Abstract
Background:
Currently, there is no consensus on the adequate management of irreparable rotator cuff tears. Arthroscopic superior capsule reconstruction (SCR) seems to be an alternative treatment option.
Purpose/Hypothesis:
The purpose of this study was to evaluate patient-reported outcomes up to 2 years after the treatment of irreparable rotator cuff tears with SCR using an acellular porcine dermal xenograft. It was hypothesized that SCR treatment with an acellular porcine dermal xenograft would not result in a significant clinical improvement or pain relief in patients with irreparable rotator cuff tears if the xenograft fails.
Study Design:
Case series; Level of evidence, 4.
Methods:
A total of 26 consecutive patients with irreparable rotator cuff tears were enrolled in the study between 2015 and 2019. All patients underwent SCR with acellular porcine dermal xenograft. Patient-reported outcome measures including visual analog scale (VAS) for pain, the American Shoulder and Elbow Surgeons (ASES) score, the Veterans RAND 12- Item Health Survey (VR-12), and the Single Assessment Numeric Evaluation (SANE) were followed up for 2 years. For statistical analysis, the 1-way analysis of variance was used to compare means for VAS, ASES, VR-12, and SANE results between pre- and posttreatment. Magnetic resonance imaging (MRI) records were obtained at 1 year postoperatively to evaluate graft integrity.
Results:
In total, 22 patients were included in the patient-reported outcomes with 4 being lost at final follow-up. The mean VAS score decreased from 4.2 ± 2.5 to 1.0 ± 1.4 (P < .001) from pretreatment to 2 years postoperatively. The mean ASES – index score improved significantly from 47.7 ± 15.3 to 86.4 ± 12.9 (P < .001) and the SANE score improved from 34.0 ± 20.4 to 77.3 ± 20.2 (P < .001). In addition, a clinically important difference in the patients’ quality of life was achieved, as shown by the mean changes in the VR-12 physical (+4.3) and mental scores (+9.3). Based on postoperative MRI, the dermal graft on the humeral side was intact in 15 (68.2%) patients after surgery.
Conclusion:
Our arthroscopic SCR with an acellular porcine dermal matrix showed significant and continuous improvement in pain and clinical scores up to a 2-year follow-up in patients with irreparable rotator cuff tears, even with structural graft failure. However, further studies and evaluation of larger patient groups are needed to evaluate the long-term effect of this procedure.
Keywords
The management of symptomatic irreparable rotator cuff tears can be a clinical challenge for orthopaedic surgeons. Various surgical techniques have been proposed in the literature for their treatment,§ including debridement, biceps tenotomy, partial repair, rotator cuff grafting, tendon transfer, and reverse shoulder arthroplasty.7,28,34,36,58,64
Arthroscopic debridement of irreparable rotator cuff tears is a commonly used technique. Studies have shown good midterm results with improvements in patient-reported outcome scores and pain, although not in the long term.18,26 Arthroscopic partial repair may be a safe and effective alternative treatment for irreparable rotator cuff tears when complete repair cannot be performed. Subjective clinical and functional scores improve postoperatively, but the rate of structural failure of the partial repair can be high, although partial repair leads to superior results when compared to debridement alone.6,31,35,57
The biological patch graft has been used in the literature to augment a deficient rotator cuff. This treatment option provided patients with decreased pain and increased function, but the graft was only attached to the remaining edges of the rotator cuff. 8
Inadequate treatment of a rotator cuff tear increases the risk of progressive superior humeral head migration 29 and rotator cuff tear arthropathy.46,52
The superior capsule of the shoulder provides superior stability to the glenohumeral joint and is often damaged in patients with irreparable rotator cuff tears.29,43 Recently, superior capsule reconstruction (SCR) has gained attention. 14 SCR is a surgical technique that aims to reinforce the superior capsule to restore the native biomechanics of the glenohumeral joint by attaching tissue from the glenoid to the greater tuberosity. The concept of SCR using a fascia lata autograft was first published by Mihata et al 40 and demonstrated a high return of shoulder function in a 5-year analysis, increasing recreational and work activity.2,14,24,39,41,42,49
Despite promising biomechanical and clinical results, the choice of graft in SCR for the treatment of irreparable rotator cuff tears is still unclear. A previous study has shown that the most performed technique for SCR is an acellular dermal allograft among others, although clinical outcomes remain uncertain due to a lack of comparative studies on graft selection. 16 A match-pair analyses between SCR and partial rotator cuff repair when using an acellular porcine dermal xenograft 13 showed improvement but no significant difference in clinical scores. 20
Other authors reported a high complication and revision rate, even with acute rejection of the porcine dermal xenograft. 50 Another study showed that structural failure with respect to the location of the graft tear has a significant impact on clinical outcomes within 1 year of follow-up. 17
Therefore, the purpose of this study was to evaluate patient-reported outcomes over 2 years and to assess whether clinical outcomes will deteriorate over time after treatment of irreparable rotator cuff tears with SCR using an acellular porcine dermal matrix.
