Abstract
Background:
Confusion persists regarding the ideal dosage of platelet-rich plasma (PRP) injection for knee osteoarthritis (OA).
Purpose/Hypothesis:
The purpose of the study was to compare the efficacy of 2 different single-injection PRP dosages in patients with early knee OA—a conventional 4 mL dose and a superdose of 8 mL. It was hypothesized that 8 mL of PRP would be superior to 4 mL of PRP in this patient population.
Study Design:
Randomized clinical trial; Level of evidence, 1.
Methods:
Patients with early knee OA (Kellgren-Lawrence grades 1 and 2) who met the inclusion criteria were randomly divided into 2 groups: Group A (n = 50 knees) received a 4-mL PRP injection, and group B (n = 49 knees) received an 8-mL PRP injection, both prepared using the same procedure. Patients were evaluated at the baseline, 6 weeks, 3 months, and 6 months using the Western Ontario and McMaster Universities Arthritis Index (WOMAC), the WOMAC-Pain subscale, the visual analog scale for pain, the Knee injury and Osteoarthritis Outcome Score, and patient satisfaction, and results were compared between the groups.
Results:
The baseline characteristics of the 2 groups were comparable (group A: mean age, 51.96 ± 6.93 years; group B: mean age, 49.12 ± 9.8 3 years). Leucocyte-depleted PRP with 3.5 times concentration (final product platelet concentration, 706.74 × 103–μL) was injected. The mean absolute platelet count injected was 2.82 ± 0.0012 billion in group A and 5.65 ± 0.0022 billion in group B. All patient-reported outcome scores improved significantly in both groups from the baseline to the final follow-up (P < .001), with overall trends and results significantly better in group B than in group A (P < .001). Patient satisfaction at the 6-month follow-up was also better in group B (96%) compared with group A (68%). Short (2 to 7 days) self-limiting complications, such as pain and stiffness, occurred more often in group B (P < .001).
Conclusion:
Patients with early knee OA had significantly better improvement in pain and function when treated with an 8-mL injection of PRP compared with a 4-mL injection of PRP. The larger dose of PRP had approximately twice the number of platelets.
Registration:
CTRI/2020/02/023403 (Clinical Trials Registry-India identifier).
Osteoarthritis (OA) of the knee—the leading cause of musculoskeletal disability—is a clinically heterogeneous entity characterized by progressive joint cartilage destruction and changes in the synovial membrane. 23 Autologous platelet-rich plasma (PRP) contains multiple growth factors that play a pivotal role in maintaining joint homeostasis, having an anti-inflammatory effect, and possibly remodeling chondrocytes, and it may have the potential to modify the disease progression. 16 Multiple studies3,4,6 have documented the beneficial effects of PRP for early knee OA; however, limited literature is available on documenting the efficacy of other treatment modalities such as cortisone, hyaluronic acid, or their combination. These injectables can control pain inside early arthritic knee joints; nonetheless, they have limited effects on the pathogenesis of OA. PRP can upregulate the anabolic cascades inside the knee joint and, hence, directly act on the pathways to change the joint microenvironment.1,3,10,11
There are many debated variables pertaining to PRP with respect to dosage, frequency, type, and the need for activators. Recent evidence and understanding seem to favor multiple injections over a single injection7,10 and leucocyte-poor over leukocyte-rich PRP. 6 The other important variable pertaining to PRP is the ideal dose per injection. The most commonly used dose is around 4 mL, partly because most commercially available PRP preparation systems yield 3 to 5 mL. On the other hand, Kon et al 11 and Patel et al 14 used 8 mL of PRP in their studies. Dhillon et al 5 referred to 8 mL of PRP as “superdose PRP” and recommended it for use in knee OA. The rationale for using 8 mL of PRP is that the greater the PRP volume, the greater the number of platelets, and thereby, the availability of growth factors. With the knee being a relatively large joint, 8 mL of PRP easily diffuses throughout the joint and reaches all the synovial folds. A 2020 study by Hahn et al 9 showed the positive and dose-dependent effects of PRP on human chondrocytes. Another study showed the dose-dependent effects of PRP on human mesenchymal stem cells. 22 A single injection of high-volume (5-9 mL) autologous pure PRP for knee OA was used by Guillibert et al 8 and Bec et al 2 in their series, with optimal results. Bansal et al 1 concluded that a requirement of an absolute count of 10 billion platelets in the PRP formulation had long-term sustained chondroprotective effects.
