Abstract
Background:
Autologous chondrocyte implantation (ACI) and matrix-induced autologous chondrocyte implantation (MACI) are 2-stage procedures requiring an index full-thickness cartilage biopsy. Only a portion of patients ultimately undergo second-stage ACI/MACI.
Purpose:
To identify patients with articular cartilage defects who underwent arthroscopic debridement with biopsy for ACI/MACI and compare those who did with those who did not proceed with implantation within 2 years after biopsy. Additionally, the authors sought to identify why patients did not proceed with implantation.
Study Design:
Case-control study; Level of evidence, 3.
Methods:
Patients who underwent arthroscopy and autologous chondrocyte biopsy from January 1, 2015, to December 31, 2019, and who had minimum 2-year follow-up data were grouped into those who proceeded with second-stage ACI/MACI (implant group; n = 97) and those who did not (biopsy group; n = 63). Demographic factors, cartilage defect characteristics, and preoperative International Knee Documentation Committee (IKDC) scores were analyzed. Patients in both groups were evaluated postoperatively using the IKDC, Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), Single Assessment Numeric Evaluation (SANE), and visual analog scale (VAS) for pain, and patients who did not undergo implantation were asked for their reasoning.
Results:
Body mass index (BMI) (P < .001) and Outerbridge grades at index arthroscopy (P = .047) were significantly higher for the implant group than the biopsy group. Both groups had significantly improved IKDC scores from their initial presentation to final follow-up (implant group: 46.4 ± 16.2 preoperative vs 69.6 ± 20.6 postoperative [P < .001]; biopsy group: 47.2 ± 15.9 preoperative vs 70.7 ± 19.1 postoperative [P < .001]); however, the level of improvement did not differ significantly between groups. Postoperative WOMAC, SANE, and VAS pain scores were also similar between groups. In the biopsy group, 23 patients (37%) cited symptom resolution or activity level improvement after initial arthroscopy as the reason for not proceeding with implantation.
Conclusion:
Patients who proceeded to the second stage of chondrocyte implantation via either ACI or MACI had higher-grade articular defects and higher BMI compared with those who underwent biopsy with concomitant debridement chondroplasty alone. Postoperative outcomes were similar between the groups.
Keywords
Focal articular cartilage defects of the knee are exceedingly common 11 and present a difficult challenge for orthopaedic surgeons. Full-thickness osteochondral defects (OCDs) are associated with pain, swelling, and mechanical symptoms and may predispose patients to future osteoarthritis development.32,33 Symptomatic cartilage lesions leading to diminished knee function appear to progress over time, as articular cartilage has a poor intrinsic capacity for healing. Surgical options for treatment of these lesions are variable and include debridement, microfracture, autologous osteochondral transplantation, and cartilage regeneration.
Autologous cartilage implantation (ACI) was first performed in human patients in 1987. 5 The 2-stage procedure involves an index arthroscopic full-thickness cartilage biopsy, which is then transferred to a medium allowing for cellular expansion over a period of 3 to 5 weeks. The patient then returns for the second stage, which involves an open operation and direct implantation of the suspension of chondrocytes into a debrided chondral defect. 5 Originally, this involved the use of a periosteal patch that is harvested from the patient and sutured over the defect. The cultured cells are then injected under the periosteal patch into the defect. 28 The second generation of ACI was developed to address the complication of periosteal patch hypertrophy and improve the ease of the procedure. With this technique, the chondrocyte culture suspension is covered with a bioabsorbable types I and III porcine collagen membrane instead of a periosteal patch, which is then sewn over the defect. 28
ACI has subsequently been modified through development of biodegradable scaffolding seeded with chondrocytes able to be implanted via the matrix-induced autologous chondrocyte implantation (MACI) technique. 7 MACI has the added advantage of being minimally invasive, and as the implant is fixed with fibrin glue, it avoids the arduous process of sewing the periosteal patch. 10 Outcomes for these procedures are promising, with a recently published randomized controlled trial reporting that 83.3% of patients were satisfied with their ability to return to sport and 93.3% were satisfied with pain relief at 10 years. 9 ACI/MACI have also been shown to have a propensity for long-term durability, with a reported reoperation rate between 0.02% and 42.5% at 5 years.6,14,17,30,31,34 The rehabilitation process for ACI/MACI, however, is quite long; it has been reported that it may take up to 15 months before patients’ functional outcome is fully defined. 3
Typically, during the index surgery at the time of biopsy, the cartilage defect is debrided of any unstable flap or edge. There is a paucity of literature discussing the benefit of debridement for a small articular cartilage lesion, especially if the debridement was not part of a combined operative cartilage procedure including meniscal and ligamentous repair. 1 Patients who do not undergo the second stage of the ACI/MACI procedure are typically cited as “lost to follow-up”; however, specific patient and/or cartilage defect factors may influence the decision to not proceed with implantation. This may be due to defect size and/or location, baseline level of symptoms, concomitant knee pathology, social factors, or a combination of all of these.
