Abstract
Background:
Pulsed electromagnetic field (PEMF) therapy is a potential nonoperative treatment for Achilles tendinopathy, but the current published literature on its effects is sparse.
Purpose:
To evaluate the therapeutic efficacy of PEMF therapy and eccentric exercise for Achilles tendinopathy.
Study Design:
Randomized controlled trial; Level of evidence, 1.
Methods:
A total of 65 participants met the inclusion criteria and were randomly assigned to either the active PEMF group (n = 33) or the sham group (n = 32). The PEMF group received biweekly PEMF therapy (10 minutes per session) for 8 weeks, plus eccentric exercise for 12 weeks. The sham group received the same eccentric exercise for 12 weeks minus active PEMF exposure. The primary outcome was the Victorian Institute of Sports Assessment–Achilles (VISA-A) questionnaire. The secondary outcomes were numeric pain rating scale (NPRS) score, tendon neovascularity, and total time spent on sports per week. A physical therapist assessed all outcomes at baseline and then 4, 8, and 12 weeks after the commencement of the PEMF treatment. The level of tendon neovascularity was determined by the Öhberg score (range, 0-3).
Results:
VISA-A scores improved significantly from baseline to 12 weeks posttreatment in both the PEMF group (from 57.9 to 71.7) and the sham group (from 55.1 to 66.8) (P < .001 for both), with no significant difference in scores between treatment groups throughout the 12 weeks (P = .527). NPRS scores during sports also improved significantly from baseline to 12 weeks for the PEMF group (from 6.05 to 2.95) and the sham group (from 6.45 to 3.85) (P < .001), with no group differences (P = .346).
Conclusion:
PEMF therapy, in addition to eccentric exercise, did not improve outcome scores or tendon neovascularity compared with eccentric exercise alone in participants with Achilles tendinopathy.
Registration:
NCT05316961 (ClinicalTrials.gov identifier).
Keywords
Achilles tendinopathy is a clinical syndrome characterized by activity-related Achilles tendon pain and swelling, leading to functional impairment. 16 It is a common overuse injury of the foot and ankle, affecting the athletic and sedentary population.6,12,15,23 An inability to heal is commonly the underlying pathology of Achilles tendinopathy, whereby damage can occur at the midportion or insertion of the tendon.5,7
Previous systematic reviews for the nonoperative management of Achilles tendinopathy have provided valuable assessments of available therapies such as exercise, electrotherapy, soft tissue therapies, and orthosis, generally implemented as the first-line management for Achilles tendinopathy.2,17,25 In some cases, pharmacological and injectable agents are used with other physical therapies, such as platelet-rich plasma, corticosteroid, and prolotherapy. Surgical treatment is often reserved as a second-line therapy when nonoperative treatments have failed to provide adequate relief.
A treatment focus has been isolated eccentric exercise, which has proven beneficial for tendinopathy by giving appropriate loading to the tendon and is associated with clinical effectiveness. 4 Three previous studies investigating the clinical effects of eccentric exercise for managing Achilles tendinopathy found that approximately 60% of participants returned to sports after completing a 12-week eccentric exercise program.11,33,35 In addition, Alfredson et al 3 reported that all participants returned to sports 12 weeks after being treated by heavy-loading eccentric exercise. Ackermann et al 1 and Maffulli and Longo 24 reported that nonathletic and female individuals exhibited lower return-to-sports rates than athletic individuals. A systematic review confirmed the benefits of eccentric exercise in promoting muscle strength gains by achieving higher forces during eccentric muscle actions. 40
Despite various available treatments, no modality or therapeutic approach has stood out as a definitive solution to improve pain and function for Achilles tendinopathy. Pulsed electromagnetic field (PEMF) therapy could treat these aspects of Achilles tendinopathy. Evidence suggests that PEMF therapy can improve pain and function and enhance cellular activity, tendon healing, and repair.14,29,36,38 PEMF therapy is drug-free, nonthermal, and low-risk. Previous studies have assessed the efficacy of PEMF therapies in several musculoskeletal disorders. PEMF therapy tended to stimulate bone growth in acute fractures and efficiently relieved pain and enhanced bone formation in osteoporosis. 8 Hu et al 14 proposed that PEMF therapies may represent a noninvasive, safe, and effective way to promote tendon regeneration.
