Abstract
Background:
High tibial osteotomy (HTO) is a treatment option for younger, active patients with medial compartment knee osteoarthritis. Clinical results of HTO have been shown to deteriorate over time despite initial satisfactory results, requiring patients to eventually undergo conversion to total knee arthroplasty (TKA). Evidence monitoring survivorship of HTO remains scarce and potentially outdated.
Purpose:
To investigate the impact of concomitant cartilage repair procedures, conversion to TKA, and associated complications for HTO.
Study Design:
Systematic review; Level of evidence, 4.
Methods:
We searched MEDLINE, Embase, Cochrane Library, and SCOPUS from inception to July 18, 2023, for studies reporting on survivorship and associated complications after medial opening-wedge HTO. Pooled analysis of conversion to TKO was categorized as occurring at <5 years, 5 to 10 years, or >10 years postoperatively. Further subgrouping was performed on studies reporting on HTO with concomitant cartilage repair procedures.
Results:
Overall, 59 studies comprising 5162 patients were included. Rates of conversion to TKA were 4.5% at <5 years, 8.3% at 5 to 10 years, and 11.2% at >10 years. When comparing patients with isolated HTO versus HTO with concomitant cartilage procedures (including mesenchymal stem cell augmentation, osteochondral allograft transplantation, microfracture, abrasion arthroplasty, and autologous chondrocyte implantation), there was no significant difference in survival rates at <5 years (relative risk, 0.78 [95% CI, 0.45-1.33]; P = .36) or 5 to 10 years (relative risk = 0.76 [95% CI, 0.32-1.83]; P = .55). The overall complication rate was 12.1%; intraoperative lateral hinge and tibial plateau fractures had pooled complication rates of 1.6% and 2.0%, respectively. The rate of nonunion was 1.7%, and pooled rates of superficial and deep infections were 2.6% and 2.0% respectively.
Conclusion:
Rates of conversion to TKA and complications were low and acceptable, although survival rates decreased with time. Concomitant cartilage repair procedures as a whole did not significantly improve survivorship; however, more high-quality studies are warranted to determine the impact of individual concomitant cartilage repair procedures.
Surgical options for management of medial compartment osteoarthritis generally include total knee arthroplasty (TKA), unicompartmental knee arthroplasty (UKA), or high tibial osteotomy (HTO). 62 For relatively younger and active patients, treatment may be challenging, as patients seek to remain involved in sporting activities, and invasive procedures such as knee arthroplasty do not meet their expectations for continued highly active lifestyles; additionally, patients may be unwilling to deal with subsequent revision arthroplasty procedures. 50 These patients are often <60 years, with early- to midstage knee osteoarthritis (Kellgren-Lawrence grades 1-3). 50
Knee realignment procedures may allow for arthroplasty to be avoided or delayed while preserving native joint mechanics. 49 The most common procedure is an HTO to offload the affected medial compartment. 49 The procedure alters the mechanical axis of the lower limb to offload the arthritic medial compartment and relatively increase the load on the unaffected lateral compartment, thereby reducing pain and improving function. 62 Key indications for HTO are patients with significant varus malalignment with medial-side knee osteoarthritis (KOA) and younger, more active patients. 58
The 2 most common techniques of HTO include a medial opening-wedge HTO (MOWHTO) and a lateral closing-wedge HTO. 54 MOWHTO offers several additional advantages—it is an easier surgical technique with less risk of neurovascular injury and offers the possibility of performing biplanar corrections, which is vital to restoring knee stability.80,89 Thus, MOWHTO has now become an established and popular operative procedure for the correction of varus deformity of the knee. To reflect the current landscape in relation to performing HTOs, the present study focuses on MOWHTOs only.
Despite its popularity, clinical results of HTO have been shown to deteriorate over time despite initial satisfactory results, requiring patients to eventually undergo conversion to TKA. 81 Moreover, the survival rate reported across several studies is highly variable, with reported 5-year survival of HTO ranging from 71% to 95% and 10-year survival ranging from 51% to 98%. 99 This causes an inherent degree of confusion and uncertainty for patients in selecting their choice of procedure. Therefore, the need for pooled, surmised rates of TKA conversion is evident. The estimates provided in this study aim to aid the surgeon-patient conversation and decision making through the use of a time-based, comprehensive meta-analysis of conversion to TKA.
While a 2012 Finnish registry study reported survivorship to be 89% at 5 years and 73% at 10 years when taking conversion to TKA as an end point, 69 evidence monitoring outcomes and survivorship of HTO remains scarce and is potentially outdated. 62 Most current reviews report on clinical, functional, or biomechanical outcomes after HTO 13 ; however, survivorship as a whole and at specific time periods during postoperative follow-up remains largely uninvestigated. Thus, the primary aim of this systematic review and meta-analysis was to determine survivorship of MOWHTO performed for medial compartment KOA at various postoperative time points and to report any associated complications. Secondarily, we investigated the effect of concomitant cartilage repair procedures on outcomes. It was hypothesized that survivorship of MOWHTO would be high but would decrease with time and that concomitant cartilage repair would improve survivorship.
Methods
Data Sources and Search Strategy
This study was conducted in adherence with PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. 67 The study protocol was registered with the PROSPERO international prospective register of systematic reviews. We searched electronic databases MEDLINE, Embase, Cochrane Library, and SCOPUS from inception, on July 18, 2023, for relevant studies. Utilizing Medical Subject Headings (MeSH) and title abstract (tiab), our search terms were as follows: ((KOA [tiab] OR Knee Osteoarth*[tiab] OR Knee Arth* [tiab] OR Knee Degenerat* [tiab]) AND (Knee [MeSH] OR Knee Joint [MeSH])) AND (High Tibial Osteotomy [tiab] OR HTO [tiab] OR Proximal Tibial Osteotomy [tiab] OR Tibial Plateau Osteotomy [tiab] OR Tibial Realignment [tiab] OR Medial Opening Wedge Osteotomy [tiab] OR Osteotomy [MeSH]). We limited our search to only articles written in English. Our full search strategy is found in the Supplementary Material (available separately).
Study Selection
Studies reporting on survival outcomes and associated complications in patients undergoing MOWHTO with or without concomitant cartilage repair procedures for medial compartment KOA were included. Study selection criteria were based of a priori relating to study population, intervention, outcomes measured, and study design (Table 1). Studies were included when they reported outcomes at specific time points postoperatively or at a mean follow-up rather than reporting a time-to-event survival analysis.
Inclusion and Exclusion Criteria for Studies a

HTO, high tibial osteotomy; PEEK, polyether ether ketone.