The purpose of this study was therefore to retrospectively identify patients with articular cartilage defects who underwent arthroscopic debridement with biopsy for ACI or MACI and compare those patients who did with those who did not proceed with implantation. We evaluated clinical outcomes for each group at minimum 2-year follow-up. For those patients who did not undergo implantation, we additionally sought to identify the factors that influenced their decision.
Methods
Study Groups
Following institutional review board exemption, a list of patients who underwent arthroscopy with autologous chondrocyte biopsy (Current Procedural Terminology [CPT] codes 28970, 29874, and 29877) at a single institution from January 1, 2015, to December 31, 2019, was obtained from the institution's database. All patients underwent surgery by 1 of 6 fellowship-trained sports medicine orthopaedic surgeons (K.B.F, S.H.), all with >10 years of experience. The electronic medical records of the patients were reviewed, and sex, age at biopsy, and body mass index (BMI), as well as injury characteristics including laterality, duration of symptoms (subdivided into “acute,” or following an injury; and “insidious,” indicating a slow progression of pain without specific injury/incident), and prior treatment were recorded. All patients completed the International Knee Documentation Committee (IKDC) subjective knee form on initial preoperative consultation. All patients had preoperative magnetic resonance imaging showing ≥1 articular cartilage defect, and this was confirmed intraoperatively before obtaining a full-thickness cartilage biopsy for possible future ACI or MACI. Operative notes were reviewed for number of chondral defects, Outerbridge classification of defect, size of defect, and defect location. 25 Some patients had concomitant pathology and additional procedures performed at the time of biopsy, which were also recorded. Those who underwent subsequent ACI or MACI were identified (CPT code 27412), and the time between initial biopsy and implantation was recorded. Patients were evaluated for ≥2 years after undergoing either biopsy alone (biopsy group) or proceeding with ACI/MACI implantation (implant group). Complications, including infection, arthrofibrosis, venous thromboembolism, or any other perioperative surgical or medical complication requiring medical treatment, hospitalization, or return to surgery, were recorded.
Eligible patients were contacted to complete functional outcome surveys, which included the IKDC, Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), Single Assessment Numeric Evaluation (SANE), and visual analog scale (VAS) for pain. Those patients who had another surgical procedure to the knee between the time of biopsy (biopsy group) or implantation (implant group) were excluded from the functional outcome survey analysis. Patients in the biopsy group were also given a custom survey inquiring about the factors that led to their decision not to proceed with implantation (Appendix Figure A1). Patients who were unable to be contacted were excluded as well as those who filled out surveys incompletely.
Operative and Rehabilitation Protocol
Biopsy Technique
Full-thickness cartilage biopsy was obtained using an arthroscopic curette on either the superolateral portion of the intercondylar notch or the proximal, nonweightbearing portion of the medial or lateral femoral condyles. The biopsy was then transferred to a Cartilage Biopsy Transport Kit (Vericel) and subsequently shipped to the Vericel cell-processing facility in Cambridge, Massachusetts. At the time of index arthroscopy, the cartilage defect was visualized, sized, probed, and assessed and, if determined to be unstable or at risk of propagating, a debridement chondroplasty was performed. Unstable flaps were trimmed with a combination of arthroscopic curettage and shaving, with care taken to leave the subchondral plate intact. Patients were prescribed formal physical therapy for 6 weeks after surgery, which involved knee range-of-motion exercises, strengthening, and then return to functional activities.
