Abstract
Background:
Understanding the factors contributing to willingness to participate in randomized clinical trials (RCTs) after anterior cruciate ligament reconstruction (ACLR) is crucial to optimizing recruitment and understanding whether interested participants represent the patient population that may benefit from the studied treatment.
Purpose:
To understand patients’ willingness to participate in a future RCT of an oral medication to prevent posttraumatic osteoarthritis (PTOA) after ACLR.
Study Design:
Cross-sectional study; Level of evidence, 3.
Methods:
A total of 103 patients aged 18 to 45 years who were either planning to undergo ACLR in the next 4 months or had undergone ACLR within 1 year of the screening date were recruited from 2 institutions. The patients viewed a video explaining the trial and completed a questionnaire that included demographic characteristics, pain intensity, activity level, willingness to participate in the hypothetical trial, and their perceived risk (on a scale of 0%-100%) of developing knee PTOA (next 10 years or lifetime).
Results:
Within the cohort, 31% stated they were “definitely willing,” 38% were “probably willing,” 17% were “unsure,” and 14% were “unwilling” to participate in a hypothetical trial. Willingness did not differ by pain or activity level; however, younger patients stated they were less willing to participate. The most common reasons for unwillingness to participate included not wanting to take a medication daily (59%) and concerns about medication risks or side effects (59%). Respondents who indicated a definite willingness to participate in the trial had higher perceptions of their own PTOA risk over the next 10 years than those who indicated they would not participate (70% vs 50%).
Conclusion:
In this prospective preference assessment, 69% of survey respondents expressed a willingness to participate in an RCT involving an oral medication to potentially alter the progression of PTOA after ACLR. The results suggest that an RCT in this study should include clear and concise information on the risk of developing PTOA after ACLR and the safety and tolerability of study medications in the recruitment materials.
Knee osteoarthritis (OA) is a costly, disabling problem that affects >30 million adults in the United States.36,51 Posttraumatic osteoarthritis (PTOA) accounts for nearly 12% of these cases 57 and occurs as a result of injuries, such as anterior cruciate ligament (ACL) rupture, meniscal tear, or intra-articular fracture. On average, PTOA develops approximately 10 years earlier than primary knee OA. 29 Therefore, sustaining an ACL injury early in adulthood is associated with greater lifetime risk, longer period of disability, and earlier onset of knee OA. 11 In addition, PTOA is more likely than primary OA to cause functional disability and work loss among younger individuals.4,38,58 Studies have demonstrated that around 60% to 75% of patients who sustained ACL injury demonstrate early evidence of knee OA and around 50% of patients who undergo ACL reconstruction (ACLR) develop OA within 12 to 14 years of injury.4,24,35,52 Patients who have sustained an ACL injury, even more so if the injury is accompanied by a meniscal tear, have a higher conversion rate to total knee replacement as compared with controls without these injuries.25,28,56
Although ACLR offers many benefits, it can neither treat nor prevent the development of PTOA.10,57,58 Ferrero et al 19 found that the relative risk (RR) for developing knee OA was higher after ACLR compared with nonoperative treatment (RR = 1.72), while Cuzzolin et al 16 reported no significant differences between operative and nonoperative ACL treatment in the risk of developing knee OA within 5 years after ACL injury. The earlier age of onset for PTOA compared with primary OA emphasizes the importance of the need to develop treatments to prevent and delay the development of OA after injury. It is also critical for researchers to unravel the biochemical and biomechanical processes that occur after traumatic injuries and the development of symptomatic OA. Results of several clinical studies have reported that metformin could potentially act as a disease-modifying drug for the development of OA.31-33,37,42,59 Given the important limitations of previous studies, such as retrospective design and confounding by indication, a well-designed and adequately powered clinical trial is warranted.
Multiple studies have researched patients’ willingness to participate in clinical trials that have been helpful for trial planning and optimizing patient recruitment.1,8,21,49,55 Our goal was to understand patients’ willingness to participate in a hypothetical placebo-controlled randomized control trial (RCT) designed to establish the efficacy of metformin to prevent or delay PTOA development and improve future trial designs and optimize recruitment methods. Therefore, we conducted a prospective preference assessment (PPA) of a pharmacological clinical trial to alter the progression of PTOA after ACLR. The PPA was performed in conjunction with designing the Preventing Injured Knees from Osteoarthritis: Severity Outcomes (PIKASO) trial—a quadruple-blinded randomized placebo-controlled trial that will establish the efficacy of the oral medication metformin in delaying the onset of PTOA after ACLR (ClinicalTrials.gov identification NCT06096259).