Here, we extend these assertions and postulate that PEMF therapy could benefit patients with Achilles tendinopathy. Achilles tendinopathy could be caused by a shift of the homeostatic set point of proinflammatory and proregenerative immune cells after a tendon injury, and PEMF exposure may reset the immune response back to normal function and prevent an acute phase of inflammatory response from becoming chronic in the Achilles tendon. In this study, we aimed to examine the effect of PEMF therapy on self-reported pain, function, tendon neovascularity, and time spent in sports participation in patients with Achilles tendinopathy undergoing an eccentric exercise program. We hypothesized that participants undergoing eccentric exercise plus PEMF therapy would exhibit additional improvements in functional scores compared with participants who did not undergo PEMF therapy.
Methods
Study Design
This was a prospective, randomized, double-blinded, placebo-controlled superiority trial with 2 parallel groups and a 1:1 allocation ratio that investigated the treatment effects of PEMF therapy for participants with Achilles tendinopathy. All participants were randomized into 1 of 2 groups: an intervention group (active PEMF therapy and eccentric exercise [PEMF group]) and a control group (sham PEMF and eccentric exercise [sham group]). Researchers oversaw all self-reported scoring and functional and ultrasonographic outcomes during baseline assessment and at the 4-, 8-, and 12-week follow-ups after PEMF treatment. The final assessment point was set at 12 weeks after all treatments, including PEMF therapy and eccentric exercise. The outcomes taken at this point may reflect the short-term effects of PEMF plus eccentric exercise for Achilles tendinopathy.
Participants were recruited from the Department of Orthopaedics and Traumatology at the Prince of Wales Hospital in Hong Kong and from multiple university sports teams at The Chinese University of Hong Kong from July 2021 to February 2023. Individuals were referred to the Sports Injury and Biomechanics Laboratory for eligibility assessment by team orthopaedic surgeons and researchers.
Study Participants
Men and women 18 to 70 years of age with insertional or midportion Achilles tendinopathy and duration of pain >6 weeks were included. Achilles tendinopathy was diagnosed clinically and confirmed using ultrasound. Participants needed to be capable of providing informed consent.
Patients were excluded if they had a history of major injury or surgery on the affected lower limb in the past year; had mental or physical limitations hindering the participant's ability to complete assessments, including severe cognitive impairment and psychiatric disorders (active medical or musculoskeletal problems could affect the ability to complete assessments); had active electronic implants such as pacemakers and defibrillators; or were pregnant.
Sample-Size Calculation
Based on a previously published study using laser therapy as an adjunct treatment to eccentric exercise therapy for Achilles tendinopathy, 42 we calculated that a minimum improvement of 16 clinically significant points in Victorian Institute of Sports Assessment–Achilles (VISA-A) scores would substantiate efficacy. The power calculation to detect the minimal clinically important difference was conducted using the G*Power program at a type 1 error of 5% and power of 80% (correlation value, 0.03; pooled SD, 20.25). 43 Twenty participants per group (allowing 20% dropout) were calculated to detect a significant difference in VISA-A scores among the 2 study groups if a 2-way repeated-measures analysis of variance (ANOVA) was proposed. Thus, the final study sample size was 40 participants in total.
Randomization
Randomization was performed using an online research randomizer (https://www.randomizer.org). Participants were randomized into 1:1 allocation in blocks of 10. An orthopaedic surgeon (S.K.K.L.) and a physical therapist (V.M.C.K.) recruited participants. The physical therapist completed the assessment and assigned participants to the interventions. The participants were given a unique radiofrequency identification (RFID) card assigned to either the PEMF or sham treatment group. Participants and assessors were blinded, as participants used the RFID card to complete the treatment without knowing the treatment group to which they belonged. The randomization results were disclosed to the orthopaedic surgeon and physical therapist after the 12-week assessment.
Interventions
A registered physical therapist and a researcher (V.M.C.K., S.S.C.) were responsible for administering the interventions.
PEMF Therapy
As previously described, the PEMF was administered using a commercial device (QuantumTX Pte). 45 The delivery of PEMF to the leg did not produce heat or cause any sensation, which allowed the participants to be blinded to the treatment. Participants in the control group received sham exposure with the same PEMF device. The injured leg was exposed to either PEMF or sham therapy for 10 minutes per session, and the treatment regime ran twice a week for 8 weeks, for a total of 16 sessions. The PEMF device was activated by the RFID card, which was coded to deliver either the active or sham treatment. The PEMF treatment procedure was as follows: The participant was seated in a 90° position on a chair, and the entire foot, ankle, and lower calf muscles were placed within the solenoids of the PEMF device (Figure 1). When active, the device delivered uniform 1-mT, 50-Hz PEMFs to the injured lower leg for 10 minutes. There appears to be no gold standard of the PEMF protocol applied to tendon regeneration. Low-energy PEMFs have been previously demonstrated to promote muscle regeneration by activating mitochondrial respiration and invoking systemic homeostasis.38,41,47,48 We postulated that this setup could promote the return to homeostasis between pro- and anti-inflammatory cytokines. 36

Demonstration of pulsed electromagnetic field treatment setup.
