Abstract
Background:
Reverse total shoulder arthroplasty (RTSA) is the gold standard surgical procedure for irreparable posterosuperior rotator cuff tears. Superior capsular reconstruction (SCR) with acellular dermal allograft (DA) has demonstrated good short-term outcomes in younger patients. Studies that compare the 2 procedures are lacking.
Purpose/Hypothesis:
The purpose of the study was to compare the outcomes of these 2 surgical techniques at a minimum 5-year follow-up to evaluate their efficacy, durability, and complication profiles. It was hypothesized that RTSA would result in superior outcome scores and higher survival rates.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
This study retrospectively analyzed data from 69 consecutive patients <70 years of age who underwent either SCR with DA (n = 34) or RTSA (n = 35) for isolated irreparable posterosuperior rotator cuff tears without advanced osteoarthritis, with a minimum follow-up of 5 years. Failure was defined as revision surgery or clinical failure. Complications were reported. Kaplan–Meier survivorship analysis was performed. Patient demographics and pre- and postoperative patient-reported outcome measures (PROMs), including the American Shoulder and Elbow Surgeons (ASES) Score, Single Assessment Numerical Evaluation (SANE), the shortened version of the Disabilities of the Arm, Shoulder and Hand (QuickDASH) score, the 12-item Short Form Health Survey (SF-12) Physical Component Summary (PCS) and Mental Component Summary (MCS), satisfaction, and sports participation were assessed and compared between groups.
Results:
Eleven patients in the SCR group (32.4%) and 3 patients in the RTSA group (8.6%) experienced failure. The RTSA group had superior survivorship (90.1%) at 5 years compared with the SCR group (75.0%) (P = .018). Follow-up of the remaining patients was achieved in 91.3% (21/23) in the SCR group and 81.3% (26/32) in the RTSA group, with significantly different mean follow-up durations (RTSA, 9.5 ± 2.4 [range, 5.0-16.3] years; SCR: 6.7 ± 1.4 [range, 5.0-9.0] years; P < .001). The respective mean ages at the time of surgery differed significantly (RTSA, 63.3 ± 4.8 [range, 46.1-70] years; SCR, 56.5 ± 6.7 [range, 41.2-67.2] years; P < .001). Aside from failures, no complications were reported in either surgical group. Preoperative PROMs were similar between groups (all P > .05). At follow-up, all PROMs in both treatment groups improved significantly from preoperative values (P < .001). When comparing postoperative PROMs between groups, ASES score, SANE, and SF-12 MCS did not significantly differ between groups (all P > .05). QuickDASH (RTSA, 24.3 ± 19.5; SCR, 11.3 ± 12.0; P = .009) and SF-12 PCS (RTSA, 44.3 ± 12.2; SCR, 52.1 ± 7.8; P = .016) did differ significantly, favoring SCR. Sports participation was moderately below the preinjury level in both groups (RTSA, <25% to 50%–65%; SCR, 25%–49% to 75%–99%) but improved significantly only in the SCR group (P = .009). Satisfaction was similar in both groups, with a median of 9 (range, 1-10).
Conclusion:
In treating irreparable posterosuperior rotator cuff tears in younger patients, RTSA demonstrated superior survivorship (90.1%) compared with SCR with DA (75.0%) at mid-term follow-up. However, in patients without treatment failure, both RTSA and SCR had equal improvements in most PROMs at ≥5 years of follow-up, with some scores favoring SCR in nonfailure cases. Higher sports participation, PCS, and lower disability were noted in the SCR group.
Rotator cuff (RC) tears are considered technically irreparable when direct fixation of the torn tendon to its insertion site is not achievable. In contrast, a functionally irreparable tear refers to cases where the tendon can be reattached, but the muscle itself is no longer viable. Chronic, large RC tears are difficult to treat because of tendon retraction, reduced elasticity, muscle atrophy, and fatty infiltration that make primary repair not possible. 15 Various surgical methods have been employed to address irreparable posterosuperior RC tears, such as arthroscopic debridement with tenodesis of the long head of the biceps tendon, partial repairs, tendon transfers, and patch augmentations. However, these treatments are not considered ideal as they have shown inferior outcomes compared with primary repairs.16,29 In response to these challenges, Mihata et al 24 introduced superior capsular reconstruction (SCR) using a fascia lata autograft as a potential treatment for symptomatic, massive, irreparable posterosuperior RC tears in patients without glenohumeral osteoarthritis (GHOA) aiming to achieve better outcomes and still preserve the glenohumeral joint. Their research demonstrated excellent clinical and radiographic outcomes, a high rate of return to sports, and a low complication rate in active, young patients. The SCR technique was later modified to use acellular dermal allografts (DAs) in place of autografts, reducing procedural time and avoiding the morbidity associated with harvesting tissue from a donor site.28,36 Early to short-term follow-up results have shown substantial improvement in functional outcomes.17,18
Reverse total shoulder arthroplasty (RTSA) is a reliable treatment option for patients with irreparable posterosuperior RC tears, provided the deltoid muscle remains functional. 10 While RTSA was initially used primarily for older patients, its use has now expanded to younger individuals. Younger patients have reported significant improvements in clinical and functional outcomes, although concerns about high complication rates and long-term durability still persist.10,12,13 Given the concerns regarding long-term survivorship after RTSA in younger patients, SCR offers a promising joint-preserving alternative for treating irreparable posterosuperior tears in individuals without GHOA. However, it remains unclear whether the outcomes of SCR are comparable with or better than those of RTSA, especially in the mid- and long term. These results are scarcely reported, and most published studies report the outcomes of this procedure using a DA with a follow-up duration of 2 to 5 years, with conflicting results that were overall favorable. We previously reported our 2-year clinical outcomes after SCR compared with RTSA in patients suffering from irreparable posterosuperior RC tears. 17 These results showed similar postoperative functional outcomes in both groups with a minimum 2-year follow-up. The objective of the present study is to compare survivorship, clinical outcomes, sports participation, patient satisfaction, and complication profiles at ≥5 years postoperatively in patients <70 years without GHOA who received either RTSA or SCR with DA for the treatment of irreparable posterosuperior RC tears. We hypothesize that RTSA would lead to better improvement in clinical outcomes, return to sports, patient satisfaction, and higher survival rates compared with SCR.
