Abstract
Background:
Previous studies have sought to determine the effect of inpatient ketamine therapy on postoperative pain in a variety of surgical specialties.
Purpose:
To determine the effects of postoperative ketamine analgesia after periacetabular osteotomy (PAO) and/or derotational femoral osteotomy (DFO) on opioid requirements, pain, and discharge time.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
Prospective data were collected on 145 patients who underwent PAO and/or DFO by the senior author between January 2021 and December 2022. Hip arthroscopy was performed 3 to 10 days before addressing any intra-articular pathology. In 2021, patients (n = 91 procedures; control group) received a traditional postoperative multimodal pain regimen. In 2022, postoperative low-dose ketamine (0.1-1 mg/kg/h) was added to the multimodal analgesic approach until 24 hours before discharge (n = 81 procedures; ketamine group). The ketamine and control groups were matched based on procedure type. Total opioid consumption was collected using milligram morphine equivalents (MMEs) for both groups. Postoperative pain was measured using the Defense and Veterans Pain Rating Scale (DVPRS), which was analyzed as the mean score per day. Data on the mean MME and DVPRS were analyzed for up to 7 days postoperatively. Linear mixed statistical analysis was performed to determine the significance of low-dose postoperative ketamine on postoperative pain and opioid utilization.
Results:
Patients who did not receive ketamine after PAO and/or DFO utilized a mean of 181 ± 335 MMEs and had a mean DVPRS score of 4.18 ± 1.63. Patients who received postoperative ketamine required a mean of 119 ± 291 MMEs and had a mean DVPRS score of 4.34 ± 1.61. The ketamine group was found to consume a significantly lower total MME dose per day (P < .001). No significant difference was found in the mean DVPRS score between the ketamine and control groups (P = .42). Also, no significant difference was found on the day of discharge (P = .79).
Conclusion:
Patients who received postoperative ketamine after PAO and/or DFO had a significant decrease in MME dose when compared with a control group of patients who did not receive ketamine. Surgeons should consider adding ketamine to their postoperative multimodal pain control protocol to decrease opioid consumption while adequately addressing postoperative pain.
Periacetabular osteotomy (PAO) is a joint-preserving surgical treatment for predominately opioid-naïve young adults (mean age, around 29 years) with symptomatic acetabular dysplasia. 25 PAO surgery reduces pain associated with instability-related labral tears and cartilage damage and may delay or prevent the need for a future hip replacement. Sometimes, a derotational femoral osteotomy (DFO) is utilized in conjunction with PAO to correct excessive femoral ante- or retroversion commonly occurring with hip dysplasia. These procedures are associated with significant postoperative pain, and pain control is critical for gaining early postoperative function. Poor control of postoperative pain has been shown to be a critical factor for adverse surgical outcomes such as cardiopulmonary complications and chronic surgical pain.31,35
Current options utilized for pain control after PAO and/or DFO include opioid analgesics, lumbar or spinal epidural anesthetic, patient-controlled analgesia (PCA), multimodal nonopiate oral medications, anxiolytics, and nerve blocks in conjunction with muscle relaxants. Opioids have been one of the most utilized and effective postoperative analgesics for many years. However, opioids are associated with a myriad of side effects—including constipation, respiratory depression, paradoxical hyperalgesia, and addiction. 5 Currently, it is a national priority to decrease opioid use because of the opioid epidemic in the United States. 36 There was a 124% rise in unintentional opioid overdose deaths between 1999 and 2007, and with orthopaedic surgeons being the third highest prescriber of opioids since 2010, it is imperative to investigate alternative analgesic options.6,30 In addition to the addictive potential of opioids, research has shown that opioid use is associated with worse clinical outcomes in several orthopaedic conditions. 30
The clinical utility of ketamine is wide-ranging, and it is commonly used during general anesthesia. At subdissociative doses, ketamine provides pain relief and can be used in conjunction with a typical multimodal pain regimen.21,27,34 Ketamine is in the phencyclidine drug class. It is a noncompetitive antagonist of N-methyl-
To our knowledge, the addition of ketamine to a multimodal analgesic regimen after PAO and/or DFO has not been investigated. This study aimed to determine the effects of postoperative ketamine analgesia after PAO and/or DFO on opioid requirements, pain, and discharge time. We hypothesized that there would be a significant reduction in postoperative opioid requirements, with moderately improved pain control among patients given postoperative ketamine in addition to the preexisting analgesic treatment regimen.