There has not been a direct comparative clinical study evaluating different dosages of injectable PRP in the published literature; hence, we conducted this study to compare the efficacy of 2 PRP dosages—the conventional 4 mL and the superdose 8 mL. The hypothesis was that a single superdose of 8-mL PRP would be superior to conventional 4-mL PRP for early knee OA.
Methods
Sample Size
This prospective, triple-blinded, randomized clinical trial, which was registered with the Clinical Trials Registry of India, compared 2 treatment groups with an allocation ratio of 1 to 1. The study protocol received clearance from our ethics committee, and all included patients provided written informed consent. Our sample size was based on an assumed study power of 80% (β = 0.2), a false-positive rate of 5% (α = .05), and a predicted difference of 1.5 ± 1.5 points on the visual analog scale (VAS) pain score. Using these parameters, and adjusting the α value for multiple comparisons, we estimated that we would need approximately 23 patients per treatment arm (ie, 46 knees). The sample size was calculated based on the protocol of previous PRP studies in our center.5,14
Study Participants
Patients attending the orthopaedics outpatient department at a single institution between February and July 2020 were considered for the study. A total of 100 patients with early-onset unilateral and bilateral knee OA on routine knee radiographs—diagnosed according to the American College of Rheumatology criteria and graded using the Kellgren-Lawrence classification—were screened and assessed for eligibility. Of the patients, 50 met the predefined inclusion criteria: Kellgren-Lawrence grades 1 or 2 knee OA without significant deformity (<10° varus/valgus deformity, <10° flexion deformity, or no evidence of inflammatory arthritis). Before receiving PRP injections, the patients were given a trial of nonsteroidal anti-inflammatory drugs and started on a physical therapy protocol that included quadriceps-strengthening exercises and a hip abductor strengthening program. The remaining 50 patients had 1 of the following exclusion criteria: OA secondary to inflammatory arthritis; crystalline arthropathy; associated metabolic disease (eg, type 2 diabetes mellitus); coexisting back pain; or advanced and late-stage OA. Patients who received intra-articular injections in the past 3 months, those with a history of thrombocytopenia, and those on anti-coagulant or antiplatelet treatments were also excluded. Also, 1 excluded knee had a previous history of trauma leading to a stiff knee with a valgus deformity of 20°.
Intervention and Randomization
The participants were randomly divided by a computer-generated random number chart into 2 groups—group A, with 25 patients (n = 50 knees), and group B, with 25 patients (n = 49 knees). Patients in group A received 4 mL of PRP injection, and patients in group B received 8 mL of PRP injection. Randomization was performed by nonclinical staff, and the researcher involved in the intervention (S.P.) was unaware of the group to which a patient belonged until the day of the intervention. A flowchart of the participant allocation process is shown in Figure 1.

A CONSORT flowchart of participant inclusion in the study. CONSORT, Consolidated Standards of Reporting Trials.
A single surgeon (S.P.) performed the standard injection procedure and was not involved in any other aspects of patient recruitment or the study follow-up. The patient recruitment, follow-up, results, and analysis were performed by 3 researchers (S.G., MSD., D.K.). The volunteer participants, the outcome scores evaluator (S.G.), and the statistician were all blinded and unaware of the treatment provided to the 2 groups. The surgeon (S.P.) who had provided the injection and was aware of the groups revealed the nature of treatment received by both groups after the data results and conclusion. For the ease of conducting this randomized controlled trial (RCT), we randomized patients, not their individual knees. In our practice, patients usually reported to the clinic if their knee OA was considered Kellgren-Lawrence grades 2 or 3. There was not much variation in symptoms between the included knees.