A patient’s having undergone prior knee procedures before HTO was not a criterion for exclusion. HTOs that were performed with external fixation devices (eg, Taylor Spatial Frame, dynamic axial fixator, Ilizarov fixator) were excluded in our study, since external fixation is a separate technique from the traditional internal fixation method, with possible variations in postoperative management (eg, weightbearing protocols, physical therapy regimen, and monitoring) and documented increased rates of pin-site infections. 94 Hence, to maintain consistency and generalizability of our results, we focused on traditional plate-and-screw HTOs.
The inclusion of articles was evaluated by 3 independent blinded authors (R.W.K.L., Y.K.C., and Y.H.L.), with any disagreements being resolved by obtaining consensus of the senior authors (B.W.LT. and J.H.P.H.).
Risk of Bias and Quality Assessment
Working independently, the same 3 blinded researchers assessed the risk of bias of the included studies. This was performed using the Cochrane risk-of-bias tool for randomized controlled trials and the ROBINS-I (Risk Of Bias In Non-randomized Studies–of Interventions) tool for nonrandomized observational studies. 85 For studies found to have moderate or serious risk of bias, sensitivity analyses were performed to ascertain the robustness of the extracted data. Studies with critical risk of bias were excluded.
Data Extraction and Outcomes
Data were extracted by the same 3 researchers independently, with discrepancies resolved by the 2 senior authors. For each included study, the following characteristics were recorded: first author, publication year, study design, level of evidence, sample size, mean patient age, sex, body mass index (BMI), follow-up duration, osteoarthritis grading, any concomitant cartilage procedure, and angle of correction. Primary outcomes considered were survival outcomes (conversion to TKA due to progression of KOA or persistence of symptoms at various mean time points postoperatively, and time to TKA). As secondary outcomes, overall complication rates, other specific complications, and time to return to work were noted when reported.
Means and standard deviations were extracted for the pooling of continuous outcome data. When means and standard deviations were unavailable and data were instead presented as medians with ranges, we derived the means and standard deviations in accordance with the method indicated by Wan and colleagues. 95 Binary outcome data were extracted in the form of number of events occurring per sample size.
Statistical Analysis
Statistical analyses were performed using RStudio (Version 2022.12.0+353). We performed a single-arm random-effects meta-analysis to synthesize observational data for continuous and binary outcomes using the respective metamean and metaprop functions of the R meta package. Continuous baseline outcome data were pooled using the weighted mean approach with random effects, and the DerSimonian and Laird estimator was applied for between-study variance. Single-arm meta-analyses of proportions were conducted for primary and secondary outcomes using random-effects modeling. The lower and upper confidence limits of the 95% CIs were estimated using the Clopper-Pearson method, with the DerSimonian and Laird estimator applied for between-study variance. The P value was calculated directly based on the estimated proportions and their standard errors using the Z test.
We assessed statistical heterogeneity by visual inspection of forest plots, I2, and τ2. I2 values of 25%, 50%, and 75% were thresholds for low, moderate, or high heterogeneity, respectively. Given the single-arm nature of our study, we expect greater heterogeneity observed. To assess heterogeneity more comprehensively, we performed prespecified subgroup analyses according to study design (prospective or retrospective) and risk of bias (low, moderate, or serious). Further sensitivity analysis was performed on studies with moderate or serious risk of bias.
Subgrouping was performed within studies reporting on HTO with concomitant cartilage procedures to assess the impact of additional cartilage repair on desired outcomes. Pooled analysis of conversion to TKA was categorized as occurring at 3 different time frames: <5 years, 5 to 10 years, or >10 years postoperatively. Additionally, TKA conversion was analyzed by age group (<40, 40-49, 50-59, or 60-69 years) to assess the effect of age as a factor influencing survival, given most HTO patients are highly active in sports. 50
Publication bias was assessed by visual inspection of the funnel plot of primary outcomes.
Results
Our search strategy yielded 2713 articles, of which 895 duplicate records were removed, leaving 1818 records for screening. We excluded 1608 studies based on study title and abstract, leaving 210 full-text articles for full-text review. Of these 210 articles, 151 were excluded for the following reasons: biomechanical study (n = 3), review (n = 3), non-English article (n = 3), surgical technique article (n = 2), inappropriate outcomes per inclusion criteria (n = 49), inappropriate procedure/technique per inclusion criteria (n = 58), and full text unavailable (n = 33). Thus, 59 articles fit the inclusion criteria for this review (Figure 1). A summary of the characteristics of each included study is available in Appendix Table A1.

PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) diagram of the study selection process.
Of the included articles, 35 had a retrospective study design and 25 had a prospective study design, with 4 randomized controlled trials.19,26,27,32 The majority of the studies were classified as having a low risk of bias. Only 2 studies44,77 were deemed to have moderate risk of bias and hence underwent further sensitivity analysis before inclusion in the review, where it was deemed that the baseline characteristics and outcomes were comparable with those of other studies. A summary of the risk-of-bias and sensitivity assessments can be found in the Supplementary Material (Tables S1 and S2).
The funnel plot for publication bias is shown in Figure 2. Visual inspection of the symmetrical funnel plot indicated that no publication bias was present.
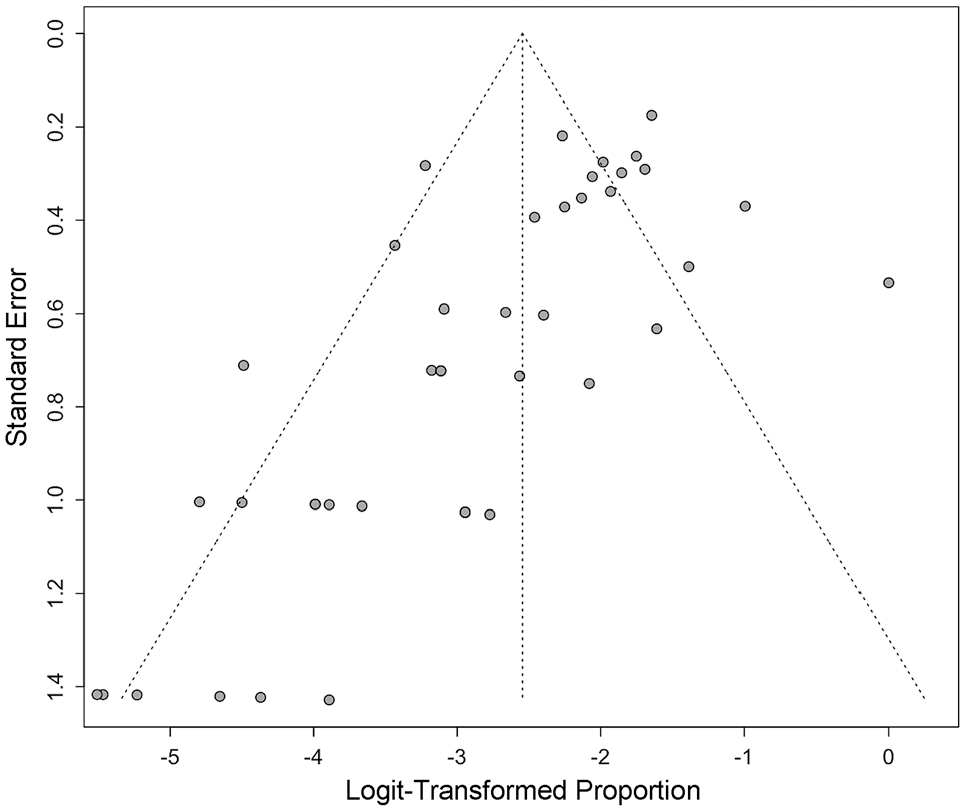
Funnel plot for publication bias.
