Abstract
Background:
Dual-energy x-ray absorptiometry (DEXA) is a widely used imaging modality in sports medicine and orthopaedics due to its accuracy in assessing bone mineral density (BMD) and body composition. Tracking these parameters provides valuable insights into the recovery process after musculoskeletal injuries, which are prevalent among athletes.
Purpose:
To evaluate how DEXA imaging has been used to guide rehabilitation in both recreational and competitive athletes by monitoring changes in BMD and body composition after musculoskeletal injuries.
Study Design:
Scoping review; Level of evidence, 4.
Methods:
A comprehensive literature search was conducted in Ovid MEDLINE, EMBASE, Cochrane Central, and SportDISCUS databases (2000-2024). Studies were included if they used DEXA to measure BMD or body composition changes in athletes after musculoskeletal injuries. Titles, abstracts, and full texts were screened independently by 2 reviewers, with discrepancies resolved through discussion. Data were synthesized qualitatively, and major trends were reported and identified.
Results:
Of 1132 unique records, 12 studies met inclusion criteria, involving 319 athletes (34% female) with injuries such as anterior cruciate ligament (ACL) tears, lumbar stress fractures, femoroacetabular impingement, and Achilles tendon ruptures. Athletes represented a range of sports, including soccer, basketball, cricket, and triathlon, at both competitive and recreational levels. DEXA identified significant BMD declines after injury, particularly in the affected limb. For instance, up to a 7% BMD reduction was observed in surgical limbs after ACL reconstruction, persisting for up to 2 years in some cases. Rehabilitation strategies incorporating blood flow restriction therapy or combined running and isometric exercises better preserved lean mass and BMD compared with conventional programs focused solely on closed and open kinetic chain exercises.
Conclusion:
The review demonstrated that DEXA imaging is a promising tool in orthopaedic sports medicine for assessing injury-related changes in BMD and body composition. It provides detailed insights into recovery processes and aids in tailoring rehabilitation strategies and return-to-sport decisions. Some studies incorporated advanced rehabilitation methods such as blood flow restriction therapy, which appeared to accelerate recovery as measured by DEXA outcomes. Future studies should explore integrating DEXA findings into athlete-specific rehabilitation protocols with validated thresholds for return to play.
Dual-energy x-ray absorptiometry (DEXA) imaging is widely recognized for its accuracy in measuring bone mineral density (BMD) and body composition, providing crucial data on skeletal health in various medical contexts. DEXA scans use low-dose x-rays to quantify the mineral content per unit area of bone, offering precise assessment of BMD, bone mineral content (BMC), and bone area. Furthermore, DEXA scans can provide precise measurements in lean and nonlean mass in specific areas of the body, allowing for an understanding of asymmetry. In addition to collecting this scope of data, DEXA is a rapid imaging modality and uses little ionizing radiation (effective radiation dose of 3 µGy per scan). 16 Although traditionally used in the diagnosis and management of osteoporosis, DEXA has increasingly found applications in broader clinical contexts such as sports medicine and orthopaedics, particularly in the evaluation and management of musculoskeletal injuries sustained by athletes.5,10 Given the high physical demands placed on athletes, maintaining optimal bone and muscle health is critical, especially during the rehabilitation and recovery phases after an injury or surgery. 22 Athletes face unique recovery challenges not encountered in the general population, including the pressure for expedited return-to-play timelines, intense physical loads, and sport-specific biomechanical demands. For example, sports that routinely require asymmetrical performance between left and right sides, such as throwing sports, stick/racquet/club sports, and soccer, may necessitate tailored rehabilitation strategies that target limb-specific recovery. Additionally, training volume, seasonal competition schedules, and psychological performance pressures often require clinicians to use highly sensitive tools like DEXA to monitor microlevel changes in musculoskeletal health that could affect performance or motivation or predispose the athlete to reinjury. The precise, rapid, and noninvasive nature of DEXA makes it particularly attractive for monitoring changes in bone and muscle composition over time, offering potential benefits in individualizing rehabilitation strategies.
