Abstract
Background:
Conventional cortical suspensory fixation devices (CSFDs) are made of metal. Most adjustable-loop CSFDs originally intended for femoral-side fixation are also used on the tibial side during “all-inside” anterior cruciate ligament (ACL) reconstruction. The T-Button-A is a novel adjustable-loop polyether ether ketone (PEEK) CSFD intended specifically for tibial-side fixation during ACL reconstruction using the “full-tunnel” technique.
Purpose/Hypothesis:
The purpose of this study was to compare the biomechanical characteristics of the T-Button-A with those of 6 commercially available fixed- or adjustable-loop CSFDs. It was hypothesized that the T-Button-A would be comparable with conventional metallic CSFDs intended for femoral-side fixation in terms of its biomechanical properties.
Study Design:
Controlled laboratory study
Methods:
Ten implants each, of T-Button-A, Tightrope RT, Proloop Ultra, Procinch RT, Infinity, Infiloop, and Endobutton CL Ultra were biomechanically tested using an axial and torsion testing machine, and an incremental load-based testing protocol comprising 3500 cycles. Elongation and load to failure on cyclic and pull-to-failure loading, respectively, were recorded for each implant and compared across all devices. The consistency of implants in completing the 3500 test cycles, and demonstrating cyclic elongation <3 mm as well as load to failure >650 N, was compared across the tested devices.
Results:
The T-Button-A demonstrated a mean elongation of 2.09 ± 0.43 mm, which was less than that of Infinity (P = .007), but comparable with the rest of the devices. Its mean load to failure was 1370.02 N ± 110.11 N, which was greater than that of Tightrope RT (P = .02), Infinity (P < .001), and Endobutton CL Ultra (P = .03), less than that of Procinch RT (P = .002), and comparable with that of Proloop Ultra and Infiloop. All 10 T-Button-A, Tightrope RT, Proloop Ultra, Infiloop, and Endobutton CL Ultra consistently completed the testing protocol and delivered elongation and load-to-failure values <3 mm and >650 N, respectively.
Conclusion:
The biomechanical performance of the T-Button-A is comparable with most fixed- and adjustable-loop metallic CSFDs and is beyond what is considered acceptable to support ACL reconstruction grafts during early postoperative rehabilitation.
Clinical Relevance:
The PEEK T-Button-A is a viable alternative to conventional metallic CSFDs, for tibial-side fixation during ACL reconstruction.
Anterior cruciate ligament (ACL) reconstruction is among the most commonly performed orthopaedic surgeries.26,31 Despite advances in ACL reconstruction surgery, tibial-side fixation remains the weakest link due to the near-parallel force vector acting on relatively lower-density metaphyseal bone.19,22,26 Cortical suspensory fixation on the tibial side, when compared with aperture fixation using interference screws, results in lower revision rates after ACL reconstruction using hamstring grafts. 28 Among cortical suspensory fixation devices (CSFDs), fixed-loop devices are considered the benchmark due to their low cyclic displacement, 27 but they cannot be used on the tibial side. Most CSFDs originally intended for use on the femoral side are also being used on the tibial side while performing “all-inside” ACL reconstruction. 28
Conventional CSFDs are metallic, made of stainless steel or titanium. Orthopaedic implants made of polyether ether ketone (PEEK) are increasingly being used because they offer several advantages over their metallic counterparts. 20 PEEK offers an elastic modulus close to that of cortical bone, thereby reducing the risk of implant loosening through osteolysis of the surrounding bone,17,20,25 while retaining high mechanical strength and rigidity. 29 It is corrosion-resistant,18,23 nontoxic,16,37 and radiolucent. 35 It is also compatible with magnetic resonance imaging and results in minimal distortion and artifacts. 20 Several biomechanical studies comparing the properties of metallic CSFDs meant for femoral-side fixation can be found in the literature.3,5,11,13 -15,27,33 To the authors’ knowledge, however, adjustable-loop PEEK CSFDs intended specifically for tibial-side fixation during ACL reconstruction have not been biomechanically tested or compared with metallic CSFDs in the past.
The T-Button-A (Sironix Arthroscopy Solutions; Healthium Medtech Limited) is a novel adjustable-loop PEEK CSFD intended for tibial-side fixation during ACL reconstruction using the “full-tunnel” technique. 8 We hypothesized that the T-Button-A would be comparable with conventional metallic CSFDs in terms of its biomechanical properties. The aims of this study were to compare the biomechanical characteristics of the T-Button-A with 6 commercially available fixed- or adjustable-loop metallic CSFDs using the following parameters: (1) ability to complete 3500 cycles based on a predefined incremental load–based testing protocol, (2) elongation on cyclic loading, (3) ultimate load to failure, and (4) consistency in demonstrating an elongation <3 mm as well as a load to failure >650 N on cyclic and pull-to-failure loading, respectively.