Methods
Patient Selection
This study was a retrospective analysis of data from patients who underwent either RTSA or arthroscopic SCR using DA to treat irreparable posterosuperior RC tears as assessed by the senior surgeon (P.J.M.). The surgeries were performed between October 2014 and September 2018 for SCRs and between January 2006 and September 2018 for RTSAs by the senior surgeon. The indications for irreparable posterosuperior RC tears included tendon retraction (Patte classification 25 ), muscle atrophy (Thomazeau classification 35 ), and fatty infiltration (Goutallier 14 ) graded ≥3 before surgery. In certain cases, an intraoperative evaluation was visually done by the senior author (P.J.M.) before the RC tear was deemed irreparable. Surgical treatment was indicated because of persistent pain, loss of strength, and functional impairment of the affected shoulder with no significant improvement of symptoms with nonoperative treatment. All patients had a negative belly press sign and preserved teres minor with a negative hornblower sign. Patients underwent 1 of 2 treatment options: SCR or RTSA. The choice between SCR and RTSA was based on patient-specific factors including age, activity level, and functional demands. The indications for both procedures were similar but SCR was generally preferred for younger, more active patients while RTSA was selected for older patients. To compare the outcomes of SCR versus RTSA in patients where either treatment was a valid option, included patients had to meet the following requirements: (1) choose SCR or RTSA as their treatment, (2) be aged <70 years, and (3) have an intact or reparable subscapularis tendon. Exclusion criteria were: neurological disorders of the affected upper extremity, deltoid weakness or deficiency, revision SCR or RTSA, previous arthroplasty, irreparable subscapularis tear, advanced GHOA (Hamada grade 3 ≥2 or Samilson grade 4 ≥2), presence of infection, or RTSA because of humeral fracture. Approval for this research was received from WCG (Protocol Number 20244664).
Surgical Techniques
SCR Surgical Technique
The technique for SCR has been previously outlined by Petri et al. 28 All patients were in the beach-chair position after general anesthesia. Diagnostic arthroscopy confirmed the irreparability of the torn supra- and infraspinatus tendons. If the proximal long head of the biceps tendon was intact, the intra-articular portion was tenotomized, and a subpectoral biceps tenodesis was performed with an interference screw. Extensive debridement of the rotator interval and superior glenoid was carried out, along with the lysis of intra-articular and subacromial adhesions. The greater tuberosity and superior glenoid were prepared using a motorized shaver. The labrum was preserved superiorly if in good condition, or removed if damaged. An arthroscopic measuring device was used to determine the graft size, which was based on the anteroposterior and mediolateral dimensions of the tear. A 3 mm–thick human acellular DA (ArthroFlex) was sized to cover 7 to 8 mm medially over the superior glenoid and 15 to 18 mm laterally over the RC footprint on the humerus. Using a Neviaser portal, the first superior glenoid anchor (4.75 mm; SwiveLock) was placed at the 12 o'clock position, ensuring no violation of the articular cartilage. Sutures were then passed through the graft via the anterolateral portal while it remained extra-articular. Two additional sutures were placed on the lateral aspect of the graft, which was shuttled into the shoulder with the help of a knot pusher. After confirming the graft's correct orientation, the medial glenoid anchor was tied to secure the graft. The graft was then fixed to the glenoid with 2 additional 3.0-mm SutureTak (Arthrex) anchors placed at the 10 o'clock and 2 o'clock positions, respectively. Sutures from these anchors were tied to fix the graft to the superior glenoid. Lateral fixation of the graft was performed with a crossing knotless, double-row anchor technique using 4 to 6 anchors (4.75-mm SwiveLock), with 2 or 3 anchors placed medially at the cartilage-bone border and 2 or 3 anchors placed laterally, 1.5 to 1.8 cm away. The graft was also secured to the infraspinatus or teres minor (posterior) and subscapularis (anterior) tendons with margin convergence sutures from the posterior and anterior medial row anchors. The rotator interval was not closed. Finally, posterior side-to-side fixation was performed between the graft and the remaining infraspinatus or teres minor using a free suture, completing the fixation of the graft medially on the glenoid, laterally on the greater tuberosity, and posterior to the remaining intact RC (Figure 1).