Methods
A retrospective analysis was performed on prospectively collected data to evaluate the effectiveness of ketamine on post-PAO and/or -DFO analgesia (institutional review board approval, 12-1385). A total of 145 patients who underwent PAO and/or DFO between January 2021 and December 2022 were analyzed in this study (Table 1). All PAO and DFO procedures were performed by the senior author (O.M.D.). Participants of any age, race, and sex who had the diagnosis of symptomatic hip dysplasia and/or femoral torsion abnormalities who underwent PAO and/or DFO were included in the analysis. Clinical diagnosis of acetabular dysplasia was determined according to accepted pathomorphologic signs and measurements. 18 The following aided in establishing a diagnosis of symptomatic hip instability: history of hip pain; positive findings on provocative hip tests indicating intra-articular hip pain; radiographic evidence of hip dysplasia (lateral center-edge angle of ≤25°, sourcil angle of ≥10°); excessive acetabular version and/or femoral antetorsion; interruption of the Shenton line on the weightbearing anteroposterior pelvic radiograph; and magnetic resonance imaging (MRI) findings of labral hypertrophy and tears, articular cartilage thickening and/or inside-out chondral flaps, or a ligamentum teres tear.8,15,17,18 Patients selected for surgery underwent preoperative computed tomography and MRI to assess the acetabular version, femoral torsion, and femoral-head sphericity as well as cartilage, labral, and subchondral bone integrity.
Number of Procedures and Procedure Types a

DFO alone represents patients who underwent DFO >7 days before PAO and had the data analyzed separately. The total PAO analyzed represents the total number of PAOs performed, including those performed in patients who underwent DFO. The patients who underwent DFO and PAO <7 days apart are included in the number of PAOs analyzed. DFO, derotational femoral osteotomy; PAO, periacetabular osteotomy.
Data on daily opioid usage and pain scores were collected until discharge. Patients hospitalized for >7 days (often secondary to social/insurance aspects rather than medical reasons) were considered outliers, and only data up to postoperative day 7 were analyzed. All patients underwent hip arthroscopy 3 to 10 days before PAO to address any intra-articular pathology. 25
Study Protocol
Patients who underwent PAO and/or DFO between January 2021 and December 2021 received the standard analgesic treatment and were used as the control group (91 procedures). The standard multimodal pain regimen included preoperative acetaminophen (1 g), preoperative celecoxib (400 mg), preoperative gabapentin (900 mg), intraoperative intrathecal morphine, and spinal anesthesia. No local anesthetic was utilized at the operative site because of the need for sciatic nerve function testing postoperatively. Standard postoperative pain control consisted of scheduled oral oxycodone (10 mg every 6 hours), oral acetaminophen (1 g every 6 hours), and oral naproxen (220 mg 3 times daily) or celecoxib (200 mg 3 times daily). As-needed medications for uncontrolled pain included oral oxycodone (5-10 mg), oral hydromorphone (1-2 mg), intravenous hydromorphone (0.5-1 mg), and oral diazepam for muscle spasms (5 mg). Although rarely utilized, there was an option for PCA with hydromorphone. Pain medication utilization was delegated to the discretion of the nursing staff, floor physician assistant, and anesthesia team based on patient requirements. Discharge pain medications did not differ between the experimental and control groups.
