Abstract
Background:
Matrix-induced autologous chondrocyte implantation (MACI) was developed for knee cartilage restoration involving seeding autologous chondrocytes onto a collagen membrane. Maintaining chondrocyte viability and achieving proper membrane size and contour are crucial for successful outcomes. Scissor cutting (Sc) has traditionally been used to shape the membrane, but recently, custom cutting (CC) have gained popularity for their enhanced preparation efficiency.
Purpose:
To determine the difference in chondrocyte viability using the Sc versus CC methods.
Study Design:
Controlled laboratory study.
Methods:
Using Sc and CC methods, 15 mm–diameter circular specimens were cut from 5 residual membranes. Membranes were stained, and confocal microscopy was used to visualize live/dead cells. Three zones were defined: the impact zone (the outermost zone at the cut edge), the central zone (the least manipulated zone), and the adjacent zone (the intermediate zone between the central and impact zones). For each circular specimen, the following measurements were recorded: the dimension of the impact zone, cell viability (percentage of live cells among total) for each zone, and the total number of cells within each zone (as cell density ×105/cm2).
Results:
The width of the impact zone (mean ± standard error) was 355 ± 31 μm and 342 ± 24 μm for CC and Sc, respectively, and the impact zone occupied approximately 10% of the specimen. With both cutting techniques, cell viability was significantly lower in the impact zone (mean ± standard error: CC, 36.42% ± 3.85%; Sc, 40.94% ± 2.85%) compared with the adjacent zone (CC, 77.69% ± 2.97%; Sc, 74.17% ± 2.8%). The cell density from all zones varied from 5.84 ± 0.26 to 6.49 ± 0.34 × 105/cm2, with no significant difference in cell viability in the impact zones between the cutting methods.
Conclusion:
Both cutting techniques led to a significant reduction in cell viability in the impact zone compared with other zones of the MACI specimen. There was no significant difference in chondrocyte viability or cell density for membranes cut by Sc or CC.
Clinical Relevance:
This evaluation of the newly introduced CC method on the chondrocyte viability of the MACI membrane will enable surgeons to make a more informed decision regarding cutting techniques.
Keywords
Focal chondral lesions of the knee are challenging to treat because of the articular cartilage's unique biological and histological properties, which give it limited innate regenerative potential. 9 As a result, surgical treatment is often required, with several well-established techniques to stimulate new cartilage growth and regeneration to restore the joint's normal function. 10 Lars Peterson introduced autologous chondrocyte implantation to treat knee chondral lesions in 1987. The procedure aimed to regenerate hyaline-like cartilage by injecting cultured autologous chondrocytes underneath a periosteal membrane attached to the debrided chondral lesion.2,3,6 This technique later evolved into matrix-induced autologous chondrocyte implantation (MACI). 4 This is a staged treatment in which, first, autologous chondrocytes are harvested by way of a small cartilage biopsy specimen from the patient's knee, usually from the intercondylar notch. The harvested chondrocytes are then expanded in culture, seeded on a collagen membrane, and subsequently implanted. 5
The lesion edges are debrided before placing the MACI membrane to form stable, viable, and vertical cartilage walls to accommodate the membrane implantation. Subsequently, the MACI membrane is precisely shaped to fit the contour of the defect before the actual implantation. Traditionally, preparation is done freehand, with the lesion prepared using shavers or curettes and the membrane shaped by scissor cutting (SC). More recently, custom cutting (CC) has made preparation easier and more efficient. Using predesigned or custom-sized cutting guides, the guide is malleted into the lesion, and everything within the boundary outlined by the guide is removed. The MACI membrane is then cut using the same cutting guide and thus sized to match the prepared lesion site exactly.
A significant barrier to MACI integration and successful chondral repair is cell survival, particularly in the area along the edge of the repair, which is known to carry many dead cells due to the debridement and preparation procedures.7,13 Owing to its avascular nature, the articular cartilage has a poor intrinsic reparative response to damaged tissues. 12 These dead cells and surrounding biomass become the physical barrier to forming continuous hyaline cartilage. For this reason, cells delivered to the defect edge through graft or implantation must be as viable as possible for the cartilage integration to succeed. It is known that Sc preparation of the MACI membrane significantly affects cell viability on the outmost cutting edge. 5 The effect of CC membrane preparation on cell viability is unknown to date.
This study aimed to investigate the effect of these 2 cutting techniques, Sc and CC, on chondrocyte viability and density within the MACI membrane.