Injuries to the upper or lower extremities are common in athletes and can significantly affect performance, in both the short and long term. 31 Effective rehabilitation aims not only to restore function but also to optimize physical conditioning and nutrition to prevent future injuries. DEXA imaging offers a unique advantage in this context by providing detailed insights into changes in BMD, muscle mass, and body composition, which are essential parameters in the recovery process. 18 Early and accurate assessment of changes in bone density and muscle mass during the recovery process could provide critical information to guide rehabilitation, ensuring a return to optimal function and minimizing the risk of reinjury or long-term deficits. 27
This scoping review examined the existing evidence on the use of DEXA imaging to guide rehabilitation and recovery in athletes who have sustained musculoskeletal injuries. The review focused on how DEXA has been used to assess the effect of these injuries on bone and muscle composition, its role in informing and adjusting rehabilitation protocols, and its effectiveness in improving recovery and return-to-sport outcomes. By synthesizing the current literature, we aimed to clarify the value of DEXA in sports rehabilitation, highlighting its potential benefits and identifying areas for future research.
Methods
Study Design
The purpose was to summarize available evidence on the use of DEXA in evaluating recovery after sports injuries in athletes, identify gaps in the literature, and make recommendations for future use. Specific outcomes of interest included changes in BMD and body composition (lean mass and nonlean mass) during the recovery process as well as return-to-play and functional/biomechanical outcomes. The PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) extension for scoping reviews (PRISMA-ScR) checklist was followed for the reporting of items in this review. 29
Search Strategy
A comprehensive search was conducted in the Ovid MEDLINE(R), EMBASE, Cochrane Central Register of Controlled Trials, and SportDISCUS from 2000 to 2024 with no restriction on language of publication. An experienced medical librarian developed the database-specific search strategies (Table 1). The reference lists of all eligible studies and related systematic reviews were also searched for additional eligible studies.
Characteristics of Included Studies a

ACL, anterior cruciate ligament; ACLR, anterior cruciate ligament reconstruction; DEXA, dual-energy x-ray absorptiometry; LSF, lumbar stress fracture; MTSS, medial tibial stress syndrome; NR, not reported.
Eligibility Criteria
Primary studies that met the following inclusion criteria were included in this scoping review: (1) The population studied consisted of athletes who sustained musculoskeletal injuries; (2) DEXA measurements were performed at baseline (shortly after injury); (3) at least 1 follow-up DEXA measurement was taken at any time point after the baseline measurement; (4) DEXA measurements were performed near the site of injury. This was defined anatomically as within the specific bone or muscle group directly affected by the injury (eg, the distal femur in the case of ACL injuries, the proximal tibia in tibial stress injuries, or lumbar vertebrae L1-L4 in lumbar spine injuries) to ensure that measurements reflect localized changes in bone and muscle composition relevant to the injury site.
Review papers or studies that did not meet the inclusion criteria above were excluded.
Data Screening and Extraction
Two reviewers (R.D. and U.T.) independently screened titles, abstracts, and full texts for eligibility using Covidence Systematic Review Software (Veritas Health Innovation). Disagreement was resolved by group discussion, with arbitration by a third author (A.H.W.C.) where differences remained. We calculated interrater agreement at the full-text screening stage using the kappa (κ) statistic. The κ value was close to 1, indicating strong agreement between reviewers. The data were extracted and recorded using Microsoft Excel spreadsheets. The following data were extracted from eligible studies: country of publication, study design, population studied, type of injury sustained, intervention details, demographic information (ie, sex, age, body mass index [BMI]), participant sport and level of competition, baseline data (ie, time of baseline testing and data on BMD, body composition, or other notable values), and follow-up testing (ie, BMD, body composition, and other notable outcomes).
Narrative Analysis
This scoping review used a narrative synthesis of the included studies due to heterogeneous study designs, interventions, outcomes, and populations, which precluded meta-analysis. Findings were narratively synthesized and organized by themes related to the research questions, intervention types, and outcome measures, specifically focusing on injury or surgery type, BMD, and body composition trends, and return-to-sport or functional outcomes. Key patterns, similarities, and differences were noted, with attention to study contexts. Factors such as study populations, type of intervention, and outcome assessment methods were explored to explain variations and contradictions in findings.