Starting in January 2022, low-dose ketamine was added to the standard postoperative pain regimen for patients undergoing PAO and/or DFO (81 procedures). Ketamine was initiated postoperatively in the operating room and was continued on the floor until 24 hours before expected discharge to ensure patient tolerance for home pain medications. The starting dose of ketamine was 0.1 mg/kg/h, and the anesthesia team could titrate the dose up by 0.1 mg/kg/h at a time to a maximum dose of 1 mg/kg/h. If a patient reported inadequate pain control and was tolerating the ketamine therapy, the dose of ketamine was increased. Most patients were satisfied with a dose of 0.1 to 0.3 mg/kg/h, and higher doses were rarely needed.
To standardize the amount of postoperative opioid consumption, opioid requirements were converted to the morphine milligram equivalent (MME) dose received per day. 13 Pain scores for all patients undergoing PAO and/or DFO were measured using the Defense and Veterans Pain Rating Scale (DVPRS). 7 The DVPRS score was measured on a scale of 0 to 10 points, with lower values indicating less pain. Pain measures were obtained by a registered nurse throughout each day of hospitalization and were analyzed as the mean score per day. Concurrent audits on ketamine use were completed to monitor for adverse events, safety, and efficacy. Discussions with the floor treatment team were held to monitor for extra potential burdens associated with the addition of ketamine to the pain regimen.
Discharge criteria were based on pain control with oral medications, working with physical and occupational therapy, and clearing postoperative radiographs. Physical and occupational therapy milestones for discharge were based on the patient's ability to clear stairs, transition from bed to walking, bathroom usage alone, and safe movement between rooms.
Matching Protocol
Patients who underwent PAO and/or DFO were matched with controls who best matched their surgical path. Patients who underwent only PAO or DFO were matched with controls who only had PAO or DFO. Patients who scheduled DFO then PAO >7 days apart for logistical reasons were considered separate cases and were analyzed individually. Patients who had DFO together with PAO were analyzed as 1 procedure, and they were matched with a control who had the same procedural scenario. There were nearly identical surgical scenarios between groups to control bias as much as possible.
Statistical Analysis
MME and DVPRS score data collection began on the date of the PAO and/or DFO. In this analysis, we used linear mixed models to compare the total MME and DVPRS scores over time between patients who did and did not receive low-dose ketamine after PAO and/or DFO. These models are helpful in handling repeated measurements within the dataset, considering both fixed and random effects to minimize bias in estimating mean parameters. We chose restricted maximum likelihood estimation, effective for unbiased parameter estimates in repeated measurements. We also selected a heterogeneous compound symmetric covariance structure to address within-subject and between-subject variability, ensuring result integrity. Importantly, including patients as a random effect addresses repeated measurements and accommodates variations associated with individual patients and multiple surgeries. We assessed model assumptions, which address the expected lack of independence in longitudinal data and confirm the normality assumption through diagnostic plots. The chosen statistical methodology—including mixed linear models and thorough assessments—is robust and appropriate for drawing valid conclusions about differences in total MME and DVPRS scores over time in patients who received low-dose ketamine.
To preserve the assumption of normality of errors, the log transformation of the total MME was taken rather than modeling the untransformed total MME. For the DVPRS score, no transformation was needed. To determine differences in the day of discharge between groups, an independent t test was utilized. Summary statistics are reported as means and standard deviations for continuous numeric variables and counts and relative frequencies for categorical variables. Statistical significance was set at P < .05. R Studio (Version 4.3.2) was used for all analysis.
Results
Participants and Descriptive Data
The total number of patients analyzed in this study was 145, with 172 surgeries analyzed (study group: 81 procedures, control group: 91 procedures) (Table 1). Four patients underwent 1 hip surgery as part of the control group (ie, no ketamine was used), and their other hip was treated with the addition of ketamine to the inpatient pain management protocol. Table 2 demonstrates the demographics of both groups. The mean opioid consumption was 181 (SD, 335) MMEs for the control group and 119 (SD, 291) MMEs for the ketamine group. The mean pain score per day for the control group was 4.18 (SD, 1.63) and 4.34 (SD, 1.61) for the ketamine group.
