC Subjective Knee Form (r = 0.56) was demonstrated. 13 The strong correlation between the BPII 2.0 and IKDC can be explained by some similarities in questions and the functional status of patients after a knee injury. Except for physical condition questions, in both questionnaires, there are questions about quality of life. Compared with the original version of the BPII 2.0, in our study a moderate correlation with the NPI score (rho = –0.615) was observed.
The present investigation is the second study, after the Norwegian validation of the BPII 2.0, to report the SEM of the BPII 2.0 in a cross-cultural adaptation. The SEM was calculated by multiplying the standard deviation observed in the first application of the BPII by square root of 1 minus the ICC as the measure of test-retest reliability
This Polish validation of the BPII 2.0 is, to the best of our knowledge, the first study to date to include an evaluation of the MCID of the BPII 2.0. The MCID is estimated at 3.028 based on the distribution observed in the current study. It is important to consider the limitations of distribution-based methods of calculating the MCID; the difference is not patient reported, and it might be sample or cohort specific. 33 It is necessary to emphasize that MCID calculation methods lead to highly heterogeneous values, 28 which significantly affect the percentage of patients achieving the MCID in a given population. 6
There were some limitations in this study. The inclusion of surgically treated patients in the study does not represent the full range of patients with PFJ instability. In addition, the study population may have influenced the floor or ceiling effects assessment. A study with a cohort of operated and nonoperated patients with PFJ instability should be assessed in the future. The second limitation of the study could be the lack of use of the kinesiophobia scale, which was included in the Norwegian study. 13 It has been demonstrated that patients evaluated with patellar instability have moderate to high levels of kinesiophobia 10 and may avoid physical activity. 9 In future research, correlation of the BPII 2.0 with the kinesiophobia scale should be evaluated. 9
Conclusion
Our study demonstrated that the BPII 2.0 has been successfully translated and adapted to the Polish population. The findings of this study indicate that the Polish version of the BPII 2.0 is a reliable and valid PROM of PF instability and can be used to evaluate treatment results for the Polish population.
Supplemental Material
sj-docx-1-ojs-10.1177_23259671251391815 – Supplemental material for Polish Validation and Language Adaptation of the Banff Patellofemoral Instability Instrument 2.0
Supplemental material, sj-docx-1-ojs-10.1177_23259671251391815 for Polish Validation and Language Adaptation of the Banff Patellofemoral Instability Instrument 2.0 by Alicja Fąfara, Jarosław Feluś and Kinga Żmijewska-Jasińska in Orthopaedic Journal of Sports Medicine
Footnotes
Acknowledgements
A.F. expresses sincere appreciation to Laurie Hiemstra, PhD, and Sarah Kerslake, BPhty, MSc, from the Banff Sport Medicine Foundation for the official recommendation and permission to adapt and validate the Polish BPII 2.0. A.F. is grateful for the invaluable support provided by Sarah Kerslake, BPhty, MSc, research director of the Banff Sport Medicine Foundation, throughout the research process. The consultations and provided instructions were extremely helpful and instrumental in completing this research project and paper successfully.
Final revision submitted October 7, 2025; accepted October 9, 2025.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the ethics committee of Jagiellonian University Medical College, Kraków, Poland (ID 1072.6120.213.2020).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