Eccentric Exercise
All study participants performed 12 weeks of eccentric exercise that coincided with the first 8 weeks of PEMF therapy and 4 weeks afterward. A physical therapist (V.M.C.K.) instructed participants on eccentric exercise in the first session and reviewed the exercises with participants if needed. Participants performed the eccentric exercise independently based on the exercise guidelines with graphic demonstrations and written instructions.
The first step was stretching exercises for the calf muscles. 37 The stretching was a static stretch of the gastrocnemius (knee in extension) and soleus (knee in flexion). The participants were instructed to hold these for at least 30 seconds and to repeat each exercise 3 times. There was a 1-minute rest between each stretch.
Next, participants were asked to stand in an upright body position with a straight leg. They started with a bilateral heel raise with the majority of weight on the noninjured leg and then gradually transferred all body weight to the injured side from the heel-raise position and lowered the heel to the floor level as slowly as possible. The eccentric exercise was performed on the floor level, so no load was placed on the ankle in dorsiflexion. Participants loaded the calf muscles only eccentrically, and the injured leg did no concentric loading throughout the exercise. Three sets of 10 repetitions of the eccentric exercise were carried out once daily for 6 weeks. After 6 weeks, the participants were instructed to carry out 3 sets of 10 repetitions 3 times a week for 6 more weeks. The intensity of the exercise should be such that pain, or discomfort, was experienced in the last set of 10 repetitions. They were allowed a 1-minute rest between each set.
Every session ended with the same static stretch exercise as in step 1. If a participant could not complete 3 sets of 10 repetitions, that participant was instructed to start with fewer repetitions and sets (minimum of 2 sets of 10) and progress to the total amount as able. All participants could contact the physical therapist during the study period if they had questions about the training program. This study did not pose any restrictions on sports and pain medication, but relevant information was monitored and recorded at all assessment points.
Outcome Measures
Patient data collected at baseline were age, sex, height, weight, body mass index, and symptom duration. All outcome measures were taken in person at baseline and at the 4-, 8-, and 12-week assessments. The primary outcome measure was the validated Chinese version of the VISA-A, which consists of 8 questions and covers the 3 domains of pain, function, and sporting activities. 22 VISA-A scores range from 0 to 100, with 100 being equivalent to asymptomatic. As a secondary outcome measure, a 10-point numeric pain rating scale (NPRS; where 10 represents maximal pain) was used, on which participants could indicate their worst pain on the day of review (NPRS-worst) and pain during initiation of sports activities (NPRS-sports). In addition, the type of sports and time spent on sports participation (minutes per week) were recorded to assess the progress of returning to sports.
Neovascularity of the Achilles tendon was measured using ultrasound and reported using the Öhberg score. 44 During the ultrasound assessment at all assessment points, the transducer was placed perpendicular to obtain a sagittal view of the Achilles tendon on palpation at the most painful part. The upper limit of the color box was placed on the dorsal side of the tendon. Pressure from the transducer was kept to a minimum to prevent the occlusion of neovascularization. The assessor screened the tendon for the area of maximum Doppler flow during the preparation phase for 1 minute. The transducer was gently moved to medial and lateral over the area where Doppler flow was present. A sonographic image was taken when the maximum Doppler flow location was identified. The Öhberg score was then determined, in which scores were defined as 0 (no neovascularization), 1 (mild neovascularization, a few solitary blood vessels), 2 (moderate neovascularization, moderate quantity, most transversal blood vessels), or 3 (several, mostly horizontal blood vessels spread in the whole depth of the tendon). 39 A higher score indicates more Doppler flow in the peritendinous and intratendinous tissues. 46
Achilles tendinopathy was categorized into midportion or insertional tendinopathy, as assessed on musculoskeletal ultrasonography by a single musculoskeletal physical therapist (V.M.C.K.), who specialized in sports medicine and regenerative therapy. Midportion tendinopathy was diagnosed by local thickening and a color Doppler flow of at least 1 on the Öhberg score at 2 to 6 cm proximal to the Achilles tendon insertion. 39 Insertional tendinopathy was diagnosed by local thickening and color Doppler flow of at least grade 1 on the Öhberg score within 2 cm of the Achilles tendon insertion.
