Abstract
Background:
While an association between femoroacetabular impingement (FAI) and osteoarthritis (OA) has been reported, the mechanistic differences and transition between the 2 conditions is not fully understood. In FAI, cartilage lesions at the femoral head-neck junction can sometimes be visualized during hip arthroscopy.
Purpose/Hypothesis:
The purpose of this study was to describe a unique dimpled pattern of superficial fissured cartilage lesions on the femoral head-neck junction at impingement site in patients with FAI syndrome (FAIS) and to evaluate the clinical, histological, and genetic phenotype of this cartilage. We hypothesized that the cartilage lesions may indicate risk for, or predict occurrence of, OA.
Study Design:
Controlled laboratory study.
Methods:
Six hips (6 patients; mean age, 34.2 ± 12.9 years; range, 19-54 years) with dimpled or fissured cartilage were included among patients who underwent hip arthroscopy for treatment of FAIS from October 2020 through December 2021. This affected cartilage (dimple-pattern group) and normal cartilage (control group) on the femoral head-neck junction were collected from the same patients and evaluated for histological quantification by Mankin scores and expression of proteins related to cartilage degeneration (eg, matrix metalloproteinase [MMP]-1, MMP-2, MMP-3, MMP-10, and MMP-12, tissue inhibitor of metalloproteinase [TIMP]-1 and TMP-2, aggrecan neopepitope CS846, and hyaluronic acid [HA]) with the use of Milliplex Multiplex Assays.
Results:
All 6 hips were of the mixed FAI subtype. Preoperatively, 4 of 6 hips had Tönnis grade 1 radiographic changes, which was associated with greater femoral head chondral damage visualized intraoperatively. Mankin scores for the normal cartilage group and the dimple-pattern group were 0.67 ± 0.82 and 3.3 ± 0.82, respectively. Dimple pattern fissured cartilage showed a significant increase in Mankin score (P = .031) and a significant increase in protein expression of CS846 (P = .031) compared with normal cartilage. There were no significant differences in MMPs, TIMPs, or HA levels between the 2 groups.
Conclusion:
The dimple pattern fissured cartilage, compared to normal cartilage, showed histologically significant cartilage degeneration and a significant increase in protein expression of CS846, a biomarker for early OA.
Clinical Relevance:
This lesion serves as helpful visual indicator of early degeneration of the cartilage of femoral head-neck junction caused by FAIS.
Hip osteoarthritis (OA) is a leading cause of disability in the United States and estimated to affect more than 41.1 million Americans by 2030. 19 Femoroacetabular impingement (FAI) is characterized by bony impingement of the acetabulum and femoral neck and has a strong clinical association with the development of premature OA. FAI is mainly classified into 3 subtypes: the cam type, characterized by osseous incongruity at the femoral head-neck junction; the pincer type, with excessive coverage of the acetabulum; and the mixed type, with a combination of cam and pincer lesions.3,5 The cam deformity is distinct from osteophytes at the femoral head-neck junction and similar to osteoarthritic-articular cartilage due to the elevation of inflammatory, anabolic, and catabolic genes at the impingement site. 11 , 24
Good clinical results and high patient satisfaction with arthroscopic osteochondroplasty and acetabular labral repair for FAI syndrome (FAIS) 8 have been reported, leading to a significant increase in the number of hip arthroscopy procedures for FAIS in recent years.17,18,21 When performing hip arthroscopy on patients with FAIS, surgeons encounter various patterns of cartilage surface fissures at the femoral head and head-neck junction. We previously reported unique vertical chondral fissure, the “crevasse lesion,” located on the posterior femoral head that was not detectable on magnetic resonance imaging (MRI) in 7 patients with mixed-type FAI out of 702 patients with FAIS. 22 The crevasse lesions involving the deeper layers of cartilage are presumed to be caused by adverse concomitant motion related to FAI. In this report, we further identified a dimpled golf-ball-like cartilage surface fissures on the femoral head-neck junction in FAIS patients, which exhibited cartilage surface irregularities. These cartilage lesions have not yet been reported, and their cause and significance are currently unknown.
The purpose of this study was to report on FAIS patients with this unique dimple pattern and compare the histological and gene expression in cartilage from the same FAIS patients with both macroscopically normal cartilage and cartilage lesion with this pattern over the cam deformity. We hypothesized that cartilage exhibiting these morphological patterns would have unique histological and molecular features commensurate with early OA development, including loss of proteoglycans and elevated expression of OA-related markers.
