Abstract
Background:
Evaluation of glenoid bone loss is critical in preoperative planning, as bone loss >13.5% has been associated with worse clinical outcomes. While 3‐dimensional computed tomography (3D CT) and the Pico method have been the gold standard in bone loss evaluation, it is unclear how most orthopaedic surgeons evaluate for bone loss in practice.
Purpose:
To investigate the techniques used by orthopaedic surgeons to measure glenoid bone loss.
Study Design:
Cross-sectional study.
Methods:
A 12‐question survey was sent to members of the AOSSM and Arthroscopy Association of North America. This survey included questions regarding the participants’ demographic information, typical surgical practice and volume, and glenoid bone loss identification method. Friedman testing with multiple comparisons using Bonferroni correction was used to evaluate the ordinal variable, use frequency, while Kruskal-Wallis testing and Spearman correlations were used to evaluate the role of surgeon demographic factors on method usage.
Results:
Overall, 171 orthopaedic surgeons responded to the survey. Participating surgeons were from a variety of locations, practice settings, and years in practice; 91.1% of respondents said that they routinely measure glenoid bone loss. The imaging modality most commonly used for bone loss evaluation was 3D CT, followed by 2‐dimensional (2D) magnetic resonance imaging, then 2D CT and radiographs. Percentage diameter loss was the most commonly used method of bone loss evaluation. The intraoperative probe length method was used occasionally, the Pico method was used rarely, and the chord length method was never used. There was no significant effect of time in practice or practice location on bone loss methods used. Surgeons in private practice (P = .022) or other settings (P = .015) had a higher frequency of using the intraoperative probe length method compared with those in the military.
Conclusion:
Overall, this study highlights the inconsistency in surgeon evaluation of glenoid bone loss in shoulder instability, despite good evidence of its clinical importance, especially in preventing recurrent instability. This lack of consistency may be mitigated with the establishment of best practice guidelines or accepted algorithms for evaluation and assessment of glenoid bone loss in shoulder instability.
Keywords
Glenoid bone loss increases the risk of recurrent shoulder dislocation in the presence of underlying shoulder instability and has been shown to occur in 86% of patients. 13 Presence and severity of bone loss is relevant in the surgical approach to a variety of shoulder procedures including arthroplasty and instability surgery, as unrecognized or unaddressed bone loss can negatively influence outcomes.30,35 Specifically, in shoulder instability surgeries, bone loss constitutes a risk factor for inferior clinical outcomes and recurrent postoperative instability and may be an indication for an osseous augmentation procedure.4,32 While traditionally reserved for patients with glenoid bone loss >25%, more recent studies have suggested that bony augmentation procedures may be considered in lieu of a soft tissue stabilization when glenoid bone loss is >13.5%.8,31,33
