Abstract
Background:
Many patients with developmental dysplasia of the hip (DDH) are young and highly active but often become limited by activity-induced pain. Characterizing how the abnormal bony geometry of DDH affects hip joint loading and multiplanar kinematics during sport-specific motions relevant to active patients may provide insight into injury mechanisms and inform optimal treatment options.
Purpose/Hypothesis:
The purpose of this study was to determine how hip joint loading and kinematics are altered in DDH during 2 common sport activities: running and multidirectional hop-cutting. It was hypothesized that superior and medial joint-reaction forces (JRFs) would be elevated during running and that medial JRFs would be elevated during hop-cutting when compared with controls.
Study Design:
Controlled laboratory study.
Methods:
Using patient-specific musculoskeletal modeling, both running and hop-cutting motions were analyzed for female patients with symptomatic DDH (DDH group) and female controls without hip disorders who were enrolled from October 2017 to August 2022. Hip joint angles, JRFs, and muscle forces were compared between the groups using 1-dimensional statistical parametric mapping.
Results:
Overall, 19 women in the DDH group and 19 female controls were included in the running analysis, and 13 women in the DDH group and 10 female controls were included in the hop-cutting analysis. During the running motion, medial hip JRFs and gluteal muscle forces were significantly elevated in the DDH group, while rectus femoris muscle forces were reduced. During the hop-cutting motion, medial hip JRFs, gluteus minimus, and gluteus medius muscle forces were elevated, while gluteus maximus forces were reduced in the DDH group. There were no significant group differences in hip joint angles for either activity.
Conclusion:
The lack of kinematic differences between women with symptomatic DDH and controls suggests that the JRF differences between groups stemmed from the abnormal joint geometry in the DDH group. These activities may have greater damaging potential than previously studied lower impact activities such as walking.
Clinical Relevance:
Better understanding of how loading varies from activity to activity can help patients with DDH, and clinicians understand the mechanistic causes of DDH-related hip pain and damage and plan appropriate intervention strategies.
Developmental dysplasia of the hip (DDH) is a bony disease characterized by a shallow acetabulum that develops in childhood and leads to reduced coverage of the femoral head.4,40 The abnormal relationship between the femur and acetabulum alters joint loads at the hip 16 and may lead to chondrolabral damage, including labral tears and accelerated cartilage degeneration.3,17 This damage ultimately increases the risk of early osteoarthritis, 29 with 48% of patients presenting for premature (age <50 years) total hip replacement having indicators of DDH. 3
Many patients diagnosed with symptomatic DDH are adolescents or young adults who regularly engage in demanding exercise or sport but become limited by insidious activity-induced pain. 28 In fact, symptom onset and corrective surgery often occur at younger ages in highly active individuals with DDH than those who are less active.23,28 Subsequent loss of ability to participate in the demanding exercise or sports may then contribute to reports of elevated anxiety, depression, and reduced quality of life in this population.14,32
Given the active nature of many young individuals with DDH, it is important to broaden the current scope of activities during which joint loading in dysplastic hips is assessed. Most of the evidence of abnormal loads in dysplastic hips, including joint-reaction forces (JRFs), has come from studies focused on gait.15,21,22,36 However, sport activities frequently require hip movements outside of normal ranges of motion such as hyperflexion/extension and excessive external rotation that can increase forces on the acetabular rim.19,27 Likewise, studies in healthy hips have shown that joint forces almost double in running when compared with walking. 12 The few published studies on running in individuals with DDH have focused primarily on sagittal plane joint kinematics and moments and have not considered frontal plane kinematics or joint forces.21,22 Characterizing how the altered bony geometry in DDH affects hip joint loading and multiplanar kinematics during higher demand/range of motion activities may provide insight into injury mechanisms and inform optimal treatment options.
The objective of this study was to determine how hip joint loading and kinematics are altered in DDH during 2 common sport activities: running and multidirectional hop-cutting. We hypothesized that, similar to what has been shown in walking, medial and superior hip JRF components would be elevated in patients with DDH during running when compared with controls and that these JRF differences would be more pronounced than during walking due to the larger loads in running. Additionally, we hypothesized that due to the lateralized hip joint centers and shorter abductor moment arms in patients with DDH, as well as the abducted and externally rotated position of the loaded limb during the cutting phase, muscle-driven medial/lateral JRFs would be elevated during hop-cutting motion when compared with controls.
