Abstract
Background:
Elevated body mass index (BMI) is a primary factor contributing to osteoarthritis of the knee, but data are lacking on the relationship between BMI and cartilage properties shortly after a traumatic injury.
Purpose/Hypothesis:
The purpose of the study was to relate BMI to patellofemoral cartilage properties after a patellar dislocation, using magnetic resonance imaging–based T1ρ relaxation times to assess cartilage. It was hypothesized that high BMI is correlated with long patellofemoral cartilage T1ρ relaxation times, indicating cartilage degradation, in both control knees and knees after patellar dislocation.
Study Design:
Cross-sectional study; Level of evidence, 3.
Methods:
T1ρ relaxation time mapping was performed for underweight to obese (BMI < 40 kg/m2) patients after a first-time patellar dislocation (n = 26; 19.8 ± 6.7 years old; 46 ± 36 days since dislocation), after multiple dislocations (n = 15; 18.4 ± 5.1 years old; 63 ± 33 days since most recent dislocation), and for healthy controls (n = 20; 20.1 ± 6.6 years old). For medial, lateral, and central regions of patellofemoral cartilage, mean T1ρ relaxation times were correlated against BMI. Statistical significance was set at P < .05.
Results:
For the control group, long T1ρ relaxation times were significantly correlated with high BMI for every region of patellofemoral cartilage except the lateral patella (P < .025; r2 = 0.26-0.63). On the contrary, for the first-time and multiple dislocations groups, long T1ρ relaxation times were significantly correlated with low BMI (P < .05; r2 = 0.24-0.50) for the medial patella (multiple dislocations only), central patella, lateral patella, medial trochlear groove (first-time dislocation only), central trochlear groove (first-time dislocation only), and lateral trochlear groove (first-time dislocation only).
Conclusion:
Long patellofemoral cartilage T1ρ relaxation times were related to high BMI for healthy knees, but unexpectedly related to low BMI after patellar dislocation. The results do not indicate a heightened risk of progressive cartilage degradation to osteoarthritis for overweight and obese patients immediately after patellar dislocation. For patients at a healthy weight and underweight, the traumatic injury is associated with early cartilage degradation, which could potentially initiate progressive degradation to posttraumatic osteoarthritis.
A lateral patellar dislocation is a traumatic injury associated with patellofemoral cartilage lesions and an elevated risk of patellofemoral osteoarthritis (OA).30,35 The injury is most common for young, active patients. The rate of dislocation peaks at approximately 150 per 100,000 person-years from 14 to 18 years of age, dropping to 50 and 20 per 100,000 person-years for 19 to 24 and 26 to 35 years, respectively. 34 Within 5 years of initial injury, >35% of patients dislocate the injured patella again,2,33,41 and approximately 5% dislocate the contralateral patella. 33 Cartilage lesions are identified on the medial or central patella for a majority of patients after a first-time dislocation.30,32,38 By 25 years after a patellar dislocation, approximately one-half of patients develop symptomatic patellofemoral OA. 35
High body mass index (BMI) is a primary factor contributing to OA of the knee. 5 High BMI has been related to heightened risk of progressive cartilage degradation to OA after traumatic knee injuries other than patellar dislocation.6,29,17 Quantitative magnetic resonance imaging (MRI) has also related elevated BMI with patellofemoral and tibiofemoral cartilage degradation for knees that have not experienced an injury and knees being treated for pain.7,8,19,22,36,37,40
Quantitative MRI–based relaxation time mapping allows evaluation of cartilage at stages along progressive degradation to OA, with long T1ρ and T2 relaxation times for cartilage indicating degeneration of the cartilage matrix.3,24,27,31 Quantitative MRI studies have identified cartilage matrix degeneration in the initial months after a patellar dislocation, particularly for adolescents.8,11,39 After a first dislocation, quantitative MRI has related patellar malalignment based on height and lateral shift to patellofemoral cartilage degradation. 11 Data relating BMI and other characteristics of patients to cartilage degradation after patellar dislocation are lacking. A better understanding of the relationship between patient characteristics and cartilage degradation is needed to determine mechanisms causing cartilage degradation after patellar dislocations and identify patients at highest risk of progressive cartilage degradation to OA.
The current study was performed to identify characteristics of patients associated with early cartilage degradation after a patellar dislocation, including BMI. The study utilized quantitative MRI to evaluate cartilage properties and included correlations between T1ρ relaxation times and characteristics of the patients for both injured and uninjured knees. The hypothesis of the study is that high BMI is correlated with long patellofemoral cartilage T1ρ relaxation times for both injured and uninjured knees.
Methods
Participants
All patients were prospectively recruited from November 2018 to January 2023. Data collected from the injured patients and controls included age, sex, BMI, and time from most recent dislocation. Patients enrolled after a patellar dislocation were divided into 2 groups: a first-time patellar dislocation group and a multiple dislocations group. The distinction was made due to the energy of dislocation and trauma of the injury decreasing from a first to recurrent dislocations 28 and the higher risk of patellofemoral OA for multiple dislocations than for a single dislocation. 35 Healthy control participants were recruited from the general population to represent the distribution of age, BMI, sex, and activity level of the dislocation patients. The study was approved by the institutional review board of Cleveland Clinic. All patients (plus a guardian for minors) signed a consent or assent form before participating.