Methods
CSFDs Tested
Following an institutional echics Committee waiver, the T-Button-A was compared with 6 different CSFDs: (1) Tightrope RT (Arthrex), (2) Proloop Ultra (Sironix Arthroscopy Solutions; Healthium Medtech Limited), (3) Procinch RT (Stryker Corp), (4) Infinity femoral adjustable button (Conmed Corp), (5) Infiloop with a 20-mm loop (Sironix Arthroscopy Solutions; Healthium Medtech Limited), and (6) Endobutton CL Ultra with a 20-mm loop (Smith & Nephew) (Figure 1 and Table 1). Ten implants of each device were biomechanically evaluated. The T-Button-A is available in a single universal size; it comprises a PEEK button made of a flat disc (that sits flush on the tibial metaphysis) with a thickness of 1.5 mm and a cylindrical stub (that is seated in the tunnel) with a diameter of 7.5 mm. It comes preassembled with braided, nonabsorbable, ultra–high molecular weight polyethylene adjustable loops (Figure 1A). Among the tested devices, Infiloop and Endobutton CL Ultra are fixed-loop devices, while T-Button-A, Tightrope RT, Proloop Ultra, Procinch RT, and Infinity femoral adjustable button are adjustable-loop devices.

Cortical suspension devices tested included (A) T-Button-A, (B) Tightrope RT, (C) Proloop Ultra, (D) Procinch RT, (E) Infinity, (F) Infiloop with 20-mm loop, and (G) Endobutton CL Ultra with 20-mm loop.
Specifications of the Tested Devices a

PEEK, polyether ether ketone; UHMWPE, ultra-high molecular weight polyethylene; USP, United States Pharmacopeia.
Biomechanical Testing
Biomechanical evaluation was conducted using a custom-made rig fixed to a 5-kN, 30-N·m axial and torsion testing machine (Instron; Illinois Tool Works India Pvt Ltd) (Figure 2A). A hook with a diameter of 8 mm was attached to the bottom of the mount. At the top of the mount, a steel plate of 3-mm thickness was attached to a 5000-N load cell (Figure 2B). To replicate bone tunnels in the in vivo setup, holes of 8 mm–diameter or 4.5 mm–diameter were drilled in 2 plates, corresponding to the manufacturers’ recommendations for the T-Button-A and the rest of the devices, respectively. A constant loop length of 20 mm was used for all adjustable-loop devices, to correspond to the fixed-loop devices with 20-mm loops. The free suture ends of adjustable-loop devices were secured using 8 half-hitch knots. The devices were then fed through the tunnel and secured against the surface of the plate, which now simulated cortical bone. The free ends of the loop were passed through a plastic cylinder to eliminate friction from the metal edges and then passed over the hook at the bottom of the mount.

(A) The biomechanical testing setup comprised a custom-made rig fixed to a 5-kN, 30-N·m axial and torsion testing machine. (B) A hook was attached to the bottom of the mount. At the top of the mount, a stainless steel plate of 3-mm thickness was attached to the 5-kN load cell.
All implants of the different device types were then assessed using a predefined testing protocol. The testing protocol comprised 4 stages: preloading, cyclic preconditioning, cyclic incremental loading, and pull to failure (Table 2). It started with the deployment of the device and preloading with 20 N at 2 N/s. This was followed by cyclic preconditioning at 10 N to 50 N, at a frequency of 1 Hz, for 10 cycles. At this point, the displacement of the construct was reset to zero. Cyclic loading comprised 500 cycles at loads of 10 N to 50 N, followed by incremental sinusoidal loading from 50 N to 650 N, using 50 N increments every 250 cycles, until the completion of 3500 cycles. The displacement at the end of 3500 cycles was documented. The devices were then tested for load to failure at a displacement-controlled rate of 50 mm/min. The ultimate load to failure was documented.
Summary of the Biomechanical Testing Protocol

NA, not applicable.