Methods
Study Participants
After receiving institutional review board approval and informed consent, we recruited 20 female patients with symptomatic DDH (DDH group) and 22 female controls who were enrolled from October 2017 to August 2022 as part of a larger group of studies investigating the relationship between the bone and the muscle in DDH.16,34,36,37,42 Female participants were recruited, not only because symptomatic DDH is considerably more prominent (79%) in women 2 but also to eliminate geometrical differences at the pelvis that may occur due to sexual dimorphism.1,6 All patients with DDH were recruited through a single clinic and were diagnosed based on a lateral center-edge angle of <20° and the presence of unilateral hip/groin pain for ≥3 months. The female control participants were recruited from flyers placed in the community and had no hip pain or indication of hip conditions based on clinical examination and review of magnetic resonance imaging (MRI). Participants were included if they had a body mass index of <27 and self-declared that they were able to complete tasks including walking, squatting, running, hopping, and cutting. Other eligibility criteria included no prior hip surgery/fracture; no history of osteoarthritis (Kellgren-Lawrence grade ≥2) 24 ; no pain/injury to low back, knee, or ankle; and no neurological or other medical conditions that may influence coordination or balance.
Data Collection
All study participants visited our motion capture laboratory, where they were outfitted with an established set of 70 reflective markers. 10 Whole-body kinematics were collected using a 10-camera 3-dimensional motion capture system (Vicon) while participants ran at 2.5 m/s (200 Hz) and performed a 60° sidestep hop-cutting motion (100 Hz) (Figure 1). 20 Running speed was selected based on criteria for recreational runners8,11 as well as reported mean female running speed, 38 and was collected over the course of 5 seconds after a 90-second warm-up period. The hop-cutting motion consisted of a forward hop followed by a sidestep cut-and-run at 60° from the vertical in an unanticipated direction (Figure 1). Ground-reaction forces were recorded at 2000 Hz during the activities using either an instrumented treadmill (running) or embedded force plates (hop-cutting) (Bertec). Additionally, surface electromyography (2000 Hz) was used to record muscle activity from 8 muscles of the lower limb and back (Motion Lab Systems). Participants also underwent an MRI scan from the psoas origin to the knee joint line using a 3.0-T scanner (VIDA; Siemens AG) with T1-weighted gradient-echo sequences (1 mm3; voxels) as described previously. 36

The hop-cutting motion consisted of a forward hop onto 2 embedded force plates (1 to 2), followed by a 60° sidestep cut-and-run (3). The tested limb is the left limb in the figure shown.
Musculoskeletal Models
A 23 degrees of freedom, 98 musculotendinous actuator, generic OpenSim musculoskeletal model 25 was modified for each participant to create patient-specific models, as described previously. 36 Briefly, 3-dimensional patient-specific pelvic and femoral reconstructions from the MRI scans were aligned to generic OpenSim geometries and were incorporated into the generic model, and hip joint centers were updated based on a sphere that was fit to the reconstructed femoral heads. 36 All other bony model segments were scaled using distances between reflective skin markers to match participant anthropometrics. Additionally, muscle origins and insertions on the pelvis and femurs were customized for each patient based on the MRI scans and 3-dimensional bone reconstructions. 36 Each model’s musculotendinous properties (ie, optimal fiber length and tendon slack length) were scaled based on changes to muscle lengths. 41
Statistical Analysis
Joint angles were compared for all running gait cycles (heel strike to heel strike) across the 5-second trial, and a single representative trial was chosen for analysis. The symptomatic limb was analyzed for the DDH group (11/20 limbs dominant) and the dominant limb for the controls. Hop-cuts were analyzed across the stance phase from the initial hop landing to the instant the push-off foot left the force plate, with the push-off limb being the symptomatic side for the DDH group and the dominant side for the controls (Figure 1). Joint angles were calculated using a combination of inverse kinematics and a residual reduction tool. 39 OpenSim Moco, a direct collocation solver, was used to solve for muscle activations given the prescribed kinematics, while also incorporating the electromyography signals to improve consistency between experimental muscle activations and model activations. 5 These muscle activations were then used to calculate muscle forces and JRFs. The JRFs were analyzed as resultant forces and were broken down into their anatomic directional components (anterior/posterior, superior/inferior, and medial/lateral).