Patients were recruited for a primary analysis comparing T1ρ relaxation times between injured knees and healthy controls,8,11 with characteristics correlated with T1ρ relaxation times added as a supplemental analysis. Patients were recruited for evaluation of injured knees ≤6 months after a first-time or recurrent traumatic lateral dislocation of the patella from the trochlear groove. Six months was selected to approximate the length of time cartilage partially recovers from the traumatic impact of a patellar dislocation. 8 A bone bruise on the medial patella or lateral femoral condyle on MRI confirmed all dislocations.
Several exclusion criteria were set to compare cartilage properties between groups. Patients were excluded for previous surgery of either knee or surgery scheduled before the injured knee could be evaluated. Patients were excluded for patellar dislocation of the contralateral knee, injuries to soft tissues other than the medial retinacular structures of either knee, previous traumatic injury of either knee other than patellar dislocation, autoinflammatory disease, and fractures of the femur, patella, or tibia other than small loose bodies. Patients were also excluded for existing OA or moderate to large chondral or osteochondral defects identified on previous MRI scans that would hinder characterization of T1ρ relaxation times throughout patellofemoral cartilage. Patients were not included in the analysis if they were considered severely obese (BMI ≥ 40 kg/m2) or otherwise had a knee considered too large to fit comfortably in the knee coil. 4 The original age range was ≥13 years. After October 1, 2021, enrollment prioritized adolescents (13-19 years) for the next phase of the study due to more consistent differences in T1ρ relaxation times between injured knees and healthy controls for adolescents than patients of all ages. 8 Exceptions for bilateral dislocations and contralateral surgery were made when patients who were already evaluated for the study experienced a contralateral first-time dislocation or second ipsilateral dislocation. To be included, these second evaluations had to occur ≤6 months after the most recent dislocation, with ≥6 months of recovery since a contralateral dislocation or surgery. The second evaluations were part of a separate aim related to longitudinal analysis of cartilage with additional dislocations and included in the present analysis to benefit the sample size. The control group included patients with no history of previous surgery, injury, or OA for either knee, targeting a distribution of age, BMI, sex, and activity level similar to the patellar dislocation population.
Activity Level
Patients completed online questionnaires corresponding to each MRI evaluation. Responses were stored in REDCap (Vanderbilt University). Activity level was characterized based on the Hospital for Special Surgery Pediatric Functional Activity Brief Scale (HSS Pedi-FABS). 10 The scale measures activity level in terms of frequency of activities such as running and cutting and level of athletic competition. The scale ranges from 0 to 30, with 30 representing the most active.
Magnetic Resonance Imaging
Injured and control knees were scanned on a 3T MRI scanner (Prisma; Siemens) using a 1Tx/15Rx knee coil (Quality Electodynamics). The imaging sequence included a 3D fat-saturated dual-echo steady-state (DESS) scan to segment and reconstruct cartilage surfaces (Table 1). A fat-saturated T1ρ magnetization-prepared angle-modulated partitioned k-space spoiled gradient-echo snapshots (MAPSS) imaging sequence quantified relaxation times for patellofemoral cartilage, with long relaxation times indicating cartilage degradation.21,23
3D MRI Acquisition Parameters a

3D, 3-dimensional; DESS, dual-echo steady-state; MAPSS, magnetization-prepared angle-modulated partitioned k-space spoiled gradient-echo snapshot; MRI, magnetic resonance imaging; VFA, variable flip angle (range, 4°-70°).
Time of spin-lock for T1ρ, 0, 10, 30, and 70 ms; spin-lock frequency, 500 Hz.
Cartilage Mapping
Cartilage was evaluated for medial, lateral, and central regions of the patella and trochlear groove (Figure 1). Cartilage was segmented from the DESS scans with a previously trained deep learning model that combines conditional generative adversarial networks and convolutional neural networks to identify borders at the articular surface and underlying bone. 13 Automated segmentation was corrected manually as needed. The Blumensaat line along the roof of the intercondylar notch on a sagittal view was projected through the femur to separate the trochlear groove from the femoral condyles. On axial slices through the length of the patella, an automated closed-contour mapping algorithm identified the most medial and lateral points on the patella and the most prominent point on the patellar ridge. Points were identified at one-third of the distance from the central ridge to the medial and lateral edges of the patella to divide patellar cartilage into medial, central, and lateral regions.8,11 Points were similarly identified at the medial and lateral edges of cartilage in the trochlear groove and the deepest point of the trochlear groove to set medial, central, and lateral regions. Points selected automatically were examined and corrected as necessary.
