Abstract
Background:
Aberrant gait biomechanics—ie, lower knee abduction moment (KAM) impulse— are linked to the development of posttraumatic osteoarthritis after anterior cruciate ligament (ACL) injury and ACL reconstruction (ACLR). There is a clinical need to identify modifiable factors, such as kinesiophobia and pain, that may contribute to aberrant gait development after ACLR to advance multimodal rehabilitation strategies.
Purpose/Hypothesis:
This study aimed to determine associations between preoperative kinesiophobia and pain and gait biomechanics linked to posttraumatic osteoarthritis development at 2 and 4 months after ACLR. We hypothesized that worse preoperative kinesiophobia and pain would be associated with lower KAM impulses in the ACLR limb but not the uninjured limb at 2 and 4 months after ACLR.
Study Design:
Cohort study; Level of evidence, 2.
Methods:
Patients within 6 weeks of ACL injury and planning to undergo ACLR with bone-patellar tendon-bone autografts were recruited for the study. Preoperatively, participants completed the Tampa Scale of Kinesiophobia (TSK-11) and Knee injury and Osteoarthritis Outcome Score Pain (KOOS Pain) subscale surveys to assess kinesiophobia (ie, psychological component to pain) and knee pain, respectively. Participants returned at 2 and 4 months after ACLR to complete a 3-dimensional gait biomechanics analysis. KAM impulses during the stance phase were calculated (N*m*s/N*m) for both limbs. Associations of preoperative TSK-11 and KOOS Pain scores with KAM impulses in ACLR and uninjured limbs were analyzed using separate linear regressions.
Results:
A total of 36 participants (58% women; mean age, 21.4 ± 4.31 years; body mass index, 24.1 ± 3.59 kg/m2 ) completed 3 study visits. Higher preoperative kinesiophobia was associated with lower KAM impulses in the ACLR limb (R2 = 0.14; P = .02) but not the uninjured limb (R2 = 0.01; P = .58) at 4 months after ACLR. Preoperative KOOS Pain scores were not associated with KAM impulses in the ACLR and uninjured limbs at 2 and 4 months after ACLR (ΔR2 range, <0.01-0.02; P range = .53-.90).
Conclusion:
Preoperative kinesiophobia, but not pain, was weakly associated with lower KAM impulses during early to midphases of clinical recovery at 4 months after ACLR.
Aberrant gait biomechanics are linked to the risk of developing posttraumatic osteoarthritis (PTOA) after anterior cruciate ligament (ACL) injury. Between 6 and 12 months after ACL reconstruction (ACLR), patients exhibit persistent lesser knee abduction moment (KAM) impulses in the ACLR limb during gait, which is recognized as a surrogate measure of lesser medial knee joint loading, compared with the contralateral limb 44 and the limbs of uninjured sex- and age-matched controls. 16 Walking with lower frontal plane moments (ie, peak KAM and KAM impulse) after ACLR is associated with early cartilage degeneration,16,36,47 catabolic cartilage metabolism, 37 and the development of radiographic PTOA after ACLR. 46 Therefore, it was hypothesized that a low KAM impulse would contribute to deleterious medial knee joint loading that fails to promote healthy knee cartilage tissue metabolism and composition in patients who had ACLR.16,46 The normalization of gait biomechanics after ACL injury and ACLR may improve long-term joint health. Thus, determining the factors that contribute to the development of lower KAM impulse profiles is critical to elucidate the mechanisms that can be targeted by early interventions and normalize gait outcomes linked to PTOA disease development. Previous longitudinal studies have demonstrated that preoperative pain and early postoperative kinesiophobia predict long-term outcomes (ie, knee symptoms and secondary ACL injury)35,43 and return to play,11,45 respectively, in ACL-injured patients. Conversely, little is known regarding how preoperative pain and kinesiophobia are associated with aberrant gait biomechanics linked to PTOA disease development.