Our hypothesis was that SCR treatment with an acellular porcine dermal xenograft would not result in a significant clinical improvement or pain relief in patients with irreparable rotator cuff tears if the xenograft fails.
Methods
This retrospective single-center study included a consecutive series of 26 patients enrolled between September 2015 and February 2019. In total, 22 patients were left for evaluation after the first and second years of follow-up.
The inclusion criteria were an irreparable posterosuperior rotator cuff tear (which represents a tear involving the complete supraspinatus and part of infraspinatus) of at least grade 3 according to Bayne et al 5 and advanced grade 3 retraction according to Patte. 47 Other inclusion criteria were muscle atrophy and fatty degeneration ≥50% (Goutallier grade 3-4) 19 on preoperative magnetic resonance imaging (MRI) and a humeral head migration according to the Hamada classification 23 grades 1 to 3 on preoperative radiographs. The exclusion criteria were an irreparable infraspinatus and/or subscapularis tendon or no evidence of osteoarthritic changes in the shoulder joint.
The decision to perform SCR was made intraoperatively when a complete repair of the posterosuperior rotator cuff to the anatomic footprint was not possible after mobilization. The matrix used was DX Reinforcement Matrix (Arthrex Inc), a porcine dermal extracellular matrix xenograft developed as a biocompatible scaffold for soft tissue reinforcement and repair.22,27
Patients had to agree to participate in the clinic's prospective data registry (Surgical Outcomes System [SOS]), which was approved by the Ethics Committee of Hamburg Medical Association (Ärztekammer Hamburg, Ethics Committee No. 2021-300050-WF). The study was conducted in accordance with the ethical standards of the Declaration of Helsinki of 1964 and its subsequent amendments or comparable ethical standards.
Surgical Procedure
Two experienced surgeons (A.I. and J.H.) performed the arthroscopic SCR. The patient was placed in either the beach-chair position (A.I.) (with TRIMANO FORTIS Support Arm (Shoulder); Arthrex Inc) or the standard lateral decubitus position (J.H.). The subacromial bursa, degenerative tissue, and sutures from previous repairs were thoroughly removed.
If necessary, repair of infraspinatus and subscapularis tendon tears was attempted and biceps tenodesis (6.75 BioComposite Tenodesis Screw; Arthrex Inc) or tenotomy was performed depending on the quality and stability of the tendon. The glenoid and tuberosity bone beds were prepared with a bone cutter to maximize graft healing. Two 3-mm BioComposite SutureTak anchors (Arthrex Inc) were placed just medial to the superior labrum in the superior glenoid percutaneously spanning the glenoid from anterior to posterior. Two 4.75-mm BioComposite SwiveLock anchors (Arthrex Inc) with FiberTape sutures were placed in the articular margin, depending on the size of the tear. Four dimensions were measured between the 4 anchors were placed. The graft size was extended at least 5 mm around the anchors to prevent the suture from cutting through.
The graft was then double folded and sutured to increase the thickness to 3 mm, it was sized, and the anchor positions were marked. The sutures from the medial anchors were shuttled through the graft, a double-pulley knot configuration was used to help maneuver the graft onto the glenoid, and a static surgeon's knot was used to secure the graft medially to the glenoid (Figure 1A).