PRP Preparation Technique
Our PRP preparation technique has been in development since 2012 and was performed by specialists from the Department of Transfusion Medicine. We understand the need to preserve the integrity of the platelets. We even look into finer aspects, such as an atraumatic single prick to draw blood.
The PRP was prepared on the day of the procedure. Under aseptic precautions, 50 to 60 mL blood was drawn from the anticubital vein of the patient and collected in a blood bag (Terumo Penpol) with citrate-phosphate-dextrose and adenine as an anticoagulant preservative solution. Constant efforts were made throughout the procedure to avoid irritation and trauma to the platelets, which are in a resting state. The whole blood was then transferred from the blood bag into the sterile tube using a blood transfusion set inside a biosafety cabinet, class 2A (BIOAIR Safe flow1.2; Euroclone). The PRP was prepared using the double-spin platelet pellet method. The first spin lasted for 15 minutes at 1300 rpm using a tabletop centrifuge (Remi Lab Instruments). The supernatant plasma, which has all the platelets and a few white blood cells, was extracted through a pipette and transferred to another sterile tube. It was then subjected to a second spin at 2300 rpm for 5 minutes. The whole platelets settled down at the bottom as a platelet pellet. The supernatant platelet-poor plasma at the top was pipetted out, leaving 16 mL of plasma behind. The platelet button was resuspended with the remaining plasma, the final PRP product was dispensed in 2 10-mL syringes (8-mL PRP per knee), and no extra plasma was added.
The total leucocyte count and platelet count were measured from the patient’s peripheral blood and in the final PRP (Table 1). The PRP was leucocyte-poor PRP, with fewer leucocytes than at the baseline, and the platelets concentrated approximately 3.5 times the baseline. The mean platelet count achieved by our method was 706.74 × 103–μL ± 10.01, and the mean number of platelets injected per knee was 2.82 ± 0.00012 billion in group A (4-mL PRP) and 5.65 ± 0.000022 billion in group B (8-mL PRP). The procedure for PRP preparation was the same for both groups, and the concentration of platelets was high for both groups (Table 1).
Baseline Platelet and Leucocyte Concentrations Between Whole Blood and Final PRP a

Data are reported as mean ± SD. PRP, platelet-rich plasma.
PRP preparation was the same for both study groups.
Intra-articular Injections
The injections were given within 30 minutes of PRP preparation. The patients were positioned supine with their knees in full extension. Under aseptic precautions, the PRP injections were administered with an 18-gauge needle into the suprapatellar pouch through a superolateral approach. The knees were flexed and extended 10 times, and the patients were discharged after 30 minutes of observation. Because of financial constraints and because our principal investigator (S.P.) had more than 5 years of experience in performing intra-articular knee injections, we did not use ultrasound to confirm the intra-articular joint position.
Outcome Measures
All study patients were evaluated with the Western Ontario and McMaster Universities Arthritis Index (WOMAC), the WOMAC-Pain subscale, the VAS for pain, and the Knee injury and Osteoarthritis Outcome Score (KOOS) before injection (baseline) and at 6 weeks, 3 months, and 6 months after injection. The primary efficacy criterion was the change from the baseline in joint pain measured using the WOMAC-Pain score, and the secondary efficacy measure was the change in the WOMAC total score from the baseline. Patients were also assessed for satisfaction (satisfied, partly satisfied, or not satisfied) at the final (6-month) follow-up.