Patient Characteristics a

Age and BMI are presented as mean (SD). BMI, body mass index.
A linear mixed model using the log transformation of the total MME over time was constructed to evaluate the differences in opioid consumption between the ketamine and control groups (Figure 1). Patients in the study group used significantly less total MME per day than the control group (P < .001). There was also statistical evidence that the experimental group required less MME on postoperative day 1 and less MME over time, indicating an effect of ketamine compared with the control and time (P = .035).

Model of daily mean MME utilization over time in the experimental and control groups. Error bars indicate 95% CIs. DOS, date of surgery; MME, morphine milligram equivalent.
A second linear mixed model was utilized to evaluate the differences in DVPRS scores over time between the study and control groups (Figure 2). No statistical difference was found between the experimental and control groups with respect to DVPRS scores (P = .42). The mean discharge day for the control group was 4.96 (SD, 1.13) days and 5.02 (SD, 1.37) days for the study group (P = .79).
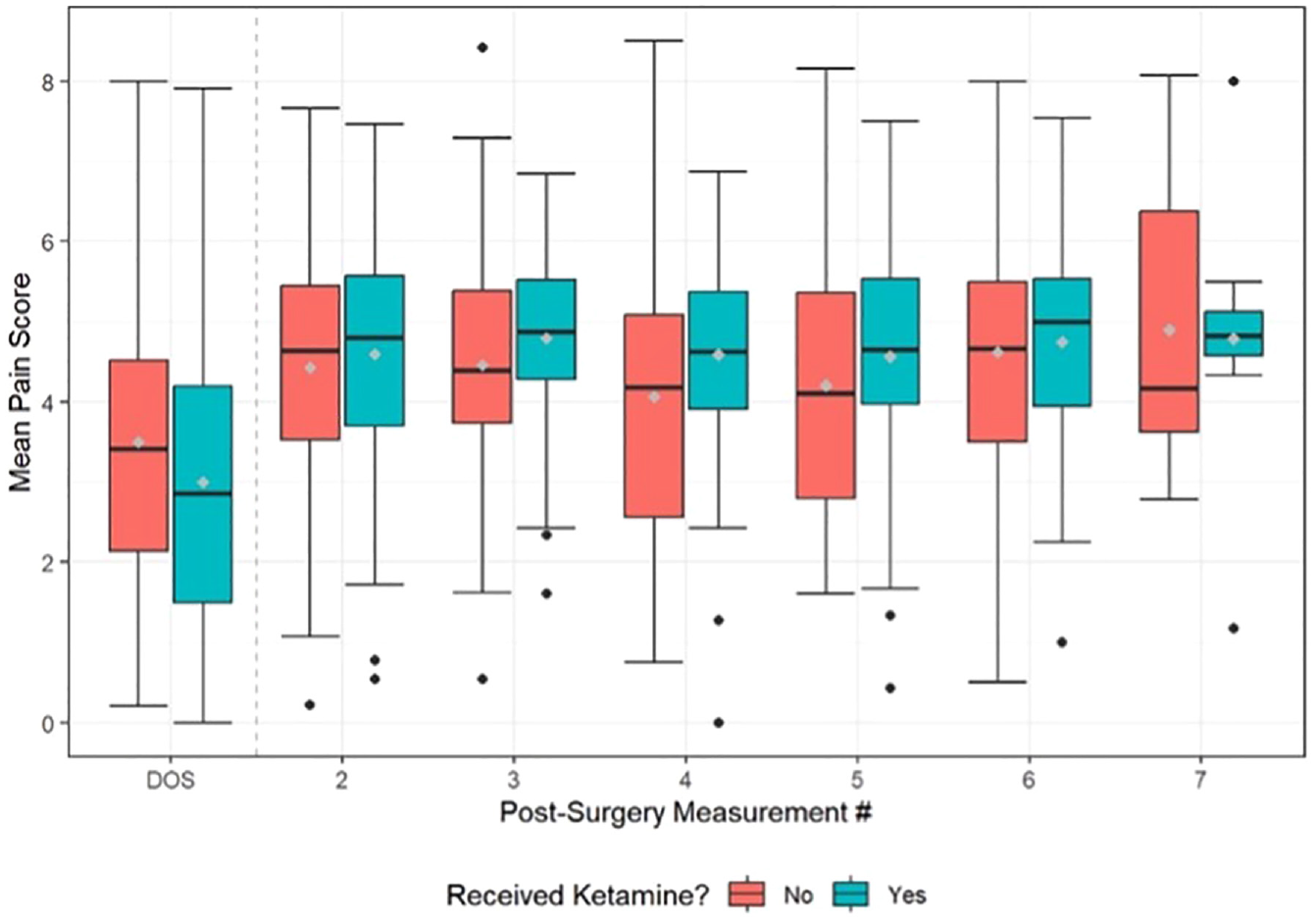
Model of daily mean DVPRS over time in the experimental and control groups. Error bars indicate 95% CIs. DOS, date of surgery; DVPRS, Defense and Veterans Pain Rating Scale.
There was only 1 documented adverse event from ketamine in this study: a patient experienced severe dysphoria and dissociation. However, in this case, it was found that the ketamine drip was inadvertently started at the highest setting (1 mg/kg/h), and this patient's side effects likely occurred because of the dosing error. The inpatient physician assistant reported anecdotal evidence of dysphoria from a handful of patients. However, this did not require or result in any changes to the protocol. The floor treatment team did not report any added difficulties to postoperative care after the addition of low-dose ketamine therapy to the protocol.
Discussion