Knee pain has been hypothesized to be a contributing factor in the pathogenesis of biomechanical alterations after knee injury. 2 Experimental knee pain models have demonstrated mechanistic links between knee pain and acutely altered KAM during gait. Specifically, a hypertonic saline injection into the infrapatellar fat pad in uninjured individuals initiates a reduction in peak KAM during gait,22,39 which is consistent with KAM profiles exhibited in the injured limb after ACLR. Lower frontal plane moments (ie, peak KAM and KAM impulse) during gait in patients persist for years after injury and surgery despite decreases in knee pain preoperatively to the first 3 months after ACLR. 23 Regardless, knee pain in the early stages after an ACL injury may have lasting effects on gait biomechanics.25,44 Kinesiophobia, or fear of movement, is based on the fear-avoidance model applied in the context of musculoskeletal injury, 29 which suggests that kinesiophobia may facilitate neurophysiological changes that alter an individual’s movement patterns. 3 Two supporting constructs of kinesiophobia have been identified—somatization and activity avoidance.19,38 Exploring associations between kinesiophobia constructs and gait biomechanics may highlight a need for construct-focused psychological rehabilitation approaches for future research. 7 Somatization refers to an individual’s fixation on pain and symptoms associated with musculoskeletal injury while activity avoidance is defined as a concern that a specific activity will cause injury or pain. Both somatization and activity avoidance are associated with self-reported disability in those with idiopathic osteoarthritis 38 but have not been explored after ACLR. It is necessary to determine whether pain after an ACL injury and kinesiophobia have extended effects on gait biomechanics after ACLR for timely implementation of pharmacological or nonpharmacological interventions to reduce pain and kinesiophobia and subsequently improve gait recovery.
Most studies exploring the relationships between pain, kinesiophobia, and aberrant knee joint loading are cross-sectional13,33,50 or occur 6 months after ACLR24,40 despite recommendations of gait retraining for normalization of walking patterns starting as early as 1 to 2 months after ACLR. 1 Pain 23 and kinesiophopia 7 are most prevalent in patients preoperatively compared with any postoperative time point. Accordingly, preoperative rehabilitation may be an optimal time point to address pain and kinesiophobia. If preoperative pain and kinesiophobia are linked to aberrant gait development in postsurgical recovery, then addressing pain and kinesiophobia in prehabilitation may help maximize the effectiveness of recommended gait retraining interventions as patients return to walking after ACLR. Identifying modifiable factors associated with the development of aberrant gait biomechanics early in the ACL injury recovery process (ie, preoperatively) in a more robust longitudinal cohort study is an important step in determining targets for treatment to improve gait recovery after ACLR. Therefore, this study aimed to determine the associations between preoperative pain and kinesiophobia as measured by the Knee injury and Osteoarthritis Outcome Score (KOOS) Pain (KOOS Pain) subscale, the Tampa Scale of Kinesiophobia-11 (TSK-11) questionnaire, and the KAM impulse throughout the stance phase of gait in the ACLR and uninjured limbs 2 and 4 months after ACLR. We hypothesized that greater preoperative pain and kinesiophobia would be associated with lower KAM impulses in the ACLR limb during gait at 2 and 4 months after ACLR but not in the uninjured limb.
Methods
This study was part of a larger, prospective longitudinal cohort study. For the present study, participants completed 3 visits: preoperatively (days after an ACL injury (23.9 ± 12.9 days); 2 months after ACLR (2 ± 0.2 months); and 4 months after ACLR (4.1 ± 0.2 months). Participants completed patient-reported outcome (PRO) questionnaires—including the KOOS Pain subscale and the TSK-11 to assess knee pain and kinesiophobia, respectively, at the preoperative visit. Participants aged 16 to 17 years completed the questionnaires autonomously without assistance from their parents or guardians. At the 2- and 4-month after ACLR follow-up visits, participants completed 3-dimensional assessments on gait biomechanics. Injury and surgery information (ie, meniscal and chondral injury, meniscectomy, meniscal repair, and date of surgery) were extracted from electronic medical records to aid in characterizing the cohort (Table 1). All study procedures and recruitment methods were approved by the institutional review board, and to review relevant health information from patients’ medical records, they were asked to give permission under the Health Insurance Portability and Accountability Act. The methods and results of this study are reported in compliance with the Strengthening of Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 42 Participants ≥18 years old provided written informed consent while those <18 years old provided written informed assent and their parents provided written parental permission before participation.