Methods
Sample Collection
All protocols used in this study received institutional review board approval, and all included patients provided informed consent to participate. The MACI specimens included in this study were from residual membranes obtained from 4 female donors and 1 male donor (mean age, 27.2 years; range, 20-32 years) undergoing MACI for knee cartilage defect.
A cartilage biopsy sample was used to create the MACI membrane after the instructions from the manufacturer (Vericel Corp). After quality control tests, a 3 × 5–cm piece of the MACI membrane was delivered to the surgeon for implantation. The membranes were collected after surgery, and 3 specimens were obtained from each membrane: Sc, CC, and control (Figure 1A). The control specimen was obtained from the top corner of the membrane, which, in theory, represents the area that was least manipulated by the MACI membrane during the surgery itself. For the CC specimen, a circular-shaped custom cutter 15 mm in diameter with a thick, high-density pad underneath to support the mallet punch was used (Figure 1B). The Sc specimen was created using a pair of surgical scissors (Figure 1C) with an effort to create a similar size of circular shape. A single surgeon (S.L.S.) performed all the cutting. Immediately after the surgery, all specimens were prepared and kept in the original shipping container with transport media (Figure 1D) and were transported to the research laboratory for further processing.

(A) A schematic diagram demonstrates different cuts and imaging areas from a MACI membrane. The dotted circles indicate the locations for imaging. The photographs show the collected specimens, including (B) CC and (C) Sc. The cuts were made with the MACI membrane sitting on the top of a piece of sterile supporting sheet to avoid any additional impact from handling with fingers or forceps. The MACI membrane is outlined for better visibility. (D) CC, Sc, and control specimens are stored in the shipping container with the medium. CC, custom cutting; MACI, matrix-induced autologous chondrocyte implantation; Sc, scissor cutting.
Sample Staining
All specimens were stained with a LIVE/DEAD Viability/Cytotoxicity Kit for mammalian cells (Invitrogen) within 2 hours of collection. The live/dead assay was conducted as described by Abrams et al 1 according to the manufacturer's instructions. The staining solution was prepared by adding 2 µM calcein AM and 4 µM ethidium homodimer-1 in Dulbecco's Modified Eagle Medium supplemented with 10% fetal bovine serum, 1× antibiotic/antimycotic (Thermo Fisher Scientific). Each specimen was stained in this solution for 45 minutes at 37°C in the dark with gentle shaking, followed by 3 washes with 1× phosphate-buffered saline for 5 minutes each.
Image Collection and Analysis