Results
Of 1132 unique search results, 12 studies that used DEXA to monitor BMD and other body composition changes in a total of 319 athletes (34% female) sustaining various musculoskeletal injuries were included in this scoping review (Figure 1). # The following injuries were noted by the included studies: anterior cruciate ligament reconstruction (ACLR) (n = 7 studies),2,9,12-14,19,32 unspecified “knee injury” (n = 1 study), 11 lumbar stress fracture (n = 1 study), 1 femoroacetabular impingement with labral tear (n = 1 study), 20 medial tibial stress syndrome (MTSS) (n = 1 study), 17 and Achilles tendon rupture (n = 1 study). 28 The weighted mean age and BMI of athletes across the included studies were 24.5 ± 4.4 years and 25.1 ± 1.2 kg/m2, respectively. The type of sport played by athletes, as well as level of competition, varied across included studies, with some studies including a diverse range of athletes. The sports played by athletes in the included studies were American football, soccer, basketball, spirit, track and field, crew, softball, volleyball, wrestling, tennis, cricket, and ironman triathlon, as well as unspecified recreational and competitive athletes. A detailed summary of characteristics of included studies can be viewed in Table 1. Furthermore, an overview of outcomes reported by included studies is provided in Table 2.

PRISMA flowchart with exclusion details: studies excluded due to lack of injury (n = 15), lack of baseline bone mineral density (BMD) measurement (n = 7), lack of injury site-specific BMD measurement (n = 5), follow-up BMD measurements not performed (n = 2).
Overview of Outcomes Reported by Included Studies
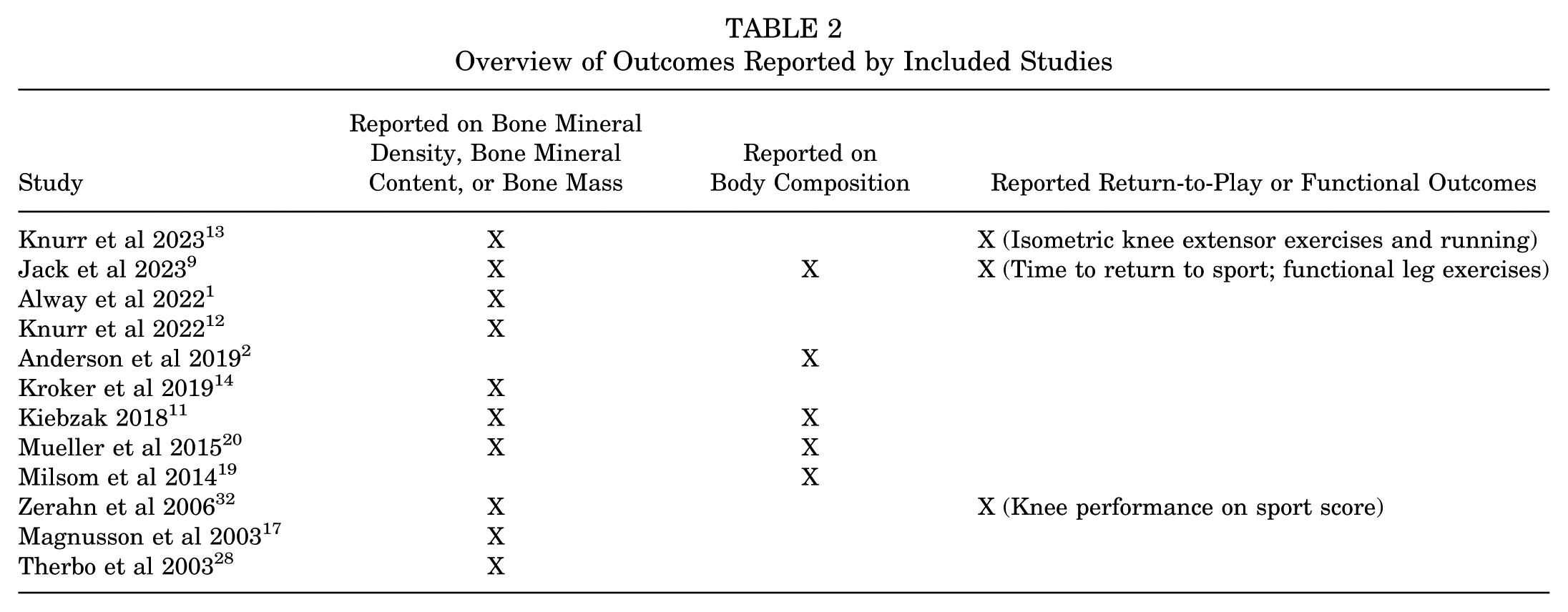
Bone Mineral Density Changes
Ten of the included studies reported findings on BMD changes using DEXA, examining a range of injuries including ACL tears,9,12-14,32 lumbar stress fractures, 1 femoroacetabular impingement with labral tear, 19 MTSS, 17 Achilles tendon ruptures, 28 and unspecified knee injuries. 11 In most studies, BMD initially decreases substantially after injury, with varying degrees of recovery over time depending on injury type and intervention. For knee injuries, regions of interest (ROIs) that were found to be reproducible among studies to take BMD measurements were centered at 5% and 15% (F5, F15) of femoral length, measured from the intercondylar notch. 13 In ACL tears,9,12-14,32 multiple studies showed that BMD decreased in the surgical limb, particularly at the 5% mark of the femur,12,13 with varying recovery patterns based on rehabilitation type. In general, BMD typically decreased by approximately 4% to 10% within the first 6 months after injury or surgery, with a rapid decline of approximately 0.8% to 1.2% per month during this period.9,12-14,32 This range represents the mean monthly percentage decline in regional BMD reported across 4 studies examining ACL injuries. Compared with athletes who received traditional rehabilitation with a combination of closed and open kinetic chain exercises, athletes who received blood flow restriction (BFR) therapy showed greater BMD and lean mass measurements by 12 weeks, with lean mass typically recovering faster than BMD. 9 Furthermore, a rehabilitation protocol consisting of running and isometric knee exercises after ACLR led to partial or near-complete recovery in BMD by 25 months. 