The intraoperative arthroscopic appearance of the SCR through a lateral view portal in a right shoulder. (A) SCR using a dermal xenograft after fixation to the medial glenoid. (B) Lateral fixation of the xenograft to the greater tuberosity using the SpeedBridge technique. (C) Dermal xenograft with side-to-side sutures linked between the graft and the infraspinatus tendon. SCR, superior capsule reconstruction.
The FiberTape sutures were pulled out through the lateral portal and individually shuttled according to predetermined markers on the graft while the graft was tensioned over the greater tuberosity in neutral rotation and 20° to 30° of abduction. The graft was later fixed to the humerus using a knotless SpeedBridge (Arthrex Inc.) repair with 2 lateral 4.75-mm BioComposite SwiveLock anchors, and the remaining suture limbs were cut (Figure 1B). In all cases, 2 to 3 side-to-side sutures were passed to the remaining infraspinatus tendon (Figure 1C).
Postoperative Management and Rehabilitation
Postoperative pain control was provided by an interscalene block or interscalene nerve catheter and an overlapping oral analgesic. Rehabilitation initially focused on passive range of motion without restrictions, with no active abduction allowed for 6 weeks. An abduction sling was used for 6 weeks postoperatively. Active-assisted rehabilitation in abduction and mobilization started at 7 weeks postoperatively, and strengthening was allowed after 3 months.
Patient-Reported Outcome Measures
Patient-reported outcomes were assessed using the SOS, a global orthopaedic surgery and sports medicine registry (Arthrex Inc). Patients received questionnaires via email for the visual analog scale (VAS) for pain, the American Shoulder and Elbow Surgeons (ASES) score, the Veterans RAND 12-Item Health Survey (VR-12), and the Single Assessment Numeric Evaluation (SANE) before and at specified timepoints after the surgery (Table 1). Any patient without email access completed their survey by telephone. In addition, patients who had undergone surgery before SCR were compared with patients who had not undergone surgery in terms of the effect of preoperative treatment on the patient-reported outcome measures.
Data Collection Timepoints a

Data are presented as mean ± SD unless otherwise indicated. ASES-FS, American Shoulder and Elbow Surgeons – function score; ASES-IS, American Shoulder and Elbow Surgeons – index score; MS, mental score; PS, physical score; SANE, Single Assessment Numeric Evaluation; VAS, visual analog scale for pain; VR-12, Veterans RAND 12-Item Health Survey.
All follow-up visit windows are calculated from the day of primary operation. Days ± months are displayed in the header. In the cells under Total, n, starting at VAS, the values are displayed in means ± SD. Blank cells indicate n/a.
Imaging
The 5-grade evolutionary classification of Hamada et al 23 was used to analyze the preoperative radiographic findings in all patients. All patients obtained postoperative imaging with MRI at 1-year follow-up.
The graft status was categorized as healed (type 1) or unhealed, classified depending on the site of failure (medial, glenoid-sided graft tear, type 2 or lateral, humeral-sided graft tear, type 3). 44
Statistical Analysis
Statistical analysis was performed using JMP (Version 14, Statistical Analysis System Institute Inc). Patient-reported outcome data before and after the surgery were analyzed using the 1-way analysis of variance with pairwise comparison via the Tukey-Kramer test. The level of significance was defined as P≤ .05. The Tukey-Kramer test was used as a post hoc test to compare the different surgical procedure positions.
Results
Between September 2015 and February 2019, 26 patients (20 men and 6 women) with an irreparable superior rotator cuff tear and a mean treatment age of 64.8 ± 8.5 years (range, 52-78 years) were included in the study; 22 patients were available for follow-up at 1 and 2 years after surgery.
All patients underwent arthroscopic SCR using a porcine dermal xenograft. Biceps tenodesis was performed in 6 cases, biceps tenotomy was performed in 12 cases, and the biceps tendon was absent in 8 cases. A subacromial decompression and side-to-side sutures between the infraspinatus and the posterior aspect of the graft were performed in every case. The infraspinatus tendon was also repaired if required. No subscapularis tendon repair had to be performed in our series.