Participant Characteristics and Surgical Information (N = 36) a

Data are presented as mean ± SD or n (%). ACL, anterior cruciate ligament; ACLR, anterior cruciate ligament reconstruction; BMI, body mass index; lab, laboratory; postop, postoperative; preop, preoperative.
Participants
Participants seeking care within 6 weeks of ACL injury were recruited from a university-affiliated orthopaedic clinic based on a referral from 1 of 3 sports medicine surgeons (J.T.S., R.A.C., G.K.). Only participants who planned to undergo ACLR with a bone-patellar tendon-bone (BPTB) autograft were included in our cohort. The exclusion criteria were as follows: a previous history of ACL injury or ACLR in either knee; a displaced fracture at the time of injury; pediatric ACLR surgical approaches (ie, physeal-sparing or partial transphyseal procedures); severe cartilage damage (ie, >3A on the International Cartilage Repair Society criteria) 34 ; or physician-diagnosed osteoarthritis. Participants planning to undergo multiligament surgery or removal of more than one-third of the meniscus were also excluded. Participants underwent an arthroscopic-assisted single incision BPTB autograft reconstruction using a single-bundle technique as previously described.32,37 All participants were referred for structured postoperative rehabilitation adapted from evidence-based guidelines by Wright et al 49 lasting 6 to 9 months under the supervision of an athletic trainer or a physical therapist, as previously described.32,37 Participant recruitment, enrollment, and follow-up are reported in a STROBE flowchart in Figure 1.

A STROBE 42 flowchart describing participant recruitment, enrollment, follow-up, and analysis. ACLR, anterior cruciate ligament reconstruction; Preop, preoperatively; STROBE, Strengthening the Reporting of Observational Studies in Epidemiology.
Sample Size Justification
A previous study reported strong associations (R2 = .53; P = .002) between higher kinesiophobia (ie, higher TSK-11 scores) and knee joint underloading (ie, second peak vertical ground reaction force asymmetry of the ACLR limb to the uninjured limb) during walking gait at 6 months after ACLR. 40 In addition, a study reported moderate associations (R2 = 0.35) between worse pain 1 month after ACLR and lower peak KAM during running gait 6 months after ACLR. 24 Similar to these studies, our analyses would be powered to detect at least moderate associations between kinesiophobia or pain and KAM impulses (ΔR2, ≥0.19) with 36 participants (α = .05; 1-β = 0.80).
Preoperative Patient-Reported Outcomes
Assessment of Kinesiophobia
The TSK-11 is a valid assessment of kinesiophobia in patients after ACLR. 19 The survey includes 11 items that are scored on a 4-point Likert scale from 1 (strongly disagree) to 4 (strongly agree). Scores from each question are summed together and range from 11 to 44, with higher scores indicating higher kinesiophobia. Two constructs of the TSK-11—somatization and activity avoidance—have been previously validated. 38 TSK-11 somatization (range, 5-20) and activity avoidance (range, 6-24) subscale scores are calculated based on scores summed from 5 and 6 of the questions included in the TSK-11 survey, respectively. 38 Higher scores indicate higher somatization and activity avoidance.
Assessment of Knee Pain
The KOOS is a valid assessment of self-reported knee function in patients after an ACL injury and consists of 5 subscales. 10 The KOOS Pain subscale quantifies the extent of pain that patients experience in their knee during activities of varying difficulty. The subscale consists of 9 total questions on a 5-point Likert scale from 0 (no pain) to 4 (extreme pain). Scores range from 0 to 100, with 100 indicating no pain with any of the activities.