The present study describes the PPA—designed to understand both the level of interest in and reasons for refusal of participation in a hypothetical RCT. We hypothesized that the most common reasons for not wanting to participate would be medication burden and efficacy concerns. The PPA will improve our understanding of both the factors associated with a willingness to participate and the reasons given by individuals who are unwilling to participate, provide an estimate of the anticipated participation rate of the full-scale RCT, and assist in optimizing and improving study materials and recruitment for the trial.
Methods
Location and Population
Patients from 2 study sites (Boston, Massachusetts, and Lexington, Kentucky) were recruited between March and July 2023. Both clinics routinely treat a heterogeneous group of patients that is representative of the general population who sustain these injuries. One site recruited participants from 8 different practicing surgeons, while the other conducted recruitment from 4 orthopaedic surgeons. Patients aged 18 to 45 years who were either planning to undergo ACLR surgery in the next 4 months or had undergone ACLR surgery within 1 year of the date of recruitment were eligible for recruitment. The exclusion criteria comprised the following conditions: a Kellgren-Lawrence grade of 3 or 4 as determined by standing bilateral radiographs taken before ACL surgery; ACL injuries treated nonoperatively or not scheduled for surgery; previous ACLR on the index knee (the knee that underwent surgery) that occurred outside the eligibility timeframe; metformin use within the previous 6 months; diabetes, dementia, or chronic renal impairment; and non-English-speaking patients. The protocol for this study received institutional review board approval from both study sites.
Eligible patients were identified through screening electronic medical records and physicians’ clinic schedules. Potential participants were either sent a letter with a link to the survey or were approached in-clinic during a visit with an orthopaedic provider. Letters were sent through the hospital-based electronic medical record system with a link to the survey and through the postal system with a scannable quick-response (QR) code to the survey.
Survey Design
The study consisted of viewing a video and completing a questionnaire. The video (available as a Video Supplement) begins with 2 physicians (E.G.M. and M.H.J.) briefly describing the pathology, symptoms, and cause of knee OA and PTOA. The video does not mention common treatments for knee OA, such as injections or total knee replacement, but states that the damage to the joints by OA cannot be reversed, only potentially slowed. It then provides an animation that explains the hypothetical trial in greater detail to help individuals make a more informed decision about their willingness to participate in a future trial. The video introduces metformin—the study medication—as a widely used, safe medication that was approved to treat people with diabetes, but has been shown to slow aging, assist with weight loss, and potentially delay the development of OA.31-33,37,42,47,53,59 The video describes the treatment groups, emphasizing that both groups receive identical treatments (including physical therapy, 6-12 months of daily medication, and 3 study visits/magnetic resonance imaging [MRIs]) with the 1 exception that 1 group takes metformin daily and the other placebo each day.
We solicited interest in participation in the hypothetical trial (modeled after the design of the PIKASO trial). Assignment to the medication groups was random, and thus each participant had an equal chance of being placed in either group. Patients were asked to indicate how likely they would be to participate in the future RCT, where the duration of daily oral medication (metformin or matched placebo) lasted 1 year. There were 5 possible responses as follows: definitely would participate, probably would, unsure, probably would not, and definitely would not. Respondents who selected unsure, probably not, or definitely not were asked additional questions to understand better why they would not wish to participate, and these same respondents were asked if they would participate in the trial if the duration of study medication use was shortened from 1 year to 6 months. All respondents were also asked whether they were willing to come to the study site for 3 study visits, each lasting 2 hours and including a knee MRI, throughout the study. Since our future study is focused on observing the development of early-stage knee OA detected with MRI, we did not ask about willingness to undergo radiographic imaging.