Patient Characteristics
Overall, 5162 patients (5478 knees) underwent either primary HTO or HTO with concomitant cartilage repair procedures; these procedures included mesenchymal stem cell (MSC) augmentation, osteochondral allograft transplantation (OCA), microfracture (MFX), abrasion arthroplasty, and autologous chondrocyte implantation (ACI). The weighted mean age of the patients was 50.4 years (95% CI, 49.9-51.0), and the weighted mean BMI was 27.3 kg/m2 (95% CI, 26.8 kg/m2-27.8 kg/m2). Male patients constituted 54.8% of the cohort. The weighted mean follow-up duration was 68.0 months (95% CI, 60.2-75.8 months). Table 2 summarizes the characteristics of the patients in the included studies and the cartilage repair procedures.
Overall Patient Characteristics a

Data are presented as mean (95% CI) unless otherwise indicated. ACI, autologous chondrocyte implantation; BMI, body mass index; HTO, high tibial osteotomy; MFX, microfracture; MSC, mesenchymal stem cell; OCA, osteochondral allograft transplantation.
There were 4647 patients who underwent isolated HTO (isolated HTO group), with a weighted mean age of 50.8 years (95% CI, 50.2-51.4 years) and a weighted BMI of 27.1 kg/m2 (95% CI, 26.3 kg/m2-28.0 kg/m2). Male patients constituted 52.5% of this cohort. The weighted mean follow-up duration was 65.9 months (95% CI, 57.4-74.4 months). Patients who underwent HTO with concomitant cartilage repair procedures numbered 515 (concomitant repair group); their weighted mean age was 46.8 years (95% CI, 41.8-51.8 years) and weighted mean BMI was 27.1 kg/m2 (95% CI, 26.3 kg/m2-28.0 kg/m2); 76.3% were male. The weighted mean follow-up was 79.1 months (47.7-110.5 months).
Survivorship Outcomes
Conversion to TKA
There were 32 studies § (2840 patients) that reported conversion to TKA with mean follow-up times. When categorized according to follow-up time frame, the rates of conversion to TKA were 4.52% at <5 years (95% CI, 2.29%-8.73% years), 8.32% at 5 to 10 years (95% CI, 5.67%-12.04% years) and 11.21% at >10 years (95% CI, 8.76%-14.23% years) (Figure 3).

Forest plots of conversion to total knee arthroplasty at (A) <5 years, (B) 5-10 years, and (C) >10 years.
All patients in the concomitant repair group underwent conversion to TKA at either <5 years or 5 to 10 years postoperatively. Comparison between the isolated HTO and concomitant repair groups indicated no significant difference in survival rates at either time frame (<5 years: relative risk [RR], 0.78 [95% CI, 0.45-1.33]; P = .36; 5-10 years: RR, 0.76 [95% CI, 0.32-1.83]; P = .55) (Figure 4).
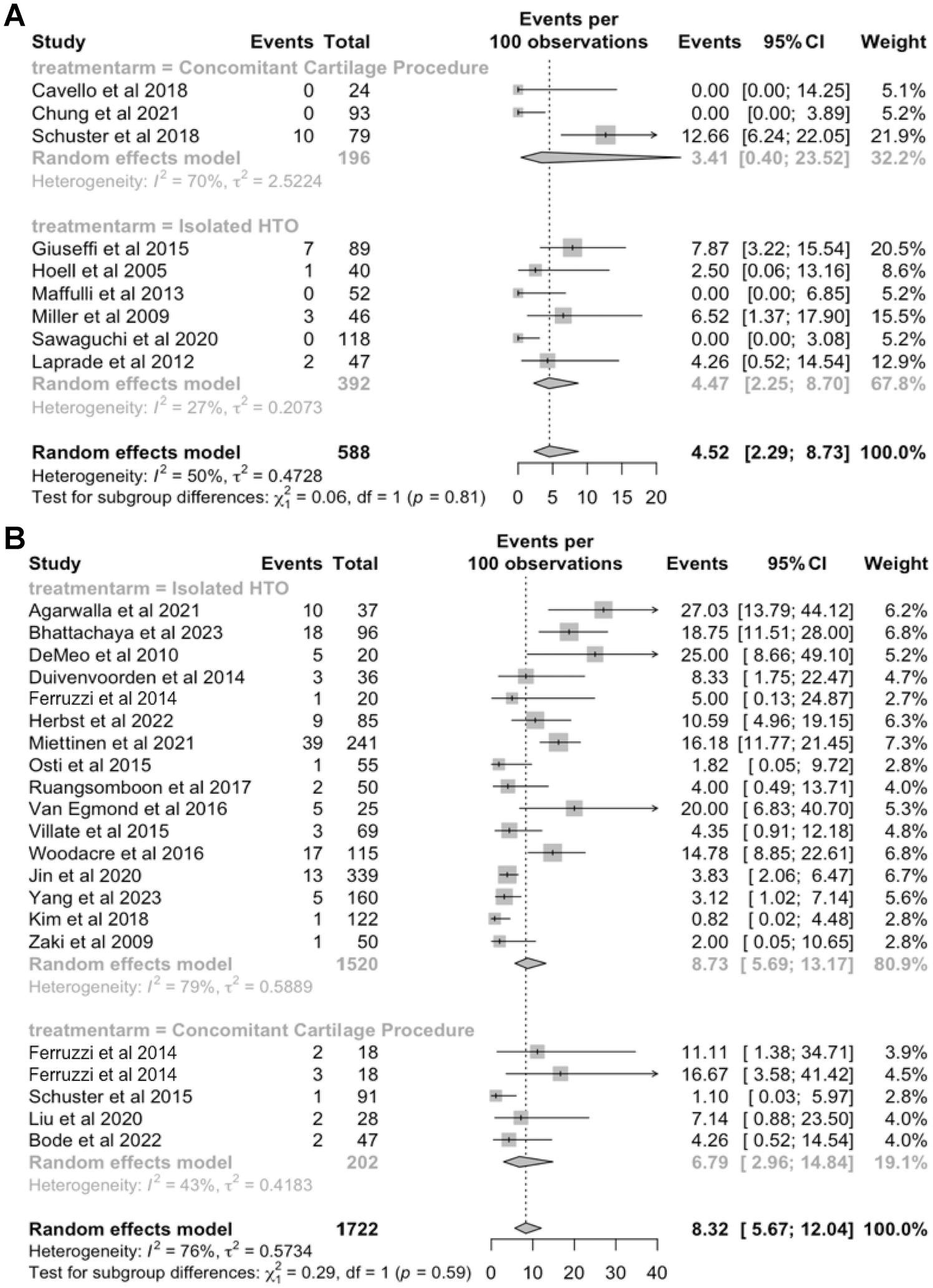
Forest plots of conversion to total knee arthroplasty after high tibial osteotomy (HTO) and HTO with concomitant cartilage repair at (A) <5 years and (B) 5-10 years postoperatively.