We calculated the minimum clinically important difference (MCID) for the Indian population by comparing the minimum difference required in the WOMAC-Pain score between the baseline and the final follow-up using patient satisfaction as the outcome variable. We determined that a 25% reduction in the WOMAC-Pain score was necessary to be clinically significant and appreciated by patients. A receiver operating characteristic curve was plotted, and the area under the curve was 0.751 (95% CI, 0.609-0.894), demonstrating fair diagnostic value (Figure 2). A cutoff percentage fall of 25% in the WOMAC-Pain score predicted clinical significance with a sensitivity of 85% and a specificity of 56%. We used the WOMAC-Pain score, as there was no conclusive data in the existing literature to define a clinically significant percentage reduction for the WOMAC total score.

The area under the ROC curve depicts the diagnostic value of the calculated MCID of 25% in the WOMAC-Pain score. MCID, minimum clinically important difference; ROC curve, receiver operating characteristic curve; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
Finally, any adverse effects related to treatment were recorded with respect to their nature, time of onset, duration, and severity.
Statistical Analysis
The outcome scores were exported to the Statistical Package for Social Sciences (Version 21; IBM) and analyzed for various tests considering the variables acquired through data collection. Measurable data were tested for normality using the Kolmogorov-Smirnov test. The normally distributed parameters were compared using the analysis of variance followed by post hoc tests such as the Student-Newman-Keuls and the Dunnett t procedure. Non-normal data were expressed as medians and interquartile ranges, and their distribution for the 2 groups was compared using the Mann-Whitney U test and Wilcoxon signed-rank tests. The associations of various categorical/classified variables, including complications were analyzed within the 2 groups using the chi-square test.
Within groups, the data on changes between the baseline and the various follow-up times (6 weeks, 3 months, and 6 months) were compared using the Student t test and paired or Wilcoxon signed-rank tests as applicable. Their difference (reported as percentage change) was compared using the Student t test or the Mann-Whitney U test as applicable. The data at the various follow-up points were analyzed using repeated-measures analysis of variance followed by post hoc tests. P < .05 was considered statistically significant for all tests.
Results
There was no difference between the 2 groups regarding baseline characteristics (Table 2). The PRP in both groups were comparable in terms of their platelet concentrations
Comparison of Baseline Variables Between Study Groups a

Data are reported as mean ± SD. BMI, body mass index; KOOS, Knee injury and Osteoarthritis Outcome Score; PRP, platelet-rich plasma; VAS, visual analog scale; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
Comparison of Baseline PRP Variables Between the Study Groups a

Data are reported as mean ± SD. PRP, platelet-rich plasma.
Outcome Measures
WOMAC-Pain
Both groups saw significant improvements in WOMAC-Pain scores from the baseline to the 6-month follow-up (Table 4 and Figure 3A). However, after the 3-month follow-up, the trend was slightly different between the 2 groups; group B experienced a continuous decrease in the WOMAC-Pain, with the lowest (ie, best) score noted at the final follow-up of 6 months (3.08 ± 1.90), whereas group A experienced a slight increase in scores at the final follow up (4.26 ± 1.01). Even though the WOMAC-Pain score decreased significantly in both treatment groups (P < .001), the change from the baseline was significantly better in group B than in group A at all time points (P < .001 for all) (Table 5).