Gait Biomechanics at 2 and 4 Months After ACLR
Gait analysis was conducted with a 10-camera motion capture system (Vicon; Nexus) integrated with 2 force plates that were embedded in the ground and positioned in a staggered manner to collect force data from the stance phase of both limbs during a single stride (FP406010; Bertec Corp). 12 A total of 26 retroreflective markers and a sacral cluster of 3 retroreflective markers were affixed to previously reported landmarks on each participant. 12 Timing gates were placed along a 6-m walkway to calculate gait speed in real time (Dashr; Dashr Systems). Participants were instructed to walk across a 6-m walkway as if they were walking “comfortably across the sidewalk” for 5 consistent trials to calculate their average habitual walking speed. Marker trajectories and force plate data were collected at 120 and 1200 Hz, respectively, during the next 5 trials and used for analysis. 12 A trial was considered successful if participants walked between 95% and 105% of their habitual walking speed, with no obvious gait deviations, and completed a single stride with both feet fully and individually contacting the corresponding force plates. Data were imported into Visual 3D (C-Motion) and filtered using a low-pass fourth-order Butterworth filter with a cutoff of 10 Hz. 12 Vertical ground reaction force (vGRF) data from the force plates were used to identify gait stance from heel strike (vGRF >20 N) and toe-off (vGRF <20 N) for the ACLR and uninjured limbs. First, net internal frontal plane joint moments for each limb were calculated during the gait stance using inverse dynamics and normalized to the product of body weight and height. The area under the frontal plane joint moment curve was determined via integration using the trapezoidal method. The internal KAM impulse was calculated as the impulse area under the abduction component only throughout the gait stance and normalized to body weight and height (N*m*s/N*m). 12 The KAM impulse was reported as a positive value to aid in interpretation.
Statistical Analysis
Primary Analysis
Participant characteristics and study outcomes were summarized as means and standard deviations or counts and percentages for descriptive purposes. Separate linear regression analyses were performed to estimate the associations of preoperative KOOS Pain or TSK-11 scores with KAM impulses during walking in the ACLR and uninjured limbs at each postoperative time point. Paired t tests were used to compare KAM impulses between the ACLR and uninjured limbs at 2 and 4 months after ACLR to aid in interpretation. R2 coefficients were reported to determine the percent variance of the KAM impulse variable explained by the preoperative PROs. Associations were considered statistically significant if P < .05. All analyses were completed in the Statistical Package for Social Sciences Software Version 28 (IBM Corp).
Secondary Analysis
Planned linear regression analyses were performed to determine associations of preoperative TSK-11 somatization and activity avoidance subscales and KAM impulses in both limbs. R2 coefficients and P values were interpreted as previously described.
Results
Participant characteristics and surgical data are reported in Table 1. Preoperative PROs and gait biomechanics at 2 and 4 months after ACLR are reported in Table 2. Patients walked with lower KAM impulses in the ACLR limb compared with the uninjured limb at 2 months after ACLR (mean difference [MD], –0.18 ± 0.40 N*m*s/N*m; P = .01) (Appendix Figure A1). The KAM impulse did not differ between the ACLR and uninjured limb at 4 months after ACLR (MD, –0.05 ± 0.31 N*m*s/N*m; P = .38) (Appendix Figure A1).
Clinical Outcomes at Preop, 2 Months After ACLR, and 4 Months After ACLR a

ACLR, anterior cruciate ligament reconstruction; KAM, knee abduction moment; KOOS, Knee injury and Osteoarthritis Outcome Score; post, postoperative; preop, preoperative; TSK-11, Tampa Scale of Kinesiophobia.
P = .01 indicating statistically significant KAM impulse differences between ACLR limb and uninjured limb.
Primary Analysis: Associations Between Kinesiophobia, Knee Pain, and Gait Biomechanics
Greater preoperative TSK-11 scores were associated with lower KAM impulses in the ACLR limb at 4 months (R2 = 0.14; P = .02); nonetheless, no statistically significant associations were observed between outcomes at 2 months after ACLR (R2 = 0.10; P = .06) (Table 3). No statistically significant associations were found between preoperative TSK-11 scores and KAM impulses in the uninjured limb at 2 or 4 months after ACLR (R2 range = 0.01 to 0.04; P range = .23-.58) (Table 3). Similarly, no statistically significant associations were observed between preoperative KOOS Pain scores and KAM impulses in the uninjured limb at 2 and 4 months after ACLR (R2 range < 0.01 to 0.02; P range = .58-.90) (Table 3).