13 However, a general trend was observed in which BMD deficits were observed around a 12-month follow-up but returned to near baseline levels by 24 months and onward.12,13,32 In 2 studies, BMD deficits at the 15% femoral level were persistent past 24 months,12,13 particularly for athletes with lower peak knee extensor movement and RTD. 13 In cricket fast bowlers, lumbar stress fractures exhibited BMD decreases on the contralateral side (the side opposite to the bowling arm) but recovered and surpassed baseline by 52 weeks. 1 A single study examining Achilles tendon ruptures found consistent decreases in BMD across various regions, with a notable 6.8% reduction at the greater trochanter after 1 year. 28 MTSS managed nonoperatively showed a significant increase in BMD in the painful region over 5 years. 17 Although a case of femoroacetabular impingement treated with hip arthroscopy initially resulted in a decrease in BMD during the 2 months of immobilization after surgery, significant recovery was observed within 6 weeks of rehabilitation involving cycling, swimming, and resistance training, with full recovery to presurgery levels achieved within 9 months. 20 Recovery timelines varied significantly, with some injuries like ACL tears showing partial recovery within 12 to 25 months, whereas others, such as MTSS, demonstrated BMD increases over several years. A detailed summary of BMD outcomes reported by included studies is provided in AppendixTables A1 and A2.
Body Composition Changes
Five of the included studies used DEXA to report on body composition changes among athletes with injuries. These studies examined athletes after ACLR,2,9,19 an unspecified “knee injury” managed through dietary and exercise interventions on a treadmill and stationary bike, 11 and femoroacetabular impingement with labral tear treated with hip arthroscopy. 20 Across the 5 studies, body composition changes showed consistent patterns after injury and rehabilitation. Lean mass typically decreased during periods of immobilization or inactivity, with the most significant losses observed in the injured limbs.2,9,11,19,20 Across studies, injured limbs showed approximately 7% to 14% lean mass reductions during immobilization, with partial to full recovery during rehabilitation, whereas unaffected limbs remained stable or experienced minimal changes (<5%).2,9,11,19,20 Certain interventions such as BFR therapy 9 and rehabilitation programs incorporating exercise and/or diet interventions led to lean mass recovery,2,11,19,20 with 1 study reporting full recovery by the end of the follow-up period of 9 months. 20 Some studies showed that dietary interventions that focused on achieving higher daily energy intakes (2500-3000 kcal/d) 2 as well as diets with high protein content (30-40 g per meal) and supplementation with a daily multivitamin, vitamin C, β-hydroxy-β-methylbutyrate, creatine monohydrate, and fish oil concentrate 19 may help prevent muscle atrophy and micronutrient deficiencies during recovery. Additionally, studies showed that interventions with early isometric/isotonic contractions 19 and a gradual progression in weightbearing activities may aid in athletes’ recovery.19,20 Fat mass generally decreased during initial recovery phases but tended to normalize or remain slightly lower after rehabilitation as seen during final follow-up periods between 5 and 12 months.2,11,19 Overall, interventions that combined structured rehabilitation and dietary adjustments facilitated improvements in lean mass and reductions in fat mass as determined by DEXA. A detailed summary of body composition outcomes reported by included studies is provided in AppendixTables A1 and A2.