Statistical Analysis
Participant characteristics are reported as means and standard deviations for continuous variables, and nominal and ordinal data are presented as numbers and percentages. All statistical analyses were performed using SPSS (Version 29.0; IBM), based on the original data collected from all assessment points. Testing was performed 2-sided, with significance set at P < .05. The differences in VISA-A scores, NPRS scores, Öhberg scores, and weekly sports participation between the PEMF and sham groups at the 4-, 8-, and 12-week follow-ups were tested by 2-way repeated-measures ANOVA.
Subgroup analysis was performed based on the adherence rate to eccentric exercise. The self-reported adherence rate was defined as the proportion of prescribed exercises performed and was divided into 2 categories: <80% (low adherence) or ≥80% (high adherence).
We also analyzed whether PEMF therapy had clinical effects on all outcome measures according to tendinopathy type (midportion vs insertional) since the number of participants was smaller in the subgroups. In addition, subgroup analysis was conducted to explore the effects of PEMF therapy among participants in different age groups (young, 18-30 years; middle-aged, 31-45 years; old, >45 years) and between the sexes. The Mann-Whitney U test was used to detect if there was a significant difference in the outcomes for each subgroup.
Results
From July 2021 to February 2023, 83 participants were screened for eligibility. Of these, 65 participants were recruited and randomly assigned to either the PEMF (n = 33) or sham (n = 32) group. The PEMF group consisted of 33 participants (13 women, 20 men) who received 12 weeks of eccentric exercise plus PEMF treatment for 8 weeks. The sham group consisted of 32 participants (19 women, 13 men) who received 12 weeks of eccentric exercise and sham treatment for 8 weeks. Recruitment was stopped when the number of participants fulfilled the sample size requirement. By the end of the study (12-week follow-up), 40 participants had completed all data points. There were 25 patients (38%) who were lost to follow-up, as 13 patients (3 from the PEMF group and 10 from the sham group) reported insufficient time to return for intervention and 12 patients (9 from the PEMF group and 3 from the sham group) discontinued the intervention because they reported full symptomatic resolution (Figure 2).
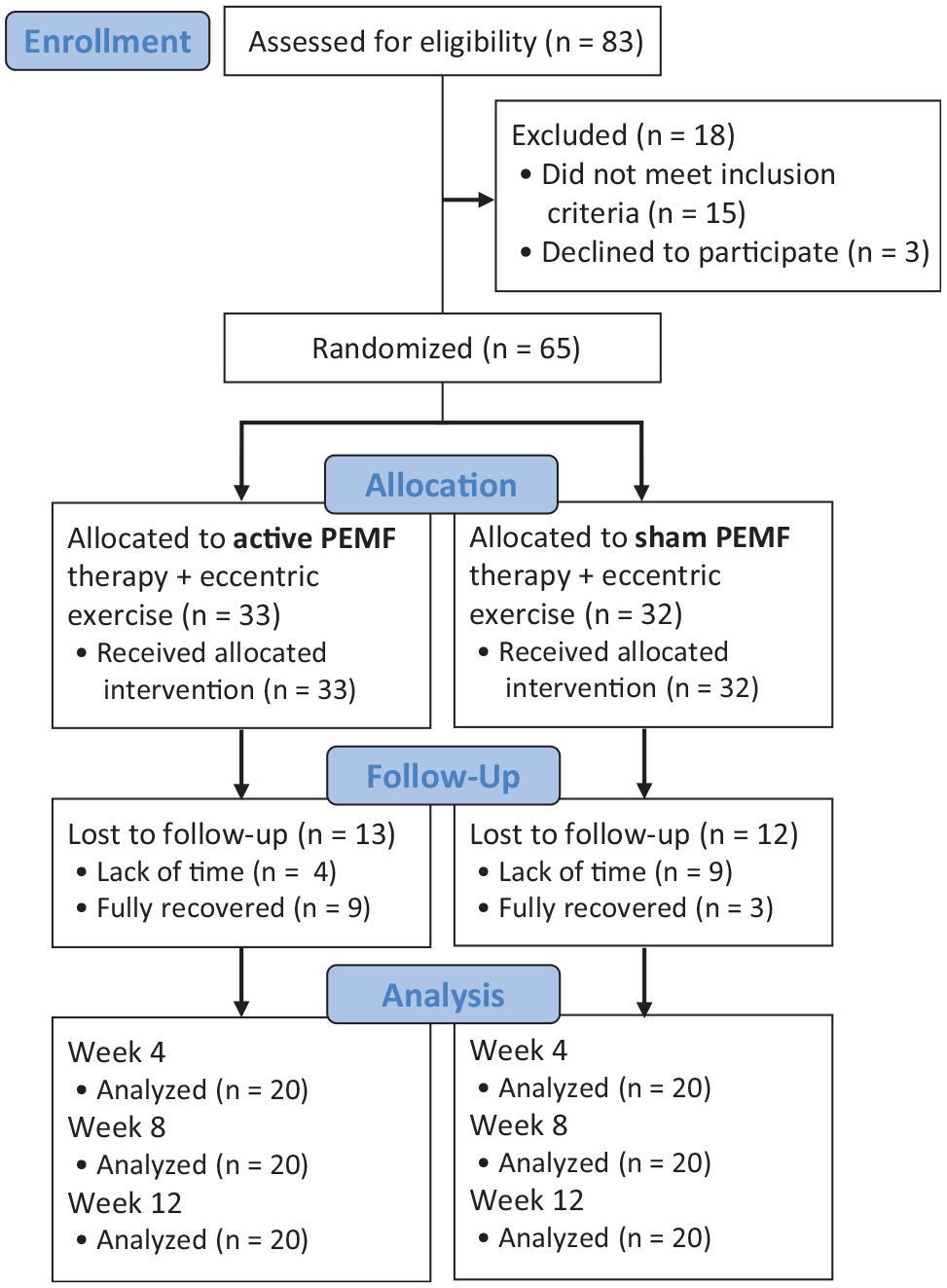
Flowchart of the study population throughout the study. PEMF, pulsed electromagnetic field.