For each MACI membrane, 3 specimens were investigated: Sc, CC, and control. To achieve the randomized selection of image fields, the specimen could only be moved in a horizontal direction once it was on the stage of a Leica upright DMi8 microscope equipped with a Leica STELLARIS 5 confocal laser scanning system and controlled by Leica Application Suite X software (see Supplemental Material for details). We used both ends of the horizontal line as the predefined locations for the periphery area and the center for the central area. Three consecutive images were collected from each location, and 6 images from the peripheral and central areas of each specimen (Figure 1A). Images taken of the edge of the Sc and CC were subsequently used to establish the impact and adjacent zones based on cell death staining (Figure 2B).
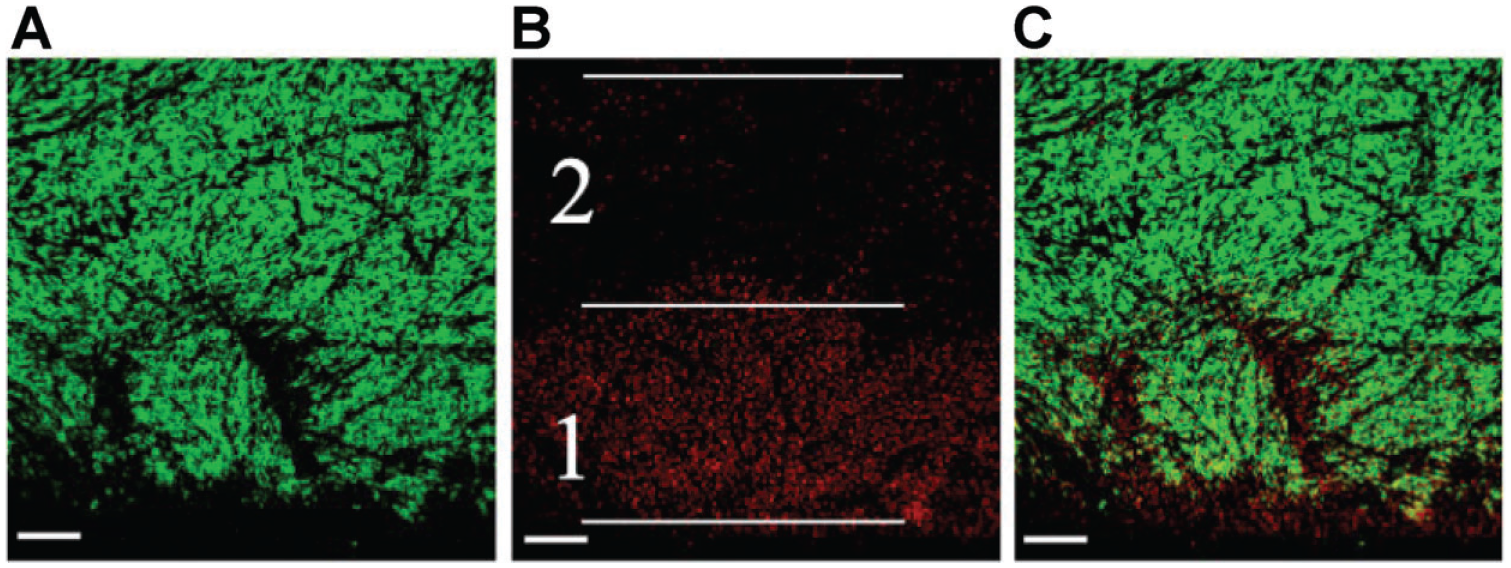
Representative results of cell staining at the peripheral location. The lower cut edge in each image serves as the demarcation for inserting the matrix-induced autologous chondrocyte implantation into the cartilage defects. (A) Live cell staining at the peripheral location. (B) Dead cell staining at the same location. The 2 white lines on either side of area 1 represented the boundaries with higher dead signals, referred to as the impact zone. Area 2, situated at the same distance from the cut edge, was defined as the adjacent zone. The central line precisely aligns with the transition between high and low density of dead cells. (C) The superimposition of the live-cell and dead-cell images. The scale bar represents 100 μm.
The live and dead cells are represented in the images by green and red colors, respectively. The area with a higher density of red signals was called the “impact zone,” and the area of identical dimension immediately adjacent to the impact zone was called the “adjacent zone” (Figure 2B). The distance spanning the boundary of dense cell death was termed the “width.” Finally, the same dimension was implemented on the center image as the “center zone.” In addition, we noticed that the width of the impact zone can be varied between 2 predefined peripheral locations of the same specimen; therefore, the average width was used to determine the size of the impact zone on specimens. The width of the impact zone (mean ± standard error) was 355 ± 31 μm and 342 ± 24 μm for CC and Sc, respectively, and the impact zone occupied approximately 10% of the specimen.
Quantification of live/green and dead/red cells was processed individually by the 3-dimensional (3D) segmentation tool from the 3DSuite 11 in ImageJ (see Supplemental Material for details). The results were presented as the percentage of live cells to total and the cell density (×105/cm2) from each zone. Furthermore, we used the 3D-reconstructed image created in ImageJ to demonstrate that the deformed edge in the impact zone was primarily filled with dead cells (Figure 3).
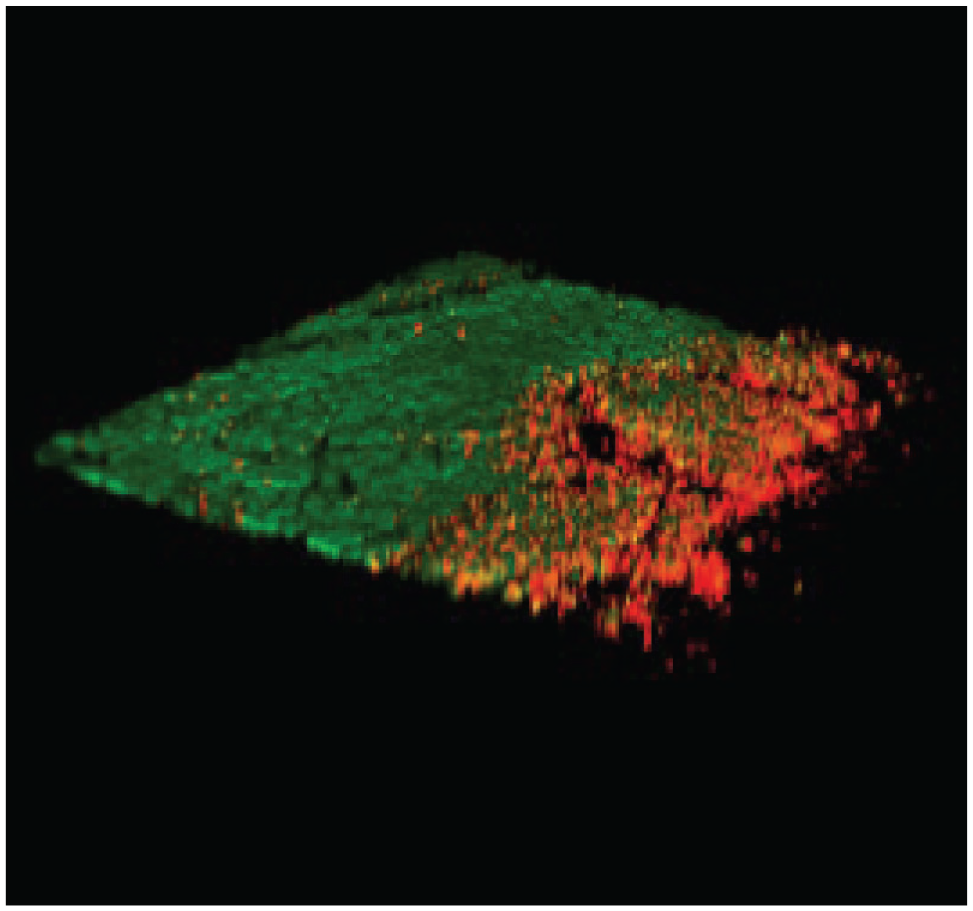
A 3-dimensional reconstructed image at the peripheral location showed the deformation of the cutting edge, with higher dead signals (red color) compared with the adjacent area.
Statistical Analysis
Statistical analysis was performed using SAS 9.4 software (SAS Institute). All data were reported as mean ± standard error. P < .05 was considered significant. Data were first subjected to the normality and homogeneity tests for comparison among multiple groups, followed by the Kruskal-Wallis test. If the P value from the Kruskal-Wallis test was significant, the Bonferroni post hoc adjustment was applied for individual P values. The paired t test was used to compare the 2 groups.