A 2-tailed general linear model was used to investigate the relationship between group (DDH vs controls) and muscle forces, JRFs, or joint angles. One-dimensional statistical parametric mapping was used to check for significant differences (P < .05) across the entire motion of interest while correcting for multiple comparisons. 31 The Hedge’s g effect sizes were also calculated for JRFs across the motions (large, ≥0.8). 7
Results
Participants
Overall, 39 female participants were included in the study: 38 participants (19 with DDH and 19 controls) were included in the final analysis of the running gait cycle, and 23 participants (13 with DDH and 10 controls) were included in the hop-cutting analysis (Table 1). Two control participants were not included in either analysis due to incomplete imaging sets, 1 DDH and 1 control participant were not included in the running analysis, and 7 DDH and 10 control participants were not included in the hop-cutting analysis due to high artificial “residual” forces in the models that resulted from discrepancies between simulated motion and real-world biomechanics. Although residual forces are typically unavoidable in musculoskeletal modeling, we excluded any models that exceeded published recommendations for residual-force thresholds. 18
Characteristics of the Included Participants a

BMI, body mass index; DDH, developmental dysplasia of the hip.
Running Gait Cycle
Medial JRFs were significantly larger in the DDH group when compared with controls during the absorption phase of stance (2%-24%; P < .001) and the double-float phase (92%-96%; P = .02) of the running gait cycle, ranging 56%-155% larger in the DDH group than controls (Figure 2). No other JRF components or the resultant JRFs were significantly different between the groups (Figure 2). Additionally, there were large Hedge’s g effect sizes for the medial/lateral JRF component at 1%-27% and 87%-98% of the running gait cycle, indicating that the DDH group had substantially larger medial JRFs than controls.

Mean JRF components across the running gait cycle for the DDH and control groups. Shaded areas represent ±1 SD. Black boxes indicate times of group differences in JRF, and black lines with asterisks represent large Hedge’s g effect sizes. Medial joint forces were larger in the DDH group during the stance phase and the float phase. BW, body weight; DDH, developmental dysplasia of the hip; JRF, joint-reaction force.
Muscle forces from the gluteus minimus were significantly larger in the DDH group at 2% (P = .03), 6%-8% (P = .01), 13%-21% (P < .001), 41% (P = .04), 60%-72% (P < .001), and 88%-98% (P < .001) of the running gait cycle; and the gluteus medius at 1%-10% (P < .001), 14%-18% (P = .003), 53%-54% (P = .03), and 90%-99% (P < .001) (Appendix Figure A1). The magnitude of muscle force differences ranged from 15%-79% larger in the DDH group for the gluteus minimus and 9%-165% larger for the gluteus medius. The DDH group also had significantly larger gluteus maximus forces at 77%-84% (P < .001) and 93%-98% (P < .001) and smaller rectus femoris forces at 7%-17% (P < .001) of the running gait cycle (Appendix Figure A1). During those times, gluteus maximus forces were 44%-146% larger in the DDH group than controls, and rectus femoris forces were 52%-66% smaller. There were no other significant group differences in any muscle forces.
There were no significant group differences in any of the hip joint angles during the running gait cycle (Figure 3). Hip flexion/extension ranged from 34.81° ± 7.66° flexion to −14.02° ± 7.81° extension in controls and 37.47° ± 8.31° flexion to −13.92° ± 4.88° extension in the DDH group. Hip adduction/abduction ranged from 13.43° ± 2.64° adduction to −8.52° ± 4.21° abduction in controls and 11.74° ± 2.29° adduction to −7.16° ± 2.68° abduction in the DDH group. Hip rotation ranged from 2.85° ± 4.49° internal to −5.14° ± 7.81° external in controls and 3.93° ± 5.72° internal to −5.05° ± 4.88° external in the DDH group.