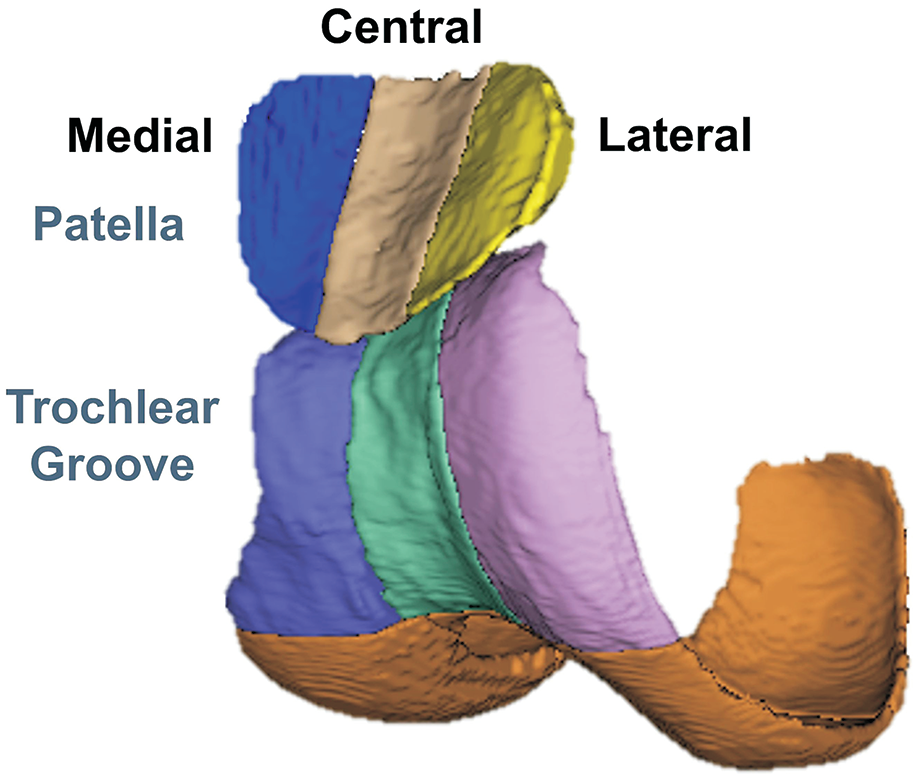
Three-dimensional reconstruction of the medial, central, and lateral regions of cartilage on the patella and within the trochlear groove for a knee model.
To map T1ρ relaxation times to cartilage,8,11 the first echo of the T1ρ images was rigidly registered with images from the DESS scan, using piecewise rigid registration to adjust for variations in the positions of the bones. A template mask was generated for the knee, with an intensity-based, multiresolution pyramidal approach used for nonrigid registration between the first echo and the mask. The resulting transformation was used for rigid registration of the later echoes of the T1ρ images to the first echo. A 2-parameter, monoexponential Levenberg-Marquardt algorithm related image signal to exponential decay based on time of spin-lock to T1ρ relaxation time. T1ρ relaxation times were mapped to each voxel (Figure 2). Voxels with extreme values (>200 ms) were discarded to avoid partial volume averaging effects. Mean T1ρ relaxation time was quantified for each region of cartilage. For this technique, the scan-rescan root mean square coefficient of variation for T1ρ values has been reported as <5%.21,25
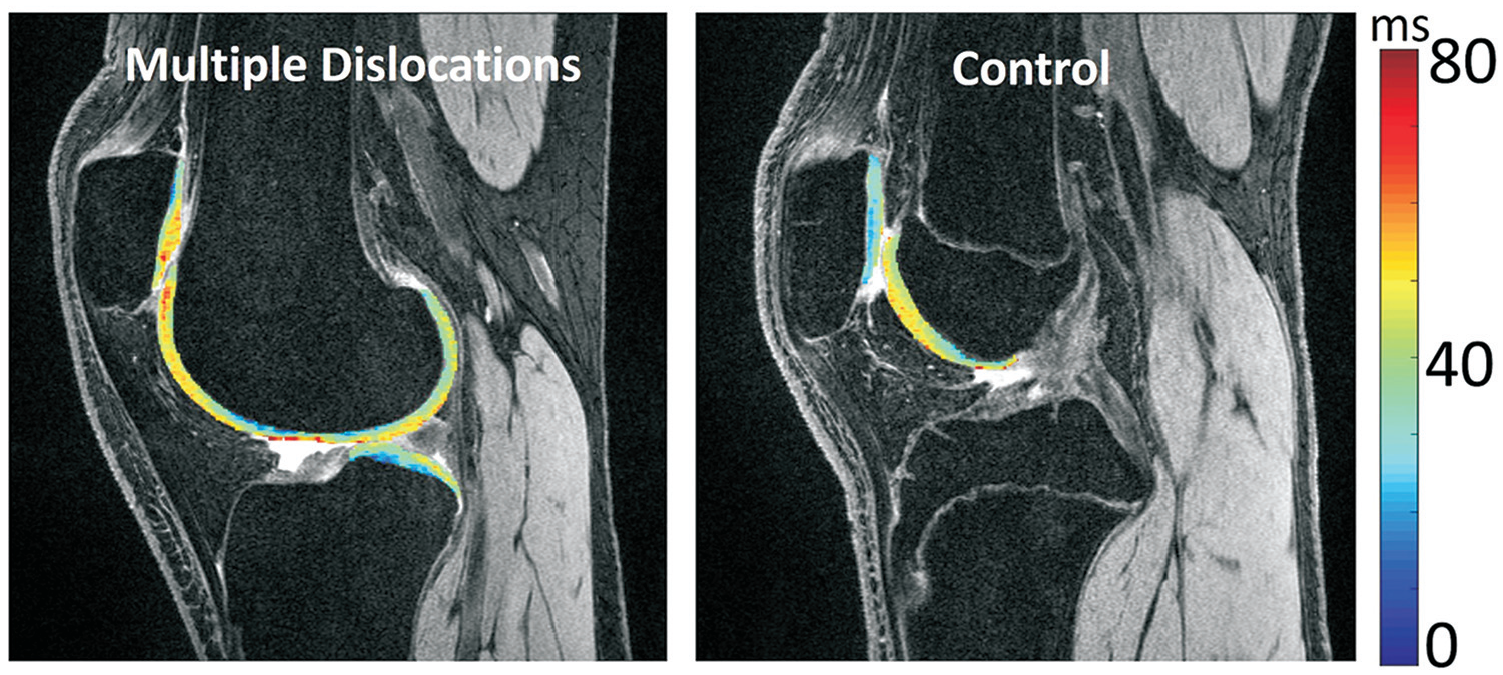
T1ρ relaxation times mapped to patellofemoral cartilage dual-echo steady-state scans for patients with a low body mass index (BMI) from the multiple dislocations group (BMI, 18.6 kg/m2) and the control group (BMI, 19.7 kg/m2).