The most important finding from this study was that patients who received postoperative ketamine after PAO and/or DFO required significantly fewer postoperative opioids during hospitalization without compromising pain control. The novel findings of this study demonstrate a beneficial role for postoperative ketamine after PAO and/or DFO. This also adds to the growing body of literature supporting the role of ketamine in postoperative opioid-sparing pain control.
There is significant evidence that intraoperative and postoperative use of ketamine reduces postoperative opioid consumption in both opioid-naïve and opioid-dependent patients after a variety of procedures. ¶ Multiple systematic reviews have demonstrated that ketamine therapy is beneficial for both pain control and limiting opioid use after orthopaedic surgeries.19,24,32 Riddel et al 32 performed a meta-analysis on 20 randomized controlled trials that investigated the use of ketamine for up to 48 hours after surgery—including joint arthroplasty, spine surgery, and arthroscopic surgery. The authors found that ketamine was a useful adjuvant for decreasing pain and opioid requirements after orthopaedic surgery.
To our knowledge, the use of ketamine postoperatively for adjuvant pain control after PAO and/or DFO has not been investigated. Previous studies have investigated the use of epidural anesthesia, local infiltration site analgesia, and transversus abdominis plane block for pain control after PAO.2,12,16,20 For patients undergoing hip or knee arthroplasty, epidural anesthesia was associated with postoperative nausea and vomiting, and it was found to be associated with decreased pain for only the first 4 to 6 hours after surgery.1,9 In patients undergoing PAO, the removal of the epidural earlier in the hospitalization (ie, postoperative day 1 vs day 2) has been associated with decreased postoperative pain and less opioid utilization. 10 The use of local infiltration site analgesia did not show a significant reduction in opioid usage after PAO. 2 Transversus abdominis plane block was associated with a significant decrease in opioid use and pain during the initial 48 hours after PAO. 20 Our study demonstrated the utility of postoperative ketamine after PAO and/or DFO for reducing opioid requirements for up to 7 days after surgery, without elevation of pain scores compared with the control group.