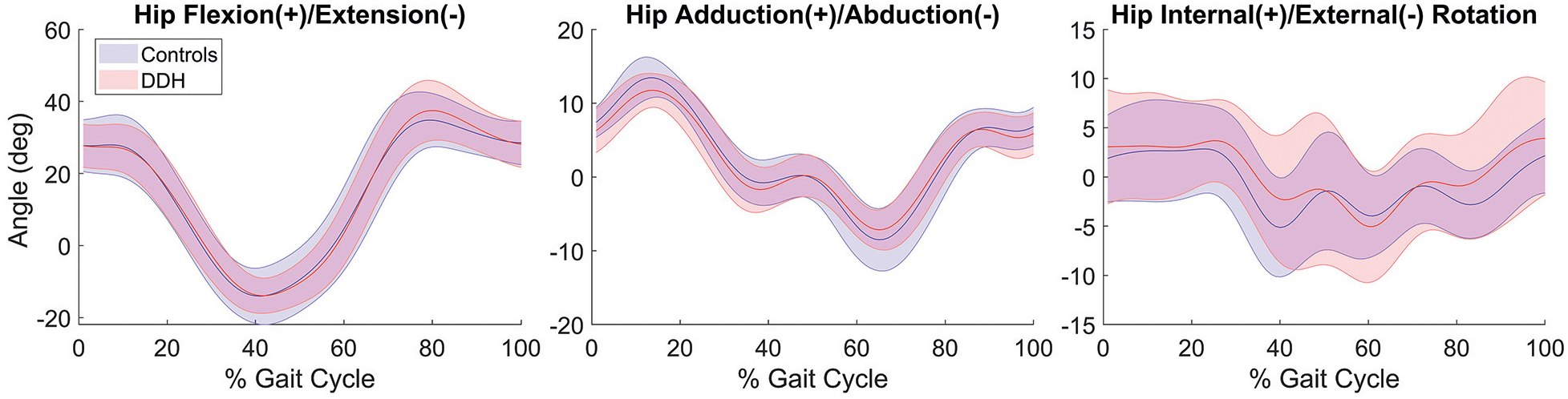
Mean hip joint angles across the running gait cycle for the DDH and control groups. Shaded areas represent ±1 SD. There were no significant group differences in hip angles. DDH, developmental dysplasia of the hip.
Hop-Cutting Motion
There were no statistically significant differences between the DDH group and controls in any hip JRF components or hip joint angles during the hop-cutting motion (Figure 4). However, there were large effect sizes, with the DDH group generating larger medial JRFs during the hop landing phase into the early propulsion phase (2%-4% and 13%-37%) of the motion and controls generating larger superior JRFs during single-limb propulsion (71%-73%). During those times, the magnitude of medial JRFs ranged 25%-43% larger in the DDH group than controls, while the superior JRFs ranged 12%-13% smaller in the DDH group.

Mean JRF components across the hop-cutting motion for the DDH and control groups. Shaded areas represent ±1 SD. Black lines with asterisks represent large Hedge’s g effect sizes. Medial joint forces were larger in the DDH group during the hop landing and superior forces were smaller during early propulsion. BW, body weight; DDH, developmental dysplasia of the hip; JRF, joint-reaction force.
Gluteus minimus muscle forces were 28%-81% larger in the DDH group at 1%-6% (P = .01), 14%-18% (P = .02), 28%-37% (P = .049), and 91% (P = .001) of the motion (Appendix Figure A2). Gluteus medius forces were also larger in the DDH group at 1% (P = .049), 16%-18% (P = .04), and 33%-34% (P = .045), whereas gluteus maximus forces were smaller at 77%-89% (P < .001) of the hop-cutting motion (Appendix Figure A2). During those times, gluteus medius forces ranged 27%-91% larger in the DDH group, while gluteus maximus forces ranged 35%-67% smaller in the DDH group versus controls. No other muscle forces had significant differences between groups.
There were no significant group differences in any of the hip joint angles during the hop-cutting motion (Figure 5). Hip flexion/extension ranged from 38.02° ± 13.29° flexion to −5.19° ± 9.94° extension in controls and 40.77° ± 8.64° flexion to −4.94° ± 7.93° extension in the DDH group. Hip abduction ranged from −24.81° ± 5.99° to −8.34° ± 4.43° in controls and −25.13° ± 5.46° to −9.36° ± 3.24° in the DDH group. Hip rotation ranged from 5.69° ± 9.07° internal to −21.85° ± 6.37° external in controls and 4.84° ± 6.99° internal to −21.14° ± 6.60° external in the DDH group.

Mean hip joint angles across the hop-cutting motion for the DDH and control groups. Shaded areas represent ±1 SD. There were no significant group differences in hip angles. DDH, developmental dysplasia of the hip.
Discussion
In this study using patient-specific musculoskeletal modeling, we found that, as hypothesized, medial hip JRF components were elevated in women with DDH during running and hop-cutting when compared with controls, although this difference was not significant during the hop-cutting exercise. These findings advance our understanding of abnormal DDH biomechanics by expanding previous reports of joint loads during slower tasks (eg, walking and squatting) to higher demand sport-related tasks during which risk for pain and joint damage may be elevated.