Statistical Analysis
Characteristics of the population were compared between the groups. The level of significance was P < .05 for each analysis. Age, body mass index, and activity level were compared between the control, first-time dislocation, and multiple dislocations groups, and time from most recent dislocation was compared between the first-time and the multiple dislocations groups. To determine if the assumptions for parametric analyses were met, normality of the dependent variables was evaluated with a Shapiro-Wilk test and homogeneity of variances was evaluated with a Levene test (SPSS Version 24; IBM Corp). Analysis of variance was used for parametric analysis (t test for time from dislocation), with Kruskal-Wallis tests used for nonparametric analyses (Mann-Whitney U test for time from dislocation). Distribution of sex was compared between the 3 groups with a chi-square test after checking the expected frequency for all cells.
The primary analysis correlated patellofemoral cartilage T1ρ relaxation times against BMI, age, time from latest injury, and activity level for each of the 3 groups. Each region of cartilage was evaluated for relationships between T1ρ relaxation times and characteristics of the patients. The assumptions for linear regressions were checked with a Shapiro-Wilk test for normality of unstandardized residuals and a White test for heteroscedasticity of the variance of the residuals. When multiple patient characteristics were significantly correlated with T1ρ relaxation times, the relative contribution of each independent variable was assessed with multivariable linear regressions, and variance influence factors were quantified to rule out multicollinearity. Spearman rank-order correlations were performed to address nonnormal distributions of the residuals, and weighted least squares regressions were performed to address heteroscedasticity. Coefficients of determination (r2) were quantified for each regression, with standardized beta (β) coefficients quantified as measures of the strength and direction of each correlation. The study population was planned to achieve a power of 0.8 for a sample size of 14 with a coefficient of determination of 0.4 or for a sample size of 21 with a coefficient of determination of 0.3. For each group, T1ρ relaxation times were compared between male and female participants with t tests or Mann-Whitney U tests, as appropriate.
T1ρ relaxation times were also compared between the control, first-time dislocation, and multiple dislocations groups for each region of patellofemoral cartilage. If the assumptions for parametric analyses were met, relaxation times were compared between groups with analysis of variance and post hoc Student-Newman-Keuls tests. Kruskal-Wallis and post hoc Dunn-Bonferroni tests were used for nonparametric analyses.
Results
The study population consisted of 26 and 15 patients in the first-time and multiple dislocations groups, respectively, and 20 healthy controls. The patient population was drawn from 434 patients evaluated ≤6 months after a patellar dislocation, with 274 excluded based on ≥1 exclusion criterion (Figure 3). The first-time dislocation group included 3 patients previously evaluated for the contralateral knee. For 2 patients, the originally injured (contralateral) knee was treated nonoperatively, with 7 and 8 months passing between the most recent dislocation of the contralateral knee and the knee that experienced a recent dislocation. For the third, the contralateral knee was treated with MPFL reconstruction approximately 12 months before evaluation of the knee that experienced a recent dislocation. The multiple dislocations group included 2 patients evaluated for a second dislocation who were previously evaluated for a first dislocation. For one of the 2, approximately 9 months passed between the first dislocation and evaluation for the second dislocation. The other also had a contralateral growth plate fusion between the first and second dislocations, approximately 8 months before evaluation for the second dislocation. Of the 26, 15, and 20 participants in the first-time dislocation, multiple dislocations, and control groups, respectively, activity level surveys were completed by 24, 14, and 18 participants.

Flowchart showing enrollment of patellar dislocation patients.
Patient characteristics were similar between the 3 groups. No significant differences in age, BMI, activity level, or distribution of sex were identified between the groups (P > .25) (Table 2). The time since most recent dislocation tended to be larger for multiple dislocations than first-time dislocations, but the difference was not statistically significant (P = .056).
Demographic Characteristics for the 3 Groups a

Values are presented as mean ± SD or n.
Mann-Whitney U test.
Kruskall-Wallis test.
Obesity Categories From US Center for Disease Control and Prevention.
Long cartilage T1ρ relaxation times were associated with a high BMI for healthy knees (Figure 4). For the control group, BMI was the only parameter significantly correlated with T1ρ relaxation time for all regions of patellofemoral cartilage except the lateral patella (P < .001–P = .02) (Table 3). At the lateral patella, only decreasing activity level from the HSS Pedi-FABS was significantly correlated with increasing T1ρ relaxation time (P = .03).

Variation in T1ρ relaxation time with body mass index (BMI) for the 3 groups for cartilage on the central patella. The regressions were statistically significant for each group.