Bone loss can be evaluated using a myriad of imaging modalities such as plain radiographs, magnetic resonance imaging (MRI), 3‐dimensional (3D) MRI, computed tomography (CT), and 3D CT. Studies have identified 3D CT as the gold standard imaging modality for measuring bone loss, as it has the highest interobserver reliability.5,10,19,27 3D MRI with specific sequences can also produce a similar estimate of bone loss to 3D CT.20,36,40,42 When evaluating 3D CT or MRI, a variety of methods can be used to estimate the percentage glenoid bone loss (PBL), including the following 4 commonly used techniques:
In the chord method, a circle is drawn over an en face view of the glenoid and a chord is added based on the location of bone loss. Bone loss is calculated with the equation
In the percentage diameter method, a best‐fit circle is based on the posteroinferior glenoid contour. The PBL is then calculated using the equation PBL = w/D× 100%, where w is the defect width and D is the maximal diameter of the drawn circle (Figure 1B).26,37 This approach has been shown to overestimate bone loss. 3
The Pico method is an option for measuring glenoid bone loss using the contralateral glenoid as a reference. 2 The PBL is calculated by drawing a best‐fit circle using the lower glenoid margin of the intact contralateral glenoid and then comparing this circle with the ipsilateral glenoid circle. 34 The area lacking bone on the injured glenoid is then calculated (Figure 1C). The Pico method can be viewed as the gold standard technique due to its simplicity, accuracy, and high interobserver reliability, especially when using 3D CT. However, it does require imaging of the contralateral shoulder, with consequent additional radiation exposure.2,6
The intraoperative measurement of PBL with a 3‐mm probe was proposed by Burkhart et al7,8 to approximate the amount of bone loss from the central bare area to the anterior (A) and posterior (P) rim of the glenoid using the equation PBL = (P – A)/(P× 2) (Figure 1D). However, the area of bone loss has been shown to change location, thus decreasing the accuracy of this method.17,18
While 3D CT is often considered the optimal imaging modality for estimating glenoid bone loss, institutional and surgeon preferences on how to evaluate glenoid bone loss and which imaging modality to use continue to persist.5,6,26,37 The purpose of this study was to assess the frequency with which these 4 glenoid bone loss evaluation methods were used by orthopaedic surgeons. We hypothesized that despite the available literature, most orthopaedic surgeons are not utilizing the Pico method or 3D imaging routinely in practice.
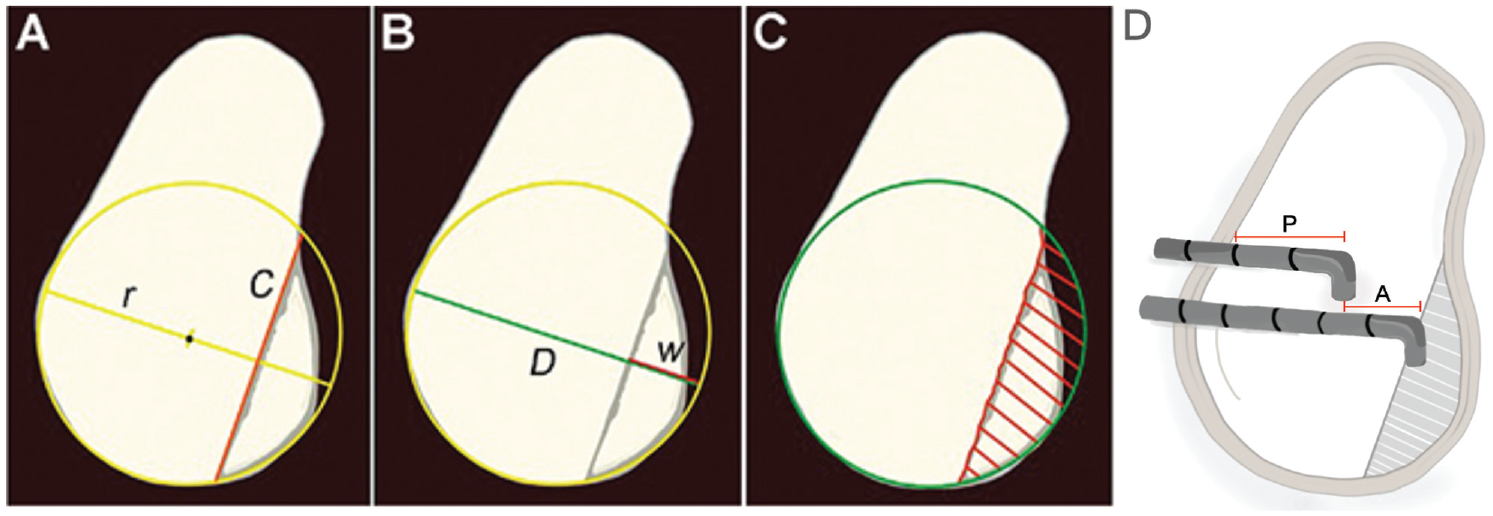
Methods of measuring percentage of glenoid bone loss (PBL). (A) Chord method: a circle is drawn over an en face view of the glenoid and a chord (red line) is added based on the location of bone loss; bone loss is calculated with the equation
Methods
A survey was constructed to evaluate the 4 common methods for measurement of glenoid bone loss: chord length, percentage diameter, Pico method, and intraoperative probe length as described in Figure 1. We also requested basic demographic information, training background, and typical surgical volume of relevant procedures. The survey asked practicing surgeons for preferences and tendencies of use of imaging studies and methods of bone loss measurements routinely acquired when evaluating patients with shoulder instability when there was a concern for glenoid bone loss.