Seven patients (26.9%) had previously undergone surgical treatment. Of the 7 patients, 4 (15.4%) had a previously failed rotator cuff repair, 2 had undergone arthroscopic subacromial decompression, and 1 had undergone arthroscopic debridement without rotator cuff repair. The number of patients who completed the questionnaires at each timepoint is shown in Table 1. Follow-up after surgery was up to 2 years. Three patients were lost to follow-up after the first year due to unavailability and 1 patient decided to withdraw from the clinic's prospective data registry 1 year after surgery.
All clinical outcome scores for pain, function, and quality of life improved after arthroscopic SCR at the final 2-year follow-up. The mean VAS pain score decreased significantly from 4.2 ± 2.5 before treatment to 2.0 ± 2.4 (P < .001) at 3 months and 1.6 ± 1.7 (P < .001) and 1.0 ± 1.4 (P < .001) at 1 year and 2 years after treatment (Figure 2).

Patient-reported visual analog scale for pain results before and after arthroscopic superior capsule reconstruction. Each error bar represents 1 SD from the mean. *P value between .01 and .05 is significant, P > .05 is not significant. m, months; Pre, pretreatment.
Functional improvement of the patients’ shoulders after SCR was assessed using the ASES – function score (ASES-FS), which increased significantly in all patients from a mean of 11.1 ± 5.3 to 23.8 ± 5.4 (P < .001) at 1 year and to 24.8 ± 5.0 (P < .001) at 2 years (Figure 3).
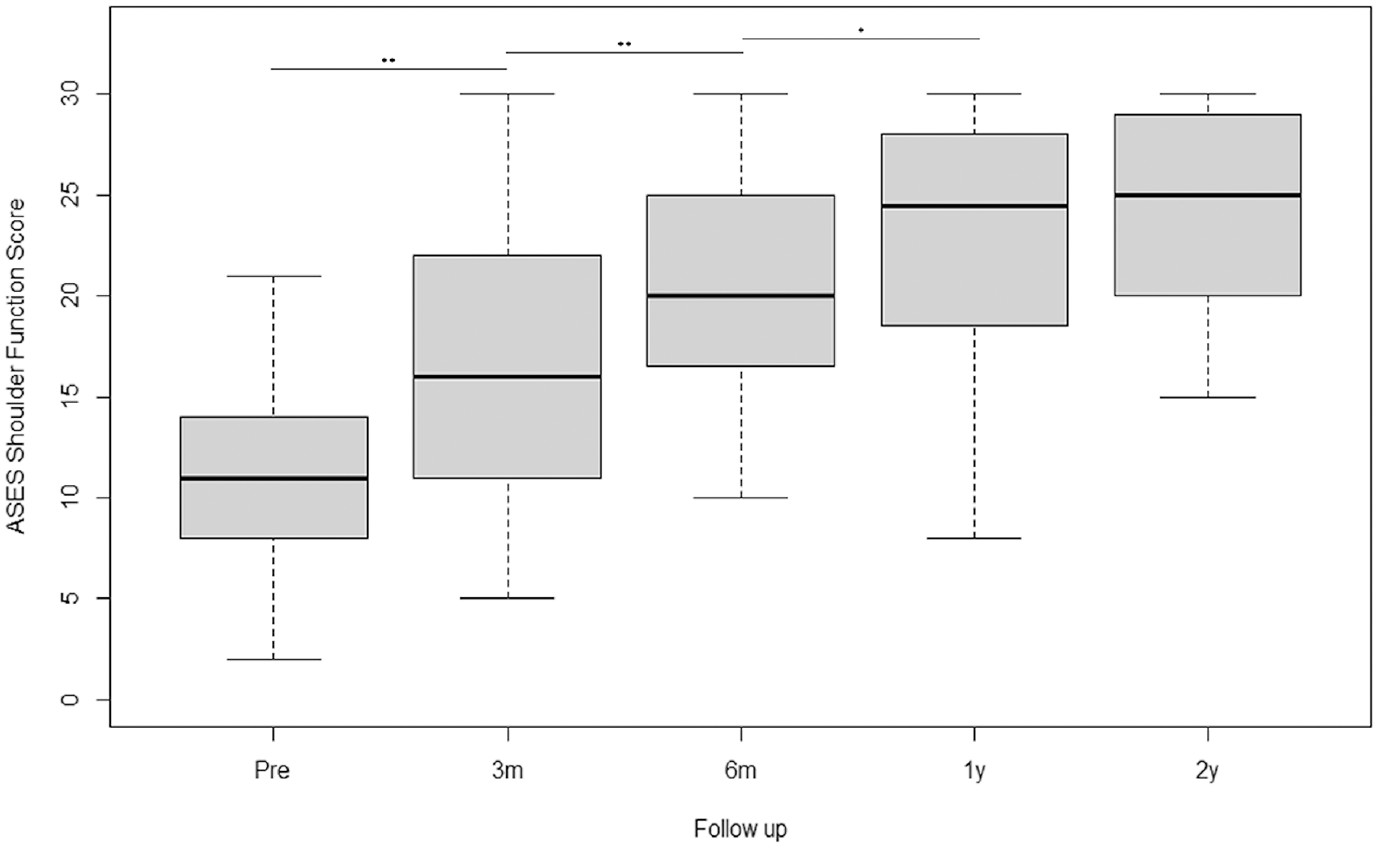
Patient-reported outcomes for the American Shoulder and Elbow Surgeons (ASES) – function score before and after arthroscopic superior capsule reconstruction. Each error bar represents 1 SD from the mean. **P value between .001 and .01 is very significant, *P value between .01 and .05 is significant, P > .05 is not significant. m, months; Pre, pretreatment.
The mean ASES – index score (ASES-IS), which combines pain and function, improved as well in all patients from 47.7 ± 15.3 to 81.8 ± 15.6 and 86.4 ± 12.9 at 1 year and 2 years, respectively (P < .001) (Figure 4).

