Abstract
Background:
Deltoid muscle detachment and atrophy have been reported to occur after shoulder surgery.
Purpose:
To investigate the 2-year changes in deltoid muscle structure and function after arthroscopic rotator cuff repair (ARCR) using magnetic resonance imaging (MRI) and electrophysical examination.
Study Design:
Case series; Level of evidence, 4.
Methods:
A total of 72 patients (72 shoulders) who underwent ARCR between 2015 and 2020 were enrolled. Whole deltoid muscle volume and regional (anterior, lateral, and posterior) muscle thicknesses were determined on T2-weighted MRI scans of both shoulders taken preoperatively and at 1, 3, 6, 12, and 24 months postoperatively, and their correlations with compound muscle action potentials (CMAPs), shoulder abduction muscle strength, and Constant scores were investigated. Comparison between groups was performed using paired or Student t tests, and the relationship between deltoid muscle volume and various factors was determined using Pearson correlation analysis.
Results:
The volume of the deltoid muscle on the affected side decreased from 44,369 ± 12,371 mm3 preoperatively to 38,139 ± 10,615 mm3 at 1 month postoperatively (P < .05), representing a 14% decrease. The deltoid muscle volume of the contralateral side also significantly decreased during the same time frame, from 43,278 ± 12,248 to 40,273 ± 11,464 mm3 (P < .05), representing a 7% decrease at 1 month postoperatively. Subsequently, the deltoid muscle volume on both sides recovered to preoperative levels at 12 months and was maintained at 24 months. Only the thickness of the anterior part of the deltoid was markedly decreased, from 13.9 ± 3.7 mm preoperatively to 12.0 ± 3.2 mm at 1 month postoperatively (P < .05), representing a 14% reduction. The CMAP amplitude showed a significant decrease at 1 month postoperatively; however, no significant difference was observed after 12 months when compared with the preoperative values or the values on the contralateral side. Positive correlations were found between deltoid muscle volume and CMAP amplitude at 24 months as well as between deltoid muscle volume and shoulder abduction muscle strength (R2 = 0.698; P < .05) and Constant score (R2 = 0.133; P < .05).
Conclusion:
Our study demonstrated that the early structural and functional decline of the deltoid muscle after ARCR was fully recovered within 1 year, confirming that this procedure does not negatively affect the deltoid muscle.
The deltoid muscle originates from the anterior border of the lateral one-third of the clavicle, the lateral border of the acromion, and the posterior border of the spine of the scapula.14,23 The deltoid muscle stops at the deltoid tuberosity in the middle of the lateral humerus and contributes to shoulder flexion, abduction, and extension and further functions as a dynamic stabilizer of the glenohumeral joint.13,16,20,22 Deltoid muscle detachment and atrophy have been reported to occur after various shoulder surgeries, such as rotator cuff repair, shoulder arthroplasty, or acromioplasty.4,7-10 Recently, deltoid preservation has become increasingly considered, as a functioning deltoid is required for reverse total shoulder arthroplasty (RTSA), which is increasing in popularity. 3
The purpose of the present study was to investigate the structural and functional alterations of the deltoid muscle after arthroscopic rotator cuff repair (ARCR) using magnetic resonance imaging (MRI) and electrophysical examination. We hypothesized that ARCR would be a minimally invasive procedure for the deltoid muscle.
Methods
Patients
This retrospective study was approved by the ethics committee of our hospital. A total of 102 consecutive patients with rotator cuff tears who underwent arthroscopic repair between 2015 and 2020 were reviewed. Thirty patients were excluded because of ARCRs on the contralateral shoulder during the observation period (n = 8), loss to follow-up (n = 6), incomplete data (n = 5), reoperation on the affected shoulder caused by infection or pullout of suture anchors (n = 4), retearing of the repaired rotator cuff (n = 3), cardiac pacemaker implantation (n = 3), and a history of cervical spine surgery (n = 1). Ultimately, 72 patients (46 male and 26 female) were included in this study.