Significant Correlations Relating T1ρ Relaxation Times to Patient Characteristics a
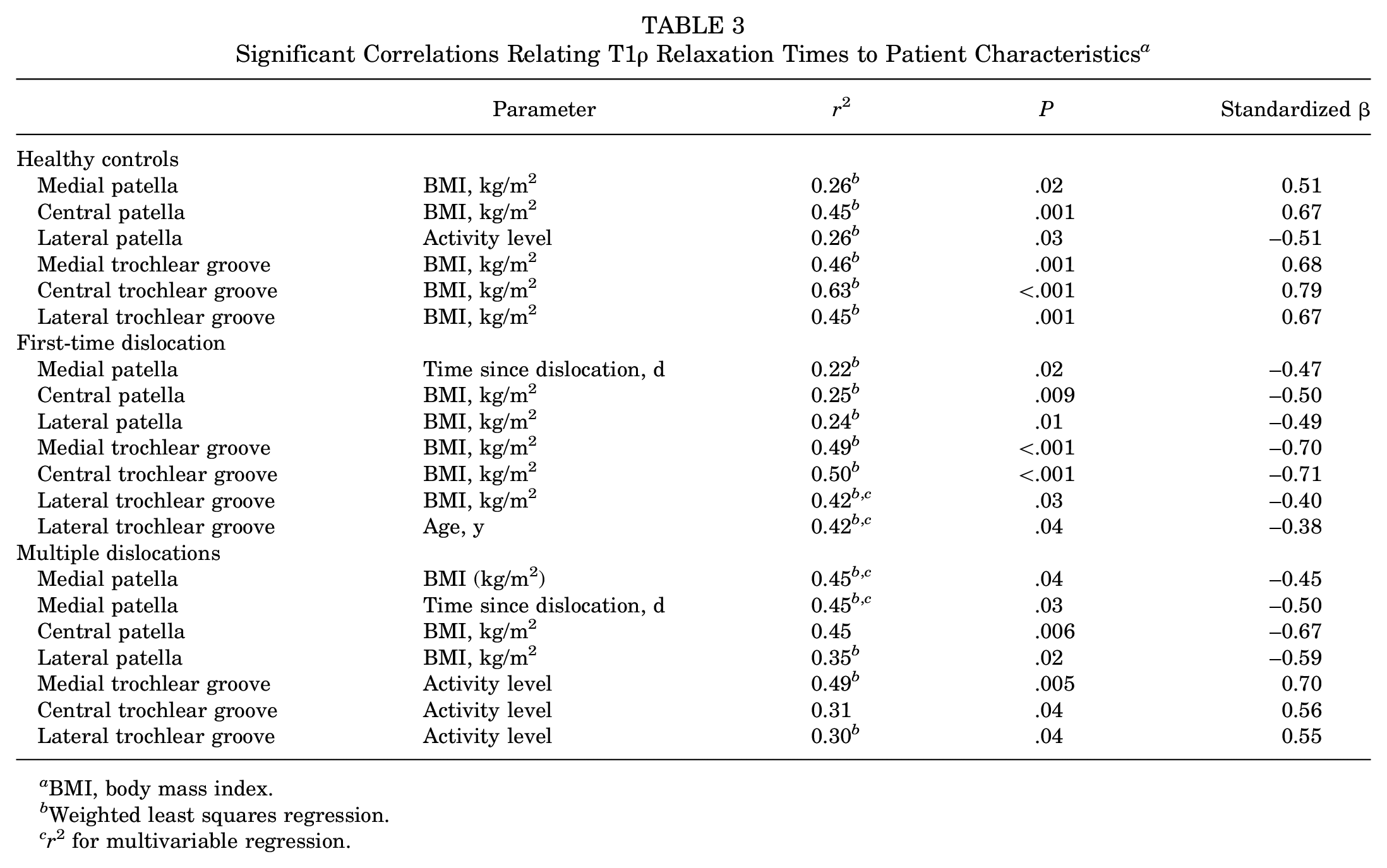
BMI, body mass index.
Weighted least squares regression.
r 2 for multivariable regression.
On the contrary, long cartilage T1ρ relaxation times were associated with a low BMI for the first-time dislocation group. For first-time dislocations, increasing cartilage T1ρ relaxation times were significantly correlated with only decreasing BMI for the central and lateral patella and medial and central trochlear groove (P < .001–P = .01). At the lateral trochlear groove, decreasing BMI (P = .03) and age (P = .04) were both significantly correlated with increasing T1ρ relaxation times. At the medial patella, decreasing time from injury was significantly correlated with increasing T1ρ relaxation times (P = .02) (Table 3).
In contrast to the control group, for the multiple dislocations group, long cartilage T1ρ relaxation times were associated with a low BMI and high activity level. For the multiple dislocations group, decreasing BMI was the only parameter significantly correlated with increasing cartilage T1ρ relaxation times for the central (P = .006) and lateral (P = .02) patella. Decreasing BMI (P = .04) and time since dislocation (P = .03) were both significantly correlated with increasing T1ρ relaxation times at the medial patella. Increasing activity level was the only parameter significantly correlated with increasing T1ρ relaxation times for the 3 regions in the trochlear groove (P = .005–P = .04) (Table 3).
Relationships between sex and T1ρ relaxation times varied between the groups. T1ρ relaxation times were significantly longer for female than male patients for the first-time dislocation group at the central and lateral patella (P < .05) (Table 4). T1ρ relaxation times were significantly longer for male than female patients for the multiple dislocations group at the central and lateral trochlear groove (P≤ .03).