Arthroscopic visualization of the final superior capsular reconstruction of a right shoulder using an acellular human dermal allograft fixated with 3 medial anchors to the glenoid and a crossing double-row anchor fixation laterally to the greater tuberosity. ISP, infraspinatus; DA, dermal allograft.
RTSA Surgical Technique
The surgical technique for RTSA was previously described. 17 All patients were positioned in the beach-chair position after general anesthesia. A deltopectoral approach was made starting from the lateral coracoid toward the deltoid insertion. Blunt dissection was used to release adhesions in the subacromial space, and the deltoid was retracted laterally. The biceps tendon, if present, was identified and the tendon sheath was opened. The tendon was followed through the rotator interval to its origin on the superior glenoid, where it was tenotomized, held at the appropriate length, and sutured to the upper border of the pectoralis major using No. 2 Ethibond sutures (Ethicon Inc) to perform a soft tissue tenodesis. The subscapularis tendon was then peeled using monopolar electrocautery, elevating it from the bone. The torn posterosuperior RC was confirmed as irreparable. The humeral head was fully dislocated, and a humeral head osteotomy was performed using an oscillating saw to prepare for implantation of a Grammont-style prosthesis with a 155° neck-shaft angle. A capsular release was done for glenoid exposure, and the labrum was removed. The glenoid surface was prepared with a curette, followed by drilling a center guide pin and reaming for insertion of a medialized baseplate at neutral version and inclination. Locking and nonlocking screws were placed, and excellent fixation was confirmed. The glenosphere was affixed and locked in place. The humeral head was then sequentially reamed and broached to an appropriate size, with the humeral stem impacted into place until stable. The liner was selected based on deltoid tension and stability. If possible, the subscapularis tendon was closed after humeral stem implantation. A postoperative radiograph of the prosthesis was obtained and is shown in Figure 2.

Postoperative anteroposterior radiograph of a right shoulder showing the result after implantation of a reverse Grammont-style shoulder prothesis (DELTA XTEND; DePuy Synthes) with 155° shaft-neck angle and medialized baseplate at neutral version and inclination.
Postoperative Rehabilitation
Patients who underwent SCR were placed in an abduction pillow and immobilized for 6 weeks. At 6 weeks postoperatively, they began full passive and active-assisted range of motion exercises as tolerated. Between 8 and 10 weeks after surgery, patients were permitted to progress to full active range of motion and begin strengthening exercises. Patients who were pain-free and had regained good function were cleared to return to full and recreational activities without restriction at 4 to 5 months postoperatively.
Patients who underwent RTSA began full passive range of motion the day after surgery, with external rotation limited to 30° for the first 3 weeks if the subscapularis was repaired (n = 7). At 3 weeks postoperatively, patients progressed to full active range of motion as tolerated. At 7 weeks, initial resistance strengthening was allowed. Pain-free patients with good function were cleared to return to full activities and sports without restriction at 4 months postoperatively.
Outcome Assessments
Several assessments were carried out preoperatively (previously recorded before surgery) and at final follow-up via email surveys. These included the American Shoulder and Elbow Surgeons (ASES) Standardized Shoulder Assessment Form score, 21 Single Assessment Numeric Evaluation (SANE) score, 34 the shortened version of Disabilities of the Arm, Shoulder and Hand (QuickDASH) score, 6 the 12-item Short Form Health Survey (SF-12) Physical Component Summary (PCS) and Mental Component Summary (MCS), 8 and patient satisfaction (on a 1-10 scale, with 10 indicating "very satisfied"). All scores were completed directly by the patients themselves; no blinded evaluator was involved in scoring. Additional optional questions were asked to evaluate patients’ participation in sports, both before and after surgery. These questions assessed the ability to perform recreational sports, with the following response options: “unable,”“very difficult,”“somewhat difficult,” and “normal.” All responses, except for “unable” and “very difficult,” were considered as ability to participate. Any complications, revision surgeries, or clinical failures were documented. Clinical failure was defined as a recurrence or persistence of function loss compared with the preoperative state and/or a recurrence or persistence of pain equal to or greater than the preoperative level. Kaplan-Meier survivorship analysis was also conducted. Furthermore, the minimal clinically important difference (MCID), Patient Acceptable Symptom State (PASS), and substantial clinical benefit (SCB) for ASES and SANE scores were calculated for both groups. 7
Statistical Analysis
Statistical analyses were performed using SPSS Version 31.0 (IBM Corp). For normally distributed variables, an independent or paired t-test was used and for nonparametric data, the Mann-Whitney or chi-square for bivariate tests was performed. Survivorship analysis was performed using Kaplan-Meier survival curves for progression to revision repair or shoulder arthroplasty on the index shoulder as an endpoint. Due to the rarity of the injury, all eligible patients were considered, and a power analysis was not performed. A P value of <.05 was considered statistically significant.
Results
Patient Demographics and Failures
A total of 44 patients who underwent SCR with DA between October 2014 and September 2018 met the inclusion criteria. Of those, 10 patients were either deceased (n = 2) or had refused to participate in the study (n = 8). The remaining 34 patients were included in the final cohort and had a mean age at surgery of 56.5 years (range, 41.2-67.2). Eleven patients (11/34; 32.4%) failed, 9 of whom underwent revision SCR (2/34; 5.9%) or progressed to RTSA (7/34; 20.6%). Two patients were lost to follow-up. Outcome assessment of the remaining patients (21/23; 91.3%) was obtained with a mean follow-up of 6.7 ± 1.4 years (range, 5.0-9.0 years) (Figure 3).