Outcome Scores at Different Timepoints a
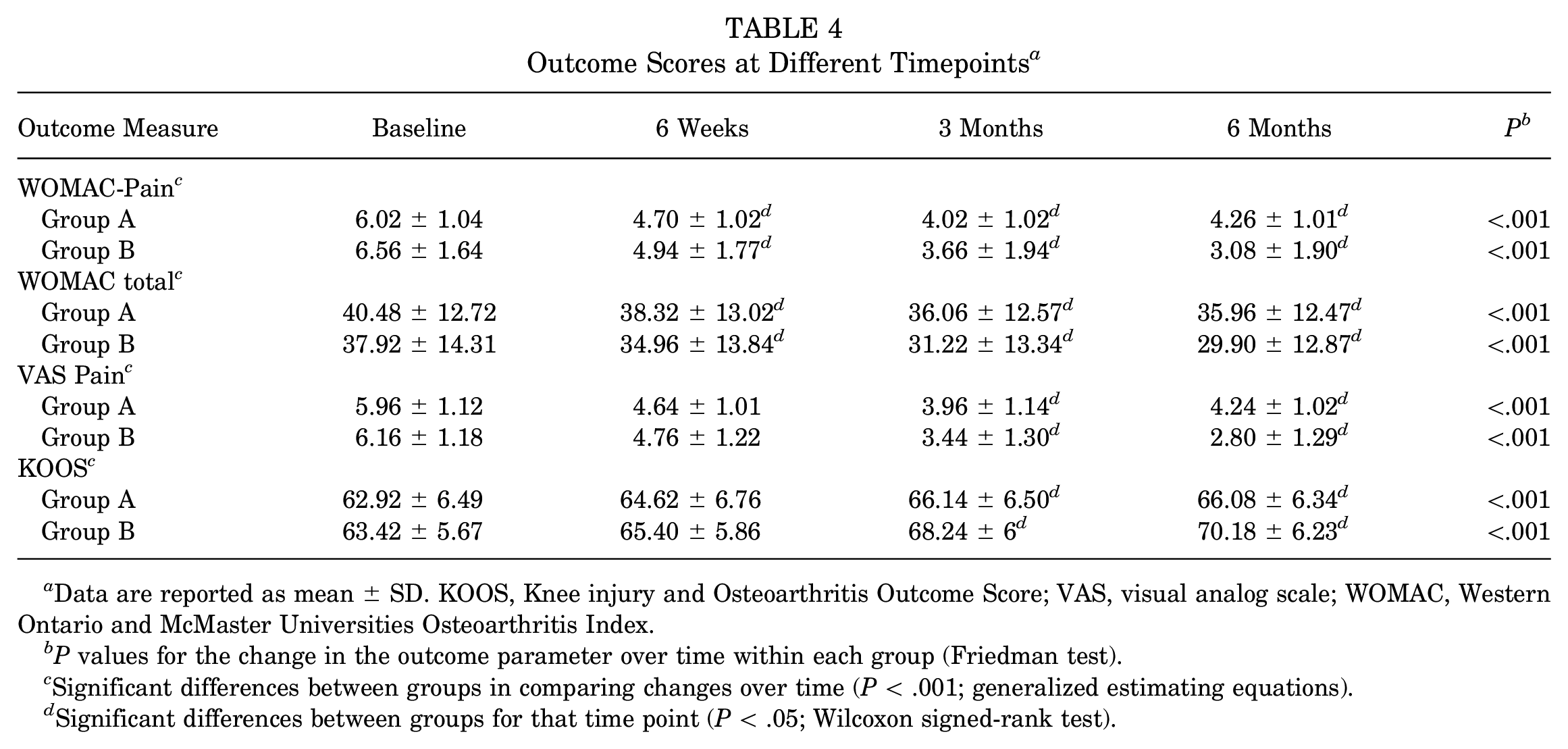
Data are reported as mean ± SD. KOOS, Knee injury and Osteoarthritis Outcome Score; VAS, visual analog scale; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
P values for the change in the outcome parameter over time within each group (Friedman test).
Significant differences between groups in comparing changes over time (P < .001; generalized estimating equations).
Significant differences between groups for that time point (P < .05; Wilcoxon signed-rank test).

Trends in the WOMAC-Pain, WOMAC total, VAS pain, and KOOS scores, respectively, over sequential follow-up. KOOS, Knee injury and Osteoarthritis Outcome Score; VAS, visual analog scale; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index. *Significant differences between groups for that time point (P < .05, Wilcoxon signed-rank test).