A final survey of 12 questions was created (see the Appendix). The first 4 questions of this survey investigated surgeon demographic information including years in practice, fellowship training, location of practice, and practice type (eg, private or academic). The second 4 questions inquired about relevant surgical volume. This included number of instability procedures performed per year, percentage of instability procedures performed arthroscopically, number of osseous augmentation procedures per year (eg, Latarjet, Bristow, distal tibial allografts, and other bone block procedures), and percentage of osseous augmentation procedures where there was no detectable bone loss. The final part of the survey investigated how the surgeons evaluated glenoid bone loss. Surgeons were asked what imaging modalities they typically used to estimate bone loss, and how frequently they utilized previously published and validated methods (chord length, percentage diameter, Pico method, and intraoperative probe length), with frequency options that included never, rarely, occasional, routinely, and always use. The survey was then sent out via email to all current members of the AOSSM (N = 3797) and Arthroscopy Association of North America (AANA) (all society members; nearly 4000 surgeons). All survey data were included in the study except for 2 surveys that were duplicates and 1 survey response from a resident physician.
Statistical Analysis
Completed surveys were analyzed using descriptive statistics in Microsoft Excel. Outlying data that were presumed to be typing errors were removed for 3 data points. Friedman testing with multiple comparisons using Bonferroni correction was conducted in SPSS (Version 22; IBM) to evaluate the ordinal variable and use frequency of each of the 4 methods. Kruskal-Wallis testing with post hoc comparisons and Bonferroni correction was used to compare surgeon factors and their influence on method frequency. Spearman correlation analysis was used to evaluate the relationship between years in practice and method use frequency. Significance was set at P <.05.
Results
Surgeon Characteristics
A total of 171 orthopaedic surgeons participated in the survey. The characteristics of the participating surgeons are shown in Figure 2. Of these surgeons, 19 did not respond to the question regarding years in practice (89% response rate). Responding surgeons were in practice for a mean of 14.6 ± 16.6 years. Regarding fellowship training (96% response rate), 135 participants completed a sports medicine fellowship, 18 completed a shoulder and elbow fellowship, 10 completed both a sports medicine and a shoulder and elbow fellowship, 1 completed a sports medicine and pediatric sports medicine fellowship, and 1 completed a sports medicine and trauma fellowship. Respondents were from a variety of geographic locations: 19% were from the Northeast, 32% were from the Midwest, 27% were from the South, 10% were from Mountain states, 11% were from the West Coast, 1 was from Hawaii, 1 was from Alaska, 1 was from Colombia, and 1 practiced in Italy (98%, 168/171 response rate). With respect to practice setting, 45% of respondents were in private practice, while 33% of practices were affiliated with an academic institution. An additional 16% reported a hospital‐based practice, 2% reported military affiliation, 2% were part of a private practice with academic affiliations, and 2% were in other practices such as medical groups or multispecialty practices (100% response rate).

Demographic information of the orthopaedic surgeons who responded to the survey. (A) Years in practice, (B) fellowship training, (C) practice location, and (D) practice setting.
Practice Characteristics
The mean number of instability surgeries performed each year was 42.0 ± 35.0 (97% response rate). On average, surgeons reported performing arthroscopic soft tissue stabilization surgery 88.0% ± 20.1% of the time. Osseous augmentation procedures were reported as 7.3% ± 8.0% of procedures annually. When asked the percentage of osseous augmentation procedures performed when there was no detectable bone loss, 108 respondents cited 0%. A total of 62 surgeons reported performing osseous augmentation procedures when there was no detectable bone loss 17.4% ± 16.4% of the time.