Patient flow diagram. PRO, patient-reported outcome; RTSA, reverse total shoulder arthroplasty; SCR, superior capsular reconstruction; TSA, total shoulder arthroplasty.
A total of 107 patients <70 years of age underwent RTSA between January 2006 and September 2018. A total of 70 patients were omitted due to different criteria, including previous arthroplasty, humeral fractures, and not being indicated for SCR. Additionally, 2 patients refused to participate. The remaining 35 patients were included in the final cohort and had a mean age at surgery of 63.3 years (range, 46.1-70.0). Three patients experienced failure (3/35; 8.6%) due to component loosening and were revised. Outcome assessment of the remaining patients (26/32; 81.3%) was obtained, with a mean follow-up of 9.5 years (range, 5.0-16.3) (Figure 3). Patient baseline characteristics for the groups are summarized in Table 1. Both groups significantly differed in age (RTSA group significantly older; P < .001), and length of follow-up (RTSA group had a significantly longer follow-up; P < .001), as seen in Table 1.
Baseline Patient Characteristics of the SCR Group and RTSA Group a
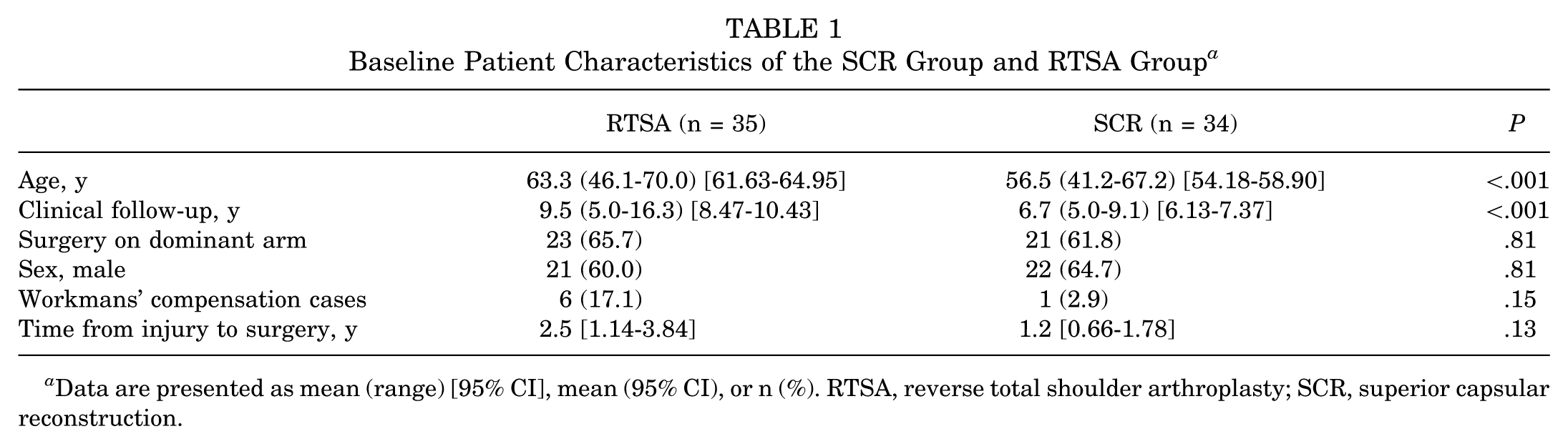
Data are presented as mean (range) [95% CI], mean (95% CI), or n (%). RTSA, reverse total shoulder arthroplasty; SCR, superior capsular reconstruction.
Kaplan-Meier survivorship analysis (Figure 4) demonstrated a clear divergence between the 2 procedures over time. At 5 years postoperatively, the RTSA group showed significantly higher survivorship of 90.1% compared with 75.0% in the SCR group (log-rank test; P = .02). The survival curve for RTSA remained relatively stable throughout the follow-up period, whereas the SCR group demonstrated a steeper decline in the early postoperative years.