Statistical Analysis
The G*Power software (Version 3.1.9.7) 9 was used for calculations related to study power and sample size requirements. Due to the lack of published literature on the biomechanical characteristics of the PEEK T-button-A system of implants, a pilot study was conducted using 3 implants from each group of the studied devices to derive the data required for a priori power analysis. With significance set at .05, for a study power of 90%, it was determined that a ≥5 implants per device group would need to be tested to detect differences in elongation on cyclic loading and load to failure. A larger sample size of 10 implants from each group was chosen for the study (including the first 3 samples used for a priori power analysis) to further characterize group means, distinguish groups during post hoc analysis, and account for the possibility of implant failure. The observed effect sizes of load to failure and elongation on cyclic loading were used for a post hoc reanalysis, wherein the chosen sample size was found to be adequate.
The IBM SPSS software (Version 29.0) was used for data analysis. Means and standard deviations were calculated for numerical variables. Proportions were assessed for categorical variables. The Shapiro-Wilk test was used to assess normality of distribution for numerical variables. Levene test was used to assess homogeneity of variance. Means of numerical variables were compared using the analysis of variance (ANOVA) test, where >2 groups existed. Where ANOVA results demonstrated a statistically significant difference, a post hoc Games-Howell analysis was conducted to uncover significant differences between group means. Proportions of categorical variables were compared using the Pearson chi-square test.
Results
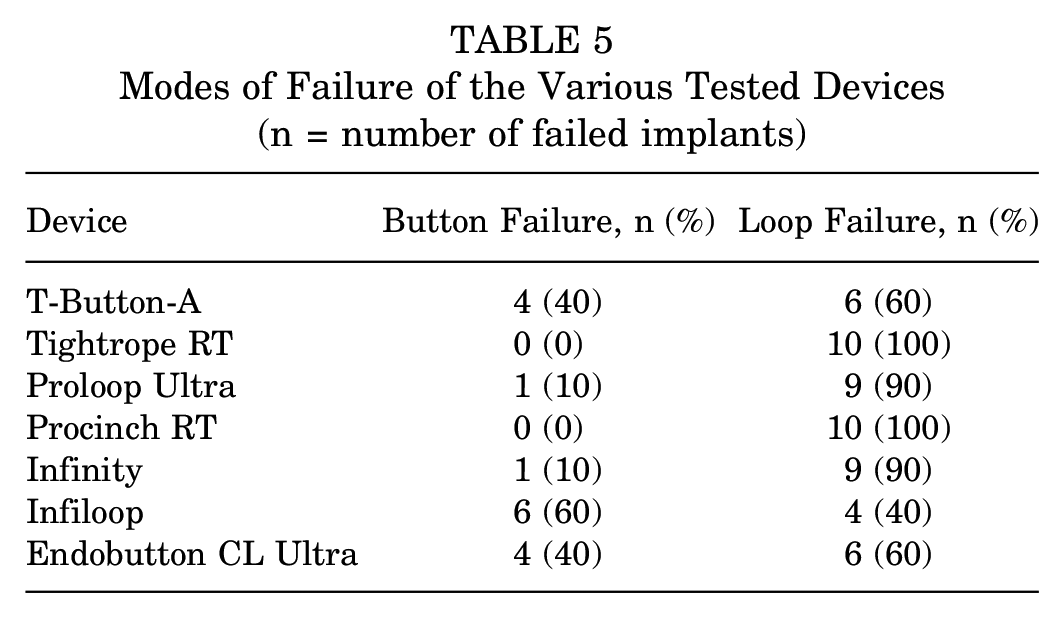
All 10 T-Button-A, Tightrope RT, Proloop Ultra, Procinch RT, Infiloop, and Endobutton CL Ultra implants, and 5 Infinity femoral adjustable buttons completed 3500 loading cycles as per the test protocol. The mean elongation on cyclic loading and load to failure of the tested devices, and interdevice post hoc comparisons are summarized in Table 3 and Table 4, respectively. All tested devices, except the Infinity femoral adjustable button, demonstrated a mean elongation <3 mm on cyclic loading and a mean load to failure >650 N (Table 3). Two modes of device failure were noted: button failure and loop failure (Figure 3). The proportions of implants from each device category that failed through either mechanism are summarized in Table 5. Table 6 summarizes the consistency of implants from the various device groups in completing 3500 cycles of the test protocol and demonstrating an elongation <3 mm as well as a load to failure >650 N, on cyclic and pull-to-failure loading, respectively.
A Summary of the Results of the Biomechanical Testing a

Post hoc interdevice comparisons are provided in Table 4. LTF, load to failure.
Values are in mm.
Values are in N.
P values are indicative of the result of the overall comparison of means using the analysis of variance test.
Post Hoc Interdevice Analyses of Elongation on Cyclic Loading and Ultimate Loads to Failure a

EB, Endobutton CL Ultra with 20-mm loop; IF, femoral adjustable button; IL, Infiloop with 20-mm loop; PC, Procinch RT; PL, Proloop Ultra; T-BA, T-Button-A; TR, Tightrope RT.