Evaluation of Bone Loss
Overall, 91.1% of respondents said that they routinely measured glenoid bone loss. In response to preferred imaging modality used to determine severity of bone loss, 3D CT was cited by 124 physicians, 2‐dimensional (2D) CT was cited by 59, radiographs alone was listed by 68 respondents, while 86 preferred 2D MRI, and 11 mentioned 3D MRI (Figure 3). Many surgeons cited using a combination of these imaging modalities.

Imaging modalities used by the surgeons to detect glenoid bone loss. 2D, 2-dimensional; 3D, 3-dimensional; CT, computed tomography; MRI, magnetic resonance imaging; XR, radiograph.
The most frequently reported method for determining glenoid bone loss was percentage diameter loss (median: routinely used) (Figure 4). This was followed by the intraoperative probe length method (median: occasionally used). Less frequently used methods included the Pico method (median: rarely used) and the chord length method (median: never used). Eight respondents reported other preferred bone loss measurement methods (eg, glenoid track method, CTs/3D CTs of contralateral glenoid, and surface area in Image J software (US National Institutes of Health)). There was a significant difference in rate of use between glenoid bone loss approaches (P < .001). Specifically, post hoc testing demonstrated all groups to have a significantly different distribution of usage.

Frequency of each of the 4 methods to evaluate glenoid bone loss (chord length, percentage diameter loss, Pico method, intraoperative probe length) as indicated by the survey respondents.
Relationship Between Surgeon Characteristics and Method Choice
The relationship between bone loss assessment methods utilized and surgeon demographic information indicated that there was no significant effect of location, years in practice, or practice setting for use of the chord, percentage diameter, or Pico technique. There was a significant relationship between practice setting and frequency of using the intraoperative probe method (P < .001). Post hoc comparisons demonstrated that those in the private practice setting (median: routinely used; P = .022) or other settings (median: always used; P = .015) were more likely to use the intraoperative probe method than surgeons with military affiliations (median: never used).
Discussion
The most important finding of this survey study was that there was significant inconsistency in evaluation of glenoid bone loss in anterior shoulder instability among practicing orthopaedic surgeons. Based on our results, approximately 10% of surgeons in the United States do not routinely measure glenoid bone loss when dealing with shoulder instability, despite clear evidence of its importance on outcomes and recurrent instability.7,22 Further, there was a lack of consistency in preferred methods of both assessment and calculation of bone loss. We believe this is important, as surgeons may change their operative plan based on the presence and degree of bone loss.
It is generally accepted that 3D CT imaging with humeral head subtraction is the gold standard for preoperative evaluation of glenoid bone loss.5,6,27,37 Based on our results, 3D CT was the most frequently mentioned imaging study, whereas 3D MRI was the least mentioned, despite studies demonstrating its accuracy when directly compared with 3D CT.14,16,42 Half of orthopaedic surgeons obtained 2D MRI, which is often obtained initially to evaluate the status of periarticular soft tissues after shoulder instability events. In some studies, 2D MRI has been reported to produce similar results when compared with both CT imaging and intraoperative arthroscopic measurement; however,bone loss measurement on 2D MRI is best performed by skilled musculoskeletal radiologists given the nuances of this imaging modality.14-16,12,34,42 This becomes important as 1‐step MRI could potentially obviate the need for additional CT studies, which are associated with higher costs and increased radiation exposure. However, other studies have shown that 2D imaging modalities in general may underestimate the degree of bone loss present.5,27,42
There have been numerous proposed methods to measure glenoid bone loss, with no definitive gold standard. In a systematic review, Verweij et al 39 reported the use of 17 different glenoid measurements in 21 studies with few studies utilizing the same technique and none directly comparing all measurements. Furthermore, in a 2021 expert consensus statement, 95% of experts agreed that there are several methods that provide reliable approximation of bone loss. 28 A cadaveric study performed by Yanke et al 42 found that there was a strong correlation between imaging modalities using the Pico method performed on CT and 3D MRI when compared with actual measured defect sizes. Despite this, other studies argue that there may be no adequate radiographic measurement to assess bone loss. A finite element analysis by Moroder et al 24 that investigated Pico and Sugaya measurement techniques reported that neither measurement strongly correlated with the shoulder stability ratio, in part because of the role of glenoid concavity.