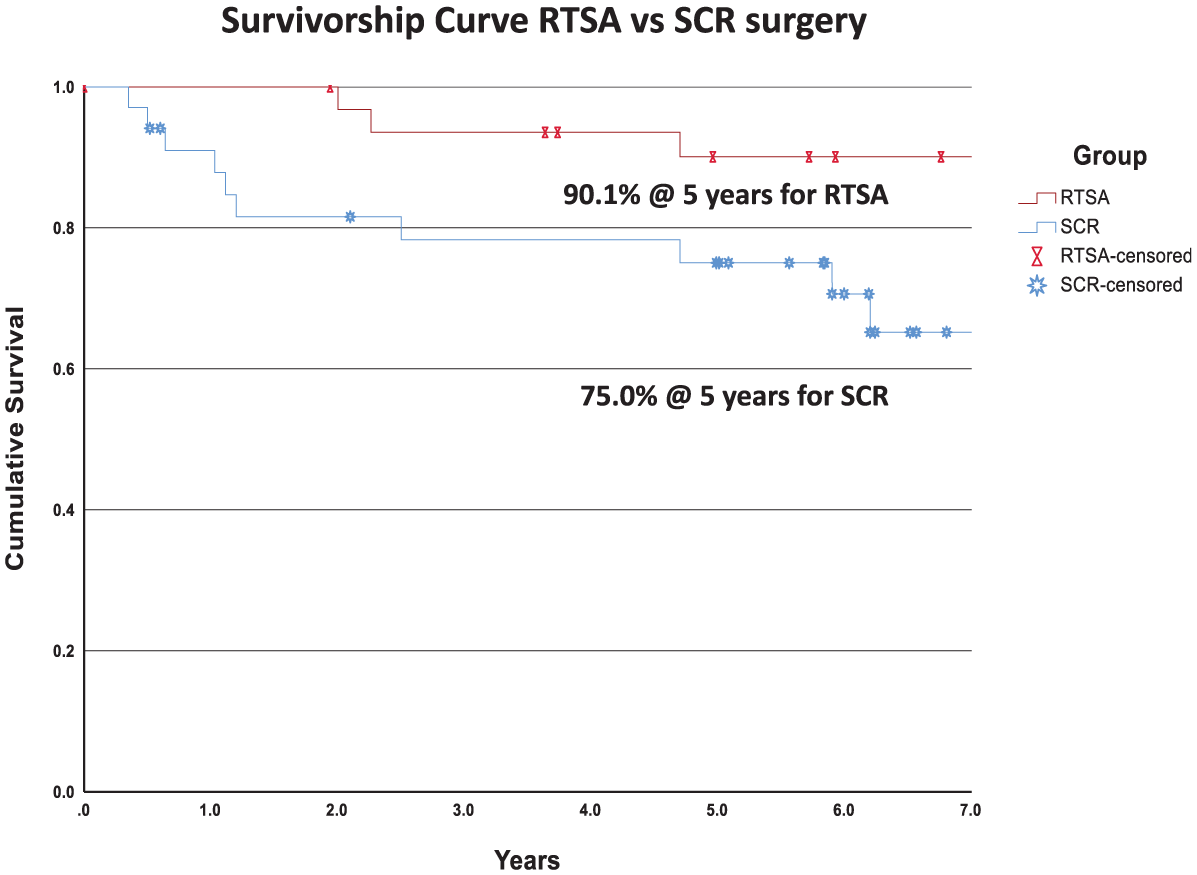
Kaplan-Meier survivorship curve comparing RTSA (95% CI, 13.6-16.4) and SCR (95% CI, 5.6-7.9). RTSA, reverse total shoulder arthroplasty; SCR, superior capsular reconstruction.
Clinical Outcome, Patient Satisfaction, and Return to Sport
Aside from mentioned failures, no complications were reported in either group. Preoperative patient-reported outcome measures (PROMs) were similar between groups (all P > .05). All postoperative outcome scores improved significantly from preoperatively to postoperatively at final follow-up in both groups, as shown in Table 2. No statistically significant differences between SCR and RTSA outcomes were detected in outcome scores, as shown in Table 3, except for QuickDASH (RTSA, 24.3 ± 19.5; SCR, 11.3 ± 12.0; P = .009), and SF-12 PCS (RTSA, 44.3 ± 12.2; SCR, 52.1 ± 7.8; P = .016), both favoring SCR. The median patient satisfaction was 9 of 10 (range 5-10) for SCR and 9 of 10 (range 1-10) in RTSA. RTS rates in Table 4 show improvement in both groups, with significant progress in the SCR group compared with minimal changes in the RTSA group for pain in competition and level of sport participation. While both groups indicated reasons such as lifestyle (RTSA, 8/25; SCR, 6/21; P = .68) and pain (RTSA, 2/25; SCR, 3/21; P≥ .99) at the same percentages. Significantly more SCR patients reported weakness (RTSA, 1/25; SCR, 11/21; P < .001) as the reason they modified their activities. The MCID for ASES in RTSA compared with SCR was 89.5% versus 88.9%; for SCB, it was 68.4% versus 88.9%; and for PASS, 43.5% versus 66.7%; no statistically significant differences were detected between groups. MCID for SANE (RTSA vs SCR, 73.7% vs 70.0%; SCB, 52.6% vs 50.0%; PASS, 68.0% vs 55.0%); no statistically significant differences were detected between groups.