When studies were further subgrouped based on the reported mean age of patients (age groups <40, 40-49, 50-59, or 60-69 years), the rates of conversion to TKA were 5.2% (95% CI, 2.8%-9.4%) for age group <40 years, followed by 14.2% (95% CI, 10.8%-18.4%) for 40 to 49 years, 6.7% (95% CI, 4.7%-9.6%) for 50 to 59 years, and 2.3% (95% CI, 1.0%-5.4%) for 60 to 69 years (Figure 5).
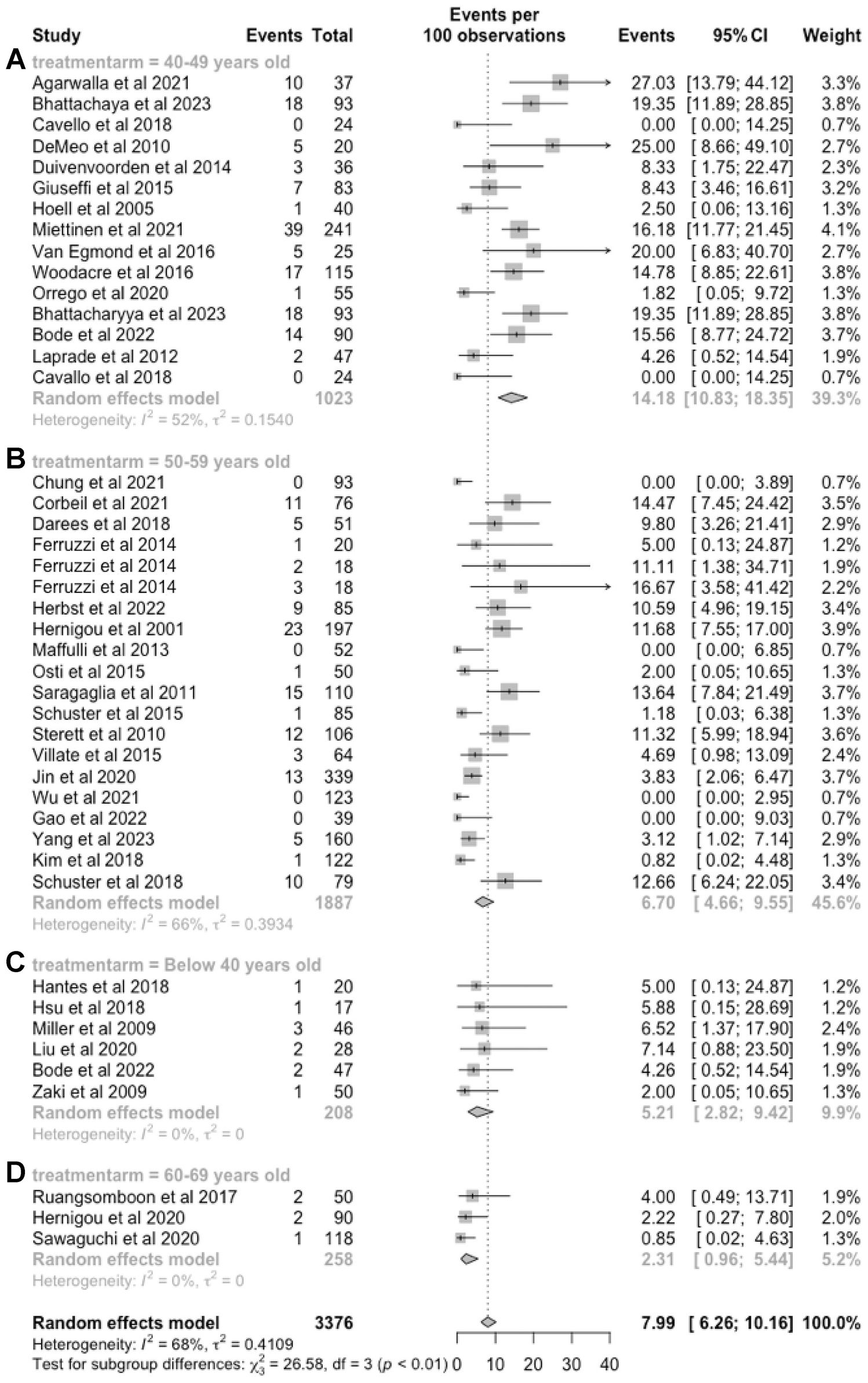
Forest plots of conversion to total knee arthroplasty after high tibial osteotomy according to age group: (A) 40-49 years, (B) 50-59 years, (C) <40 years, and (D) 60-69 years.
Time to TKA
For patients who underwent conversion to TKA, the overall pooled mean time to TKA was 5.80 years (95% CI, 4.49-7.11 years). The pooled mean time to TKA was 5.50 years (95% CI, 4.10-6.90 years) in the isolated HTO group and 7.94 years (95% CI, 5.59-10.29 years) in the concomitant repair group, with no significant group differences in time to TKA (RR, 0.69 [95% CI, 0.10-5.02]; P = .73). Heterogeneity was high in both groups (I2 = 100% for isolated HTO and 85% for concomitant repair) (Figure 6).