Changes in Outcome Scores Within and Between Study Groups a

Data are reported as mean ± SD. Bold P values indicate statistically significant differences between groups compared (P < .05). KOOS, Knee injury and Osteoarthritis Outcome Score; VAS, visual analog scale; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
WOMAC Total
The WOMAC total scores followed a similar trend in both groups. The mean WOMAC total score decreased from 40.48 ± 12.72 at the baseline to 35.96 ± 12.47 at the 6-month follow-up for group A and from 37.92 ± 14.31 at the baseline to 29.90 ± 12.87 at the 6-month follow-up for group B, with a continuous trend of improvement with the lowest mean value at the final follow up (Table 4 and Figure 3B). The change in the WOMAC total score was statistically significant at all time frames for both groups (P < .001 for all). However, the change from the baseline was significantly better in group B than in group A at all time points (P < .001 for all) (Table 5).
VAS Pain
VAS pain scores followed a similar trend as seen with WOMAC-Pain scores, with statistically significant improvements (ie, the decline in scores) in both groups and a mild deterioration in scores in group A at 6 months compared with 3 months. However, group B showed continued improvement from 3 to 6 months (Table 4 and Figure 3C). The change from the baseline to the 3- and 6-month follow-up was significantly better in group B than in group A (P < .001 for both) (Table 5).
KOOS
The KOOS score improved significantly from the baseline in both groups until the final follow-up (P < .001). The trend of KOOS scores was similar to that of the WOMAC-Pain and VAS pain scores in the 2 groups, with continued improvement noted in group B from 3 to 6 months (Table 4 and Figure 3D), and the change from the baseline to the 3- and 6-month follow-up was significantly better in group B than in group A (P < .001 for both) (Table 5).
Other Outcomes
Regarding the MCID for the WOMAC-Pain, a 32.1% decrease was observed in scores in group A, a 45.9% decrease in group B from the baseline to the 3-month follow-up, a 28% decrease in scores in group A, and a 54.3% decrease in group B from the baseline to the 6-month follow-up (Table 5). Thus, both groups achieved the desired MCID (a 25% decrease in scores) at the 3- and 6-month follow-up.
Patient satisfaction with the procedure and whether they would recommend the procedure to others was assessed at the end of the final follow-up (6 months). In our study, 68% of patients in group A and 96% of patients in group B were satisfied with the intervention.
Major self-limiting complications after injections were pain and stiffness, which were noticed in the postinjection period and lasted from 2 to 7 days, with a mean period of 4 days. Also, 80% of participants in group A and 95.3% of participants in group B had pain and stiffness. Both groups had similar self-limiting adverse reaction profiles; nonetheless, significantly more pain and stiffness were observed in group B (P < .001).
Discussion
Results of this study indicated that the larger 8-mL injection of PRP showed significant improvement in overall joint functionality and reduced arthritic joint pain when compared with the smaller, 4-mL injection. Evidence in support of this finding is provided by the results of multiple RCTs7,11,20 and meta-analyses4,12,18.
In our study, significantly greater improvement in WOMAC and KOOS scores were seen in group B (8-mL PRP) compared with group A (4-mL PRP). We also noted a different trend in the results between the groups, with a continuously improving trend of WOMAC-Pain, VAS pain, and KOOS scores from the 3-month to the final (6-month) follow-up in group B. In contrast, in group A, the best scores were noted at the 3-month follow-up, with mild worsening at 6 months—albeit still better than the baseline. A similar finding of mild deterioration at the final follow-up was noted in a previous study by our author group. 14 The most plausible reason for these findings could be the difference in the absolute platelet counts (group A, 2.82 billion; group B, 5.65 billion). Our results in the present study further support the superiority of superdose PRP in terms of improved patient-reported outcome scores 6 months after injection.