The medical records, MRI scans, electrophysiological examinations, and muscle strength data of the patients were investigated. These examinations were performed before surgery and at 1 month (mean, 30.6 days; range, 30-57 days), 3 months (mean, 91.5 days; range, 87-127 days), 6 months (mean, 184.9 days; range, 178-211 days), 12 months (mean, 367.4 days; range, 358-400 days), and 24 months (mean, 736.7 days; range, 710-827 days) postoperatively. The patients’ age at the time of surgery was a mean of 64.1 years (range, 43-78 years). The affected side was the right shoulder in 48 patients and the left shoulder in 24 patients. The right hand was dominant in 50 patients and the left in 22 patients. The tear size, according to the Cofield classification, 5 was small in 4 cases, moderate in 45 cases, large in 18 cases, and massive in 5 cases. Arthroscopic subacromial decompression was performed in 62 patients (86.1%). ARCR was performed using the single-row, double-row, or suture-bridge technique with suture anchors. Postoperatively, all patients kept their arms abducted using an UltraSling IV (ENOVIS) device for 6 weeks. Passive shoulder range of motion was initiated 3 weeks postoperatively; active shoulder range of motion was allowed after brace removal, light work after 3 months, and heavy work after 6 months.
Radiologic Evaluation
All patients underwent MRI via a 1.5-T imaging system (Magnetom Avanto, Siemens Healthineers) preoperatively and at 1, 3, 6, 12, and 24 months postoperatively. The sections had a thickness of 6 mm, and a reference line was drawn on the undersurface of both acromions on the coronal images. T2-weighted axial images (repetition time/echo time, 3700/114 ms) were used to calculate the deltoid muscle volume. MRI scans were imported into ImageJ software (US National Institutes of Health), and the length and units were configured based on the measurements embedded in the images. The polygon-selection tool was used to manually outline the deltoid muscle, and the area was automatically quantified. The same procedure was carried out on all slices from the acromion to the distal deltoid muscle attachment, and the volume was calculated using the following formula: Deltoid Muscle Volume (mm3) = Sum of All Measured Muscle Areas (mm2) × 6 mm (Slice Thickness) × (Number of Slices– 1) (Figure 1).

(A) T2-weighted coronal magnetic resonance imaging (MRI) measurement of the deltoid muscle volume of both shoulders (between the dashed lines). (B) Cross-sectional area of the left deltoid muscle on T2-weighted axial MRI at the white reference line in (A).
The deltoid muscle thickness (at the center level of the glenoid) was measured following the method of Cho et al. 4 The muscle belly thicknesses of the anterior, lateral, and posterior deltoids were measured on axial T2-weighted MRI scans. In brief, the first guideline was drawn through the anterior and posterior edges of the glenoid. The second guideline was drawn parallel to the first guideline and through the center of the humeral head, after which the anterior and posterior muscle thicknesses were measured. The third guideline was drawn perpendicularly to the second guideline and through the center of the humeral head to measure the middle deltoid thickness (Figure 2).

Measurement of the deltoid muscle thickness at the center of the glenoid level. (A) T2-weighted coronal magnetic resonance imaging (MRI) scan of both shoulders. Axial MRI slices along the white reference line were used to measure the deltoid muscle thickness. (B) The anterior, lateral, and posterior deltoid muscle thicknesses as indicated on axial MRI scan.
The inter- and intratester reliabilities of the measurements were assessed using the intraclass correlation coefficient (ICC). A random sample of 10 left shoulder MRI scans was selected from the data of the 72 patients. Each measurement was taken twice by 2 authors (T.S. and K.Y.) at a minimum of 1-week intervals. Based on the 95% CIs of the ICC estimate, values <0.5, between 0.5 and 0.74, between 0.75 and 0.9, and >0.9 indicate poor, moderate, good, and excellent reliability, respectively.
Electrophysiological Evaluation
Using Erb point stimulation, we recorded compound muscle action potentials (CMAPs) in the deltoid muscles of all participants using a Neuropack X1 (Nihon Kohden) preoperatively and at 1, 3, 6, 12, and 24 months of participation. 6 Briefly, a 10 mm–diameter disk was placed in the middle of the lateral deltoid (6 cm distal to the acromion) as the working electrode, and a reference electrode was placed at the acromion (Figure 3A). The skin was treated with an abrasive solution to reduce impedance, and a ground strap was wrapped around the wrist. The bipolar stimulation probe provided a pair of bare metal contacts whose diameter was 3 mm, with a distance of 20 mm between the electrodes. The stimulus rate was set at 1 Hz, and the stimulation duration was set at 0.2 ms. The stimulus intensity, from a minimum of 20 mA, was gradually increased until the magnitude of the recorded response no longer changed. The CMAP measurements included a baseline-to-peak negative peak amplitude.