Results
Impact on Cell Viability at the Edges
After evaluating the collected images from all MACI specimens, we noticed no difference in pattern between the CC and Sc edges (Supplemental Figure S1). As expected, the center zone demonstrated the lowest density of dead signals with smoother and flat contours (Supplemental Figure S2). The cell viability results are presented in Table 1. The center and adjacent zones for both Sc and CC showed similar viability, ranging from a mean ± SE of 74.17% ± 2.85% to 79.95% ± 1.99% without any significant difference. As expected, the impact zone demonstrated lower cell viability in the Sc group (40.94% ± 2.85%) and CC group (36.42% ± 3.85%). While the impact zone showed significantly lower cell viability compared with the adjacent and central zones (P = .0021), there was no difference in cell viability of the impact zone between the 2 cutting methods—Sc and CC (P = .2334).
Percentage of Live Cells in Each Zone a

Data are presented as mean ± standard error. CC, custom cutting; Sc, scissor cutting.
Distribution of Cell Density
The cell density results in each zone were presented in Table 2, with all the areas showing similar cell density, ranging from 5.84 ± 0.26 to 6.49 ± 0.34 (×105/cm2) without any significant difference (P = 0.77, Kruskal-Wallis test). These results suggested limited cell loss at the impact zone.
Cell density in Each Zone a

Data are presented as mean ± standard error. CC, custom cutting; NA, not applicable; Sc, scissor cutting.
Discussion
In this study, we aimed to determine the difference in chondrocyte viability using 2 different cutting methods for the MACI membrane preparation (ie, Sc vs CC). We found that the MACI membrane prepared with either cutting technique demonstrated a significant reduction in cell viability in the impact zone (ie, periphery) compared with the adjacent and central zones. Last, we found no significant difference in the overall chondrocyte density among the study groups.
When reviewing the 2 cutting methods, CC enables relatively easy formation of stable and vertical cartilage boundaries with exact size and shape match between the implantation site and the MACI membrane. On the other hand, freehand debriding and SC are more suitable in situations where the defects have an irregular shape, their locations are too close to a convex area, or when there is a concern for violating the subchondral bone. 13
Above all, it is crucial to evaluate the impact of the newly introduced custom cut method on the chondrocyte viability of the MACI membrane, enabling surgeons to make an informed decision. This is the first study to explore this question, and we have offered recommendations based on the data presented here. Although we were limited to 5 MACI membranes with 30 images from each zone (impact/adjacent, center for Sc and CC groups), we employed a paired match or dependent sample strategy to reduce variation between membranes considerably.