Patient-reported outcomes for the American Shoulder and Elbow Surgeons (ASES) – index score before and after arthroscopic superior capsule reconstruction. Each error bar represents 1 SD from the mean. ***P < .001 is highly significant, *P value between .01 and .05 is significant, P > .05 is not significant. m, months; Pre, pretreatment.
Further improvement was noted in 14 (64%) patients regarding their ASES-FS and in 15 (68%) patients regarding their ASES-IS between the first and second years of follow-up.
There was a trend toward poorer functional outcomes if the graft had failed, although this was not statistically significant due to the small data set. We compared the ASES score in patients with and without a graft tear between the 1- and 2-year follow-ups. The ASES-FS only improved for healed graft (+0.5 points) whereas the ASES-IS in both groups increased slightly (+2.23 points in the rupture group, +1.09 points in the healed group).
The SANE functional score increased by 43.3 points (34.0 ± 20.4 to 77.3 ± 20.2) (P < .001) at final follow-up (Figure 5).

Patient-reported outcomes for the Single Assessment Numeric Evaluation score before and after arthroscopic superior capsule reconstruction. Each error bar represents 1 SD from the mean. **P value between .001 and .01 is very significant, P value > .05 is not significant. m, months; Pre, pretreatment.
Finally, quality of life as measured by the VR-12 also improved after SCR. Two years after the treatment, the mean VR-12 physical score (PS) had increased from 34.5 ± 7.9 to 45.6 ± 10.5 (P < .001) (Figure 6) and the VR-12 mental score (MS) had increased from 49.8 ± 14.0 to 54.6 ± 10.8 (P = .1077) (Figure 7). Furthermore, 22 (100%) patients were satisfied with their results at 1 year postoperatively and 19 (86.4%) patients were satisfied at the 2-year follow-up.

Patient-reported outcomes for the Single Assessment Numeric Evaluation score before and after arthroscopic superior capsule reconstruction. Each error bar represents 1 SD from the mean. *P value between .01 and .05 is significant, P > .05 is not significant. m, months; Pre, pretreatment.

Patient-reported outcomes for the Veterans RAND 12-Item Health Survey mental score before and after arthroscopic superior capsule reconstruction. Each error bar represents 1 SD from the mean. *P value between .01 and .05 is significant, P > .05 is not significant. m, months; Pre, pretreatment.