The PPA also included several validated questionnaires. We assessed knee pain using the Knee injury and Osteoarthritis Outcomes Score (KOOS)–Pain subscale (0-100, where lower scores indicate worse outcomes) and knee activity level over the 12 months before their ACL injury using the Marx activity rating scale (0-16, where higher scores indicate more activity).39,50 We asked respondents to estimate their perceived risk (on a scale from 0%-100%) of developing knee OA. Finally, we asked them to estimate the risk of OA for someone of the same age without ACL injury. We asked these questions with respect to risk over the next 10 years and the respondent's lifetime. We did not provide information on the percentage of those who typically develop OA in those who did and did not sustain an ACL injury.
Statistical Methods
Patients were stratified according to the following 4 preference groups: definitely participate, probably participate, unsure, and would not participate (a combination of probably and definitely not) as well as by clinical site. Descriptive statistics are presented as medians with interquartile ranges (IQRs) for numeric variables and as counts with percentages for categorical variables. We compared demographic, clinical, activity, and perceived OA risk characteristics across the 4 participant preference categories using the chi-square and Fisher exact tests for categorical variables and Kruskal-Wallis tests for continuous variables. All analyses were conducted using SAS Version 9.4 (SAS Institute).
Results
Initially screened were 260 patients who either had an ACLR in the past year or were planning to undergo one in the next 4 months. A total of 169 (65%) patients were screened through the electronic medical records mechanism in anticipation of sending a link or QR code, and 91 (35%) patients were screened the day before their clinic visit related to their ACL injury/surgery in anticipation of recruiting them in person at their clinic visit and giving them a QR code. Of the 260 screened patients, 12 (5%) were deemed ineligible during the screening process (all from the group that was to receive the electronic link), with the most common reason being a previous ACL surgery on the index knee outside the eligibility timeframe (Figure 1).

Diagram of patient screening and enrollment. ACL, anterior cruciate ligament; OA, osteoarthritis.
Out of the 248 eligible participants, 35 (14%) were missed in the clinic and 213 (86%) were provided a questionnaire (74% of the group recruited remotely and 26% of the group recruited in person). Out of the 213 participants who received a questionnaire via an electronic link or in-person recruitment, 103 (48%) completed the survey (45% of the group recruited remotely and 57% of the group recruited in person), 20 (10%) refused participation (10% of the group recruited remotely and 7% of the group recruited in person), and 90 (42%) never completed the survey (45% of the group recruited remotely and 36% of the group recruited in person).
The median age of the participants who completed the survey was 26 years (mean age, 28 years), and 52% were women (Table 1). Most participants were White (74%), of non-Hispanic ethnicity (92%), and well-educated (67% attained at least an associate degree). We did not observe differences in participants across the 2 sites, except education level. Overall, those at the Kentucky site (site 2) were more likely to report they were in high school or college but had not completed a degree. When investigating specific age groups, we did not see a difference in education levels between the sites for participants ages ≤20 years, but there was a significant difference in education levels for participants ≥25 years. Most of the participants (n = 99) had undergone ACLR in the past year, whereas only 4 (4%) had a planned ACLR within the next 4 months.
Participant Characteristics Stratified by Study Site a

Data are presented as median [interquartile range] or n (%).
Willingness to Participate
When asked about their willingness to participate in the PIKASO trial if it was available today, 32 (31%) indicated that they would definitely participate, and 39 (38%) reported that they would possibly participate. Eighteen (17%) reported they were unsure, and 14 (14%) indicated that they probably or definitely would not participate in the hypothetical trial. Among the 14 who were not willing to participate in the future trial, 12 (86%) indicated they probably would not participate.
Patients who stated that they were unsure or probably or definitely unwilling to participate were asked to provide the reasons for their response and were also queried about their willingness if the medication duration was shortened from 1 year to 6 months. The most common reasons for unwillingness to participate for 1 year included not wanting to take a medication daily (59%), concerns about medication risks or side effects (59%), and not wanting to commit to 1 year of participation (34%). Other reasons for not wanting to participate include not wanting the treatment to be decided by chance (22%), not feeling comfortable participating in research (3%), logistical barriers such as school schedules or no longer living in the area (13%), or study logistics being too vague (9%). The reasons for not wanting to participate in the 6-month trial mirrored those from the 1-year trial and only 2 (6%) of those initially unsure/unwilling to participate changed their willingness if the duration of taking the study medication was reduced from 1 year to 6 months.