Indicates a significant difference at P < .05.

The 2 modes of device failure noted are (A) button failure and (B) loop failure.
Modes of Failure of the Various Tested Devices (n = number of failed implants)
Consistency of Implants From the Various Device Groups in Completing 3500 Test Cycles and Delivering an Elongation <3 mm as Well as a Load to Failure >650 N a (n = number of implants)

LTF, load to failure.
After completion of 3500 cycles of the testing protocol.
Criteria included completion of 3500 test cycles.
Discussion
The principal finding of this study was that all 10 T-Button-A implants completed 3500 cycles of the test protocol and demonstrated acceptable elongation (<3 mm) and loads to failure (>650 N) on cyclic and pull-to-failure loading, respectively, thereby establishing the adequacy of the biomechanical properties of the device. Hamstring tendon autografts are a popular choice for ACL reconstruction. 10 A graft diameter >8 mm is considered optimal to minimize the risk of early revision when hamstring grafts are used.21,34 Quadrupling the semitendinosus to achieve adequate graft diameter frequently results in a short graft, necessitating the use of CSFDs on the tibial as well as the femoral side. While metallic adjustable-loop CSFDs originally intended for femoral-side fixation are being used for tibial-side fixation using the all-inside surgical technique, the T-Button-A presents an alternative tibial fixation–specific option for ACL reconstruction using the full-tunnel technique. 8
All 10 T-Button-A, Tightrope RT, Proloop Ultra, Infiloop, and Endobutton CL Ultra implants, completed 3500 cycles of the test protocol (Table 3). Although multiple studies describe 1000 loading cycles,11,13 -15,27,33 some authors used test protocols with 4500 cycles, postulating that in vivo lengthening may continue under cyclical stress until graft-to-bone healing occurs and that the mechanical response of CSFDs to larger volumes of cycles would be a truer representation of the postoperative state.3,5 Moreover, some studies used loads up to 250 N,3,11,13,15,27,33 while others performed cyclical loading up to 400 N.5,14 Most rehabilitation protocols after ACL reconstruction permit weightbearing before 8 to 12 weeks, which is the expected time for autologous soft tissue graft integration.3,12,38 CSFDs are reported to require failure loads beyond 590 N during early postoperative rehabilitation, based on the peak forces experienced by the ACL during walking and isokinetic extension.14,27 With the benefit of insights from the aforementioned studies, the authors designed the present testing protocol to provide for a large volume of cycles, as well as loads slightly higher than what is considered physiological, thereby accounting for clinical requirements resulting from more diverse anthropometric measurements.
All T-Button-A implants displayed elongation <3 mm on cyclic loading, with no instances of device failure before completion of the testing protocol. Their mean elongation was comparable with those of all other devices, with the exception of the Infinity femoral adjustable button, which had a mean elongation >3 mm (Table 4). Despite their overall mean elongation being statistically comparable with that of the T-Button-A, 2 implants from the Procinch RT group displayed elongation values >3 mm. Cyclic displacement affects not only the graft’s ability to integrate, but also clinical stability on return to sport. 27 Although the exact extent of graft slippage that would result in functional failure is unclear, 3 mm of elongation was considered to indicate device failure, because a side-to-side difference of ≥3 mm in anterior tibial translation, as measured by a KT-1000 arthrometer, is considered a sensitive indicator of symptomatic ACL failure.7,14,27
All T-Button-A implants demonstrated loads to failure >650 N, rendering them capable of withstanding physiologic loads during rehabilitation after ACL reconstruction. Although statistically significant differences in the mean loads to failure were noted between the T-Button-A and Tightrope RT, Endobutton CL Ultra, Infinity, and Procinch RT, these were not clinically relevant except in the case of the Infinity femoral adjustable button, wherein the mean load to failure was <650 N, and implants started to fail beyond loads of 485.16 N (Table 4). Failure of ACL reconstruction surgery could be due to graft loosening or failure of the cortical fixation device itself, wherein the device construct either lengthens excessively before graft incorporation to a point where the graft has insufficient tension to provide near-normal ACL laxity or breaks under load.3,27 Despite the greater clinical relevance of concerns regarding displacement, 3 therefore, the acceptability of load-to-failure values of CSFDs used in ACL reconstruction remains pertinent, which is why it was given equal consideration in this study.