Patients with surgical treatment before SCR showed no statistically significant differences in the scores evaluated at 1 year compared to patients without prior surgery (VAS: P = .83, ASES-FS: P = .60, ASES-IS: P = .69, SANE: P = .52, VR-12-PS: P = .95, VR-12-MS: P = .91).
Regarding surgical position, the beach-chair group compared with the standard lateral decubitus group showed no significant difference between the 2 groups at 1 year in any of the scores used (VAS: P = .73, ASES-FS: P = .16, ASES-IS: P = .35, SANE: P = .43, VR-12-PS: P = .38, VR-12-MS: P = .33). At the 1-year follow-up, the remaining 22 patients were evaluated with postoperative MRI. Based on routine postoperative MRI scans (mean 9.5 months postoperatively), 15 (68.2%) of the dermal allografts were healed. In 7 (31.8%) patients, the graft failed to heal and showed clear graft tearing. All the graft ruptures occurred on the humeral side (Figure 8).

Magnetic resonance imaging scan showing a humeral-sided graft failure after superior capsule reconstruction (red circle).
Partial healing was difficult to assess on MRI because of the double-layer technique of the graft used for surgery. In cases of a graft tear, the graft was retracted medially, leaving a stump on the glenoid side.
In our series, we saw 2 patients with a “persistent inflammatory reaction of the operated shoulder.” The patients presented at follow-up with persistent clear secretions from the portal used for graft implantation. Regular blood tests showed a slight increase in C-reactive protein but no evidence of an elevated white blood cell count.
Swabs were taken from the wound, immediate microbiological testing/gram stain was performed, and cultures were observed for 14 days with no evidence of a bacterial infection.
There were no other general symptoms of acute infection, such as fever or signs of infection around the portal. We interpreted this condition as an acute inflammatory irritation or reaction due to necrotic subcutaneous tissue. The secretion resolved spontaneously approximately 3 to 4 weeks postoperatively, and no arthroscopic revision was required. No previous relevant medical history was apparent.
One patient had a BioComposite Tenodesis Screw dislocation and underwent arthroscopic revision 4 months postoperatively with an apparent humeral-sided tear of the dermal graft. One patient reported revision surgery with reverse total shoulder arthroplasty at another site 7 months postoperatively.
Discussion
The most important finding in this case series was that 22 (100%) patients significantly and continuously improved pain, function, and quality of life after arthroscopic SCR using an acellular porcine dermal xenograft up to 2 years postoperatively and were satisfied with the surgery.
Patients also reported significant improvements in all clinical measures but at different timepoints. Pain perception (VAS) showed a significant reduction at 3 months (P < .001), which was maintained at 2 years (P < .01). The ASES (function and index) score increased significantly from preoperatively to postoperatively. At 12 and 24 months, this was highly significant for both scores (P < .001) based on the pre-operative value. In addition, the ASES-FS improved significantly again between 6 and 12 months (P = .02). The SANE score improved significantly immediately postoperatively (P < .001) and showed a significant improvement between 3 and 6 months (P = .0027). The VR-12-PS and -MS showed a significant improvement between pretreatment and 3 months and between pretreatment and 1 year (P < .001). Only the VR-12-PS also showed a highly significant difference between pretreatment and 2 years (P < .001).
Our hypothesis that SCR treatment with an acellular dermal xenograft would not result in a significant clinical improvement in function and pain in patients with irreparable rotator cuff tears, if graft tears of the xenograft occur, cannot be entirely confirmed by the results in this study. Even in cases with failure of the graft on postoperative MRI scans, all patients improved their functional scores over 2 years, although only 54% of patients improved the ASES-FS and 58% the ASES-IS between the first and second year. This is in accordance with other studies that reported a low rate of revision surgery even if the graft had failed.11,16,19,35
SCR for the treatment of irreparable posterosuperior rotator cuff tears is increasingly used and leads to good and satisfactory clinical outcomes.9, 10 ,54 However, mid- and long-term data supporting the safety and efficacy of the procedure are limited by the small number of published Level 4 case series. 1
Mihata et al 40 published a clinical trial in 2013 in which 23 patients (24 shoulders) with massive rotator cuff tears underwent arthroscopic SCR using fascia lata allograft of 6- to 8-mm thickness, while other available dermal allo- or xenografts are less thick. In the present study, instead of fascia lata, we used an approximately 1.5 mm–thick acellular matrix of porcine dermis, which was double folded to achieve a thickness of up to 3 mm. Double folding may have advantages in the avoidance of graft harvesting, morbidity at the donor site, and a shorter operative time.12,40 Furthermore, Mihata et al 40 reported that 83.3% of fascia lata grafts healed postoperatively (3 retears and 1 graft tear). In a more recent study, Mihata et al 38 showed a 95% healing rate.