The hypothetical study proposed 7 in-person study visits that included MRI scans. Among respondents who said they would not participate in the future trial, only 21% indicated a willingness to come in for a study MRI, as compared with 100% and 97%, respectively, of those who were definitely or probably interested in participating. Finally, among respondents who were unsure about participation in the trial, 78% were willing to undergo multiple MRIs.
Demographic characteristics by willingness to participate categories revealed that among the youngest and middle-aged groups, 25% and 21% indicated they would “definitely” participate, compared with 65% of those in the oldest age group (Table 2). More of the youngest and middle age groups reported they would “probably” participate (41% and 47%, respectively), compared with 15% of those in the oldest age group.
Patient and Clinical Characteristics Stratified by Willingness to Participate a

Data are presented as n (%) unless otherwise indicated. The boldface P value indicates a statistically significant difference between groups (P < .05). KOOS, Knee injury and Osteoarthritis Outcome Score; NSAIDs, non-steroidal anti-inflammatory drugs.
n = 99.
Clinical Characteristics
Among respondents with a KOOS-Pain score of ≤80, a total of 44% of participants were definitely willing to participate in PIKASO (Table 2). In comparison, for those with a KOOS-Pain score of >80, a total of 22% of participants were definitely willing to participate. Among respondents with a Marx score of >12, a total of 47% of participants indicated they would definitely be willing to participate, while 23% of respondents with a Marx score of ≤12 stated they would definitely participate.
OA Risk Perception
Survey respondents who indicated a definite willingness to participate in the trial had higher perceptions of their own OA risk in the next 10 years (median, 70%; IQR, 50%-88%) than respondents who indicated they probably or definitely would not participate in the trial (median, 50%; IQR, 36%-73%) (Figure 2). When asked about the risk of developing OA over their lifetime, on average, survey respondents in all willingness-to-participate groups estimated that they had at least a 79% chance of developing knee OA. For all willingness levels and both time points, the respondents rated their own risk of developing OA as higher than the risk of persons their age (Figure 2).
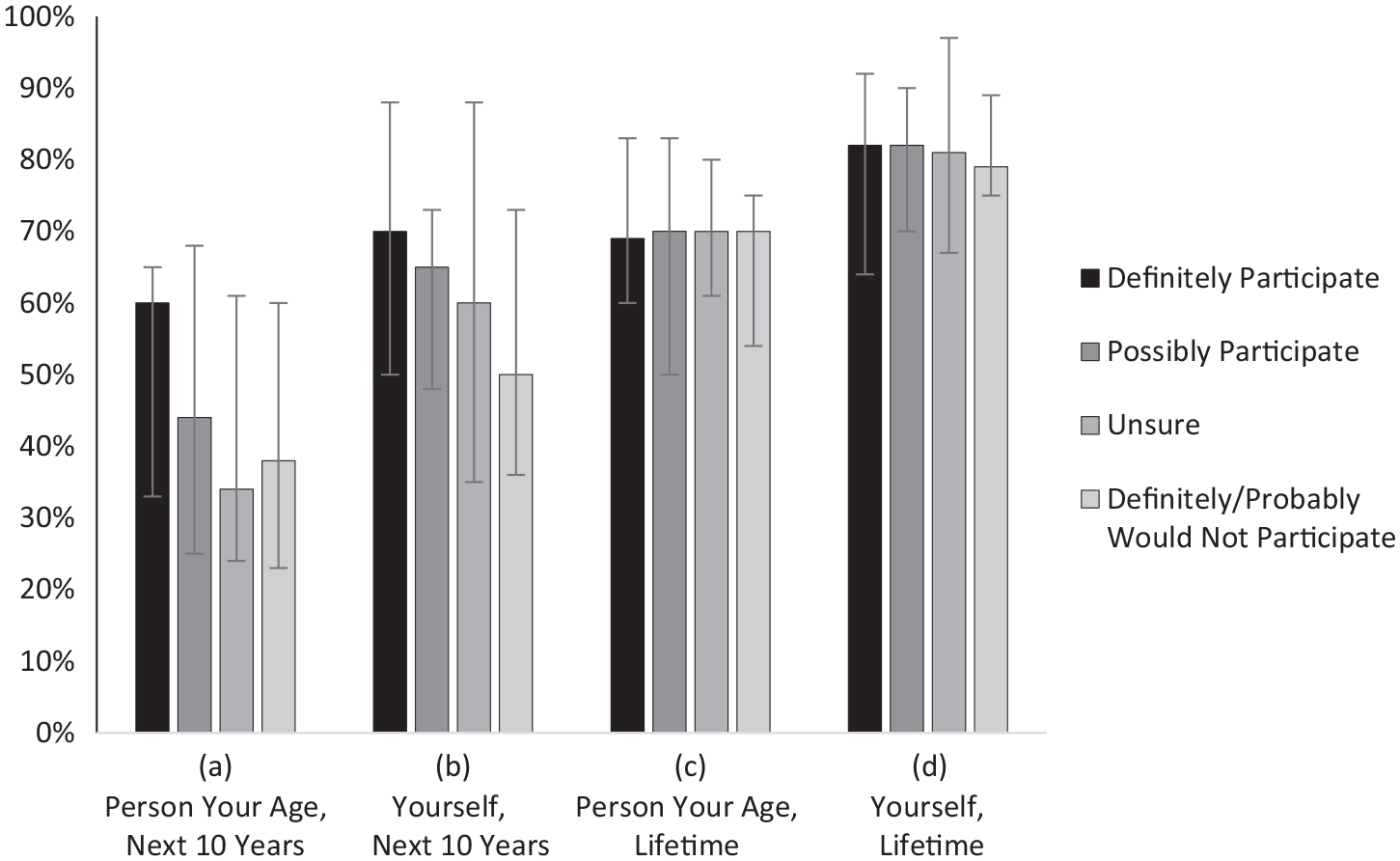
Survey respondents’ perception of the risk of OA according to the likelihood of study participation. Patients were asked to rate on a scale of 0% to 100% what they think the chances are of developing OA for (A) a person of their age in the next 10 years, (B) themselves in the next 10 years, (C) a person of their age in their lifetime, and (D) themselves in their lifetime. The results are shown are median rating values, with error bars indicating the interquartile range. OA, osteoarthritis.
Discussion