(A) Measurement of deltoid compound muscle action potentials with Erb point stimulation. (B) Measurement of shoulder abductor muscle strength.
Muscle Strength Evaluation
Bilateral shoulder abductor muscle strength measurements were performed in 49 patients in the medical record. Shoulder abduction muscle strength, which entailed the entire muscle rather than each section (anterior, middle, and posterior), was measured bilaterally using a handheld dynamometer (microFET2; Hoggan Scientific), with the patient sitting on a chair and the shoulder positioned at 90° of abduction at 24 months postoperatively (Figure 3B). The examiner fixed the dynamometer against the patient's elbow and obtained the reading while the patient abducted the shoulder as strenuously as possible against the device. This is the so-called “make test.”2,11 The maximal isometric strength was measured 3 times bilaterally. The mean force (in newtons) was calculated and then multiplied by the distance (in meters) between the center of the humeral head and the device to obtain the moment (N·m).
Statistical Analysis
A paired t test was performed to assess the differences in the preoperative and postoperative parameters of the deltoid muscle in each group. The Student t test was used to compare postoperative alterations in the deltoid muscle between the affected and contralateral sides. The Pearson correlation coefficient was used to determine the correlation between the deltoid muscle volume and clinical factors. Data are presented herein as the mean ± standard deviation, and a P value of <.05 was considered statistically significant. GraphPad Prism software (Version 9.1.1; GraphPad Software) was used for all statistical analyses.
Results
The ICC values for deltoid muscle volume showed excellent intratester reliability (ICC = 0.996; 95% CI, 0.983-0.999) and intertester reliability (ICC = 0.997; 95% CI, 0.991-0.999). The volume of the affected deltoid muscle was markedly reduced at 1 month postoperatively compared with preoperatively, from 44,369 ± 12,371 to 38,139 ± 10,615 mm3 (86.0%) (P < .05) (Figure 4). The volume improved over time, fully recovered at 1 year, and was maintained at the near-preoperative level at 2 years postoperatively (44,369 ± 12,371 vs 44,793 ± 12,469 mm, respectively; 102.3%). Interestingly, the deltoid muscle volume of the contralateral side also decreased from preoperatively to 1 month postoperatively, from 43,278 ± 12,248 to 40,273 ± 11,464 mm3 (93.0%) (P < .05) and recovered to 43,577 ± 12,219 mm3 (100.7% of preoperative level) at 2 years postoperatively.

Changes over time in the deltoid muscle volume of both shoulders relative to the preoperative (preop) value. Error bars represent standard deviations. Statistically significant difference (P < .05) *between the affected and contralateral sides, #compared with the preoperative value on the affected side, and §compared with the preoperative value on the contralateral side.
Similarly, the CMAP amplitude of the affected deltoid muscle decreased from 11.3 ± 2.5 mV preoperatively to 7.1 ± 2.0 mV at 1 month (65.3%) and recovered to 10.5 ± 2.3 mV at 12 months postoperatively (P < .05) (Figure 5). This amplitude was still maintained at 10.9 ± 2.3 mV (97.2%) at 2 years postoperatively. However, during the observation period, no change was observed in the CMAP amplitude of the unaffected side.

Changes over time in the amplitude of compound muscle action potentials (CMAPs) of both shoulders relative to the preoperative (preop) value. Error bars represent standard deviations. Statistically significant difference (P < .05) *between the affected and contralateral sides and #compared with the preoperative value on the affected side.
A positive correlation was found between the entire deltoid muscle volume and CMAP amplitude of the affected side at 1 month postoperatively (R2 = 0.1585; P < .001) and 24 months postoperatively (R2 = 0.3416; P < .0001) (Figure 6). Furthermore, a positive correlation was found between the volume of the deltoid muscle on the affected side and shoulder abduction muscle strength (R2 = 0.698; P < .05) and Constant score (R2 = 0.133; P < .05) 24 months postoperatively (Figure 7).

Relationship between deltoid muscle volume on the affected side and compound muscle action potential (CMAP) amplitude at (A) 1 month and (B) 24 months postoperatively.