Table 1 presents the baseline characteristics of the groups. Statistical analysis using 2-way repeated-measures ANOVA indicated no significant differences between the PEMF and sham groups in baseline VISA-A, NPRS-sports, and NPRS-worst scores or in tendon neovascularity. Table 2 presents the within-group and between-group differences for the effects of interventions for all outcome measures. The results of the subgroup analysis are shown in Appendix Figures A1 and A2.
Baseline Characteristics of the Study Participants a

Data are reported as mean ± SD or n (%). BMI, body mass index; PEMF, pulsed electromagnetic field.
VISA-A Scores at Baseline and 4, 8, and 12 Weeks of Follow-up in the Study Groups a

Sample sizes at 12-week follow-up: PEMF group (n = 20), sham group (n = 20). Data are presented as mean ± SD. Boldface P values indicate a significant difference between groups (P < .05, 2-way repeated-measures analysis of variance). PEMF, pulsed electromagnetic field; NPRS, numeric pain rating scale; VISA-A, Victorian Institute of Sports Assessment–Achilles.
VISA-A Scores
There was a significant improvement in VISA-A scores over time in both treatment groups (F = 12.811; P < .001), with scores improving from 57.85 at baseline to 71.70 in week 12 in the PEMF group and from 55.10 to 66.75 in the sham group. However, there were no significant differences between the treatment groups in VISA-A scores (F = 0.408; P = .527), and no significant group differences when analyzing scores based on age, sex, tendinopathy type, or adherence to eccentric exercise (Figure 3).

Victorian Institute of Sports Assessment–Achilles (VISA-A) scores at baseline and 4, 8, and 12 weeks posttreatment in the pulsed electromagnetic field (PEMF) and sham groups. Data represent mean ± SD; PEMF group (n = 20), sham group (n = 20). *Significant difference versus baseline (P < .05, 2-way repeated-measures analysis of variance).
Pain Scores
There were significant within-group improvements in NPRS-sports (F = 27.727; P < .001) and NPRS-worst (F = 24.007; P < .001) scores. NPRS-sports scores decreased (ie, improved) from 6.05 at baseline to 2.95 at 12 weeks in the PEMF group and from 6.45 to 3.85 in the sham group. NPRS-worst scores decreased from 6.70 at baseline to 3.60 at 12 weeks in the PEMF group and from 7.25 to 4.30 in the sham group. No significant group differences were found in NPRS-sports (F = 0.912; P = .346) or NPRS-worst (F = 1.345; P = .253) scores.