T1ρ Relaxation Times for Male vs Female Patients a
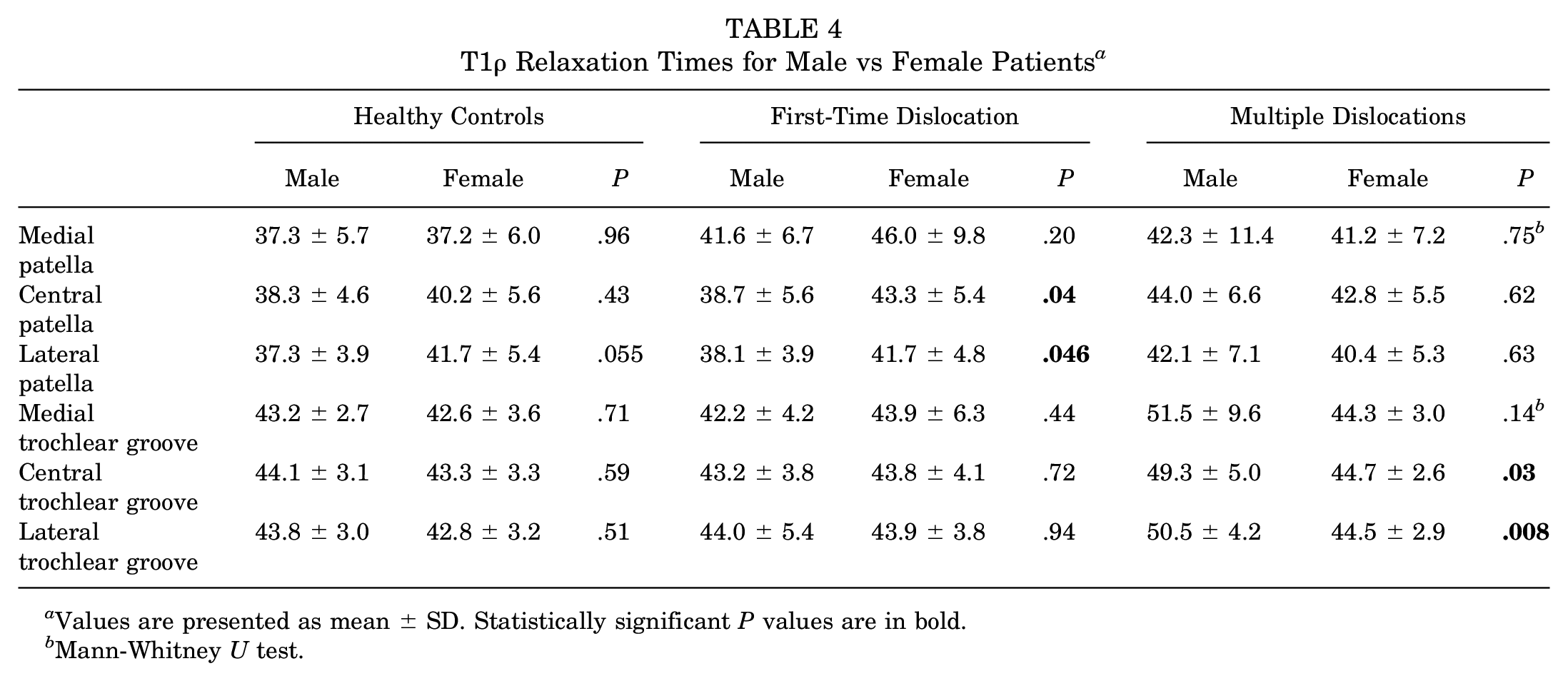
Values are presented as mean ± SD. Statistically significant P values are in bold.
Mann-Whitney U test.
The only significant variation in T1ρ relaxation times between groups occurred for cartilage on the medial patella. At the medial patella, T1ρ relaxation times were significantly longer for the first-time dislocation group than the control group (P = 0.020, Table 5). No other significant differences were identified for the medial patella or any other region of cartilage (P > 0.085).
Comparison of T1ρ Relaxation Times Between the 3 Groups a
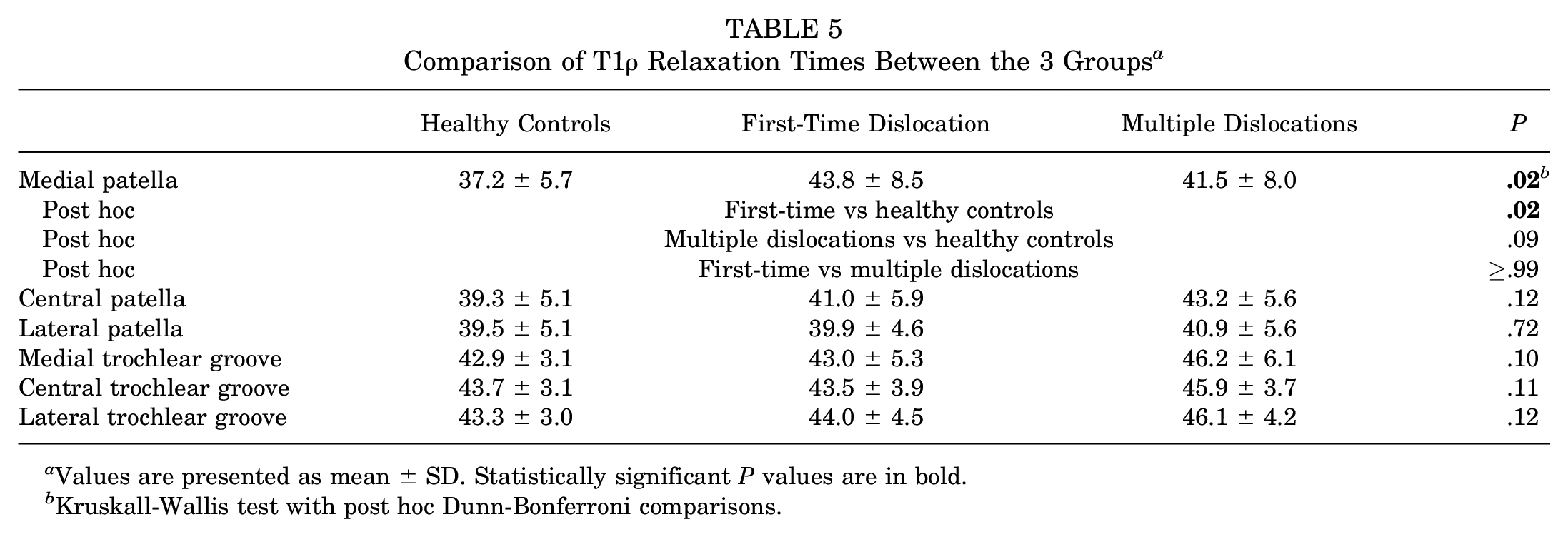
Values are presented as mean ± SD. Statistically significant P values are in bold.