Implantation Technique
For both ACI and MACI, a mini-arthrotomy was utilized to allow full visualization of the chondral defect in the appropriate compartment of the knee. The borders of the defect were then debrided to healthy, stable cartilage using a cutting template and ring curette. Patients undergoing ACI had a porcine collagen patch sutured over the defect using circumferential 6-0 Vicryl suture (Ethicon Inc.), initially leaving a small flap through which to inject the cultured cartilage cell medium. The patch was then secured with additional suture and sealed with fibrin glue. For patients undergoing MACI, the chondrocyte-seeded bioabsorbable scaffold was cut to size and appropriately positioned after the base of the chondral defect was layered with fibrin glue. Fibrin glue was then used to seal the edges of the implant. Postoperative rehabilitation protocols varied if concomitant procedures were performed at the time of implantation; however, they included ≥6 weeks of nonweightbearing.
Statistical Analysis
Continuous data were reported as means with standard deviations, and categorical data were reported as frequencies with percentages. The distribution of data was assessed with the Shapiro-Wilk test. Continuous parametric data distributed normally were compared between the biopsy and implant groups with the Student t test, while nonparametric data were compared with the Mann-Whitney U test. Categorical data were compared between the groups with the chi-square analysis. The threshold for significance was set at P < .05. All statistical analyses were performed with R Studio (Version 3.6.3).
Results
Over the study period, 90 patients were identified to have undergone a chondrocyte biopsy only (biopsy group) and 130 patients were identified to have undergone both biopsy and ACI or MACI (implant group). Of these, 63 patients (70%) in the biopsy group were available for follow-up at a mean of 3.5 years from initial biopsy, and 97 patients (75%) in the implant group were available for follow-up at a mean of 4.1 years after their initial biopsy and subsequent implantation. Implantation occurred an average of 20.9 ± 10.7 weeks after index biopsy. There was a significantly greater proportion of male patients (P < .001) and lower BMI (P < .001) in the patients in the biopsy group. Detailed baseline patient data are shown in Table 1.
Patient Demographics (N = 160 Patients) a