Regarding the subgroup analysis of the 13 participants with midportion tendinopathy, there was a significant group difference in NPRS-sports scores at week 4 (PEMF: 2.30 vs sham: 4.60; P = 0.009) and week 12 (PEMF: 1.67 vs sham: 5.14; P = .029) (Figure 4A). There was also a significant group difference in NPRS-worst scores at week 4 (PEMF: 2.90 vs sham: 5.80; P = .048) (Figure 4B).
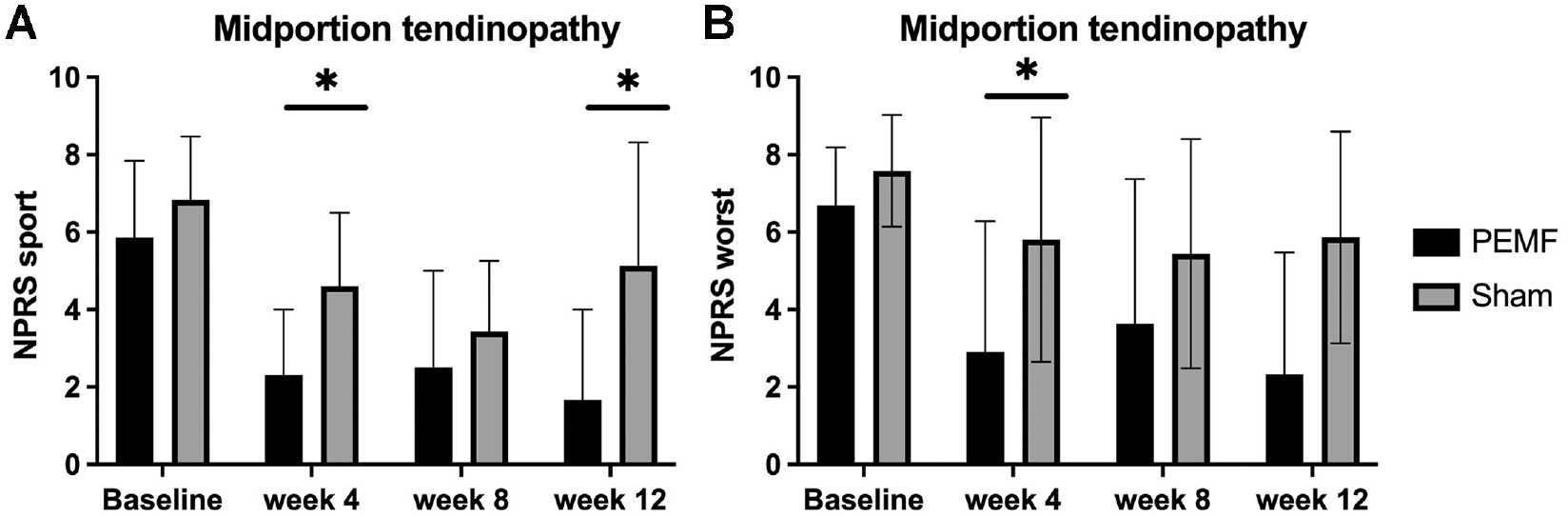
Comparison of (A) numeric pain rating scale (NPRS)–sports and (B) NPRS-worst scores between treatment groups among participants with midportion tendinopathy. Data represent mean ± SD; pulsed electromagnetic field (PEMF) group (n = 6), sham group (n = 7). We recruited 25 participants with midportion tendinopathy. However, due to dropouts, the number was reduced to 13 at week 12 of the follow-up assessment. *Significant group difference (P < .05, Mann-Whitney U test).
Tendon Neovascularity
Overall, there was a significant reduction in tendon neovascularity measured using ultrasound assessment for all participants (Figure 5). No significant group differences were observed in tendon neovascularity throughout the study period. According to the subgroup analysis, there was a significant group difference in Öhberg scores among the 13 participants in the middle-aged group (31-45 years) at week 8 (PEMF: 0.20 vs sham: 1.25; P = 0.023) (Figure 6A). In addition, a significant difference in Öhberg scores at week 8 was found among the 22 male participants included in the subgroup analysis (PEMF: 0.59 vs sham: 1.30; P = .043) (Figure 6B). We recruited 33 male participants, however, at week 12 of the follow-up assessment, it was reduced to 22 after dropout.