Kruskall-Wallis test with post hoc Dunn-Bonferroni comparisons.
Discussion
Significant correlations related long patellofemoral cartilage T1ρ relaxation times in the early months after a patellar dislocation to low BMI for 5 of the 6 patellofemoral cartilage regions for the first-time dislocation group and the 3 regions on the patella for the multiple dislocations group. In contrast, significant correlations related long T1ρ relaxation times to high BMI for 5 patellofemoral cartilage regions for the healthy control group. Despite being in opposite directions, relationships between BMI and T1ρ relaxation times were well-established for the controls and dislocation patients, with P values <.01 for most of the significant correlations and several with P values ≤.001. Regression lines for the opposing trends crossed at a BMI of approximately 25 kg/m2, which is the upper limit for a healthy weight. The results indicate that early degradation of the cartilage matrix after patellar dislocation is primarily experienced by patients considered to be at a healthy weight or underweight, with the largest difference between injured and healthy knees at low values of BMI (Figures 2 and 4).
The data for the patellar dislocation groups opposed the hypothesis that high BMI is correlated with long patellofemoral cartilage T1ρ relaxation times. Previous studies related high BMI to long patellofemoral cartilage T1ρ and T2 relaxation times for healthy knees,8,36 similar to the current study. A BMI considered overweight has also been related to progressive cartilage degradation to OA after traumatic knee injuries other than patellar dislocation.6,29,17 One previous study differed from the prevailing literature, showing a quadratic relationship between BMI and knee cartilage characteristics ≤3 years after ACL reconstruction, with both high and low BMI values related to cartilage degradation. 1 The previous studies did not focus on the period immediately after injury. The current study focused on the early months after patellar dislocation to improve understanding of the baseline condition that could initiate progressive cartilage degradation to OA. One study, with a population overlapping the present study, did not identify a significant relationship between T1ρ relaxation times and BMI after a patellar dislocation. 8 Unlike the current analysis, the previous study included severely obese patients (BMI ≥ 40 kg/m2). Severe obesity compromises fit of the knee within the knee coil and the resulting MRI data 4 and creates potential for a quadratic relationship between BMI and properties of knee cartilage, 1 all of which adversely influence linear regressions.
Activity level and age were also associated with cartilage T1ρ relaxation times. Similar to BMI, the relationship between activity level and T1ρ relaxation times reversed from healthy to injured knees. Although activity level can be related to BMI, multicollinearity of the 2 variables was not identified. For the control group, low activity was significantly correlated with high relaxation times only at the lateral patella (standardized β = −0.51), with the correlation generally weaker than identified for BMI for the other regions (standardized β = 0.51 to 0.79). After multiple dislocations, high activity was significantly correlated with long relaxation times for the trochlear groove regions (standardized β = 0.55 to 0.7). Strength of the correlations was similar to BMI for regions on the patella for this group (standardized β = −0.45 to −0.67). Significant relationships with activity level were not identified after a first-time dislocation. Previous studies have related both high and low activity levels to longer knee cartilage T1ρ and T2 relaxation times than moderate activity for healthy knees and knees at risk of OA due to pain or an injury.12,16,18,26 For the multiple dislocations group, functional loading of injured cartilage after the first dislocation may have created a relationship with activity level not identified in the early months after a single dislocation. The only significant correlation between age and T1ρ relaxation times occurred at the lateral trochlear groove for the first-time dislocation group, with a relatively low strength (standardized β = −0.38) compared with other significant regressions. Young age is generally associated with a low BMI, although collinearity was not identified between age and BMI for this correlation.
Other characteristics related to cartilage degradation were time from injury and sex. T1ρ relaxation times were longest a short time after dislocation at the medial patella, demonstrated by significant correlations for both dislocation groups. A previous study with an overlapping population similarly showed T1ρ relaxation times for cartilage on the medial patella decrease over time after a first dislocation. 8 The medial patella is the site of traumatic impact against the lateral femoral condyle during a dislocation episode. Decreasing cartilage T1ρ relaxation times over time likely represents partial recovery from traumatic injury. 8 T1ρ relaxation times were longer for female than male patients for patellar cartilage for the first-time dislocation group but longer for male than female patients for trochlear groove cartilage for the multiple dislocations group. Varying influence of sex on T1ρ and T2 relaxation times has been described previously.8,20 For the previous first-time patellar dislocation study overlapping with the current study, T1ρ relaxation times were longer for female than male patients at the lateral patella. 8 Another study of patients treated for knee pain or injury, not including patellar dislocation, showed longer patellar cartilage T2 relaxation times for male patients. 20 The relationship between sex and cartilage relaxation times may vary with type of injury and region of cartilage.