Forest plots of time to total knee arthroplasty for (A) isolated high tibial osteotomy (HTO) and (B) HTO with concomitant cartilage repair. MRAW, raw means.
Complications
Overall Complications
The overall pooled complication rate was 12.4% (95% CI, 9.8%-15.6%), with high heterogeneity (I2 = 87%). In the isolated HTO group, the rate was 12.83% (95% CI, 10.03%-16.27%), with high heterogeneity (I2 = 88%). In the concomitant repair group, the rate was 8.23% (95% CI, 3.40%-18.61%), with moderate heterogeneity (I2 = 61%) (Figure 7).

Forest plots of overall complication rates for (A) isolated high tibial osteotomy (HTO) and (B) HTO with concomitant cartilage repair.
Fractures, Nonunion, and Infections
There was a pooled rate of intraoperative lateral hinge fractures of 1.6% (95% CI, 1.0%-2.5%), with moderate heterogeneity (I2 = 71%). Intraoperative tibial plateau fractures had a pooled rate of 2.0% (95% CI, 1.4%-2.8%), with low heterogeneity (I2 = 43%). Postoperative fractures had a pooled rate of 0.9% (95% CI, 0.7%-1.3%). No heterogeneity was found in this group. The pooled rate of nonunion was 1.7% (95% CI, 1.3%-2.2%), with no heterogeneity. The pooled rate of superficial infection was 2.6% (95% CI, 1.9%-3.5%), with moderate heterogeneity (I2 = 53%), and the pooled rate of deep infection was 2.0% (95% CI, 1.5%-2.6%), with no heterogeneity. The related forest plots are available in the Supplemental Material (Figures S3-S8).
Return To Work
Four studies2,39,77,86 reported on patients’ time to return to work, all of which were from the isolated HTO group. The pooled time to return to work was 3.2 months (95% CI, 2.8-3.6 months). Heterogeneity was high (I2 = 97%) (Figure 8).

Forest plot for return to work after high tibial osteotomy (HTO) after isolated HTO. MRAW, raw or untransformed mean.
Discussion
The most important finding of this systematic review and meta-analysis was a reinforcement of the present consensus that HTO is an effective procedure that does provide low rates of conversion to TKA and complications; hence, it is a viable option along with UKA for medial compartment KOA. We found that 4.52% of patients underwent conversion to TKA within the first 5 years after HTO; this increased to 8.32% within 5 to 10 years and 11.21% after 10 years. Subgroup analyses for patients who underwent HTO with concomitant cartilage repair procedures was performed, and results reflected similar, nonsignificant rates of conversion to TKA within the <5 year and 5- to 10-year follow-up time frames, with no studies reporting patients who converted to TKA at >10 years. This main finding of survival rates decreasing with time is to be expected. The majority of patients opting for HTO are younger and more active, resulting in increased wear on an already osteoarthritic knee joint, accelerating osteoarthritis progression. Given that the medial compartment experiences 60% to 80% of the weightbearing load, 93 loss of correction and return of varus malalignment and thus osteoarthritis symptoms are inevitable. Having only approximately 1 in 10 HTOs convert to HTO after >10 years postoperatively lends support to the procedure’s effectiveness at delaying or postponing the need for arthroplasty. A previous meta-analysis by Kim et al 51 reported a 95.1% survival rate (95% CI, 93.1%-97.1%) at 5 years postoperatively, which is comparable with that of the current study.
Other options available to patients with medial compartment KOA are a TKA or UKA. Consensus has yet to be determined whether an HTO or a UKA is preferable for this group of patients. A registry study by Di Martino et al 18 found a 92.6% survival rate for UKA, slightly lower but still comparable with the HTO survival rate in the present study at 5 years postoperatively. On the other hand, a propensity score–matched study by Jin et al 45 found that UKA had superior survival rates at 10-year follow up. A meta-analysis by Huang et al 41 found no significant differences between HTO and UKA with respect to conversion to TKA. The current literature does not definitively favor one over the other, and so the choice should take into consideration patient preferences and lifestyle factors. Huang et al and Santoso and Wu 74 suggested that UKA was more appropriate for older patients, while HTO provided a better performance of physical activity for younger patients, due to a shorter rehabilitation period and quicker functional recovery. Finally, a TKA is known to have the highest survival of all options; however, not only are patients aged ≤55 years more likely to outlive their prosthesis and require revision surgery, but their higher activity level predisposes the implant to early failure. 10 Hence, a TKA may not be such a suitable alternative in this patient group.
Given that HTOs are commonly performed in a relatively younger population, it was important to assess the survival of HTOs across different age groups. This is a vital clinical point that will aid further personalizing of the surgeon-patient conversation. Interestingly, we found with the exception of patients who underwent HTO before the age of 40 years, the HTO survival was proportional to age: the highest rate of conversion to TKA was in the 40 to 49 year group, with rates of 14%, approximately double that of the 50 to 59 year group. The 60 to 69 year group saw lower rates of conversion at 2.3%. Indeed, this is likely due to progression of medial compartment osteoarthritis in the more physically active, younger demographic (older patients are documented to have reduced physical activity 64 ), which places more strain on the arthritic knee. Presently, the literature is inconclusive on the effect of age on HTO survival; the registry study by Niinimäki et al 69 suggested that older age indicated worse HTO survivorship, and other studies have deemed younger age as a predictor of HTO survival.42,48 However, it is worth noting that the latter studies defined failure as also including revision HTO or looked at early conversion to TKA—these are more indicative of patients’ ability to achieve quick and full postoperative recovery, or complications of HTO, rather than the potential of limiting the progression of osteoarthritic changes that our study sought to highlight. Taken together, we believe our data yield an important adjunctive point in clinical discussion with patients, to aid reiteration that a joint-preserving surgery such as MOWHTO is not a substitute for joint replacement, but rather delays its necessity.