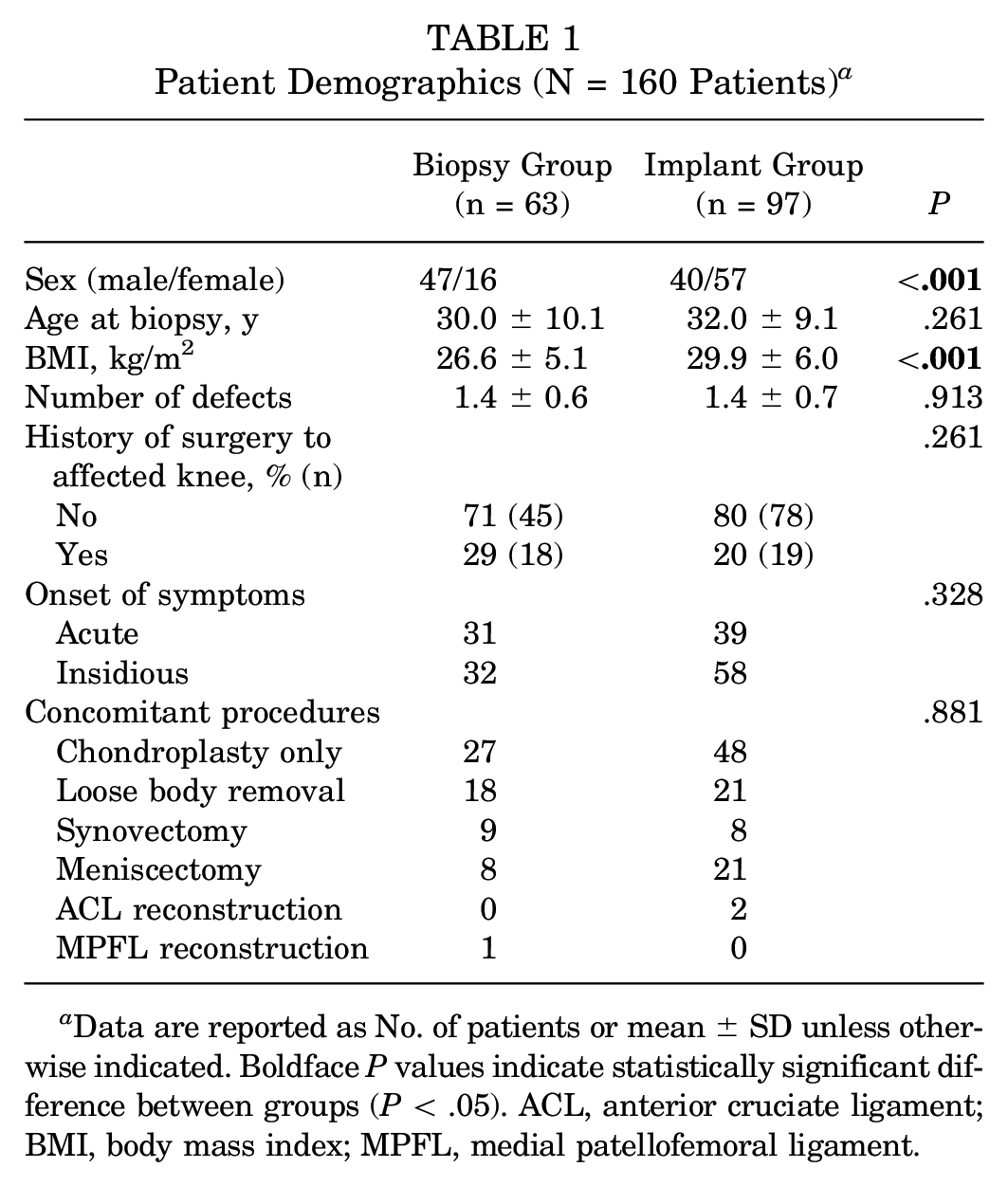
Data are reported as No. of patients or mean ± SD unless otherwise indicated. Boldface P values indicate statistically significant difference between groups (P < .05). ACL, anterior cruciate ligament; BMI, body mass index; MPFL, medial patellofemoral ligament.
In total, there were 91 chondral defects in the 63 patients in the biopsy group and 141 chondral defects across the 97 patients in the implant group. Index arthroscopy operative reports were reviewed, and characteristics of chondral defects in each group were analyzed (Table 2). There were no significant differences between the 2 groups in either the location of defect within the knee or mean size of defect; however, the defects of patients in the implant group had overall higher Outerbridge grades compared with those of patients in the biopsy group (P = .047).
Chondral Defect Characteristics (n = 232 Defects) a

Data are reported as % (n) or mean ± SD. Boldface P value indicates statistically significant difference between groups (P < .05). LFC, lateral femoral condyle; MFC, medial femoral condyle.
There were 13 patients (21%) in the biopsy group and 35 patients (36%) in the implant group with subsequent surgical procedures to the affected knee between the index biopsy/implantation and final follow-up (Table 3). These patients were included in the demographic and chondral defect characteristics analysis but excluded from the patient-reported outcome analysis to minimize confounding due to additional procedures. Both groups had significantly improved IKDC scores from their initial presentation to final follow-up (biopsy group: 47.2 ± 15.9 preoperatively, 70.7 ± 19.1 postoperatively [P < .001]; implant group: 46.4 ± 16.2 preoperatively, 69.6 ± 20.6 postoperatively [P < .001]); however, the level of improvement did not differ significantly between groups (24.4 ± 22.7, biopsy group; 27.0 ± 20.1, implant group [P = .608]). Patient-reported outcomes including the IKDC, WOMAC, VAS pain, and SANE did not significantly differ between groups (Table 4).
Subsequent Procedures in the Biopsy and Implant Groups a

MACI, matrix-induced autologous chondrocyte implantation; MPFL, medial patellofemoral ligament; TTO, tibial tubercle osteotomy.
One patient underwent 2 subsequent osteochondral allograft transplantation procedures.
One patient underwent 2 subsequent MACI procedures to separate lesions.