It is also important to consider other potential effects (ie, effects on the hospital system, patients, and floor providers) associated with the addition of ketamine to a postoperative pain regimen. Notably, the floor treatment team did not report any extra burden with the addition of low-dose ketamine therapy to the protocol. A study on patients undergoing neurosurgery showed that adding postoperative, short-duration, low-dose ketamine lowered the total medical costs associated with the hospitalization, mainly secondary to decreased length of stay. 23 In contrast, our findings did not reveal a statistical difference in the length of hospitalization.
In a systematic review, Bell et al 4 demonstrated that low-dose ketamine for postoperative pain (in nonorthopaedic surgeries) was accompanied by a small incidence of side effects. Out of 37 trials analyzed, this review reported that only 4 trials recorded some psychotomimetic side effects due to ketamine (ie, dysphoria), and 21 trials specifically stated that there were no psychotomimetic side effects observed secondary to ketamine analgesia. 4 In our study, there was only 1 formally documented side effect of dysphoria and dissociation, although it was due to a dosing error. A handful of dissociative side effects due to ketamine were anecdotally reported by the inpatient physician assistant, but these side effects were not formally documented by the treatment team because they were minor and lacked any physiologic or clinical sequela. Four patients underwent 1 hip surgery as part of the control group (ie, no ketamine was used), and their other side was done with the addition of ketamine to the inpatient pain management protocol. Although this did not constitute enough power for significance, these patients reported improved pain control with the addition of inpatient ketamine versus without. Overall, patients who underwent procedures in both the control and experimental groups reported better satisfaction with the addition of ketamine to the postoperative pain regimen.
The dissociative effects of ketamine may have inadvertently resulted in a decreased ability to request more pain medication. One patient reported struggling to advocate for more opioids as a result of dissociation from the ketamine. Further formal investigation into minor dysphoria and dissociation after ketamine analgesia is required.
Surgeons may use this information to implement alternative options for a multimodal opioid-reducing pain reduction strategy after PAO and/or DFO. Given the significant effectiveness of postoperative ketamine and the limited side effect profile, it may be considered for all patients undergoing PAO and/or DFO. Limiting opioid utilization may be effective for the mitigation of undesired side effects like hyperalgesia, addiction, constipation, and respiratory depression. As many patients who undergo PAO are young and opioid-naïve, it is of utmost importance for orthopaedic surgeons to explore alternatives to opioids for postoperative pain control to limit addiction potential. In addition, due to the osteotomy of the pelvis and severing of the abdominal muscles during this procedure, defecation can be associated with severe pain postoperatively, resulting in constipation that can occasionally require an enema. 3 Opioids exacerbate this issue by causing constipation. Limiting opioid use with postoperative ketamine may lessen constipation after PAO and benefit patient recovery and satisfaction. 28
Limitations
The limitations of this study should be noted. These involve a lack of formal randomization between interventions, a lack of detailed subjective ketamine side effect data collection, heterogeneous groups of PAO and/or DFO procedures, and possible Hawthorne effects in the experimental group. The strengths of this study include large sample size, standardization of the delivery of the intervention, and detailed and thorough objective documentation. Future studies should investigate whether there is a dose-related response to continuous ketamine and further decreases in opioid requirements in the postoperative period after PAO without undesirable side effects.
Conclusion
Patients who received postoperative ketamine after PAO and/or DFO had a significant decrease in MME dose when compared with a control group of patients who did not receive ketamine. Surgeons should consider adding ketamine to their postoperative multimodal pain control protocol to decrease opioid consumption while adequately addressing postoperative pain.
Footnotes
Final revision submitted November 21, 2023; accepted January 1, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: C.Z. owns stock or stock options in AbbVie. O.M.D. holds stock or stock options in HeapSi and MITA and receives IP royalties from Stryker. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the University of Colorado Denver (ref No. CRV006-1).