For patients with poor outcomes or progression of osteoarthritis, the time to TKA reported in the present study was comparable with those in previous studies, such as 8.1 years by Abdelaal et al 1 and 7.3 years by Haddad and Bentley. 31
With regard to complications of HTO, the prominent ones related to the HTO procedure are intraoperative fractures (particularly of the lateral cortex), nonunion, and postoperative infections. Our study found that the rates of occurrence of these complications were all extremely low, consistent with previous findings; Miltenberg et al 66 in their 2024 systematic review found a 1 in 11 chance of lateral cortex fracture intraoperatively, along with 1.9% risk of nonunion and 2.9% risk of infection. Even the overall complication rates found by our study—between 10% and 15%—is within what is known and currently accepted in the orthopaedic community in relation to HTOs. 66
Evidence has emerged that concomitant cartilage repair procedures may be effective in symptomatic pain relief.16,25,47,53,61,100 Yet, other studies have also shown that degenerated cartilage could be completely regenerated in just 2 years after isolated HTO. 46 Hence, in practice there is no established consensus on the benefits of these procedures on long-term outcomes. Our study included studies that combined HTO with ACI, MSC, OCA, MFX, or abrasion arthroplasty and found that on the whole, concomitant cartilage procedures did not significantly improve survival outcomes. A previous systematic review of 839 knees found that concomitant procedures delayed conversion to UKA or TKA and reduced odds of failure; however, that review only included HTOs combined with either ACI or OCA. 15 Another review by Lee et al 57 suggested that concomitant procedures (included were ACI, MFX, abrasion arthroplasty, and MSC) had little benefit on clinical outcomes. This discrepancy among studies suggests that different concomitant procedures have varying degrees of benefit. HTO with ACI has consistently shown benefit in outcomes across several studies,5,57,72 and MSC augmentation for HTO was suggested in a meta-analysis by Tan et al 87 to modestly improve functional outcomes, although the impact on survival outcomes was not quantified. There are few studies comparing HTO with OCA to isolated HTO, so no conclusions can be drawn at present. MFX and abrasion arthroplasty, on the other hand, have been shown to have no significant improvement on HTO survival or clinical outcomes.3,58,96,100 Although studies on survival outcomes relating to HTO with concomitant cartilage repair procedures are limited, the establishment of nonsignificant clinical improvement would likely correspond to nonsuperior survival outcomes, as poor clinical outcomes has been suggested to be a predictive factor for the need for conversion to TKA. 83
In our meta-analysis, 10 of the 79 patients who underwent HTO with abrasion arthroplasty converted to TKA before the >10-year follow-up time frame, and that number was 15 of 124 in patients who underwent HTO with MFX. Comparatively, only 4 of 65 patients who underwent HTO with ACI, 0 of 117 of patients with combined HTO and MSC, and 3 of 45 of patients with combined HTO and OCA converted to TKA. The single-arm nature of this meta-analysis means there was significant disparity in sample size between the isolated HTO and the individual concomitant repair groups, compromising generalizability of results should comparisons be made between the groups. Nonetheless, the raw proportions and pooled rates of conversion suggest the varying effectiveness of each individual cartilage procedure in affecting survival outcomes, and that likely is the reason for the nonsignificant general finding for rates of conversion to TKA in the overall concomitant repair group. Nonetheless, our study underscores the need for larger, well-balanced cohorts to further investigate and validate any observed trends in survival outcomes associated with different procedural variations in HTO.
Strengths and Limitations
There are several hypothesized prognostic factors in the literature that affect the survival rate after HTO, including BMI, osteoarthritis severity, age, degree of correction, and postoperative weightbearing protocol.30,55,98,100 In the present study, the methodological quality meant that we were unable to ascertain the significance of all of these prognostic factors. However, care was taken to ensure that all of the included studies had groups that were comparable at baseline in relation to these prognostic factors, the study design, and even the bias of each study. Heterogeneity in study design and bias in studies are other factors that affected the robustness of the present study, and so further subgrouping by study design and sensitivity analyses for studies that had moderate risk of bias were performed. Results indicated that the studies were comparable, and hence the studies were included in our review. Publication bias was also assessed to be low given the symmetry of funnel plots.
The current review also has several limitations. First, we were unable to reach conclusions on the effect of specific cartilage procedures on overall outcomes, and the associations between the isolated HTO and HTO with concomitant cartilage repair procedure group should be interpreted with caution given the relatively smaller sample size in the latter group compared with the former. Second, there was potential heterogeneity across studies. High or moderate I2 values were observed across all outcomes due to inherent properties of I2 values in single-arm meta-analysis, making it difficult to quantify between-study heterogeneity. Last, we were unable to confirm the significance of hypothesized prognostic factors on survival outcomes. This was due to the similarity in patient demographics within the studies. Although this prevented us from ascertaining prognostic factors, it helped us ascertain that those are not sources of confounding and thus assess heterogeneity more comprehensively.
Conclusion
Rates of conversion to TKA and complications were found to be low and acceptable, although survival rates decreased with time. Concomitant cartilage procedures as a whole did not significantly improve survivorship; however, more high-quality studies are warranted to determine the impact of individual concomitant cartilage repair procedures, given that the sample sizes of individual, specific cartilage repair types were too small for meaningful comparison. The provision of survivorship of HTOs at various postoperative time points gives both clinicians and patients important discussion points in selection of surgical options for KOA in younger populations, while answering the question on effectiveness of concomitant cartilage repair procedures, which is becoming increasingly popular, albeit with limited literature.