Longitudinal ultrasound images with color Doppler demonstrating changes in tendon neovascularity. (A) Tendinopathic Achilles tendon that received active pulsed electromagnetic field (PEMF) therapy showing severe neovascularity (Öhberg score, 3) during baseline assessment. (B) Same tendon as in panel A, showing mild neovascularity (Öhberg score, 1) at the 8-week assessment. (C) Tendinopathic Achilles tendon that received sham PEMF therapy showing severe neovascularity (Öhberg score, 3) at baseline assessment. (D) Same tendon as in panel D, still showing severe neovascularity (Öhberg score, 3) at the 8-week assessment.

Comparison of Öhberg scores between treatment groups among (A) middle-aged participants (age, 31-45 years; pulsed electromagnetic field (PEMF) group [n = 7], sham group [n = 6]) and (B) male participants (PEMF group [n = 14], sham group [n = 8]). Data represent mean ± SD. *Significant group difference (P < .05, Mann-Whitney U test).
Sports Participation
All participants increased the time spent on sports participation from baseline to 12 weeks (F = 9.534; P < .001), with no significant differences between the PEMF and sham groups (F = 0.323; P = .573). During subgroup analysis based on participants with midportion tendinopathy (n = 25), sports participation at 12 weeks was found to be significantly greater in the PEMF group (184.00 vs 47.14 min/wk in the sham group; P = .03) (Figure 7).

Comparison of time spent on sports participation per week between treatment groups among participants with midportion tendinopathy. Data represent mean ± SD; pulsed electromagnetic field (PEMF) group (n = 6), sham group (n = 7). We recruited 25 participants with midportion tendinopathy. However, due to dropouts, the number was reduced to 13 at week 12 of the follow-up assessment. *Significant group difference (P < .05, Mann-Whitney U test).
Adverse Effects
No adverse events occurred in the study.
Discussion
The main finding of this randomized controlled trial (RCT) was that adding PEMF therapy did not improve outcome scores over exercise alone. Both the PEMF and sham groups saw significant improvement in VISA-A scores from baseline to 12-week posttreatment (13.85- and 11.65-point increases, respectively), which exceeded the minimal clinically important difference for the VISA-A (6.5 points), 27 with no difference between groups. All participants demonstrated a significant decrease in tendon pain and tendon inflammation measured using color Doppler ultrasound after the 12-week intervention. Subgroup analysis of participants with midportion tendinopathy indicated that those who received PEMF therapy spent more time on sports at 12 weeks after treatment compared with the sham group (P = .03). To our knowledge, this is the first RCT to investigate the effects of PEMF therapy on Achilles tendinopathy with a published study protocol. 21
Subgroup analyses in patients with midportion Achilles tendinopathy showed significant differences between the PEMF and sham groups in NPRS-sports scores at week 12 and in NPRS-worst scores at week 4. Thus, active PEMF plus eccentric exercise yielded synergistic clinical benefits in pain reduction for midportion tendinopathy. Concurrent with the clinical improvement in those participants with midportion tendinopathy, PEMF therapy increased the participants’ weekly time spent on sports. Subgroup analyses in male participants and middle-aged (31-45 years) participants showed a significantly lower degree of tendon inflammation classified by Öhberg score in the PEMF group at 8 weeks.
The clinical effects of PEMF therapy on pain reduction are supported by a preliminary study by Gerdesmeyer et al 10 that compared the clinical effects of PEMF therapy with extracorporeal shock wave therapy (ESWT) for midportion Achilles tendinopathy. These authors reported a significant decrease in pain scores at the 12-week assessment point in the PEMF group compared with the control group, who were treated by ESWT. Both treatment groups in our study exhibited improved within-group NPRS-worst scores. In the PEMF group, pain scores decreased from 6.70 at baseline to 3.60 at 12 weeks, and in the sham group, scores decreased from 7.25 to 4.30.
Our results also agree with a previous study using this same PEMF technology for an older cohort of community dwellers, whereby pain relief was significantly detected in the participants with preexisting pain after 3 months of PEMF intervention. 45 The mean pain scores improved from 6.57 to 4.45 for participants with pretreatment of pain >5 points on the 10-point scale. The authors reported correlations between 12 weeks of PEMF therapy and improvements in physical activity measured using the mobility tests.