Methods
Patients and Cartilage Tissue Collection
The study protocol was approved by our institutional review board. Included were patients who underwent hip arthroscopy for FAIS between October 2020 and December 2021 and who also agreed to participate in the study preoperatively. The inclusion criteria were as follows: (1) patients who underwent hip arthroscopy for FAIS; (2) patients with superficial cartilage fissures with a golf-ball-like pattern appearance in sufficient extent on hip arthroscopy; and (3) patients for whom normal and damaged cartilage could be harvested intraoperatively. Exclusion criteria were: (1) patients who had previous open or arthroscopic hip procedures, significant acetabular dysplasia (lateral center-edge angle [LCEA] <20°), avascular necrosis, previous hip fracture or dislocation, or Legg-Calve-Perthes disease; (2) patients who did not have a golf-ball-like pattern appearance on hip arthroscopy; (3) patients for whom normal and damaged cartilage could not be obtained; and (4) patients who did not provide consent.
All patients underwent preoperative standard radiographs of the affected hip consisting of a supine anteroposterior pelvic view and a cross-table lateral view, and MRI without contrast medium. The diagnosis of FAIS was based on the patient's physical findings, including positive anterior impingement and flexion, abduction, external rotation (FABER) distance tests; radiographic findings, including crossover sign and alpha angle >50°; and activity-limiting hip symptoms. 17
Arthroscopic surgery was performed on all patients by a single surgeon (M.J.P.). Standard anterolateral and anteromedial portals were used, and both cartilage lesions (5-10 mm × 5 mm) and adjacent native cartilage were harvested if there were sufficient areas of golf-ball-like pattern cartilage lesions on the femoral head-neck junction at impingement site of FAI intraoperatively. The cartilage samples collected from the patients were separated and placed in sterile specimen cups containing saline solution. The samples were deidentified and processed for histology and protein analysis. The femoral head and acetabular cartilage status was macroscopically quantified using the Outerbridge classification system. 20 The cam and pincer deformities were resected as a standard hip arthroscopy procedure for FAIS. All patients underwent labral repair as well, and dynamic examination of the hip was performed to confirm proper tracking of the femoral head and chondrolabral junction.
Data Extraction
Patient characteristics (age, sex, body mass index [BMI], affected hip, sport, and traumatic event) were collected from patients who participated in the study. In addition, the following radiographic information was recorded: LCEA, alpha angle, Sharp angle, femoral anteversion, Tönnis grade, and FAI subtype (cam, pincer, or mixed). 26 Finally, grading of cartilage damage in the femoral head and acetabulum was extracted from the arthroscopic findings using the Outerbridge classification system. 20 The radiographic evaluations were blindly assessed by 2 orthopaedic surgeons (K.Y. and H.N.) and the evaluation of cartilage degeneration of the femoral head and acetabulum from arthroscopic findings was blindly performed by 2 orthopaedic surgeons (K.Y. and B.K.) using intraoperative arthroscopic photographs. The specific surgical procedures for FAIS performed on each patient was also obtained from the surgical records.
Histological Evaluation
All samples were fixed overnight at 4°C with 10% neutral buffered formalin. After dehydration, they were embedded in paraffin wax, and sections (6 μm) were prepared. Cartilage sections were histologically analyzed by safranin-O staining. Cartilage degeneration was independently and blindly quantified by Mankin score, a well-established histological scoring system to characterize cartilage degeneration, 15 by two orthopaedic surgeons (K.Y. and H.N.). The Mankin score is a 14-point scale consisting of structure, cellular abnormalities, matrix staining, and tidemark integrity. A score of 0 indicates normal cartilage, whereas a score of 14 indicates the most severe cartilage lesions and has been reported to have adequate intra- and interobserver reliability. 27
Gene Protein Expression Analysis