Before the surgery, all MACI membranes must pass the quality control of the manufacturer (Vericel), whose prescribing information states that the cell density is >500,000 cells/cm. 2 Our results showed the cell density from the center zone and control is in line with their claims, and the percentage of live cells at 76% is similar to a previous study. 5 With both Sc and CC specimens collected from the same membrane, we removed the variation factor from membrane to membrane, which is always the biggest challenge in this type of study. The focal point of this study is that there was no significant difference in chondrocyte viability within the impact zone between CC and Sc groups. In addition to our software-based automatic image analysis, we meticulously examined all edge images and determined that there was no clear pattern distinguishing the Sc and CC groups.
Furthermore, the mean difference in the percentage of live cells between these 2 groups in the impact zone (40.94% for Sc and 36.42% for CC) was merely 4.52%. From a practical point of view, it is arguable that such a slight difference can make a meaningful impact on the outcome of the MACI procedure even with P < .05. A randomized controlled trial for patient outcomes should be undertaken to address this question directly, as it is beyond the scope of the present study.
In this study, we adopted the previously published image analysis technique to assess the chondrocyte viability using these 2 different cutting techniques. 5 Furthermore, using a larger supporting sheet under the MACI membrane during the cutting process eliminated the need to exert an extra force usually generated from grabbing or pressing the membrane directly. While this study concentrated on the effects of 2 cutting methods on membrane chondrocytes, it is also important to consider another key interface during MACI implantation: the host chondrocytes at the lesion's edge. The death of chondrocytes surrounding the defect poses a significant obstacle to proper cartilage integration during the repair process. In an animal study by Shapiro et al, 12 chondrocyte cluster formation was observed in areas of low viability near the defect edge. This cluster formation is frequently associated with areas of cell death and tissue degeneration and serves as a histological hallmark of osteoarthritis cartilage. 8 Therefore, the death ring in the surrounding wall of native cartilage created from the debridement and preparation is the significant barrier to the success of MACI integration.7,13
In theory, the effect of a custom cutter on cell viability would be similar both for the implanted MACI membrane as well as at the host lesion border in the native tissue. However, we assessed only the effect of cutting on the MACI membrane. Further study is warranted.
In the present study, the impact zone was positioned precisely at the focal point of the cartilage walls, which were debrided as a component of the repair technique. The percentage of live cells, cell density, and the size of the impact zone of MACI membrane may play significant roles in the success of cartilage integration in the long term. Future studies are necessary to address how each of these factors contributes to the overall outcome and identify other related factors that may impact the success of cartilage integration and repair success.
Limitations
We acknowledge the limited number of specimens included in this study. The foremost reason is the procedure's high cost and finding suitable patients with the proper indications, such as focal cartilage damage involving the condyles, normal or corrected alignment, and adequate size.
Conclusion
This is the first study to investigate how CC affects the chondrocyte viability and density within the MACI membrane. Although both cutting methods led to a significant reduction in cell viability in the impact zone compared with other zones of the MACI specimen, there was no significant difference between these 2 cutting techniques.
Supplemental Material
sj-pdf-1-ojs-10.1177_23259671241308855 – Supplemental material for No Difference in Chondrocyte Viability Using Manual Versus Custom-Cutting Technique for Matrix-Induced Autologous Chondrocyte Implantation
Supplemental material, sj-pdf-1-ojs-10.1177_23259671241308855 for No Difference in Chondrocyte Viability Using Manual Versus Custom-Cutting Technique for Matrix-Induced Autologous Chondrocyte Implantation by Wen-Teh Chang, Ran Atzmon, Monica S. Vel, Kenneth M. Lin, Geoffrey D. Abrams and Seth L. Sherman in Orthopaedic Journal of Sports Medicine
Footnotes
Final revision submitted July 3, 2024; accepted July 18, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: R.A. has received education payments from Arthrex and Smith+Nephew. K.M.L. has received education payments from Arthrex and Smith+Nephew. G.D.A. has received education payments from Arthrex and consulting fees from Bioventus. S.L.S. has received grant support from Vericel; education payments from Arthrex; consulting fees from Olympus America, Biorez, Bioventus, DJO, DePuy/Medical Device Business Services, Kinamed, Linvatec, Pacira Pharmaceuticals, Smith+Nephew, Vericel, JRF Ortho, LifeNet Health, and Flexion Therapeutics; nonconsulting fees from Arthrex, Smith+Nephew, Synthes GmbH, Vericel, and Linvatec; royalties from CONMED; honoraria from Pacira Pharmaceuticals, Flexion Therapeutics, and JRF Ortho; and hospitality payments from Aesculap Biologics. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Stanford University (ref No. 56522).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