Patient-Reported Outcomes (n = 112 Patients) a
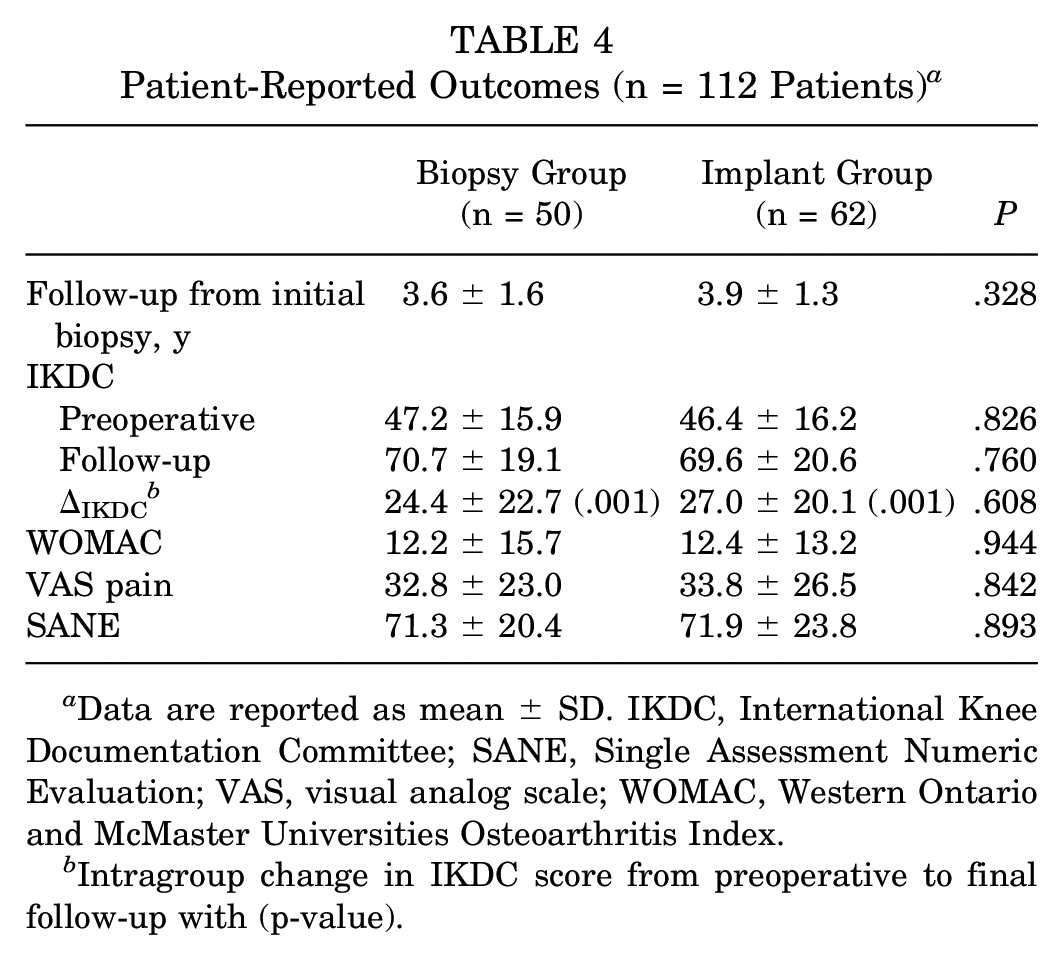
Data are reported as mean ± SD. IKDC, International Knee Documentation Committee; SANE, Single Assessment Numeric Evaluation; VAS, visual analog scale; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
Intragroup change in IKDC score from preoperative to final follow-up with (p-value).
When those who did not proceed to the second stage of implantation were asked to explain their reasoning, 23 patients (37%) cited symptom resolution or activity level improvement after initial arthroscopy, 16 patients (26%) reported that it was never scheduled, 8 patients (13%) cited reasons unrelated to their symptoms, 4 patients (6%) cited decreased activity level, and 3 patients (5%) cited physician cancellation; however, of these, 1 patient was recommended to undergo an osteochondral allograft transfer, 1 patient was recommended to continue nonoperative treatment until eventual total knee arthroplasty, and 1 patient had the implantation canceled due to the suspension of orthopaedic procedures during the COVID-19 pandemic and never rescheduled. Five patients (8%) cited “not sure,” and 17 patients (27%) cited “other.” Upon further review of the 25 patients who cited “other” and/or reported that implantation was never scheduled by their physician (8 patients cited both), 13 patients (21%) elected to proceed with different treatment options, including corticosteroid injection, viscosupplementation, osteochondral allograft transplantation, high tibial osteotomy, microfracture, and/or Fulkerson osteotomy; 11 patients (17%) deferred or declined the procedure due to the requisite recovery time, and the final patient was unable to be scheduled due to insurance and payment issues.