Proteins were extracted from cartilage samples using T-PER Tissue Protein Extraction Reagent (Thermo Fisher Scientific) and sample concentrations were normalized using the Pierce BCA Protein Assay Kit (Thermo Fisher Scientific). After homogenization, protein samples were stored at -80°C until bulk evaluation. Protein detection and quantification were performed using Milliplex Multiplex Assays (Millipore-Sigma) on a Luminex 100/200 system (Luminex). This technique is a bead-based suspension array using Luminex xMAP technology. Fluorescence-encoded beads with cytokine-capturing antibodies on their surface bind to proteins and are analyzed by flow cytometry. Magnetic bead kits (EMD Millipore) were used to measure analyte concentrations according to manufacturer's protocols. Belysa Immunoassay Curve Fitting Software System (EMD Millipore, catalog No 40-122) then determined each analyte concentration. All protocols were followed as provided by the manufacturer. In this study, a total of 9 analytes were evaluated: matrix metalloproteinase (MMP)-1, MMP-2, MMP-3, MMP-10, and MMP-12 (EMD Millipore, catalog No HMMP2MAG-55K-04); tissue inhibitor of metalloproteinase (TIMP)-1 and TIMP-2 (EMD Millipore, catalog No HTMP1MAG-54K-02); aggrecan chondroitin sulphate neopepitope CS846 (IBEX, catalog No 60-1004); and hyaluronic acid (HA; R&D systems, catalog No DHYALO). Regarding these investigated items, it has been reported that MMPs and TIMPs are involved in cartilage degeneration,13,30 HA in cartilage is reduced by OA, 32 and CS846 is a useful diagnostic marker for early OA that has received much attention in recent years.2,14
Statistical Analysis
Statistical analyses were performed using GraphPad Prism Version 9.4.0 (GraphPad Software), and data were expressed as means ± standard deviation. Comparison of Mankin scores and each protein expression between two groups of normal cartilage (control group) and affected cartilage (dimple-pattern group) was analyzed by the Wilcoxon test for paired data. Statistical significance was set at P < .05. A prior power analysis of sample size numbers was performed using G*Power (Version 3.1) from a previous study comparing Mankin scores in cartilage exhibiting FAI and normal cartilage at the previous femoral head-neck junction, 9 and the required number of sample sizes was calculated to be 5 (alpha error 0.05; statistical power 0.80).
Results
Of 281 total patients who underwent hip arthroscopy for FAIS during the study period, 6 hips (6 patients; mean age, 34.2 ± 12.9 years; range, 19-54 years) satisfied the study inclusion and exclusion criteria for collection of cartilage samples. The demographics of each patient are given in Table 1. Mean BMI was 21.9 ± 3.3 (range, 18-26.6), with a skew toward female (n = 4) versus male (n = 2) patients. All but 1 study participant had a history of sports participation.
Characteristics of the Included Hips (N = 6) a

Data are reported as mean ± SD or n. BMI, body mass index.
The results of the radiographic and arthroscopic findings are listed in Table 2. The mean LCEA and alpha angles were 36.0°± 5.5° and 67.7 ± 5.9°, respectively. Four of the 6 hips showed preoperative Tönnis grade 1 (66.7%), with intraoperative chondral evaluation demonstrating 2 hips with grade 2 and 2 hips with grade 3 femoral head cartilage damage from arthroscopic findings. The 2 patients with preoperative Tönnis grade 0 changes (33.3%) had grade 1 femoral head chondral wear identified intraoperatively. In all hips included in this study, golf-ball-like pattern cartilage lesions on the femoral head-neck junction were not identifiable using MRI. All 6 hips were of the mixed FAI subtype. All patients underwent labral repair, resection of pincer and cam lesion, acetabuloplasty and osteochondroplasty at the femoral head-neck junction, and capsular plication.
Radiographic and Arthroscopic Findings a

Data are reported as mean ± SD or n. All hips were of the mixed subtype. LCEA, lateral center-edge angle; SD, standard deviation.
Dimple-Patterned Cartilage as Gross and Histological Evidence of Degeneration
The cartilage surfaces of normal compared with golf-ball-like pattern are distinguishable by arthroscopic visualization (Figure 1). The location of the golf-ball-like pattern was predominantly in the region of impingement proximate to the cam deformity but was also frequently present over the weightbearing portion of the femoral head. The location of this golf-ball-like pattern at the femoral head-neck junction corresponds to zone 2 lateral or zone 3 lateral of the geographic zone described by Ilizaliturri et al. 12 Histological comparison of the dimple-patterned cartilage relative to normal cartilage shows a decrease in proteoglycans in the dimple-pattern group (Figure 2A and B). The Mankin score for the normal cartilage group was significantly lower (0.67 ± 0.82) than the dimple-patterned cartilage (3.3 ± 0.82; P = .031) (Figure 2C).

Dimple-patterned cartilage of femoral head-neck junction is visually identified by arthroscopy.