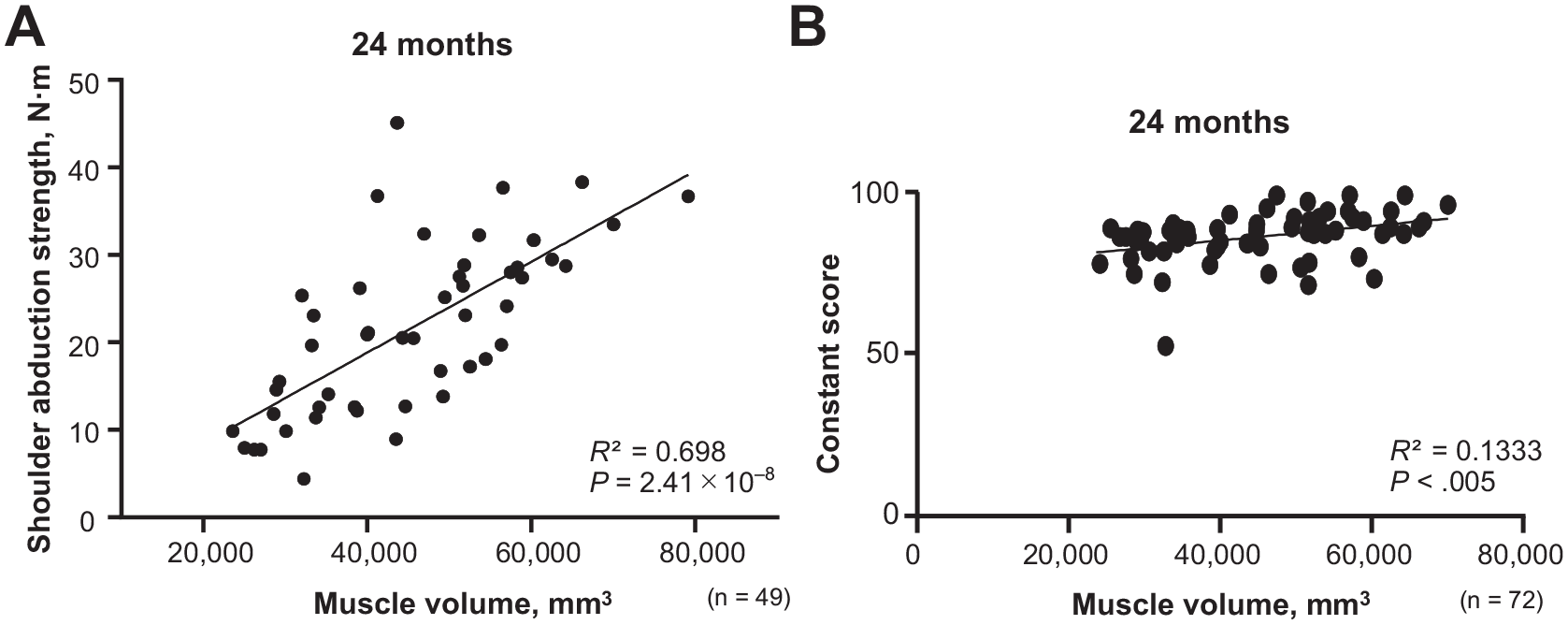
Relationship between deltoid muscle volume on the affected side and (A) shoulder abduction muscle strength and (B) Constant score at 24 months postoperatively.
Finally, the muscle belly thicknesses of the anterior, lateral, and posterior deltoids were measured to assess site-specific muscle alterations because it was difficult to divide the volume into anterior, lateral, and posterior compartments. The ICC values for deltoid muscle belly thickness showed excellent intratester reliability (0.987; 95% CI, 0.949-0.997) and intertester reliability (0.984; 95% CI, 0.938-0.996). From our measurements, only the thickness of the anterior deltoid was markedly decreased, from 13.9 ± 3.7 to 12.0 ± 3.2 mm (86.2%), at 1 month postoperatively (P < .05), and it recovered to 14.1 ± 4.1 mm (101.7%) by 2 years postoperatively (Figure 8A). Lateral and posterior deltoid muscle thicknesses were not significantly altered throughout the observation period (Figures 8, B and C).

Changes over time in the (A) anterior, (B) lateral, and (C) posterior deltoid muscle thickness of both shoulders. Error bars represent standard deviations. *Statistically significant difference (P < .05) compared with preoperative (preop) value.