The only difference between groups for T1ρ relaxation times was longer relaxation times for the first-time group than the control group at the medial patella. The previous study with an overlapping population also isolated the medial patella as the site of elevated T1ρ relaxation times after a first-time dislocation, reflecting the traumatic impact at this region. 8 Energy of injury and traumatic impact to cartilage tend to decrease from the first to subsequent dislocations due to injury to the medial retinacular structures, 28 which likely contributes to no significant differences in relaxation times between the control and multiple dislocations groups. Other studies have shown more widespread elevated T1ρ and T2 relaxation times throughout the patellofemoral joint for a population only including adolescents who experienced a dislocation.11,39 Based on the current results, relatively low BMI values contribute to consistent differences in T1ρ and T2 relaxation times between patellar dislocation and control groups for adolescents.
The mechanism relating low BMI to long T1ρ relaxation times after patellar dislocation and the implications for potential progressive cartilage degradation to OA are currently unknown. Decreasing volume of tissue surrounding the knee could potentially escalate the traumatic impact and inflammatory response of a patellar dislocation, both of which can contribute to early cartilage degradation.8,9 Regardless of the mechanism, the results do not indicate a heightened risk of progressive cartilage degradation to OA for overweight and obese patients immediately after patellar dislocation. Being overweight or obese could still contribute to longer term cartilage degradation after a dislocation. The traumatic injury is associated with early cartilage degradation for patients at a healthy weight or underweight. Early cartilage degradation could potentially initiate progressive degradation to posttraumatic OA in some cases. Further studies are warranted to identify characteristics of patients that initiate progression to patellofemoral posttraumatic OA after patellar dislocation and develop strategies to preserve cartilage.
Limitations
The primary limitation in this study is the secondary analysis from a population recruited to compare cartilage relaxation times between groups. Inclusion and exclusion criteria were developed to isolate the influence of patellar dislocation on cartilage properties to the extent possible, but some exceptions were allowed. To support ongoing longitudinal analyses, previous contralateral dislocations or surgeries that would have been exclusion criteria for initial enrollment were allowed for previously enrolled patients who experienced a second dislocation or first-time contralateral dislocation. To maximize the sample size, these patients were kept in the current analysis if the time since previous injury or surgery exceeded 6 months. The current study also includes patients from an initial evaluation of cartilage T1ρ relaxation times with no maximum age limit 8 and a follow-up with a maximum age of 19 years 11 for the benefit of the sample size, which contributed to heteroscedasticity addressed with weighted least squares regressions.
Other limitations are related to the data collected and assessment at a single time point. BMI does not account for proportion or distribution of fat or proportion of muscle mass. Increased recognition of the potential influence of body mass on cartilage properties after injury could prompt collection of more accurate measures, such as waist:hip ratio or body fat percentage.14,15 The HSS Pedi-FABS survey could underrepresent activity levels for nonathletes and athletes in sports with limited running and change of direction. 10 The study focused on characteristics of the whole patient, without including knee anatomy or alignment. A previous study 11 including an overlapping population examined measures of anatomy and alignment after a first dislocation and multiple dislocations. The only significant correlations related long T1ρ relaxation times to patellar height (central and lateral trochlear groove) and lateral shift (medial patella) after a first-time dislocation. Additional studies with a larger population are warranted to evaluate interactions between anatomy, alignment, and characteristics of the whole patient. The current data provide a baseline assessment of the relationship between postinjury cartilage degradation and BMI. Longitudinal evaluation is needed to identify factors influencing progressive cartilage degradation to posttraumatic OA and to identify patients at greatest risk. Cartilage was evaluated based only on T1ρ relaxation times related to proteoglycan concentration. Quantifying T2 relaxation times could also assess the cartilage matrix based on water and collagen content, along with organization of collagen fibrils.
Conclusion
Despite the relationship between long patellofemoral cartilage T1ρ relaxation times and high BMI for healthy knees, long T1ρ relaxation times were unexpectedly related to low BMI in the early months after patellar dislocation. A heightened risk of progressive cartilage degradation to OA is not evident for overweight and obese patients immediately after patellar dislocation. The traumatic injury is associated with early cartilage degradation for patients at a healthy weight and underweight. Early cartilage degradation could potentially initiate progressive degradation to posttraumatic OA for some healthy weight and underweight patients.
Footnotes
Final revision submitted November 27, 2024; accepted January 3, 2025.
Presented as a poster at the annual meeting of the AOSSM, Washington DC, July 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: Funding was provided by the Office of the Assistant Secretary of Defense for Health Affairs through the Peer Reviewed Medical Research Program Discovery Award under award No. W81XWH-20-1-0040 and a research grant from the Arthroscopy Association of North America. L.D.F. has received consulting fees from DePuy Synthes and hospitality payments from DJO. C.S.W. has received consulting fees from Siemens Medical Solutions. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Cleveland Clinic (No. 18-1388).
In house AI programs were used for mapping T1ρ relaxation times to cartilage. AI was used for identifying cartilage borders from MRI and divide the cartilage surfaces into sub-regions.