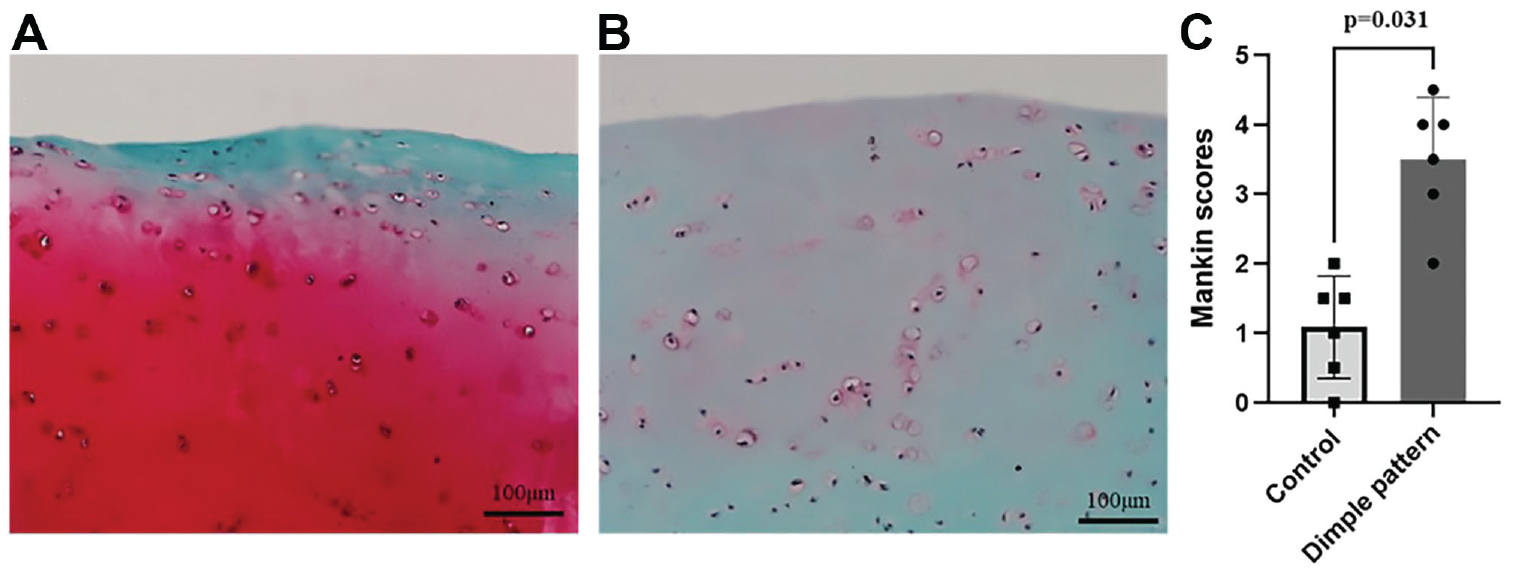
Dimple-patterned cartilage shows histological loss of proteoglycans consistent with OA phenotype. (A) Safranin-O staining of normal cartilage of femoral head-neck junction. (B) Safranin-O staining of dimple-patterned cartilage surface fissure of femoral head-neck junction. (C) Comparison of control and dimple-pattern groups in Mankin scores (P = .031).
Increased Aggrecan Neoepitope in Dimple-Patterned Tissue Consistent with Early OA
Protein expressions from the normal and dimple-patterned cartilage were measured using multiplex immunoassays from digested cartilage samples (Figure 3). The aggrecan chondroitin sulfate neoepitope CS846 was significantly higher in the dimple-pattern group compared with the control group (Figure 3A; P = .031). However, neither the MMP nor TIMP panels were statistically different at our current sample size (Figure 3B-I).