Discussion
The results of the study showed that while patients’ reasons for not returning for the second stage of the ACI/MACI procedure were variable, their postoperative functional outcome scores, including IKDC, WOMAC, VAS pain, and SANE, did not vary significantly from those who underwent both stages. Interestingly, both the biopsy and implant groups had significantly improved IKDC scores at final follow-up compared with preoperatively, with no difference in the level of improvement between the 2 groups. These findings indicate that while ACI/MACI does appear beneficial for some patients, proper patient selection is of paramount importance, as there exist several patients with articular cartilage defects who solely require debridement chondroplasty, either in isolation or with concomitant treatment of additional knee pathology to achieve equivalent results. The absence of an additional procedure could decrease costs and potential morbidity for this cohort while still improving knee symptoms, function, and outcomes.
While the benefits of ACI and MACI have been established, cellular expansion of the biopsy represents increased cost for patients who undergo implantation, and the staged nature of the procedure introduces increased risk. The findings of this study help define those patients who may benefit from ACI/MACI and those for whom the procedure may not be necessary. All patients underwent debridement chondroplasty at the time of biopsy, and several patients underwent concomitant procedures. Debridement chondroplasty in isolation has been shown to be beneficial for patients with focal cartilage lesions, as a recent case series demonstrated clinically meaningful improvements in IKDC, WOMAC, Lysholm, and Knee injury and Osteoarthritis Outcome Score values after 31.5 months. 1 Despite the large number of chondroplasty procedures performed in the United States, a direct comparison between the procedure and ACI/MACI does not yet exist, and while ACI and MACI have been shown to be beneficial in patients with larger and higher grade OCD lesions, there is not yet high-quality evidence showing long-term benefit over debridement and chondroplasty.6,15,18 An ongoing randomized controlled trial will seek to answer this question and will likely help guide surgical decision-making in the years to come. 27
Debridement chondroplasty, with or without concomitant procedure(s), appears to have adequately treated the primary generator of patients’ knee symptomatology in the biopsy group, given the improvement in IKDC from preoperative scoring to final follow-up as well as the fact that there were no significant differences in outcome between groups. This is in agreement with a recent study by Pasic et al, 26 who retrospectively analyzed 46 patients who underwent ACI or MACI biopsy and found only 9 patients (20%) went on to implantation. Almost all patients had chondroplasty, loose body removal, meniscal repair/meniscectomy, or other concomitant procedure performed at the time of biopsy, and the authors concluded that those treatments appeared to be sufficient for alleviating knee symptoms in their cohort of patients with articular cartilage defects.
The mean number of defects as well as their size, location, and symptomatic etiology (either after acute injury or via chronic progression) did not differ between the biopsy and implant groups in the current study. Of the defect characteristics we analyzed, Outerbridge classification represented the only significant factor influencing the decision of whether to proceed to implantation, as those patients who went on to implantation had significantly higher grades. Because the patients in the implant group had generally more severe cartilage damage, it is difficult to draw direct comparisons between the 2 cohorts, but the finding does emphasize a salient distinction. Despite there being no significant differences in defect characteristics, symptom onset, or preoperative IKDC score, those patients with higher grade cartilage damage were more likely to proceed with the second stage of ACI or MACI. Outerbridge classification may therefore be misleading for these patients; a surgeon may be more inclined to proceed with chondrocyte implantation given high lesion grading during index biopsy, but the surgical findings may not necessarily correlate with functional status. Notably, however, 13% of patients with grade 2 or 3 lesions went on to the second stage of the procedure while 73% of patients who did not go on to implantation had grade 4 lesions, further demonstrating that it is difficult to make an intraoperative decision that would suit all patients based solely on defect grade. Asymptomatic osteochondral defects may be ubiquitous in the population. A review of 31,516 knee arthroscopies across all age groups reported chondral defects in 19,827 patients (63%), with a mean of 2.7 defects per knee, 8 and a study of 993 consecutive knee arthroscopies found the prevalence of full-thickness cartilage lesions to be 11%.2(p993) It is impossible to know how many of these were the primary generator of a patient's knee pain and how much was due to other knee pathology. The higher mean Outerbridge grades in the implant group of this study represent a key factor in patient selection. Future study may further define this potential cohort of patients by comparing outcomes of matched patients with similar OCD scores who undergo ACI/MACI to those who undergo isolated debridement chondroplasty.