Among the compared devices, it was found that T-Button-A, Tightrope RT, Proloop Ultra, Infiloop, and Endobutton CL Ultra consistently completed 3500 cycles of the testing protocol and delivered elongation and load-to-failure values within acceptable limits for all implants tested (Table 6). The combination of these attributes differentiated these devices from the others tested. Statistical assessments based on comparison of means alone are affected by outliers but do not account for the acceptability of their values. To overcome this limitation, the attainment of an acceptable result for all tested parameters was assessed as a surrogate measure of the health of each implant tested in this study. The present study fulfilled its primary objective, which was to biomechanically evaluate the T-Button-A and compare the results with those of 6 commercially available fixed- and adjustable-loop metallic CSFDs to establish its comparable, if not superior, performance. While the suboptimal performance of the Infinity femoral adjustable button in our study is broadly relevant and consistent with the findings of Bachmaier et al, 2 an interdevice post hoc comparative analysis of the tested CSFDs other than the T-Button-A was not the aim of the present study.
Maximization of tibial-side fixation during ACL reconstruction using all available means is critical for achieving optimal outcomes after surgery. Suboptimal biomechanical properties, increased graft slippage, and greater graft length requirements associated with aperture fixation using interference screws led to the increasing use of CSFDs.3,27,30,36 The use of adjustable-loop femoral CSFDs for tibial-side fixation using the all-inside technique decreased graft length requirements and offered the potential benefits of a bone-saving tunnel preparation. 2 In a recent radiologic follow-up study, however, Okutan et al 24 found that 98.5% of the tibial tunnel was filled by bone 1 year after ACL reconstruction when a titanium button and the full-tunnel technique were used. Ammann et al 1 noted that failure loads and elongation were similar whether an adjustable-loop CSFD was used for tibial-side fixation by itself (using the all-inside technique) or in combination with a custom-made extension button (using the full-tunnel technique). Recent systematic reviews have also shown comparable clinical outcome scores with the use of the full-tunnel and all-inside techniques.4,6 Early clinical outcomes after ACL reconstruction with the T-Button-A have also proved to be satisfactory. 8
Limitations
This study has the inherent limitations of being a time-zero, in vitro, device-only analysis. The results from laboratory-based device studies may not match those of clinical studies. 32 In vivo conditions entail numerous additional confounding variables such as graft elongation, cortical bone density, and the effects of intra-articular fluid and graft healing, which could influence the success of ACL reconstruction surgery. 3 The steel plate on which the devices were rested during biomechanical testing are stiffer and stronger than cortical bone and may not be representative of the clinical scenario. Furthermore, the application of force during biomechanical testing was in line with the long axis of the tunnel, which simulates “worst-case scenario” testing, but is not physiological. Last, elongation was not assessed periodically after every 500 or 1000 cycles, because the aim of the study was to evaluate the overall viability of the tested devices, rather than to perform an assessment of the temporal mechanisms of their failure. On the other hand, the obvious constraints of in vitro testing notwithstanding, the study protocol permitted an evaluation of the properties of isolated device constructs, absent tendon or bone. The greatest strength of this study, however, lies in the novelty of the T-Button-A itself, and the scarcity of biomechanical studies focused on dedicated tibial-side fixation CSFDs in the literature.
Conclusion
The PEEK T-Button-A is a viable alternative to conventional metallic CSFDs, for tibial-side-fixation during ACL reconstruction. Its biomechanical performance is comparable with most commercially available fixed- and adjustable-loop CSFDs and beyond what is considered acceptable to support ACL reconstruction grafts during early postoperative rehabilitation and ambulation. Future in vivo studies are required to compare clinical outcomes after ACL reconstruction using the T-Button-A and other metallic CSFDs.
Footnotes
Acknowledgements
We thank Mr Jagadeeswaran Kasinathan, engineer, Sironix Arthroscopy Solutions (Healthium Medtech Limited, Bengaluru, Karnataka, India) for his guidance regarding the use of the axial and torsion testing machine as well as its software.
Final revision submitted October 26, 2024; accepted November 22, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: Healthium Medtech Limited provided the implants tested in the study and allowed the authors to use their testing facility for carrying out the biomechanical evaluation. S.A. is a paid consultant for Healthium Medtech Limited, has received royalties from Healthium Medtech Limited, and has received speaker honoraria from Healthium Medtech Limited and Conmed. He holds a patent for the T-Button (Sironix Arthroscopy Solutions, Healthium Medtech Limited, Bengaluru, Karnataka, India). K.K.E. is a paid consultant for Smith & Nephew and BioradMedisys. S.K.C.K. has received funds for research from Healthium Medtech Limited. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Medicover Hospitals (MH/2024/IEC/NOC/09).