Running Gait Cycle
Similar to what has been shown in walking, medial JRFs were elevated in individuals with DDH during running.15,36 The medial JRF differences occurred during single-limb stance of the running gait cycle, which corresponds with the timing reported during walking.15,36 However, the differences in medial forces between groups during running were more pronounced than in walking, with differences averaging 0.60×BW as opposed to up to 0.39×BW.15,36 Also, unlike walking in these same cohorts, there were no significant group differences in superior or resultant JRFs. During the stance phase of running, the gluteus medius and other abductors generate large forces to stabilize the pelvis. 26 The large medial JRFs and corresponding large gluteus medius and minimus forces support the idea that abductor muscles surrounding dysplastic hips are subject to chronic larger-than-normal demand during ambulation. 16 We previously found that these high medial JRFs during walking are associated with patient reports of pain, reduced quality of life, and a negative impact on sports activity. 42 With JRF magnitudes during running almost double that of walking, 12 it stands to reason that these abnormally high forces may contribute to the pain and abductor fatigue reported by active individuals with DDH.
While forces generated by the gluteal muscles were higher than controls, rectus femoris forces were lower during a portion of the loading response after foot strike. These reduced rectus femoris forces may be linked to previously reported alterations to this muscle’s moment arms due to the dysplastic bony geometry. 36 While the rectus femoris functions primarily to flex the hip, it has secondary roles related to abduction. 30 The abduction moment arms of the rectus femoris are reduced in individuals with DDH, 36 which could lead to the reduced muscle force generation seen as well as decreased stabilization capacity in the coronal plane. In response, the primary abductor muscles may take on additional stabilizing demands, further elevating forces from the gluteus minimus and medius during the stance phase. In the second half of the running gait cycle, the elevated gluteus minimus, medius, and maximus forces occur when resultant loading on the limb is relatively low due to the limb’s being in swing (Figure 2). Because of this timing, those elevated loads likely have less damaging potential than those in the stance phase.
Differences in hip kinematics between patients with DDH and controls can contribute to joint-loading differences. However, a consistent theme of altered hip kinematics in DDH patients has not been established.21,33,35 While some studies have reported reduced hip flexion and extension during walking in patients with DDH,33,35 no significant sagittal-plane kinematic alterations have been found during running, which we confirmed with our results. 21 Thus, for the cohorts within the current study, the lack of kinematic differences found in all planes suggests that differences in JRF between groups stemmed primarily from the abnormal joint geometry.
Hop-Cutting Motion
Although not statistically significant, medial JRFs were elevated and superior JRFs were reduced in the DDH group compared with controls during the hop-cutting motion. Group differences with large effect sizes occurred as participants landed from the hop and again as they propelled themselves into the cutting maneuver. During the hop landing, the hip is in an abducted position. As with walking, running, and single-leg squatting, the higher medial forces during this time may occur due to an increased demand for stabilization by the abductor muscles. 9 An interesting difference between the running and the hop-cutting activities was that peak medial JRFs were larger during hop-cutting. While the hop-cutting motion is less repetitive than running, it is common among athletes in various sports. We surmise that the higher magnitude medial JRFs combined with the potentially less stable flexed, abducted, and slightly internally rotated position of the hip during the first part of the hop-cutting motion may increase shearing within in the hip and increase the risk of damage. Additionally, the reduced gluteus maximus forces seen in the DDH group during the propulsion phase when compared with controls may be a pain-avoidance strategy, with DDH patients pushing off less aggressively than controls.
There were no significant hip joint angle differences between the DDH group and controls during the hop-cutting motion, once again confirming that for this cohort, differences in JRFs resulted from abnormal DDH geometry, not altered movement patterns. Although we used an established hop-cutting technique, 20 there was greater patient-to-patient variability in the hop-cutting motion than running, as can be seen by the ranges of motion and standard deviations, suggesting that participants may have used differing techniques. As a result, analyzing movement patterns on a person-to-person basis may be beneficial in this group when identifying injury risk.