Preoperative TSK-11 and KOOS Pain Scores with KAM (N*m*s/N*m) Impulses at 2 and 4 Months After ACLR a
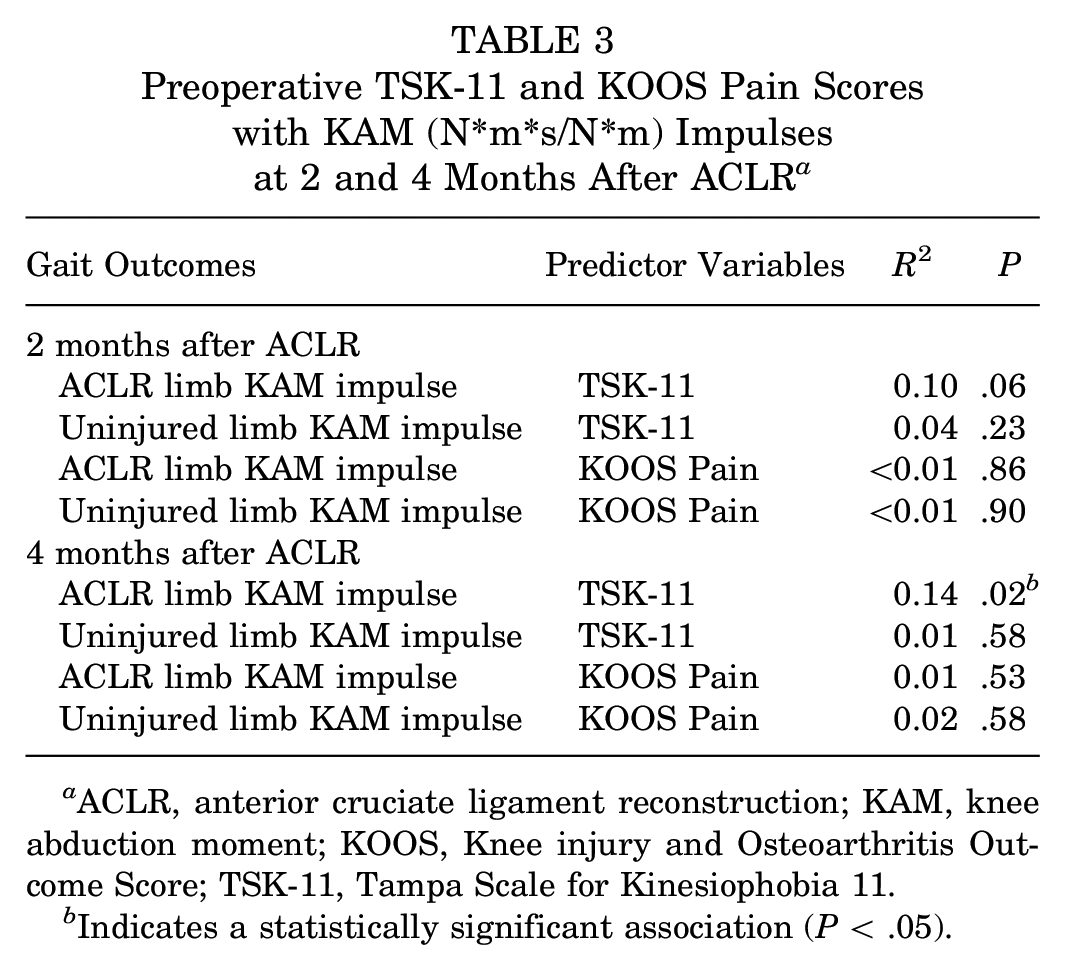
ACLR, anterior cruciate ligament reconstruction; KAM, knee abduction moment; KOOS, Knee injury and Osteoarthritis Outcome Score; TSK-11, Tampa Scale for Kinesiophobia 11.
Indicates a statistically significant association (P < .05).