Dermal allografts have also shown encouraging results, avoiding donor site morbidity. In a small series of 9 patients, Hirahara et al 25 found that the mean ASES score improved significantly from 43.54 to 86.46 (P < .00002) and mean VAS pain score decreased significantly from 6.25 to 0.38 (P < .00002), even after 2 years of follow-up. In a retrospective study of 86 patients, the outcome data from Pennington et al 48 revealed an improvement in VAS (4.0 to 1.5) and ASES (52 to 82) scores at 1 year (P = .005).
Denard et al 13 reported only 45% of healed grafts on postoperative MRI. It should be mentioned that 55.9% of patients in their study had an additional subscapularis tear that was repaired. However, there was no difference in clinical outcomes regarding the incidence of subscapularis tears. Lacheta et al, 33 who used a human dermal allograft in their study, showed graft healing rates between 76% and 100%, depending on the location of the graft (100% on the greater tuberosity, 81% on the glenoid side, and 76% on the middle of the graft), but no differences in clinical outcomes, if the graft had healed or not. Only a few studies have been published so far using acellular dermal xenografts for SCR.
The cohort series of Garofalo et al 17 showed a graft tear rate of 52% on average, depending on the thickness of the graft used for SCR. The graft tear more commonly occurred in their single-layer group (1.5-mm thickness) compared with in the double-layer group (3-mm thickness) (77% vs 33%).
Our study showed a nearly similar retear rate of the xenograft. Based on postoperative MRI scans at 1 year postoperatively, which could be obtained in our series, 31.8% of the porcine dermal grafts did not heal.
In a matched-pair analysis comparing SCR and partial infraspinatus repair, Greiner et al 20 reported on just 1 graft tear in the SCR group on postoperative MRI of 1 symptomatic patient with persistent pain and dysfunction; however, no routine evaluation by MRI scans had been performed to look at retear rates of the graft. Previous studies using a patch graft, such as porcine small intestine submucosa or porcine dermal collagen implants, in which the grafts were attached medially to the torn tendon for the treatment of massive rotator cuff tears, have also shown higher retear rates compared to our results.53,55
In a systematic review of 24 studies using various patch augmentation and interposition techniques, Steinhaus et al 56 reported an overall retear rate of 25%, with rates of 44%, 23%, and 15% for xenografts, allografts, and synthetic grafts, respectively. Our study also focused on patient-reported outcomes after SCR using the VAS, ASES, VR-12, and SANE scores. The mean VAS pain score decreased significantly from pretreatment to 2 years posttreatment from 4.2 to 1.0 point. Denard et al, 13 using an acellular dermal allograft, showed comparable results for the VAS pain score in their study after a minimum follow-up of 1 year (5.8-1.7). In our study, the ASES-FS and the ASES-IS increased significantly from 11.1 to 24.8 (+13.7) and from 47.7 to 86.4 (+38.7), respectively, 2 years after treatment (P < .001). Mihata et al 40 reported an improvement in the ASES-IS from 23.5 to 92.9 (+69.4) at the last follow-up (mean, 34.1 months). Denard et al 13 showed an improvement in the ASES-IS from 43.6 to 77.5 (+33.9) and Kalina et al 30 from 23.8 to 73.2 (+49.4) 1 year postoperatively.