In this PPA, 69% of the survey respondents expressed willingness to participate in the PIKASO trial if it were available today. The most common concerns about participation included not wanting to take a medication daily, medication risks/side effects, and not wanting to commit to a study for 6 months or 1 year. Other reported reasons for not wishing to participate included not wanting the treatment to be determined by chance, an issue that all RCTs face, or logistical barriers, such as college schedules or no longer living in the area.
Importantly, we found that individuals who were unsure or unwilling to participate appeared to report lower estimates of their own risk of developing PTOA than those interested in participating. This finding suggests that groups investigating interventions that may reduce their risk of PTOA should provide potential participants with clear and concise educational information on the risk of developing PTOA after ACL injury. Previous studies have demonstrated that many patients with ACL injury are unaware of their risk of developing PTOA and how to manage their risk.20,34 Highlighting and emphasizing OA risk awareness for those who have sustained an ACL injury may encourage patients to become more involved in risk management and prevention strategies.
Understanding hesitations for participating in clinical trials is critical for improving recruitment. A study examining orthopaedic clinical trials15,18,22 from 2007 to 2022 reported that of the 1369 discontinued trials, the most common reason for trial discontinuation was insufficient patient accrual (29%). More specifically, a systematic review of 24 surgery-controlled trials (orthopaedic surgery: n = 9; cardiovascular surgery: n = 8; neurosurgery: n = 7) found that only 3 trials were completed within the predicted timeframe, and, on average, studies took 88% more time than originally projected to reach recruitment targets. 7
In designing future clinical trials involving daily oral medications to alter the progression of PTOA after injury, one aspect to focus on is clarifying and detailing the safety of the study medications. Given that many of the hesitations for participating revolved around the nature of taking the medication, such as its frequency and possible risks/side effects, emphasizing the safety and the magnitude and duration of potential side effects of both the study drug and the placebo may encourage participation in clinical trials. It is important to help patients understand their natural history, typical progression patterns, and potential treatments so that they can make informed decisions about their health condition. For example, patients who are more knowledgeable about their care and condition often reported better treatment outcomes and higher odds of making quality decisions. 9 This knowledge allows patients to make more informed decisions about trial participation and may also improve trial retention rates.3,13 In addition, understanding the exact reasons for not wanting to take a medication daily may help to increase enrollment. For example, not wanting to take daily medication because it may be viewed as a burden versus fear of frequently taking a medication requires different ways to address the concern.