In this study, patient demographics and chondral defect characteristics were largely similar between the 2 groups; however, the implant group featured more female patients (57 vs 16 in biopsy group; P < .001) and a higher mean BMI (29.9 vs 26.6 in biopsy group; P < .001), indicating that female sex and a higher mean BMI represented the demographic characteristics of patients more likely to go on to implantation after cartilage biopsy. Interestingly, recent literature suggests that female patients are consistently more likely to undergo revision surgery after ACI.12,13,16,21 Kreuz et al 19 evaluated sex-specific differences in ACI outcomes in a 2013 study of 52 patients and found that while clinical scores improved overall in both male and female patient cohorts, male patients achieved significantly better Lysholm scores at all time intervals (6 months, 12 months, 48 months) and IKDC scores at 6 and 12 months after surgery (P < .05). They also found that female patients with patellar defects had the worst results. The authors postulated that these results might be explained by decreased proprioception and muscle imbalances across the knee joint 19 ; however, the precise explanation requires further study. Additionally, higher BMI is associated with increased cartilage breakdown, contributing to abnormal joint biomechanics and subsequent cartilage destruction in OCD lesions.4,20,22,24 Obesity has also been associated with worse functional outcomes and higher failure rates in patients undergoing ACI. These findings are similar to those in other studies that examined the role of obesity in increasing failure rates of other knee procedures, including microfracture and unicompartmental knee arthroplasty.12,18 A study examining treatment outcomes of OCD lesions demonstrated that both obesity and excision without allograft reimplantation were independent predictors of the development of osteoarthritis and need for knee arthroplasty. 29 Considering the preponderance of data demonstrating the association between higher BMI and lower functional outcomes after knee surgery, it is important to recognize this as a risk factor for failure of surgical management of chondral defects.
The current study attempted to define patient-specific reasoning for not undergoing the second stage of the ACI/MACI procedure through a custom survey. While there were a variety of reasons that patients reported that were unrelated to their symptoms, most patients either experienced symptom resolution from their index arthroscopy, cited recovery time as a significant factor in their decision, or underwent different treatment modalities, both nonoperative and operative. In the biopsy group, 13 patients (21%) underwent a subsequent procedure (other than implantation) to the affected knee following biopsy, while 35 patients (36%) who underwent implantation required a subsequent procedure to the affected knee. This again highlights the importance of patient selection when considering ACI or MACI. Etiology of knee pain may be multifactorial, with OCD lesions contributing only partially or not at all. For those patients who did not undergo implantation, it was thought that a different procedure would better address their symptoms. For those who underwent additional surgery after implantation, their knee pain was likely not completely addressed through ACI or MACI.
Limitations
The current study must be viewed in the context of its limitations. The retrospective design of this study limited the ability to match patient demographics and lesion characteristics. A total of 160 patients and 232 lesions were included in the final analysis, and while this is similar to or larger than other studies that have examined ACI implantation,19,21,23 the cohort size and follow-up rate of 70% limited the ability to account for differences within the cohort and make clinically significant conclusions. No power analysis was performed, limiting the statistical significance. Patients were encouraged on the custom survey to elaborate on reasoning for not undergoing the second stage of the procedure; however, responses were limited both by recall bias and by a likely incomplete understanding of the procedure or of additional pathology that was addressed, either during initial biopsy or with subsequent treatment. This was evident in the 25 patients who cited “other” or that their procedure was never scheduled despite clear reasoning behind those decisions. Future randomized, prospective design studies with larger cohort sizes would help to limit sampling and eliminate the recall bias and subjective nature of the survey that limit its analysis. Finally, although patients in the biopsy group had similar functional outcomes to those who underwent implantation, it is not known if this effect would be maintained with a longer follow-up.
Conclusion
Patients who proceeded to the second stage of chondrocyte implantation via either ACI or MACI had higher-grade articular defects and higher BMI compared with those who underwent biopsy with concomitant debridement chondroplasty alone. Outcomes were similar between the groups.
Footnotes
Appendix
Final revision submitted February 4, 2024; accepted February 13, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: B.R.K. has received education payments from Eclipse Technology Solutions, Liberty Surgical, and Paladin Technology Solutions. T.D. has received education payments from Liberty Surgical and Paladin Technology Solutions. K.B.F. has received grant support from Vericel; education payments from Liberty Surgical; consulting fees from Vericel, Innocoll, and Medical Device Business Services; nonconsulting fees from Vericel; and honoraria from Vericel. S.H. has received education payments from Arthrex, nonconsulting fees from Arthrex, and hospitality payments from Smith+Nephew. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was waived by Philadelphia University and Thomas Jefferson University.