Secondary Analysis: Associations Between Somatization, Activity Avoidance, and Gait Biomechanics
Greater preoperative TSK-11 activity avoidance subscale scores were associated with lower KAM impulses in the ACLR limb at 2 (R2 = 0.20; P = .01) and 4 months after ACLR (R2 = 0.16; P = .02) (Table 4). However, no statistically significant associations were observed between TSK-11 activity avoidance subscale scores and KAM impulses in the uninjured limb at 2 and 4 months after ACLR (R2 range = 0.04-0.07; P range, .12-.25) (Table 4). In addition, no statistically significant associations were observed between preoperative TSK-11 somatization subscale scores in ACLR and uninjured limb KAM impulses at 2 and 4 months after ACLR (R2 range, <0.01 to 0.05; P range, .21-.80) (Table 4).
Preoperative Activity Avoidance and Somatization Scores With KAM Impulses (N*m*s/N*m) at 2 and 4 Months After ACLR a

ACLR, anterior cruciate ligament reconstruction; KAM, knee abduction moment.
Indicates a statistically significant association (P < .05).
Discussion
In support of our hypothesis, higher preoperative kinesiophobia—specifically related to activity avoidance—was associated with lower KAM impulses in the ACLR limb but not the uninjured limb at 2 and 4 months after ACLR. Contrary to our hypothesis, preoperative pain was not associated with KAM impulses in either limb at 2 and 4 months after ACLR. Our preliminary results indicated that preoperative kinesiophobia may be more predictive than pain for the postoperative development of aberrant gait biomechanics linked to cartilage degeneration 16 but that the strength of the association is weak. While associations were statistically significant, kinesiophobia and activity avoidance only explained 14% to 20% of the variance in ACLR limb KAM impulse, suggesting that other preoperative clinical factors need to be considered to understand the development of aberrant gait biomechanics after ACLR.
The results of our study support the findings of previous cross-sectional studies that reported associations of poor psychological outcomes with aberrant gait biomechanics in patients 6 months after ACLR.40,50 Lower psychological readiness to return to sports and greater kinesiophobia are related to lower limb loading (ie, vGRF) in the ACLR limb. 40 Lower joint (ie, KAM impulse) and limb loading (ie, vGRF) are hypothesized to facilitate sustained, localized loading of the knee leading to knee joint cartilage degradation and catabolic joint tissue metabolism6,36,37,48 in the development of PTOA after ACLR. The longitudinal approach of our study linking preoperative kinesiophobia to gait biomechanics after ACLR strengthens the previous findings reporting cross-sectional associations between kinesiophobia and gait biomechanics.40,50 Together, these results highlight early kinesiophobia as a potential target for supplemental interventions in the development of aberrant gait biomechanics after ACLR. Moreover, targeting kinesiophobia preoperatively may be the optimal time point to begin psychological intervention integration. In vivo exposure interventions to fear-eliciting activities (ie, jumping and hopping) 8 or exercise 18 are recommended to help reduce kinesiophobia related to activity avoidance in the later stages of postsurgical rehabilitation. Preliminary feasibility and efficacy for reducing kinesiophobia through in vivo exposure therapy for sports-specific activities has been established in a small cohort of women after ACLR. 5 We hypothesized that the development of supplemental in vivo exposure interventions to fear-eliciting activities related to activities of daily living (ie, gait) may be helpful in the preoperative phases when patients may have more severe limitations to gait movement15,21,27 to reduce the risk of adopting aberrant gait patterns as early as possible. Future clinical trials should determine whether cognitive functional interventions (such as in vivo exposure therapy) combined with gait retraining reduce kinesiophobia and the risk of aberrant gait development after ACLR.