In many studies, 3 to 5 mL of PRP was used in knee injections, as most commercially available kits dispense only 3 to 5 mL of PRP.3,15,17,19 A growing emphasis has been placed on various PRP preparation procedures and PRP composition, as there is now a greater awareness regarding the potential of different platelet concentrates. A 2017 RCT by Bennel et al 3 found no significant efficacy of PRP over placebo; that study was controversial, and the major criticism of the study was the PRP type used. The PRP used was merely 1.6× concentrated and a single-spin system with very low yield; in fact, the product used can be labeled mere plasma and not actual PRP. We believe that the Bennel et al study is an important eye-opener, signifying that the quality of PRP in terms of platelet concentration and absolute platelet count does matter. In the present study, we used the double-spin (soft spin and hard spin) platelet plug method, a unique method devised at our institute. Using commercially available PRP kits only allows for a platelet concentration of 2 to 3 times the baseline, which, along with the low volume (3-5 mL) harvested, leads to a low cumulative absolute platelet count. The PRP we used was 3.6 times concentrated, and the mean platelet count achieved was 706.74 × 103–μL. This, along with the 8 mL volume (superdose PRP), led to a greater absolute platelet count, which imparted a better outcome. Magalon et al 13 introduced the DEPA (Dose of injected platelets, Efficiency of production, Purity of the PRP, Activation of the PRP) classification in 2016; the highlight of this classification system is that it takes into account the absolute platelet count, where in a very high dose refers to >5 billion platelets. In their study, Magalon et al observed various platelet counts in previous publications and PRP systems (range, 0.21-5.43 billion). The mean absolute platelet count in the superdose PRP group in our study was 5.65 billion (very high dose per DEPA classification) compared with 2.82 billion in the 4 mL group (medium dose per DEPA classification).
In the 2013 study by our group, 14 we used 8 mL of PRP and concluded that a single injection of PRP was as good as 2 injections of PRP. However, later studies by Görmeli et al 7 and others 10 seem to favor multiple PRP injections. We feel that this discrepancy in results may be due to the volume of PRP injection used. Bansal et al 1 recently noted that the dosage is crucial and critical for long-term clinical efficiency; their study used PRP prepared using filtration and observed 10 billion platelets as the absolute number essential for sustained chondroprotective effects. The present study achieved statistically significant results with an absolute platelet count of 5.6 billion. This is the first comparative study comparing 2 doses of PRP with respect to volume and absolute platelet counts injected, and our results support the use of a single, high-volume dose of PRP for knee OA. Future studies are necessary to see if an 8-mL injection is better than 2 separate 4-mL injections.
In this study, we also used the MCID to evaluate whether the change in the mean primary outcome parameters was clinically significant. Both of the study groups had a >25% postoperative improvement in mean WOMAC-Pain scores. At 3 months from the baseline, group B had a 45.9% decrease, while group A had a 32.1% decrease in scores; at 6 months from the baseline, group B had a 54.3% decrease, and group A had a 28% decrease in scores. These results indicate that 8 mL of PRP is superior to 4 mL of PRP in reducing knee pain related to OA, signifying its longer-lasting effect. Other clinical studies have highlighted the importance of the MCID.21,24 We are currently working on another publication evaluating 200 patients who received PRP and the MCID to define treatment success.
In this study, we observed a significant percentage of patients having self-limiting short-term complications (pain, stiffness, and swelling). This was more commonly seen in group B patients, possibly because of the greater volume of PRP (superdose). Along with platelets, leucocytes were also present (though leucocyte depleted). Other studies using leucocyte-rich PRP have also mentioned pain and stiffness as adverse effects.15,20
Limitations
A limitation of this study was the short follow-up time (6 months) and the small sample size. Another limitation is that we did not offer our patients any postinjection imaging. Duplication of patient-reported outcome measures was also a potential limitation, as we injected both knees of almost all the patients. Finally, we did not evaluate the MCIDs for the VAS pain or the KOOS between the study groups. Future studies should focus on using higher doses of PRP, as it is becoming more apparent that the dosage of PRP does matter.
Conclusion
In the present study, a single 8-mL injection of PRP with an absolute platelet count of more than 5 billion (superdose PRP) was better than a conventional 4-mL injection of PRP for early knee OA.
Footnotes
Final revision submitted August 6, 2023; accepted August 18, 2023.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Postgraduate Institute of Medical Education and Research in Chandigarh, India (ref No. NK/5635/MS/837).