Duration of exposure to the medication was also a common reason for unwillingness to participate. However, out of the 11 respondents who were unsure/unwilling to participate because of the 1-year timeline, only 2 changed their willingness to participate if the duration was shortened to 6 months. One possible reason for this concern is that patients are uncertain of the long-term consequences of taking the medication for 1 year/6 months. Conversely, others may not believe that 1 year/6 months is enough time to see clinically meaningful results, and thus are unwilling to participate. Emphasizing that trial participation is completely voluntary and providing greater clarity on the safety of the drug can help to address these concerns and increase recruitment.
In addition, to address those who have discomfort with research studies in general, it may be beneficial to use approachable language, a racially/ethnically diverse study staff, and supplemental material, such as a video or visual cues, to help with the clarity and conciseness of the explanations and to emphasize that participation is voluntary and that they are free to stop the study at any time and their decisions do not affect their medical care.2,30,48
Our results also showed that age may contribute to the likelihood of participation. When combining those who were definitely and probably willing, the differences between the age groups decreased (66% for 18-25 years, 68% for 26-35 years, and 80% for 36-45 years age groups); nonetheless, those in the oldest age group still have the highest proportion who are willing to participate. Older individuals may be more likely to participate in research trials because they have greater familiarity with OA and its risks, tend to be more highly educated, and potentially understand better the future consequences and risks of their injuries.
One strength of the present study was the clarity of the material provided. Only 3 (9%) of the 32 participants who were unsure or not interested in trial participation indicated that their reason for hesitation was that the study logistics were too vague. The inclusion of the video may have helped participants to make a more well-informed decision about trial participation. Another strength was improved generalizability by the inclusion of multiple sites, in 2 different geographical locations, which resulted in a more diverse sample population.
Our research team has found the PPA design to be an informative and important part of planning a large-scale RCT. We performed PPA studies when designing 2 multicenter RCTs that have completed enrollment, and the actual rate of enrollment in the trials mirrored the simulated rate of enrollment in the PPA. This process allows us to be more confident about our study planning activities that involve recruitment at our various sites. The PPA for the Meniscal Tear in Osteoarthritis Research (MeTeOR) trial found that 22% of eligible respondents indicated they would definitely participate while 24% indicated they would probably participate in the MeTeOR trial, 14 and the actual trial enrollment was around 30% among those eligible. 26 Similarly, the PPA for the Treatment of Meniscal Problems in Osteoarthritis (TeMPO) trial reported that 24% and 39% were definitely and probably willing, respectively, to participate in the TeMPO trial, 27 and ultimately 36% of those fully eligible were randomized into the trial (unpublished data, 2023).
Limitations
One limitation of our study is its modest sample size, which leads to lower precision around our estimates. We posed a hypothetical question to respondents (“If the trial were available today, how likely would you be to participate?”), and we acknowledge that responses of actual persons once the trial is launched may not mirror these results. In addition, although our survey results indicated that more than half of the PPA respondents would be willing to participate in the trial, these results may be biased given that 103 of the 248 eligible completed the survey. Our survey completion rate may be attributed to our target population of busy, working young adults who may not have time to respond to calls or invitations for the study, and it raises concerns about attrition rates for future studies. Interest in the PPA study may have been higher if study staff were able to communicate directly with participants about their concerns or answer any questions regarding the safety/logistics of the study. We, however, did not have approval to follow up with potential study patients about possible concerns. Still, our survey completion rate (48%) falls within the anticipated range of questionnaire response rates from previous studies5,40,43,44,46,54,60 (24%-80%).