Each technique investigated in this study has its own advantages and disadvantages. For example, both the Pico and percentage diameter loss methods rely on the best‐fit circle technique and have been shown to have high inter- and intraobserver reliability.16,23,12 However, this best‐fit circle technique may overestimate bone loss due to the slight asymmetry of the anterior and posterior glenoid. 21 The Pico method also has the advantage of avoiding the influence of glenoid bone loss position and morphology, which can significantly alter percentage diameter loss calculations. 1 The chord length measurement, described by Bhatia et al, 3 had the most consistent response within our survey, with overall infrequent use. The advantage of this approach is its accuracy of true bone loss when compared with diameter‐based measurements. However, this technique is relatively new and requires a complex trigonometric equation that may be cumbersome to some. Arthroscopic intraoperative assessment of glenoid bone loss using probe measurements based on the bare area has historically been used as the benchmark to determine appropriate surgical management in shoulder instability. This tried‐and‐true approach has the advantage of not requiring access to 3D CT or MRI. However, recent evidence has called into question the reliability of the anatomic bare area as the geometric center of the glenoid, thus affecting the accuracy of its use in probe measurements.18,38 An alternative method to arthroscopically measure bone loss, described by Detterline et al 11 and called the secant chord theory, may be more accurate than traditional intraoperative methods described. However, while large differences in measuring bone loss may have effects on surgical decision‐making, it is unclear if preoperative measurement technique or precise measurements (within 1%) have a sizable effect on clinical results.
Despite the lack of a gold standard, based on studies in the current literature, percentage diameter loss and the Pico method are the most used radiographic techniques within the orthopaedic literature to measure glenoid bone loss. The Pico method is routinely used when comparing imaging modalities.29,34 A systematic review performed by Walter et al 41 further supports this claim. Their study found that the Pico method and glenoid width were the most cited measurements in an analysis of 27 studies. However, a few studies utilized percentage bone loss diameter to analyze bone loss. 24 In contrast, to our knowledge, no contemporary studies evaluate glenoid bone loss based on chord length or intraoperative probe measurements.
It is important to consider the methodology for measurement of glenoid bone loss when applying results of clinical outcome studies to clinical practice. In addition to clinical history and presentation, anterior glenoid bone loss is a significant anatomic consideration when indicating a patient for surgery. Misapplication of bone loss estimation techniques may lead to inaccurate measurements, which can lead surgeons to proceed with soft tissue stabilization in a high‐risk patient or alternatively to an unnecessary osseous augmentation procedure. Specifically, Chalmers et al 9 reported that differences in imaging modality (CT vs MRI), measurement approach (linear and area percentage bone loss), and observer can result in differences in treatment in up to 34% of cases. However, the costs of additional imaging are high and cost analysis studies are needed to evaluate the cost‐benefit utility of higher‐level imaging studies.