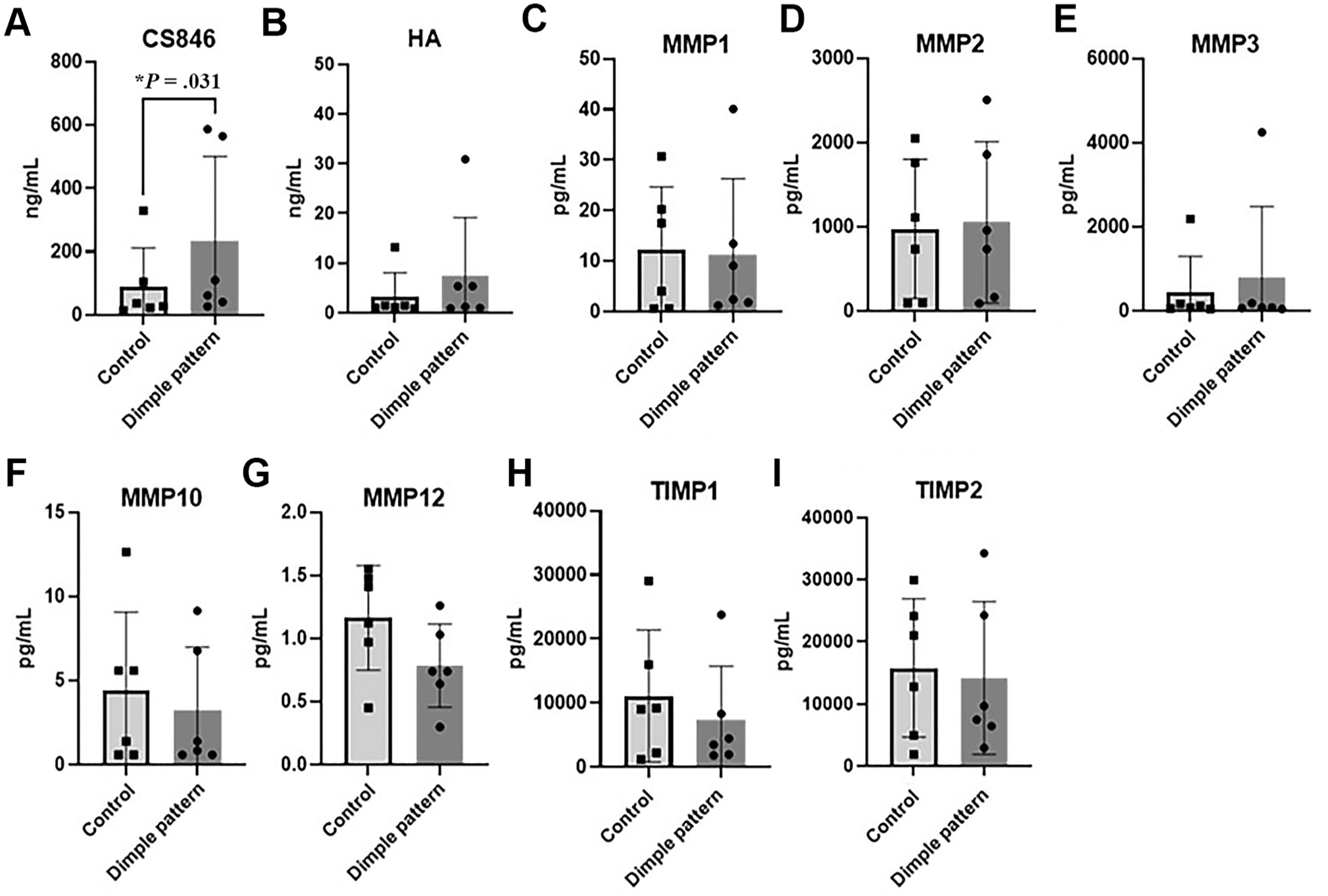
Proteomic analysis of normal and dimple-patterned cartilage. (A) CS846 was significantly higher (*P = .031) in the dimple-pattern group compared with the control group. (B) Hyaluronic acid trended higher, but was not significant (P = .44). No statistical differences were found in this sample set for the (C-G) MMP or (H-I) TIMP panels.
Discussion
In this study, we have reported a unique cartilage lesion in patients with FAIS that shows a dimpled pattern-gross morphology in the femoral head-neck junction that was found predominantly in patients with radiographic evidence of OA and arthroscopic evidence of cartilage damage. The cartilage in the dimple-patterned samples showed some OA-like signatures by histological evaluation compared with normal-appearing adjacent cartilage. This study also reports geographic differences in impingement cartilage quality where macroscopically damaged cartilage within the dimpled pattern had greater levels of both histologic proteomic degenerations compared with normal-appearing cartilage. Protein expression analysis showed no increase in isoforms of MMP or TIMP expression, which are associated with cartilage degeneration,13,30 whereas the aggrecan epitope, CS846, was increased significantly. Although there have been previous studies in which cartilage from the femoral head-neck junction, a site of impingement of FAI, was collected and evaluated for histology and gene expression, this study is the first to evaluate differential pathological attributes of the unique golf-ball-like pattern cartilage morphology.10,11,24,28 In addition, these previous studies evaluated normal cartilage from cadavers as a control group, and to the best of the authors’ knowledge, this is the first report to evaluate normal appearing cartilage from the same patient as a control group.