Return-to-Play and Functional Outcomes
Three studies commented on return-to-play and functional outcomes among the injured athletes. In the 2023 study by Knurr et al, 13 athletes with ACLR were either tested with isometric knee extensor exercises (to study peak torque [PT] and rate of torque development [RTD]) or were assessed while running (to study peak knee flexion [PKF] or peak knee extensor movement [PKEM]) so the authors could examine associations between exercise performance and biomechanics with BMD changes. No significant interactions were found between time after ACLR and PT, RTD, PKF, or PKEM for 5% femur BMD. However, significant interactions were observed for these variables with 15% femur BMD, indicating that athletes with lower RTD and PKEM at around 9 to 10 months after ACLR experienced decreases in BMD at the distal femur over time. In the study by Jack et al, 9 time to return to sport was significantly shorter in the BFR group (mean ± SD, 6.42 ± 0.31 months) compared with the routine rehabilitation group (8.30 ± 0.54 months). Functional assessments included single-leg (SL) squat, SL eccentric step-down, SL press, SL hamstring curl, and Y balance. Both groups showed similar improvements in all functional measures over 4 weeks, except for the anterior Y-balance, where only the BFR group had a significant improvement between 8 and 12 weeks. This corresponded to their higher DEXA BMD scores compared with the control group. Zerahn et al 32 found that at 24-month follow-up after ACLR and structured rehabilitation, athletes’ knee performance on sport score increased from a median of 2 (abnormal) to 1 (almost normal). Knee performance on activity of daily living increased from a median of 2 (abnormal) to 0 (normal). This improved knee performance in sports activities was associated with increases in BMD Z scores for both the medial and lateral tibia. A detailed summary of return-to-play and functional outcomes reported by included studies is provided in AppendixTables A1 and A2.
Discussion
This review represents the first effort to systematically consolidate fragmented and heterogeneous evidence on the role of DEXA imaging in sport-specific recovery from musculoskeletal injuries. Although various studies have explored DEXA in isolated contexts, no previous synthesis has integrated findings across injury types, athletic populations, and recovery outcomes. By bringing together this dispersed literature, the present review offers a comprehensive understanding of how DEXA can inform individualized rehabilitation strategies, monitor localized bone and muscle changes, and contribute to return-to-play decision making in athletes.
As DEXA can provide highly accurate, region-specific measurements of BMD and body composition changes, healthcare professionals can use it to monitor injury effects and recovery progress over time. 7 This capability is particularly valuable in sports medicine, where athletes’ ability to resume training and competition with minimized reinjury risk is essential. Furthermore, the capability of DEXA to effectively identify sport-specific bone density adaptations and asymmetry in athletes highlights its relevance in sports medicine for monitoring both physiological and pathological changes. 4
The evidence reviewed consistently shows that BMD often decreased after injury or surgery, followed by time off sport, particularly in the affected limb. For instance, ACL injuries demonstrated a notable reduction in BMD in the injured limb, with partial to near-complete recovery observed over 12 to 25 months after injury.12,13 Similarly, a case-control study by Bayar et al 3 involving recreational athletes with complete ACL ruptures demonstrated significant region-specific BMD loss in ACL-deficient knees, particularly in the lateral femoral condyles and patella, which was associated with injury duration and activity levels. This aligns with our findings and with other studies on localized BMD reduction, 24 highlighting the potential of targeted rehabilitation to mitigate these deficits. However, studies indicate that certain factors, such as lower peak knee extensor torque, can delay recovery of BMD at specific femoral sites, underscoring the importance of individualized rehabilitation that takes into account an athlete's baseline strength and injury severity. 13 In cases of lumbar stress fractures, BMD recovery exceeded baseline levels by approximately 52 weeks, suggesting that specific injury types may even benefit from targeted rehabilitation that promotes bone density gains beyond preinjury status. 1 These findings infer that DEXA's detailed monitoring of BMD may guide adjustments in rehabilitation intensity and duration to optimize recovery and minimize long-term deficits. Similarly, a prospective longitudinal study involving athletes with tibial bone stress injuries used high-resolution peripheral quantitative computed tomography to measure volumetric BMD, noting an initial decline in volumetric BMD in both injured and uninjured limbs, with recovery to baseline by 24 weeks and exceeding baseline values at 52 weeks, aligning with our observations on lumbar stress fracture recovery. 25