Associations between preoperative pain and KAM impulses after ACLR were negligible. Previous mechanistic studies reported the development of pathological gait adaptations (ie, reduced KAM throughout gait stance) in response to the onset of experimental-induced knee pain.22,39 Furthermore, healthy individuals exposed to experimental pain demonstrated similar KAM throughout the gait stance to those with mild radiographic knee osteoarthritis, suggesting an acute mechanistic effect of pain on the development of aberrant gait biomechanical profiles. 22 While mechanistic studies suggest an acute response in gait adaptations to experimental pain, the pain reported preoperatively in patients after an ACL injury was not associated with the propensity to develop aberrant gait biomechanics after ACLR in the present study. Johnson et al 24 reported an association between higher KOOS pain 1 month after ACLR and lower KAM in the ACLR limb during running 6 months after ACLR. Together, these results suggest that postoperative pain and not preoperative pain may be related to aberrant gait after ACLR, as surgical trauma and donor site morbidity 28 may influence postoperative pain. The timing of associations between knee pain and gait biomechanics needs to be considered when developing timely interventions.
While the longitudinal design of the present study strengthens results from previous cross-sectional studies,24,50 cause and effect relationships between preoperative kinesiophobia and walking KAM impulse after ACLR cannot be determined. However, we can hypothesize that preoperative neurophysiological mechanisms related to kinesiophobia would facilitate later biomechanical alterations after ACLR. Patients demonstrate altered corticospinal excitability30,31 and neuroplastic changes9,14,20 after an ACL injury and ACLR compared with uninjured individuals. Kinesiophobia has been associated with aforementioned peripheral and central nervous system alterations in populations who had ACLR. Centrally, kinesiophobia has been associated with neuroplastic changes in the emotional regulation regions of the brain after an ACL injury.3,26 Baez et al 3 reported greater corticolimbic activation in patients after ACLR compared with uninjured controls during a picture imagination task of activities of daily living (ie, walking) to elicit an emotional response. These central nervous system alterations may have downstream effects on motor control and may explain the links between kinesiophobia and biomechanics reported in the present study as well as previously reported associations between kinesiophobia and lower muscular strength, 35 altered muscle activation, 41 jump landing biomechanics, 4 and slower visuomotor reaction time after ACLR. 17 Future studies should seek to improve our understanding of the fundamental mechanistic underpinnings linking fear-related neurophysiological changes to biomechanical gait changes after ACLR.
Limitations
A few limitations should be considered while interpreting the results of the present study and should be used to inform future research. We could not determine cause and effect relationships between kinesiophobia and gait biomechanics given the observational nature of the study. However, the longitudinal design of our study builds on previous cross-sectional study designs and provides critical evidence that the magnitude of kinesiophobia after an ACL injury that predates ACLR relates to gait biomechanics evaluated at critical time points after ACLR. Patients undergoing ACLR in the present study exclusively received BPTB autografts. Thus, the results of the present study cannot be generalized to those receiving other graft types (ie, hamstring tendon or quadriceps autograft or allograft). The present study only included the TSK-11 to assess psychological outcomes after an ACL injury. However, the psychological response to injury is complex, and other psychological responses to injury—such as self-efficacy, locus of control, and pain catastrophizing—should be considered to determine potential treatment strategies for aberrant gait after ACLR. Finally, it is important to note that preoperative kinesiophobia and activity avoidance explain a small percentage of the variance in ACLR limb KAM impulse (R2 range, 14%-20%). Only the association between preoperative activity avoidance and ACLR KAM impulse at 2 months after ACLR (R2 = 0.20) exceeded the effect size that the study was powered to detect (R2, ≥0.19). The results only provide preliminary support for associations between preoperative kinesiophobia and gait biomechanics after ACLR and thus should be interpreted with caution because of the study’s small sample size.
Conclusion
Preoperative kinesiophobia, but not pain, was weakly associated with aberrant gait biomechanics during the early phases of recovery after ACLR. Our results support previous cross-sectional studies linking poor psychological outcomes with aberrant biomechanical alterations in patients after an ACL injury and ACLR. However, the weak strength of the associations between kinesiophobia and gait patterns highlights the need for future research to determine other modifiable preoperative clinical factors that may affect aberrant gait development after ACLR.
Footnotes
Appendix
Final revision submitted March 26, 2024; accepted May 6, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: Research support was received from the Arthritis Foundation. J.T.S. has received education payments from Southtech Orthopedics. G.K. has received nonconsulting fees from Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from The University of North Carolina at Chapel Hill (20-2227).