However, the minimal clinically important difference for the ASES ranges from 6.4 for various shoulder disorders to 12 to 17 points for rotator cuff problems.30,37,59 Ours measured a difference of 38.7 points, therefore demonstrating a clinically important improvement in shoulder function after arthroscopic SCR. Functional outcomes as measured by the SANE score further support this statement as it increased significantly by 43.3 points (34.0-77.3) at 2 years (P < .001). In the literature, the minimal clinically important difference for SANE scores in patients after treatment of rotator cuff tears is in the range of 11.8 to 15.60,65 Finally, we also demonstrated a clinically significant improvement of 6.8 points in quality of life, as measured by the VR-12 score.
Available data regarding the use of dermal xenografts are currently difficult to compare due to different study designs. Kalina et al 30 showed the first clinical results using the DX Reinforcement Matrix (xenograft), similar to our technique. The slightly different results may be due to the different follow-up periods or patient groups. Greiner at al 20 reported a significant improvement in patient-reported outcomes in the Constant score (49.7-77.1), Disabilities of the Arm, Shoulder and Hand score, and Western Ontario Rotator Cuff Index pre- to postoperatively at a minimum 2-year follow-up.
In a retrospective analysis, Garofalo et al 17 found a significant improvement in active range of motion for forward flexion (72.8°-120.6°), abduction (68.3°-140.2°), and external rotation (38.2°-56.7°), and the mean Constant score improved significantly from 40.4 to 73.3 at least 14 months postoperatively. Their outcomes were dependent on structural failure and graft tear location. Patients with a healed graft or a medial graft tear showed significantly better results compared with graft tears on the humeral side or entirely reabsorbed grafts.
In our series, we saw 2 patients with a “persistent inflammatory reaction of the operated shoulder” with no evidence of a bacterial infection. The xenograft was well tolerated in all other patients. In a study of 3 patients with a similar acute inflammatory condition, Polacek 50 reported that all patients underwent arthroscopic revision, and complete graft degradation was observed. However, the onset of symptoms occurred several weeks after implantation, which is more consistent with an acute immunological rejection of the xenogeneic antigens, which we believe is in contrast to our series, where the onset of symptoms was immediately postoperatively. 62 The current study examined the patient-reported outcomes of arthroscopic superior reconstruction with a xenograft and a minimum follow-up of 2 years and showed continuous improvement in clinical scores, even if the graft failed to heal. These preliminary data illustrate that SCR with porcine dermal xenograft may be a promising option for the treatment of irreparable rotator cuff tears. However, further prospective studies, including randomized controlled trials, are needed to investigate the mid- and long-term outcomes of SCR using this xenograft. The outcomes of our patients will continue to be collected through the SOS up to a follow-up of 5 years.
Limitations
We are aware that this study has limitations. First, it is a case series with a small group of patients without a control group. Therefore, we were not able to investigate whether porcine dermal xenograft SCR has a long-term advantage over conventional techniques or other surgical methods for the treatment of massive rotator cuff tears, such as partial rotator cuff repair, the use of autografts versus allografts, tendon transfers, or reverse total shoulder arthroplasty. Two surgeons performed surgery with their preferred patient positioning at the time of surgery and possibly different individual surgical techniques, which could influence the outcome, but they had a similar level of experience. In addition, we did not collect functional outcomes such as shoulder range of motion and muscle strength.
It should also be mentioned that administering patient-reported outcome measures by telephone when patients were unavailable by email may introduce a significant bias. Finally, it should be mentioned that the present study is not large enough and the follow-up too short term to be able to evaluate the complications of this type of transplant.
Conclusion
Arthroscopic SCR with an acellular porcine dermal matrix showed significant and continuous improvement in pain and clinical scores up to a 2-year follow-up in patients with irreparable rotator cuff tears, even with structural graft failure. However, further studies and evaluation of larger patient groups are needed to evaluate the long-term effect of this procedure.
Footnotes
Correction (November 2024):
Article updated online to correct the article type to Original Research.
Final revision submitted December 8, 2023; accepted February 12, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: A.I. is a paid consultant for and receives research funding from Arthrex. J.H. is a paid consultant for and receives research funding from Arthrex. S.S. receives research funding from Arthrex. R.K. receives research funding from Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Arztekammer Hamburg (ref No. 2021-300050-WF).