DEXA has also proven valuable in tracking body composition changes during recovery, especially in assessing lean muscle mass changes in injured limbs. Immobilization after injury typically leads to lean mass losses in the affected limb, whereas targeted rehabilitation, particularly those programs that incorporate resistance training or BFR therapy, can help restore lean mass. 9 The studies included in this review demonstrate that DEXA was effective in quantifying these lean mass changes and monitoring recovery, with some interventions accelerating full recovery of lean mass. However, it should be noted that previous studies have found DEXA to overestimate trunk lean mass compared with magnetic resonance imaging (MRI), particularly in individuals with high intramuscular fat, suggesting the need for careful interpretation of lean mass recovery metrics in specific populations like those with spinal cord injuries.15,26 As MRI may be impractical in certain settings, DEXA has been validated as a feasible alternative for thigh muscle mass assessment in spinal cord injuries. 15 Moreover, the use of DEXA in evaluating body composition asymmetries may be helpful in understanding injury risk in athletes. A recent cross-sectional study by Vaillancourt et al 30 found significant asymmetries in upper limb and trunk lean body mass among university soccer players, with lower limb asymmetry remaining minimal due to the symmetric demands of running. Although no significant correlations were observed between asymmetries and injury risk, marginal associations were noted between trunk and lower limb asymmetry indices and lower limb injuries, suggesting that body composition asymmetry may be a potential modifiable risk factor for injury prevention in soccer players. 30
In evaluating functional outcomes and return-to-play timelines, this review suggests that DEXA imaging may contribute indirectly by offering data that informs targeted rehabilitation strategies. For example, Jack et al 9 used DEXA to track lean mass recovery in athletes undergoing BFR therapy after ACLR. Not only did the athletes in BFR group have significantly higher lower extremity lean mass, but they also demonstrated accelerated return-to-play times compared with the non-BFR group, despite similar functional scores at the end of follow-up. 9 In a prospective cohort study of collegiate athletes, lower total body BMD and higher MRI grades (based on periosteal edema on T1- and T2-weighted images) were independent predictors of delayed recovery in bone stress injuries, emphasizing the clinical relevance of integrating BMD assessments into routine conditioning and rehabilitation planning. 21 This underscores the potential role of a comprehensive rehabilitation approach that combines DEXA imaging with functional assessments to ensure both structural and biomechanical readiness for return to sport.
Limitations and Future Directions
Although this review provides valuable insights into the utility of DEXA imaging, several limitations should be noted. The variability in study design, injury types, and rehabilitation protocols limits comparability. Although formal risk of bias assessments were not performed, given the scoping review framework, most included studies were observational in nature with small sample sizes and limited follow-up, which restricts the strength of conclusions drawn. A notable overrepresentation of ACL injury studies skews the evidence base toward lower limb pathologies, whereas upper body injuries—such as shoulder dislocations/instability, rotator cuff tears, and clavicular fractures—remain underrepresented. This imbalance restricts the generalizability of findings across the full spectrum of athletic injuries and underscores the need for further research on upper extremity rehabilitation monitored through DEXA. Additionally, heterogeneous follow-up durations may fail to capture full recovery trajectories or introduce confounding variables. Observational designs prevalent in the literature hinder causal inferences between DEXA-monitored recovery markers (eg, BMD, lean mass) and functional outcomes. Without standardized preseason, postinjury, and rehabilitation DEXA protocols, it remains challenging to establish definitive relationships between DEXA changes and recovery metrics. Inconsistent scanning protocols and preparation methods, such as hydration and positioning variability, may affect measurement reliability. Furthermore, a lack of sport-specific baseline values for BMD and body composition complicates recovery assessments and protocol adjustments.
Hind et al 8 emphasized the need for standardized DEXA scanning protocols and precision error reporting in athletic populations to ensure accurate interpretation of body composition changes during recovery. They highlight that inconsistencies in prescan preparation—such as variability in hydration, fasting, and positioning—can lead to significant errors in lean and fat mass estimates. For example, dehydration may falsely lower lean mass readings, whereas fluid overload could exaggerate them. Improper or inconsistent positioning during the scan can lead to asymmetric data, especially in limb-specific assessments, potentially masking or exaggerating recovery progress. Similarly, differences in limb-specific measurements—if the dominant or injured side is not consistently assessed—can undermine longitudinal comparisons. This reflects the importance of consistent protocols to enhance measurement reliability. 8
To address these limitations, clinicians may consider adopting provisional thresholds to guide rehabilitation progress. For instance, research indicates that asymmetries >10% in muscle strength or mass are associated with an increased risk of injury and may impede optimal performance. Actionable metrics can improve consistency in monitoring and decision making.