In addition to altering clinical decision‐making, imaging modality and measurement technique can play a role in treatment indication guidelines. For example, there is variation in the technique employed for measuring glenoid bone loss when determining what constitutes “critical” bone loss in shoulder instability. Lo et al 22 reported that 25% bone loss of the inferior glenoid may be the threshold of significant bone loss based on measurements using the intraoperative probe method. In contrast, Shin et al 32 reported “critical” bone loss of 17.3% when measuring with 3D CT imaging using the percentage diameter and reported significant differences in postoperative functional scores and failures at this degree of bone loss. Similarly, Shaha et al 31 reported that bone loss of >13.5% as measured using the percentage diameter method on 3D CT resulted in significant differences in Western Ontario Shoulder Instability Index scores, suggesting this threshold should be utilized instead. These differences in imaging techniques render it difficult to directly compare studies and deduce the most accurate threshold for defining “critical” bone loss.8,31,32
Interestingly, our study demonstrated that most surgeon demographics, including location or time in practice, had no significant effect on measurement methods chosen. However, surgeon practice setting, which included military, hospital, private practice, academics, “privademics” (mixed academic and private practice), and other (medical group or multispecialty group), played a significant role in measurement technique utilized. Specifically, those in a medical group or private practice were more likely to use the intraoperative probe measurement method. This may be because the intraoperative method has been around for the longest and is how many surgeons learned to measure glenoid bone loss during their training. In addition, there may be less access to 3D CT/MRI and/or automated glenoid bone loss techniques in these settings.
Limitations
This study is not without limitations. The first limitation is the relatively small sample size. However, the participants were from a variety of locations, had a wide range of years in practice, and were from different practice settings. Due to the diversity of our sample, we believe our results are valid and representative of sports medicine surgeons at large. In addition, this study is subject to possible sampling bias in the group of surgeons it reached. Specifically, a survey was sent out through only 2 organizations (AOSSM and AANA). It is possible that members of other sports medicine and arthroscopic organizations tend to utilize imaging modalities and the analyzed glenoid bone loss measurement techniques at differing frequencies. In addition, to limit survey burden the survey did not include further questions such as if participants had trialed other methods previously, or why they selected their utilized method. As with any survey study, there is potential for this study to be affected by a recall bias, specifically in the frequency of surgeons’ using each measuring technique. Despite these limitations, we believe our results are useful in understanding how orthopaedic surgeons assess glenoid bone loss.
Conclusion
The findings of this study highlight the inconsistency in surgeon evaluation of glenoid bone loss in shoulder instability, despite good evidence of its clinical importance, especially in preventing recurrent instability. This lack of consistency may be mitigated with the establishment of best practice guidelines or accepted algorithms for evaluation and assessment of glenoid bone loss in shoulder instability.
Footnotes
Appendix
Final revision submitted March 28, 2024; accepted April 12, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: W.C. has received grant support from Arthrex; education payments from Medwest, Peerless Surgical, Smith & Nephew, and Evolution Surgical; consulting fees from Vericel; and hospitality payments from Lima USA and Medical Device Business Services. D.A.L. has received consulting fees from AlloSource and Vericel and nonconsulting fees from Arthrex. J.C. has received grant support from Arthrex; education payments from Arthrex and Smith & Nephew; consulting fees from Arthrex, ConMed, Ossur, DePuy, RTI, Smith & Nephew, and Vericel; nonconsulting fees from Arthrex, Linvatec, and Smith & Nephew; and hospitality payments from Stryker. G.E.G. has received education payments from Medwest; consulting fees from AzurMeds, Bioventus, DePuy/Medical Device Business Services, DJO, Encore Medical, Restor3d, Stryker, Tornier, and Wright Medical; royalties from DJO, Elsevier, Encore Medical, Stryker, Tornier, and Wright Medical; hospitality payments from Exactech; and honoraria from Wright Medical; and has stock/stock options in Aevumed, CultivateMD, Genesys, Patient IQ, Restor3d, ROM 3, and Sparta Biopharma. N.N.V. has received research support from Arthrex, Breg, Ossur, Smith & Nephew, and Stryker; consulting fees from Arthrex and Stryker; nonconsulting fees from Arthrex; royalties from Arthrex, Smith & Nephew, Graymont Professional Products, and Stryker; and hospitality payments from Spinal Simplicity and Relievant Medsystems. A.B.Y. has received research support from Arthrex and Organogenesis; consulting fees from AlloSource, JRF Ortho, Olympus America, and Styker; nonconsulting fees from Arthrex; hospitality payments from Stryker; and honoraria from JRF Ortho. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