Another possible reason for the modest response rate was that study staff largely utilized remote recruitment, such as sending questionnaires via email/postal service and making cold calls. While interest levels in participating were similar by recruitment method, remote recruitment had a lower overall response rate. Given our younger population, texting may have been a better method of communication. Without being able to utilize a diverse set of communication modes, it may be difficult to recruit younger patients without employing in-person methods. While virtual screening allows for more efficient and higher volume recruitment, in-clinic screening allows for relationship building with face-to-face contact and an ability to ask questions.6,23,41,45 Finding ways to incorporate personal contact into recruitment may help increase participation and retention rates. For example, Cooksey et al 12 found that having the patients’ provider hand out study materials or having study staff offer video meetings to discuss concerns with participants helped increase recruitment numbers. In addition, patients may feel more obliged to respond and show interest in person compared with remote recruitment methods.
Another potential method is to improve the effectiveness and usability of preexisting web-based recruitment services. These services include hospital-based electronic medical record systems like EPIC, RedCap, or other online applications that researchers utilize to recruit patients. For example, Mouarbes et al 17 noted that an augmented care experience helped increase patient participation and retention rates for a series of 11 questionnaires. By using a more streamlined approach for online recruitment, patients may feel less overwhelmed about participating.
Patients will need to decide to participate in the trial before undergoing ACLR, and yet our findings are largely based on post-ACLR patients (n = 99 [96%]). Given the small proportion of pre-ACLR patients relative to the sample size (n = 4), the questionnaire results of these patients likely did not affect the presented results. Future studies must recruit a representative proportion of participants in the same manner that recruitment will be carried out to have a more complete understanding of willingness levels. Also, given that so many of our study participants were postoperative, their willingness to hypothetically participate may have been influenced by postoperative factors such as current pain levels or their overall experience with their surgery. In addition, our study only included patients who were up to 1-year post-ACLR, a time point where PTOA is not often present or the primary concern for most patients. Given that our future trial will enroll patients before ACLR, we were focused on a shorter time frame. Future studies interested in longer-term treatments should consider surveying patients at further postoperative time points (ie, 5-10 years since surgery) to assess how patients’ willingness to participate in future RCT changes as the risk of developing the disease potentially increases.
Furthermore, although our study explained the benefits of metformin in relation to PTOA development, our video and study letter did not emphasize the additional potential benefits that may be more relevant for patients in the short term, such as less pain or stiffness during the immediate postoperation period and as they return to daily activities and sports. This lack of emphasis on the benefits that may be more immediately relevant to participants than longer-term consequences (such as the risk of PTOA) perhaps also contributed to why many of those approached did not complete the survey. However, despite potentially understated explanations of pain or activity benefits of the trial, most of the participants (69%) still expressed a willingness to participate in future trials.
Conclusion
We found that 69% of our sample expressed a willingness to participate in the PIKASO trial if it were available today, with 31% definitely willing and 38% probably willing. Older respondents expressed a greater willingness than those who were younger, as did respondents with a higher perception of their individual OA risk. Individuals who were unsure or unwilling to participate reported believing that they have a lower risk of developing PTOA and also expressed more concerns about side effects or duration of treatment. These findings suggest that investigators studying pharmacological interventions to alter the progression of PTOA should provide potential participants with detailed information on the risk of developing PTOA after ACLR as well as information about what to expect regarding the expected side effects, safety, and tolerability of the study interventions. Hopefully, the implementation of these measures will increase interest in participating in PTOA intervention trials, thereby improving the generalizability of the study results.
Footnotes
Final revision submitted August 3, 2024; accepted August 28, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study received funding from the Arthritis Foundation. J.N.K. has received an honorarium from Pfizer. A.V.S. has received research and fellowship support from Smith & Nephew, Arthrex, Allosource, and the Arthritis Foundation; education payments from Legacy Ortho; consulting fees from Vericel and Bioventus; nonconsulting fees from Bioventus, Vericel, and Smith & Nephew; and hospitality payments from Stryker. E.M. has received education payments from Arthrex and hospitality payments from Medacta USA. J.S.K. is an employee of the Arthritis Foundation, the funder for this study. M.H.J. is on the scientific advisory board for Regeneron Therapeutics and Biosplice (formerly Samumed). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Mass General Brigham (reference No. 2022P003066 and 85371).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