Discussion
The most important finding of the present study was that the volume and CMAP amplitude of the affected deltoid muscle were significantly reduced 1 month after ARCR, from 44,369 ± 12,371 to 38,139 ± 10,615 mm3 (P < .05) and from 11.3 ± 2.5 to 7.1 ± 2.0 mV (P < .05), respectively; recovered at 1 year postoperatively; and were maintained at 2 years postoperatively. Several studies have reported that the deltoid muscle area or thickness is reduced within 3 to 6 months after rotator cuff repair; in fact, we confirmed that the decrease in muscle volume had already occurred 1 month postoperatively.10,15 We believe that this early muscle atrophy was mainly caused by 2 factors: namely, surgical invasion and immobilization with an orthosis.
One of the reported advantages of ARCR is the avoidance of deltoid muscle detachment from the acromion,17,24,26,27 because deltoid detachment has been reported as a complication of rotator cuff repair using a mini-open and deltopectoral approach.7,8,21 In particular, the deltoid detachment/repair technique, in which the rotator cuff is well observed, is highly invasive, and the deltoid is at risk of retearing after repair.8,21 However, recent studies have reported no difference in detachment of the deltoid muscle between mini-open and arthroscopic approaches.4,9 Thus, the differences in surgical techniques regarding postoperative deltoid detachment are still controversial. In the present study, arthroscopic subacromial decompression was performed in 86.1% of the patients. Portal creation was performed in all patients, but there were no cases of detachment, probably because only the basal and marginal osteophytes of the anterior half of the acromion were excised in order to minimize surgical invasion of the deltoid muscle belly.
Immobilization after rotator cuff repair has been found to induce deltoid muscle atrophy. In experimental animal studies, Max et al 18 reported a weight loss of 30% in rat gastrocnemius muscle after only 3 days of immobilization and a 50% loss at 15 days. Appell 1 reported that the greatest muscle atrophy occurred during the early phases of immobilization and that only a slight further reduction in fiber diameter occurred after 1 week. Therefore, the majority of the early muscle atrophy observed in the present study is likely due to disuse atrophy associated with 6 weeks of brace immobilization rather than surgical invasion.
A unique feature of this study is the measurement of CMAPs to assess deltoid muscle function, because there were no previous reports on the correlation between deltoid muscle volume and CMAP alterations after shoulder surgery. CMAPs are the sum of muscle fiber action potentials generated synchronously by activating a motor unit, which consists of motor neurons and skeletal muscle fibers. CMAP amplitudes obtained by stimulating the muscle fibers below the lesion after injury determine the degree of axonal loss and the prognosis for cervical spondylotic myelopathy/amyotrophy; however, little information is available on the significance of CMAPs in disuse muscle atrophy.5,12,25 Similar to our data, the findings by Mobach et al 19 in 2020 showed that 6 weeks of immobilization of the hand in a cast after distal radius fracture caused a reduction in CMAP amplitude for the abductor pollicis brevis (29.2%), abductor digiti minimi (19.0%), and first dorsal interosseous (24.9%). The reduction in deltoid CMAP amplitude may be primarily due to disuse atrophy rather than nerve palsy. The other novelty of the present study is that changes in the volume and CMAP amplitude of the deltoid muscle on the contralateral side were monitored for 2 years after ARCR. Interestingly, deltoid muscle atrophy was also observed on the unaffected side, whereas the CMAP amplitude did not decrease remarkably. We can only speculate that the muscle volume might have decreased because the frequency of use of the deltoid muscle on the healthy side also decreased because of limitations in the activities of daily living. Why the CMAP amplitude on the unaffected side did not decrease remains unclear.
Limitations
This study has several limitations. First, the sample size was small. Second, intramuscular fatty infiltration and fibrosis were ignored in the MRI measurements. Third, the MRI slice thickness was large (6 mm), and the deltoid volume was not examined separately for the anterior, middle, and posterior deltoid muscles in this study because it was difficult to distinguish among those fibers. Also, it was difficult to test isolated muscle strength with our methods. Future studies that directly compare ARCR with open surgery are needed to prove the minimally invasive nature of ARCR for the deltoid muscle.
Conclusion
Deltoid muscle volume after ARCR significantly decreased at 1 month postoperatively and later recovered. Moreover, the deltoid muscle volume at the 2-year follow-up was positively correlated with the CMAP amplitude, shoulder abduction strength, and clinical score. This study confirmed that ARCR does not negatively affect the deltoid muscle and minimizing deltoid injury after rotator cuff repair may improve outcomes.
Footnotes
Final revision submitted February 26, 2024; accepted March 15, 2024.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Ogori Daiichi General Hospital (ref No. 16-11).