Furthermore, future research should focus on identifying the most appropriate anatomic sites for DEXA assessments tailored to specific injury types, as the optimal site for monitoring BMD may depend on the injury location, rehabilitation goals, and bone characteristics at the affected region. 23 A need remains to establish definitive normative baseline BMD and body composition values for athletes across various sports. Dobrosielski et al 6 emphasized the importance of using DEXA to establish sport-specific baseline and recovery norms, highlighting significant differences in BMD and lean mass between athletes in high-impact sports (eg, gymnastics, basketball) and those in low-impact sports (eg, swimming, cross country). These baselines are crucial for identifying deviations during recovery, allowing for precise adjustments in rehabilitation protocols to ensure athletes return to preinjury levels of BMD and lean mass.
Conclusion
Our review demonstrated that DEXA imaging is a promising tool in orthopaedic sports medicine for assessing injury-related changes in BMD and body composition. It provides detailed insights into recovery processes and aids in tailoring rehabilitation strategies and return-to-sport decisions. Some studies incorporated advanced rehabilitation methods, such as BFR therapy, which appeared to accelerate recovery as measured by DEXA outcomes. Future studies should explore integrating DEXA findings into athlete-specific rehabilitation protocols with validated thresholds for return to play. DEXA's ability to detect subtle changes in bone and muscle health may make it a valuable tool for evaluating rehabilitation efficacy and informing clinical decisions in sports medicine.
Footnotes
Appendix
Summary of Outcomes Examined by Included Studies on Non-ACL Injuries a

| Study | Study Group | Baseline Testing | Follow-up Testing | Other Notable Results (eg, return to activity, function) | Summary | ||||
|---|---|---|---|---|---|---|---|---|---|
| Time of Baseline Testing After Injury | BMD, g/cm2, Mean ± SD | Other Notable Baseline Values, Mean ± SD | Follow-up | BMD, g/cm2, Mean ± SD | Bone Density Change Score (from baseline) | ||||
| Alway et al 2022 1 | Intervention (n = 17): injured players receiving LSF rehabilitation | 11 underwent DEXA scans on date of diagnosis, 2 scanned within 14 d of diagnosis, and 4 scanned before diagnosis | L1-L4 1.464 ± 0.150, L3 1.634 ± 0.170, CL3 1.795 ± 0.215, IL4 1.633 ± 0.184, |
Fat-free mass was 69.01 ± 3.85 kg, and career match workload was 3887 ± 4217 balls. | Short term (5-11 wk after baseline or diagnosis), medium term (15-34 wk) and long term (52-59 wk) | Depicted in graph. Raw values could not be estimated. | BMD decreased for up to 21-24 wk after diagnosis (L1-L4, 2.3%; IL3, 1.9%; CL3, 3.0%; IL4, 2.2%; CL4, 2.0%) and increased to above baseline levels by 52 wk (L1-L4, 2.2%; IL3, 0.7%; CL3, 1.8%; IL4, 1.9%; CL4, 2.1% greater than baseline). | Fast bowlers with LSF demonstrated |
Fast bowlers with LSF had lower lumbar BMD than uninjured bowlers, particularly on the contralateral side, decreasing up to 3.0% by 20-24 wk and recovering by 52 wk. |
| Control (n = 12): uninjured players | Reported above | L1-L4 1.585 ± 0.134, L3 1.703 ± 0.175, CL3 1.968 ± 0.256, IL4 1.711 ± 0.157, and |
Fat-free mass was 71.13 ± 7.40 kg, and career match workload was 3933 ± 3767 balls. | Short term (5-11 wk after baseline or diagnosis), medium term (15-34 wk) and long term (52-59 wk) | Depicted in graph. Raw values could not be estimated. | Small increases in BMD across time at L1-L4, IL3, and CL3 sites, with 1.6%, 1.8%, and 2.0% increases at 52 wk, respectively, compared with baseline. IL4 and CL4 increases observed for the first 28-30 wk (2.1% and 0.7% respectively), before decreasing to near baseline levels at 52 wk (0.5% and 0.3%, respectively). | |||