Comparison of Preoperative and Postoperative Patient-Reported Outcome Scores in RTSA and SCR a
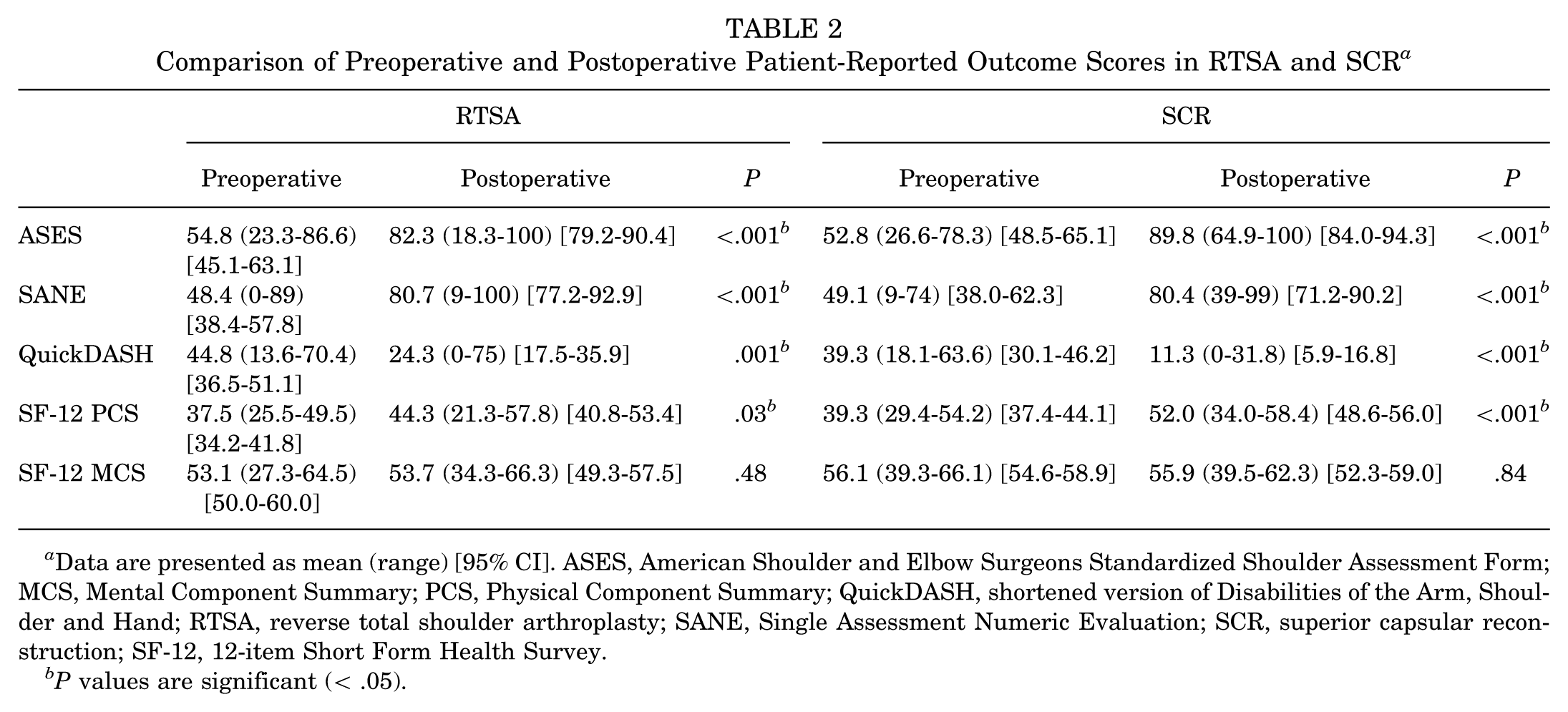
Data are presented as mean (range) [95% CI]. ASES, American Shoulder and Elbow Surgeons Standardized Shoulder Assessment Form; MCS, Mental Component Summary; PCS, Physical Component Summary; QuickDASH, shortened version of Disabilities of the Arm, Shoulder and Hand; RTSA, reverse total shoulder arthroplasty; SANE, Single Assessment Numeric Evaluation; SCR, superior capsular reconstruction; SF-12, 12-item Short Form Health Survey.
P values are significant (< .05).
Comparison of Postoperative Patient-Reported Outcome Scores Between RTSA and SCR a

Data are presented as mean (range) [95% CI] unless otherwise indicated. ASES, American Shoulder and Elbow Surgeons Standardized Shoulder Assessment Form; MCS, Mental Component Summary; PCS, Physical Component Summary; QuickDASH, shortened version of Disabilities of the Arm, Shoulder and Hand; RTSA, reverse total shoulder arthroplasty; SANE, Single Assessment Numeric Evaluation; SCR, superior capsular reconstruction; SF-12, 12-item Short Form Health Survey.
P values are significant (< .05).