Supplemental Material
sj-pdf-1-ojs-10.1177_23259671241310963 – Supplemental material for Conversion to Total Knee Arthroplasty After High Tibial Osteotomy: A Systematic Review and Meta-analysis
Supplemental material, sj-pdf-1-ojs-10.1177_23259671241310963 for Conversion to Total Knee Arthroplasty After High Tibial Osteotomy: A Systematic Review and Meta-analysis by Ryan Wai Keong Loke, Yang Kai Chan, Yao Hui Lim, Barry Wei Loong Tan and James Hoi Po Hui in Orthopaedic Journal of Sports Medicine
Footnotes
Appendix
Characteristics of the Included Studies a

| Lead Author and Year | Study Design | Isolated HTO or HTO With Cartilage Repair | No. of Patients (Knees) | Male, n | Age, y b | BMI, kg/m2 b | K-L Grade | Degrees of Correction b | Follow-up, mo b |
|---|---|---|---|---|---|---|---|---|---|
| Agarwalla 2 2021 | Retrosp | Isolated HTO | 37 (37) | 31 | 43.4 ± 7.8 | 27.3 ± 3.7 | 3 Gr 1 or 2; 34 Gr 3 or 4 | 9.9 ± 3.3 | 108 ± 39.6 |
| Asik 4 2006 | Retrosp | Isolated HTO | 60 (65) | 13 | 54 ± 9.25 | NR | NR | NR | 34 ± 10.5 |
| Bhattacharyya 6 2023 | Retrosp | Isolated HTO | 93 (96) | 71 | 47.5 ± 5 | NR | NR | NR | 107.4 ± 42 |
| Birmingham 7 2009 | Prosp | Isolated HTO | 128 (128) | 102 | 47.48 ± 9.53 | 29.5 ± 4.82 | 4 Gr 1; 32 Gr 2; 41 Gr 3; 51 Gr 4 | NR | 24 ± 0 |
| Bode 8 2022 | Prosp | (1) Isolated HTO (2) HTO + ACI |
(1) 90 (90) (2) 47 (47) |
(1) 59 (2) 29 |
(1) 46.64 ± 9.87 (2) 39.53 ± 8.76 |
(1) 27.75 ± 4.64 (2) 25.77 ± 3.62 |
(1) 1.99 ± 0.62
c
(2) NR |
(1) 9.07 ± 4.49 (2) 7.42 ± 4.04 |
(1) 120.8 ± 28.02 (2) 113.29 ± 23.73 |
| Brosset 9 2011 | Prosp | Isolated HTO | 51 (51) | 36 | 53 ± 7.5 | — | NR | 9 ± 4.36 | 24 ± 0.75 |
| Cavallo 11 2018 | Prosp | HTO + MSC | 24 (24) | 15 | 47.9 ± 12.3 | NR | NR | NR | 44.4 ± 17.7 |
| Chung 12 2021 | Retrosp | HTO + MSC | 93 (93) | NR | 56.6 ± 5.5 | 25.8 ± 3.08 | All Gr 3 | NR | 20.4 ± 7.5 |
| Corbeil 14 2021 | Retrosp | Isolated HTO | 76 (84) | 54 | 50 ± 10.5 | 30.2 ± 5.6 | NR | NR | NR |
| Darees 15 2018 | Retrosp | Isolated HTO | 51 (51) | 33 | 53 ± 7.5 | 28.9 ± 3.5 | NR | 9 ± 4.36 | 122.4 ± 5 |
| DeMeo 17 2010 | Prosp | Isolated HTO | 20 (20) | 14 | 49.4 ± 7.75 | NR | NR | 11.1 ± 2.75 | 99.6 ± 11.25 |
| Duivenvoorden 19 2014 | Prosp RCT | Isolated HTO | 36 (36) | 24 | 49.9 ± 7.9 | 27.3 ± 5.4 | — | 11.6 ± 3.4 | 87.6 ± 13.2 |
| Duivenvoorden 20 2017 | Retrosp | Isolated HTO | 112 (112) | 73 | 48.7 ± 10.1 | 28.5 ± 4.5 | NR | — | 88.8 ± 38.4 |
| El-Assal 21 2010 | Prosp | Isolated HTO | 58 (58) | 21 | 47.5 ± 10.25 | 28.5 ± 0.1 | NR | 10.7 ± 3.7 | 38 ± 0.1 |
| Esenkaya 22 2007 | Retrosp | Isolated HTO | 56 (58) | 9 | 52 ± 7.5 | NR | NR | NR | 21 ± 9.5 |
| Ferruzzi 23 2014 | Retrosp | (1) Isolated HTO (2) HTO + ACI (3) HTO + MFX |
(1) 20 (20) (2) 18 (18) (3) 18 (18) |
(1) 10 (2) 12 (3) 13 |
(1) 54 (2) 51 (3) 53 |
(1) 26 ± 4 (2) 28 ± 3 (3) 31 ± 5 |
(1) 12 Gr 3; 8 Gr 4 (2) 12 Gr 3; 6 Gr 4 (3) 10 Gr 3; 6 Gr 4 |
NR | (1) 106 ± 7.75 (2) 96 ± 5.5 (3) 92 ± 3.75 |
| Floerkemeier 24 2013 | Retrosp | Isolated HTO | 533 (533) | 367 | 49.3 ± 16.5 | 27.2 ± 4 | NR | NR | 43.2 ± 6.9 |
| Gaasbeek 26 2010 | Prosp RCT | Isolated HTO | 25 (25) | 14 | 47 ± 8.5 | 29.7 ± 4.2 | NR | 8.1 ± 4.4 | 12 ± 0 |
| Gao 27 2022 | Prosp RCT | Isolated HTO | 39 (39) | 16 | 57.84 ± 7.83 | 24.58 ± 3.08 | 1 Gr 1; 4 Gr 2; 11 Gr 3 | 9.02 ± 1.64 | NR |
| Giuseffi 28 2015 | Retrosp | Isolated HTO | 83 (89) | 62 | 48.1 ± 0.1 | NR | All Gr ≤3 | 9.6 | 48 ± 27 |
| Goshima 29 2015 | Retrosp | Isolated HTO | 60 (60) | 23 | 61.6 ± 8.6 | 24.7 ± 2.5 | 4 Gr 1; 23 Gr 2; 22 Gr 3; 5 Gr 4 | — | 51.1 ± 21.9 |
| Haghpanah 32 2021 | Prosp RCT | Isolated HTO | 46 (46) | 24 | 25.55 ± 7.44 | 24.2 ± 1.99 | NR | NR | 96 ± 0 |
| Han 33 2019 | Retrosp | Isolated HTO | 209 (209) | 29 | 56.4 ± 5.9 | 26.5 ± 3.2 | NR | NR | NR ± NR |
| Hantes 34 2018 | Retrosp | Isolated HTO | 20 (20) | 18 | 35.4 ± 4 | NR | NR | 8.3 ± 4.3 | 147.6 ± 15 |
| Herbst 35 2022 | Prosp | Isolated HTO | 85 (85) | 60 | 54.6 ± 6.4 | 28.6 ± 4.6 | NR | 6.9 ± 3.2 | 72 ± 0 |
| Hernigou 36 2001 | Retrosp | Isolated HTO | 197 (245) | 78 | 59 ± 9.5 | 0 ± 0 | NR | — | 120 ± 2.25 |
| Hernigou 37 2013 | Retrosp | Isolated HTO | 170 (170) | 76 | 62.9 ± 7.5 | 23.9 ± 3.8 | NR | 13.6 ± 4.37 | 12 ± 0 |