| Kiebzak 2018 11 | Intervention (n = 1): diet program, exercise on a treadmill and stationary bike | 12 mo | BMD 1.245 | Fat mass: 29.02 kg LM 39.61 kg |
12 mo | BMD 1.230 | NR | Fat mass 22.58 kg LM 41.98 kg |
|
| Mueller et al 2015 20 | Intervention (n = 1): hip arthroscopy, followed by immobilization and gradual increase in training | 4 y before injury (T0) and 1 wk after surgery (T1) | NR | NR | 8 wk after surgery (T2), 14 wk after surgery (T3), 9 mo after surgery (T4) | NR | Total aBMD in the left proximal femur |
Left lower extremity LM increased from T0 DEXA to T1, decreased by 4.5% during immobilization, then rose by 4.5% from T2 to T3 and by 6.9% from T3 to T4. |
The DEXA assessment showed a 22% fat reduction and 6% LM increase, reflecting successful diet and exercise. After hip arthroscopy, aBMD and LM losses in the lower extremity and lumbar spine were fully reversed after 6 wk of training, with a professional Ironman recovering presurgery levels in 4 mo and maintaining them at 9 mo. |
| Magnusson et al 2003 17 | Intervention (n = 14): athletes with MTSS | 29 mo (range, 5-120 mo) | Total body, 1.26 ± 0.05; femoral neck, 1.17 ± 0.10; Ward triangle, 1.13 ± 0.15; trochanter region, 1.06 ± 0.11; tibia region 1, 1.27 ± 0.13; tibia region 2, 1.52 ± 0.15; tibia region 3, 1.92 ± 0.23; tibia region 4, 1.39 ± 0.19; tibia region 5, 1.33 ± 0.17 | NR | Mean ± SD 5.7 ± 1.3 y | Total body, 1.30 ± 0.07; femoral neck, 1.13 ± 0.14; Ward triangle, 1.06 ± 0.20; trochanter, 1.03 ± 0.13; tibia region 1, 1.28 ± 0.14; tibia region 2, 1.56 ± 0.14; tibia region 3, 1.93 ± 0.20; tibia region 4, 1.65 ± 0.22; tibia region 5, 1.30 ± 0.15 | Change per year: |
During the follow-up, athletes with MTSS experienced a significant increase in BMD by |
The localized low BMD in the tibial region corresponding to the pain increased after recovery in athletes with MTSS. |
| Control (n = 24): nonathletes (without MTSS) | Reported above | Total body, 1.23 ± 0.07; femoral neck, 1.08 ± 0.17; Ward triangle, 1.04 ± 0.20; trochanter region, 0.96 ± 0.14; tibia region 1, 1.16 ± 0.16; tibia region 2, 1.52 ± 0.18; tibia region 3, 1.87 ± 0.18; tibia region 4, 1.57 ± 0.15; tibia region 5, 1.22 ± 0.15 | NR | Mean ± SD 4.0 ± 0.7 y | Total body, 1.24 ± 0.07; femoral neck, 1.04 ± 0.15; Ward triangle, 0.99 ± 0.18; trochanter, 0.95 ± 0.15; tibia region 1, 1.19 ± 0.16; tibia region 2, 1.55 ± 0.15; tibia region 3, 1.99 ± 0.16; tibia region 4, 1.59 ± 0.13; tibia region 5, 1.23 ± 0.13 | Change per year: |
Reported above | ||
| Therbo et al 2003 28 | Intervention (n = 12): patients with Achilles rupture undergoing surgical repair and immobilization | 7 d after surgery | BMC for proximal tibia, BMD for femoral neck and greater trochanter. Values are mean (range). |
NR | 6 wk, 3 mo, 6 mo, 1 y | Injured leg: |
Mean decrease in bone mineral 1 y after the injury was 6.4%, 2.5%, and 6.8% for measurements in, respectively, the proximal tibia, femoral neck, and greater trochanter. | NR | One year after an Achilles tendon rupture, patients showed a significant decrease in BMC of the proximal tibia and a decrease BMD at the femoral neck and greater trochanter. These findings suggest that such patients may be at an increased risk for osteoporotic fractures. |
ACL, anterior cruciate ligament; BMC, bone mineral content; BMD, bone mineral density; CL, contralateral lumbar; DEXA, dual-energy x-ray absorptiometry; IL, ipsilateral lumbar; LM, lean mass; LSF, lumbar stress fracture; MTSS, medial tibial stress syndrome; NR, not reported; ROI, region of interest.
Final revision submitted June 23, 2025; accepted July 15, 2025.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