Pre- to Postoperative Comparison of Specific Shoulder Pain and Function Questions for SCR and RTSA a
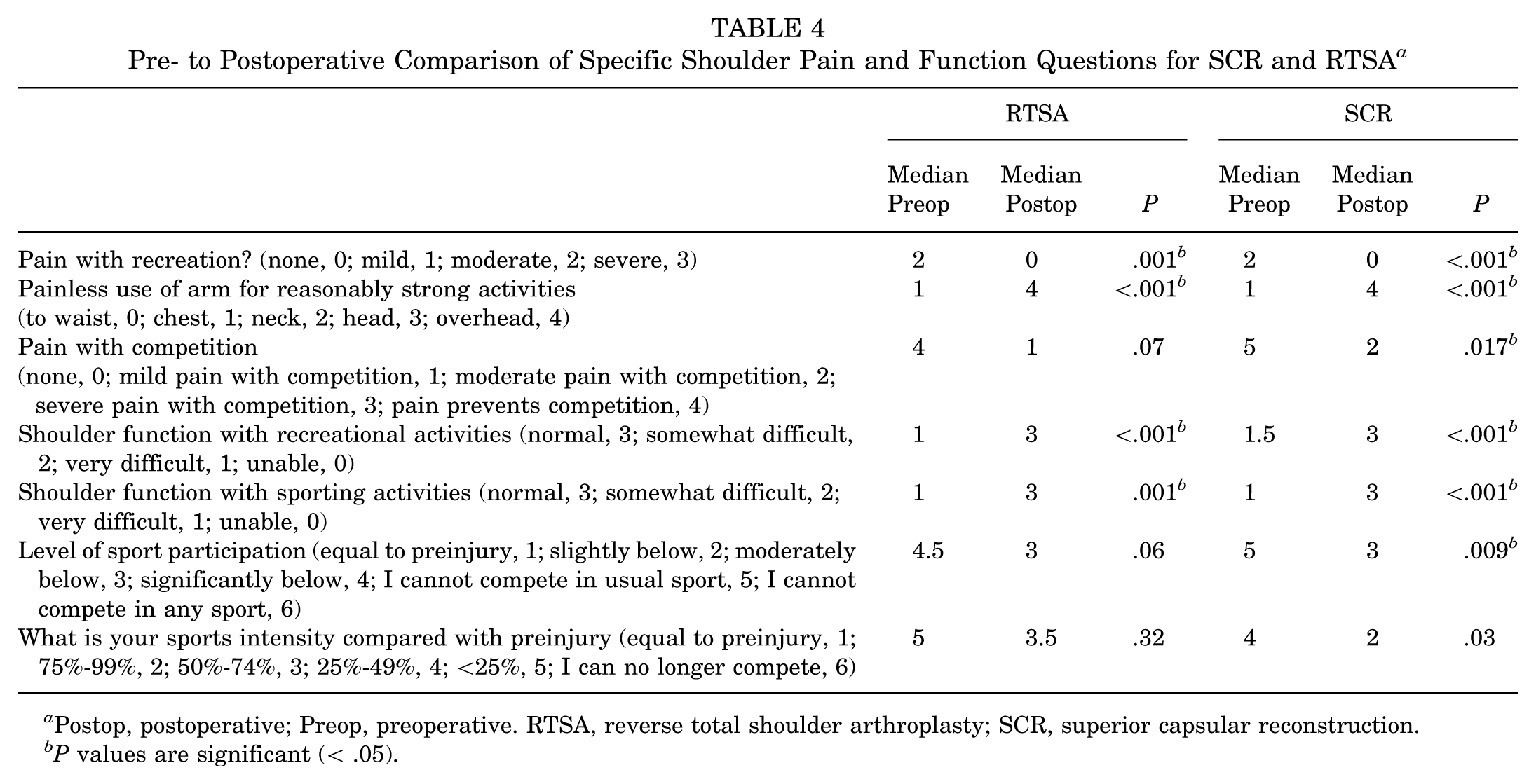
Postop, postoperative; Preop, preoperative. RTSA, reverse total shoulder arthroplasty; SCR, superior capsular reconstruction.
P values are significant (< .05).
Discussion
The most important findings in this study are that RTSA demonstrated superior durability and lower failure rate compared with the SCR group at ≥5 years. This supports our hypothesis that the outcomes would favor RTSA. In contrast, SCR did perform well in those that did not experience failure after surgery. For example, in patients whose procedure was not a failure, both procedures significantly improved all PROMs at follow-up. The SCR group actually had significantly better QuickDASH scores and SF-12 PCS scores than the RTSA group, but ASES, SANE, SF-12 MCS, and patient satisfaction were similar. The results suggest that RTSA offers better long-term durability and a lower risk of failure in patients <70 years with symptomatic, irreparable posterosuperior RC tears without GHOA, but that SCR may be a good option that preserves the joint and delays the need for shoulder arthroplasty.
The initial 2-year results comparing these 2 procedures (SCR and RTSA) from our institution, with a partially overlapping patient population, showed results that were more similar in outcome, with comparable failure rates, improvement in clinical outcomes, and high return-to-sport rate. 17 However, at mid-term follow-up, in the present study, a significant revision rate in the SCR group became evident. It should be noted that this study was done with a Grammont-style implant with a 155° neck-shaft angle and a medialized center of rotation. The senior author has since transitioned to implants with a more anatomic 135° neck-shaft angle and greater glenoid lateralization, based on accumulating evidence suggesting improved biomechanics, functional outcomes, and reduced complications with these designs. 11 This reflects an evolution in surgical technique and implant choice over time, rather than a direct conclusion from the present results. It is possible that this may result in improved PROMs in the RTSA group, and it is also possible that future technical improvements in SCR could reduce the need for revision surgery and improve survivorship.