Limitations
There are several limitations that should be considered when interpreting the results of this study. First, we only included female patients, which limited the generalizability of these findings to all patients, and we only analyzed a single running speed and a single cutting angle, which did not represent the full spectrum of activities performed by the study participants. It has been shown that increasing running speed can increase hip JRFs. 12 Therefore, we wanted to control for speed across individuals while also using a speed that all participants could complete safely. Additionally, increasing the cutting angle can increase hip rotation, 20 which may change JRFs. However, due to laboratory limitations, 60° was the largest cutting angle we could perform while maintaining an unanticipated direction cue. Second, our sample size during the hop-cutting motion was relatively small, which limited our power to detect significant differences in joint loading and kinematics. Smaller hip flexion angles have been reported in individuals with hip pain as opposed to those without during a drop-jump landing motion similar to the hop-cut landing, a relationship that may become apparent in the hop-cutting motion with increased sample sizes. 13 Third, not only did the DDH population have abnormal bony geometry, but also pain. The presence of pain has the potential to alter movement and loading. We did not find significant differences in kinematic measures, which suggests that the patients were not hindered by pain during the data collection. However, we cannot state with certainty that pain did not affect the JRF measures or did not affect the patients’ actions outside of the laboratory. Last, we incorporated electromyographic tracking into our models and followed established modeling best practices to improve confidence in our muscle force estimations. 5 We also used models that incorporated patient-specific bony geometry and muscle paths.15,36 These models allow calculation of biomechanical variables that cannot be directly measured. However, models are imperfect representations of in vivo musculoskeletal mechanics, and results should be interpreted with caution. Because of this, we only reported models that met our model validation criteria. Regardless, as both patient and control groups were run through the same modeling pipeline, we believe that our comparisons can identify valuable group differences despite the aforementioned modeling limitations.
Altered loading in dysplastic hips can vary by activity. The findings of this study add running and hop-cutting to the list of activities where JRFs have been reported. Elevated medial JRFs are most consistent across activities, being found in walking, running, and to some degree hop-cutting, but not in squatting.15,36,37 For these young, active individuals, altered loading may instigate muscle pain and fatigue. Due to the high loading and repetitive nature of running and the extreme joint angles during hop-cutting, these sports activities may also have greater damaging potential than previously studied activities. By understanding how loading varies from activity to activity, we can better help both patients with DDH, and clinicians understand the mechanistic causes of DDH-related hip pain and damage and plan appropriate intervention strategies that may preserve the hip joint.
Conclusion
The findings of this study indicate that altered hip JRFs, such as elevated medial JRF components, occur during certain sport-specific activities, including running and hop-cutting in female patients with symptomatic DDH. There was a lack of kinematic differences between the controls and patients with symptomatic DDH, suggesting that the JRF differences between groups stemmed from the abnormal joint geometry in the DDH group. While elevated JRFs have been noted during walking, sport-specific activities may pose greater risks, highlighting their significance in the understanding of DDH.
Supplemental Material
sj-png-1-ojs-10.1177_23259671251318736 – Supplemental material for Altered Joint Forces Found in Symptomatic Dysplastic Hips in Women During Sport-Specific Activities
Supplemental material, sj-png-1-ojs-10.1177_23259671251318736 for Altered Joint Forces Found in Symptomatic Dysplastic Hips in Women During Sport-Specific Activities by Molly C. Shepherd, Rannon Huo, John C. Clohisy, Jeffrey J. Nepple and Michael D. Harris in Orthopaedic Journal of Sports Medicine
Supplemental Material
sj-png-2-ojs-10.1177_23259671251318736 – Supplemental material for Altered Joint Forces Found in Symptomatic Dysplastic Hips in Women During Sport-Specific Activities
Supplemental material, sj-png-2-ojs-10.1177_23259671251318736 for Altered Joint Forces Found in Symptomatic Dysplastic Hips in Women During Sport-Specific Activities by Molly C. Shepherd, Rannon Huo, John C. Clohisy, Jeffrey J. Nepple and Michael D. Harris in Orthopaedic Journal of Sports Medicine
Footnotes
Appendix
Acknowledgements
The authors thank the Stanford Restore and Mobilize Center for their assistance with OpenSim Moco, the University of Southern California ReproRehab program (NIH R25HD105583) for their data science training, and Ke Song for his help in model development.
Final revision submitted September 10, 2024; accepted September 19, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was supported by the National Institutes of Health (grants R01AR081881, K01AR072072, and T32HD007434) and a Graduate Student Grant-in-Aid from the American Society of Biomechanics. J.C.C. has received consulting fees from MicroPort Orthopedics and Zimmer Biomet Holdings, nonconsulting fees from Synthes GmbH, royalties from MicroPort Orthopedics, and hospitality payments from Encore Medical. J.J.N. has received education payments from Elite Orthopedics, consulting fees from Smith & Nephew and Responsive Arthroscopy, nonconsulting fees from Arthrex and Smith & Nephew, royalties from Responsive Arthroscopy, and hospitality payments from International Life Sciences. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Washington University in St Louis (reference No. 201612053).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