| Hernigou 38 2020 | Retrosp | Isolated HTO | 90 (180) | NR | 61.7 ± 4.67 | 28 ± 3.33 | NR | NR | NR |
| Hoell 39 2005 | Retrosp | Isolated HTO | 40 (40) | 40 | 46.4 ± 8 | 30 ± 5.2 | NR | NR | 22.5 ± 6.125 |
| Hsu 40 2018 | Retrosp | HTO + OCA | 17 (17) | 15 | 36.5 ± 11.3 | 26.1 ± 3 | NR | NR | NR |
| Iacono 43 2020 | Prosp | Isolated HTO | 75 (75) | 46 | 45.7 ± 0.01 | NR | NR | NR | 8 ± 0 |
| Jin 44 2020 | Retrosp | Isolated HTO | 339 (339) | 91 | 56 ± 7.25 | 25.3 ± 5.88 | 67 Gr 1; 151 Gr 2; 109 Gr 3; 12 Gr 4 | NR | 115.2 ± 96 |
| Kim 52 2018 | Retrosp | Isolated HTO | 122 (122) | 14 | 55.96 ± 4.99 | 25.89 ± 2.6 | 60 Gr 2; 49 Gr 3; 13 Gr 4 | NR | 90 ± 23.4 |
| LaPrade 56 2012 | Prosp | Isolated HTO | 47 (47) | 32 | 40.5 ± 8 | 28.3 ± 7.05 | All Gr 1-2 | NR | 43.2 ± 20.7 |
| Liu 59 2020 | Retrosp | HTO + OCA | 28 (28) | 22 | 28.97 ± 7.52 | 27.92 ± 3.87 | 25 Gr 1 or 2; 3 Gr 3 or 4 | 8.48 ± 2.24 | 79.56 ± 48.72 |
| Maffulli 60 2013 | Prosp | Isolated HTO | 52 (52) | 36 | 58.75 ± 6.6 | 29.9 ± 1.8 | NR | 12.05 ± 3.5 | 41 ± 6.6 |
| Miettinen 63 2021 | Retrosp | Isolated HTO | 241 (241) | 208 | 48.84 ± 8.18 | 29.9 ± 5.2 | 51 Gr 1; 149 Gr 2; 41 Gr 3 | 8.30 ± 3.68 | 72 ± 3 |
| Miller 65 2009 | Retrosp | Isolated HTO | 46 (46) | 34 | 38.2 ± 11.75 | NR | NR | NR | 12 ± 0 |
| Nelissen 68 2010 | Retrosp | Isolated HTO | 45 (49) | 29 | 48 ± 19.9 | NR | NR | NR | NR ± NR |
| Orrego 70 2020 | Retrosp | Isolated HTO | 55 (55) | 37 | 43.25 ± 4.75 | NR | NR | NR | NR |
| Osti 71 2015 | Retrosp | Isolated HTO | 50 (55) | 33 | 54.7 ± 12.6 | 26.8 ± 3.6 | NR | 3.8 ± 3.3 | 60 ± 16.8 |
| Ruangsomboon 73 2017 | Retrosp | Isolated HTO | 50 (50) | 17 | 66 ± 5 | 25.9 ± 2.8 | 15 Gr 2; 35 Gr 3 | 10.6 ± 3.6 | 72 ± 36 |
| Saragaglia 75 2011 | Prosp | Isolated HTO | 110 (124) | 74 | 53.23 ± 10.68 | 27.1 ± 4.37 | NR | NR | 124.68 ± 23.76 |
| Sawaguchi 76 2020 | Prosp | Isolated HTO | 118 (118) | 19 | 64.7 ± 8.4 | 24.6 ± 2.8 | 29 Gr 1; 47 Gr 2; 34 Gr 3; 7 Gr 4 | NR | 24 ± 0 |
| Schröter 77 2013 | Prosp | Isolated HTO | 32 (32) | 22 | 47 ± 9 | 28.6 ± 4.7 | NR | 5.7 ± 3.6 | 77 ± 19 |
| Schuster 79 2015 | Prosp | HTO + abrasion/MFX | 85 (91) | 79 | 50.4 ± 8 | 27.4 ± 3.1 | All Gr 3 or 4 | 10.2 ± 5.3 | 67.2 ± 27.6 |
| Schuster 78 2018 | Prosp | HTO + AA | 79 (79) | 67 | 50.9 ± 7.6 | 27.1 ± 3 | NR | NR | 120 ± 14.4 |
| Song 81 2010 | Retrosp | Isolated HTO | 90 (90) | 21 | 51 ± 6.5 | 25.5 ± 2.18 | All Gr 1-3 | 26.7 ± 9 | |
| Spahn 82 2004 | Prosp | Isolated HTO | 85 (85) | 49 | 44.2 ± 11.6 | NR | NR | 9.66 ± 2.12 | NR |
| Sterett 84 2010 | Retrosp | HTO + MFX | 106 (106) | 80 | 52 ± 10.25 | NR | NR | NR | NR |
| Tabrizi 86 2013 | Prosp | Isolated HTO | 16 (16) | 13 | 36.5 ± 8.1 | NR | NR | NR | NR |
| Türkmen 88 2014 | Prosp | Isolated HTO | 38 (41) | 3 | NR | NR | NR | NR | 6 ± 0 |
| van Egmond 90 2016 | Prosp | Isolated HTO | 25 (25) | 15 | 47.1 ± 8.5 | 29.7 ± 4.2 | NR | 7.8 ± 2.6 | 94.8 ± 6 |
| van Wulfften Palthe 91 2018 | Retrosp | Isolated HTO | 14 (14) | — | — | — | — | — | — |
| Villate 92 2015 | Retrosp | Isolated HTO | 64 (69) | 43 | 51.8 ± 10.75 | 27.2 ± 6.4 | NR | NR | 90 ± 12.6 |
| Woodacre 97 2016 | Retrosp | Isolated HTO | 115 (115) | NR | 47 ± 7.5 | 29.1 ± 4.98 | NR | NR | 100.8 ± 42 |
| Wu 99 2021 | Retrosp | Isolated HTO | 123 (123) | 39 | 59.64 ± 6.64 | 27.9 ± 3.76 | 88 Gr 3; 35 Gr 4 | NR | NR |
| Yang 101 2023 | Retrosp | Isolated HTO | 160 (160) | 44 | 55.75 ± 5.45 | 26.15 ± 3.41 | 61 Gr 2; 97 Gr 3; 2 Gr 4 | NR | 96 ± 0.1 |
| Zaki 102 2009 | Prosp | Isolated HTO | 50 (50) | 50 | 39.5 ± 4.75 | NR | NR | NR | 60 ± 9 |
Dashes indicate areas unable to be calculated. AA, abrasion arthroplasty; ACI, autologous chondrocyte implantation; BMI, body mass index; Gr, grade; HTO, high tibial osteotomy; K-L, Kellgren-Lawrence; MFX, microfracture; MSC, mesenchymal stem cell; NR, not reported; OCA, osteochondral allograft transplantation; Prosp, prospective; RCT, randomized controlled trial; Retrosp, retrospective.
Data are presented as mean or mean ± SD.
Mean ± SD.
Final revision submitted July 29, 2024; accepted August 27, 2024.
The authors declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
§
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