Initially, RTSA was primarily indicated for elderly patients with RC arthropathy. However, recent trends indicate a significant increase in the use of RTSA in younger patients and for a wider range of indications. 1 Best et al 2 found that the incidence of primary RTSA increased substantially from 2012 to 2017, with the greatest increase observed in patients aged 50 to 64 years. Results of RTSA for irreparable RC tears in patients without joint arthropathy or GHOA are generally positive, with significant improvements in pain relief, shoulder function, and range of motion beyond 10 years.10,26 Nevertheless, joint preservation remains a desirable standard of care particularly for younger, more active patients. 27 Joint-preserving treatments before RTSA are also deemed more cost-effective than performing a primary RTSA. 9 SCR is still a relatively new procedure and mid- to long-term follow-up is being reported scarcely with varying results. Mihata et al 23 reported their outcomes after arthroscopic SCR using fascia lata autograft for irreparable RC tears. Their study demonstrated significant improvements in ASES and Japanese Orthopaedic Association scores and a failure rate of 10% after a 5-year follow-up. In a later 10-year follow-up study, the same group reported sustained improvements in shoulder function and high return-to-sport rates. The mean ASES score was 95.1 at 5 years compared with 91.3 in the present study, with a fascia lata autograft survival rate of 89% (32/36) after 5 to 10 years, 24 compared with the survival rate of SCR in our study of 75%. The reason SCR with DA appears to have inferior results compared with fascia lata autograft is still unclear. Lee and Shin 19 demonstrated that reduced graft volume after SCR with DA is associated with inferior outcomes, and this was reported in 42% of 81 patients in their cohort. Fascia lata allografts have also been shown to provide superior biomechanical stability compared with DA in terms of length and stiffness under physiological loads, whereas DAs tend to elongate by approximately 15% during testing, with reports of peak elongation reaching up to 171% in certain regions during shoulder movements, compared with intraoperative length.5,20,22,32 More recently, biomechanical studies have investigated the potential benefits of using a thicker DA of around 5 to 6 mm, which has a comparable thickness to the fascia lata allograft.5,31 The results showed that the thicker graft better restored native glenohumeral joint properties. 32 In terms of clinical application, Snow et al 33 recently reported their outcomes 48 months after SCR in 31 patients using both a standard and a doubled (folded) DA. The postoperative ASES score was 76.3 ± 25.1, which is relatively lower than in our study but clinical failures (n = 4) and revisions (n = 1) were all in patients with the standard DA. 32 On the other hand, it is possible that RTSA with a more anatomic 135° neck-shaft angle and a more lateralized glenoid could lead to better functional outcomes, supporting RTSA as the preferred treatment strategy. SCR still offers the advantage of joint preservation, and as seen in this patient cohort, the potential of equal or better clinical results compared with RTSA. However, utilizing it generously as a first option should be done with caution. Saad Berreta and colleagues 30 found that patients with a history of failed SCR to address RC insufficiency before RTSA had greater range of motion deficits, higher complication rates, and lower rates of achieving clinically significant outcomes than those without previous ipsilateral shoulder surgery. This presents the need for a stricter primary treatment consideration in these patients.
Limitations
This study is limited by its retrospective design, which is inherently subject to biases such as selection bias, recall bias, and lack of randomization. The retrospective nature restricts the ability to control for confounding factors that may influence the results. Additionally, there is an inconsistent follow-up durations and recruitment periods between the 2 groups (RTSA, 2006-2018; SCR, 2014-2018), which may introduce a potential temporal bias. Another limitation is the difference in study periods between the 2 cohorts. Advances in surgical techniques, implant design, rehabilitation protocols, and perioperative care over time could have influenced the outcomes, particularly in the RTSA group where earlier cases may not fully reflect current standards of practice. Conversely, the relatively short recruitment period of the SCR cohort may capture outcomes in a more contemporary surgical environment. This temporal discrepancy represents a potential confounding factor that should be considered when interpreting the comparative results of the 2 procedures. A further limitation is that adherence to postoperative rehabilitation was not systematically tracked. Although standardized protocols were prescribed, variability in compliance may have influenced outcomes. Although this proportion was relatively low, missing data could still introduce bias. Importantly, no significant differences in baseline demographics or preoperative characteristics were identified between patients who completed follow-up and those who were lost, reducing the likelihood of systematic bias. Our outcome assessment also lacked detailed data regarding notching and data on individual sports.
Conclusion
In treating irreparable posterosuperior RC tears in younger patients, RTSA demonstrated superior survivorship (90.1%) compared with SCR with DA (75.0%) at mid-term follow-up. However, in patients without treatment failure, both RTSA and SCR had equal improvements in most PROMs at ≥5 years of follow-up, with some scores favoring SCR in nonfailure cases. Higher RTS and PCS, as well as lower disability, were noted in the SCR group.
Footnotes
Final revision submitted September 29, 2025; accepted October 19, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: A.J. has received a grant from AGA outside of the submitted work sponsored by Arthrex Inc. B.M.K. has received support for education and grants from Arthrex, Smith & Nephew, DJO LLC, and Stryker Corp and reimbursement from Stryker. J.J.L. has received a grant from Smith & Nephew. M.T.P. receives royalties from Arthrex Inc, Arthrosurface, Responsive Arthroscopy, and Anika Therapeutics Inc; has received consulting fees from Arthrex Inc, Joint Restoration Foundation, Zimmer Biomet Holdings, and Arthrosurface; has received grants from the Department of Defense, the National Institutes of Health, and DJO; and has received honoraria from Flexion Therapeutics. P.J.M. has received royalties, consulting fees, and research support from Arthrex and holds stock options in VuMedi. A.J., M.P.H., M.H., C.J.H., C.S.D., and J.J.L. are supported by the Steadman Philippon Research Institute (SPRI), which is a 501(c)(3) nonprofit institution supported financially by private donations and corporate support. SPRI exercises special care to identify any financial interests or relationships related to research conducted here. During the past calendar year, SPRI has received grant funding or in-kind donations from Arthrex, Canon, DJO, Icarus Medical, Medtronic, Ossur, Smith & Nephew, SubioMed, Stryker, and Wright Medical. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the WCG institutional review board (protocol Number 20244664).