The benefits of electromagnetic field therapy have been demonstrated in other types of tendinopathy. A notable evidence level 1 RCT by Klüter et al 19 provided evidence for a distinct management effect of PEMFs in rotator cuff tendinopathy. In total, 86 patients with rotator cuff tendinopathy were randomized to undergo ESWT sessions combined with either electromagnetic transduction therapy (EMTT) or sham EMTT. 19 Both groups had significantly reduced pain and improved shoulder function, with the EMTT group having more significant improvements in pain scores compared with the sham EMTT after 24 weeks. 19 Future research may investigate the effects on tendons after different delivery modalities and doses of electromagnetic field therapy.
Eccentric exercise was chosen to be part of the treatment program in our study, as it is the most common nonoperative treatment among various Achilles tendinopathy treatments.20,32 Overall, eccentric exercise with or without PEMF therapy had similar effects in improving self-reported pain and function, tendon inflammation, and time spent in sports participation. Similar within-group improvements were observed in participants receiving PEMF therapy plus eccentric exercise and eccentric exercise only throughout the study period. There were no significant differences in outcome scores between the PEMF and sham groups after the 12-week intervention. Two recent systematic reviews have been published that investigated the effects of eccentric exercise on midportion Achilles tendinopathy 31 and insertional Achilles tendinopathy. 20 Prudêncio et al 31 compared eccentric exercise with another type of exercise or control group (rest/wait-and-see), which supported using eccentric exercise as an integral component in managing midportion Achilles tendinopathy. One of the included studies, by Rompe et al, 34 reported a successful outcome for 50% to 60% of participants in the eccentric exercise group. The authors concluded that eccentric exercise was more effective than other exercises in treating midportion Achilles tendinopathy. In another systematic review by Ko et al, 20 6 RCTs were included to compare eccentric exercise with other interventions for insertional Achilles tendinopathy. All included studies9,13,18,26,27,30 reported a significant within-group reduction in tendon pain and improvements in VISA-A scores after the 12-week eccentric exercise program.
Limitations
There are several limitations to the current study. First, tissue sample analyses may have better allowed the detection of changes in collagen content, cross-link composition, and fibril composition of the Achilles tendon associated with PEMF treatment. Biopsy collection was not possible within the confines of the study. Second, our RCT relied on community recruitment and represented a heterogeneous group of recreational athletes or sedentary individuals. Selective recruitment of professional athletes or sedentary individuals would have improved the generalizability of the results. Third, this study reported only the short-term consequences of PEMF plus eccentric exercise on Achilles tendinopathy. Future studies should investigate the carryover longer-term effects of PEMF therapy.
A fourth limitation was that, although participants were completely blinded to the PEMF treatment, the loss to follow-up was differential across the study groups: 31% for the sham group and 5% for the intervention group. Fifth, direct exposure to the injury site may not have been the most effective therapeutic approach with this technology. Previous studies have shown that direct PEMF exposure may aggravate inflamed tissues, while metabolic improvements were observed at the systemic level. 38 It may have thus proven more effective to invoke a cross-feeding therapeutic strategy by exposing healthy neighboring muscle or the contralateral leg, if healthy. This precaution arises from the mitohormetic characteristics of this specific form of magnetic therapy, whereby low levels of oxidative stress are used as an adaptive stimulus to invoke secretome activation with systemic regenerative and metabolic consequences.28,48,45,47 Finally, the Öhberg score lacks a display of the magnitude of vessels; a quantitative evaluation of the area of vessels may help explore the role of vascularization in the context of Achilles tendinopathy.
Conclusion
PEMF therapy, in addition to eccentric exercise, did not improve outcome scores or tendon neovascularity compared with eccentric exercise alone in participants with Achilles tendinopathy. Both the PEMF and sham groups showed statistical and clinically significant improvement in VISA-A scores after 12 weeks. Further research is required to determine the optimal therapeutic approach of PEMF therapy in the management of Achilles tendinopathy.
Footnotes
Appendix
Final revision submitted March 14, 2024; accepted April 5, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was fully funded by the Health and Medical Research Fund (HMRF), Food and Health Bureau of the Government of Hong Kong SAR (reference No. 10210186). A.F.O. holds a patent (WO 2019/17863 A1; System and Method for Applying Pulsed Electromagnetic Fields) and is a cofounder of QuantumTX Pte Ltd. S.K.K.L. has received consulting fees from Smith & Nephew. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Joint Chinese University of Hong Kong–New Territories East Cluster (reference No. 2021.150-T).