Few reports have been published on the morphological pattern of cartilage lesion of femoral head-neck junction associated with FAI. A prior study from 2003 by Wagner and colleagues evaluating impingement cartilage from the proximal femur found a range of histologic damage with Mankin scores ranging from 2 to 12 compared with healthy cartilage which had Mankin scores ranging from 0 to 2 (P = .007) and osteoarthritic samples ranging from 3 to 14 (P = .01). 28 These findings indicate that FAI impingement cartilage bears histologic features of osteoarthritic cartilage that progress over time and severity of the disease. Identification of unique risk factors for OA progression would be valuable for prognostic discussions and potentially therapeutic interventions. We previously reported a unique crevasse lesion, a vertical cartilage injury of the posterior aspect of the femoral head. 22 The crevasse lesions involving the deeper layers of cartilage are presumed to be caused by adverse concomitant motion related to FAI. In addition, Zaltz and Leunig 31 reported a rare cartilage defect within the posterior superior quadrant of the femoral head associated with FAI and assumed that the injury was shear stress induced. The superficial cartilage fissures in the present study, based on histological evaluation of the Mankin scores, showed OA-like changes, indicating a distinct difference in the shape, location, and mechanism of the injury. Further, the appearance of the golf-ball-like pattern in the weight-bearing region of the femoral head, separate from the focal zone of impingement, may be a marker of progressive chondral injury signifying disease progression.
Although the molecular mechanisms by which FAI contributes to OA progression are not fully understood, local inflammation has been reported to contribute to the progression of OA throughout the hip joint through the production of inflammatory cytokines and matrix proteases. 7 The main components of articular cartilage are water and extracellular matrix (ECM) consisting of type 2 collagen and proteoglycans. 16 In early OA, proteoglycan loss has been reported to occur before type 2 collagen degradation, 16 and the histological evaluation in this study showed loss of proteoglycans in dimple-patterned fissured cartilage compared with normal cartilage. Due to the covalent bonding of glycosaminoglycans, proteoglycans are negatively charged, and this property attracts cations such as sodium, which draws water into the tissue and generates cartilage swelling pressure for mechanical resilience. 29 The characteristic golf-ball-like morphology of cartilage lesion presented in this study may be observed as a dent in the cartilage due to loss of water as well as loss of proteoglycans. In the evaluation of molecular gene expression associated with cartilage in FAI, elevated inflammatory and catabolic markers such as interleukin (IL)-1β and MMP-13 have been reported in the cartilage of the femoral head-neck junction in patients with FAIS compared with normal cartilage.10,11 In this study, there were no significant differences between normal cartilage and dimple-patterned fissured cartilage in protein expression of the six types of MMPs, two types of TIMPs, and HA investigated at the sample size tested. Considering that most of the hips with golf-ball-like pattern fissured cartilage lesion in this study showed radiological and arthroscopic signs of OA, this protein expression result may suggest that the histologically normal looking cartilage might have been subjected to the same stresses as the adjacent golf-ball-like fissured cartilage, resulting in the same level of expression of catabolic markers.
Despite a largely similar gene expression profile, this study found that CS846 was significantly elevated in dimple-patterned fissured cartilage. CS846, a byproduct of proteoglycan metabolism, is a highly sensitive and specific biomarker that reflects cartilage degradation and has attracted attention as a useful and promising tool for early OA diagnosis.2,14 CS846 is present only in newly synthesized aggrecan 6 and has been reported to increase as part of the response of cartilage to damage.23,25 This finding of a significant elevation of OA-specific markers in the dimple pattern cartilage over normal looking cartilage is supported by histological evaluation with Safranin O staining between the 2 groups. This has important prognostic implications given the increased rate of OA progression and inferior outcomes following hip arthroscopy in patients with significant femoral head chondral damage.1,4 Unlike previous studies, which identified primarily focal defects, this study reports on a chondral lesion that is more diffuse and possibly related to the progression of osteoarthritic changes affecting the entire joint.
Limitations
There are some limitations to this study, the first being the small number of patients included. Although a prior power analysis was performed from previous studies and the required sample size number was reached, further data accumulation is needed in the future. Second, there is no control group of cartilage harvested from the same site in patients without golf-ball-like pattern fissured cartilage. The control cartilage in this study was different from the normal cartilage harvested from cadavers evaluated in most of previous studies of FAI-associated cartilage.9-11 Arthroscopically and histologically normal appearing cartilage may have been subjected to the same stresses as the golf-ball-like pattern fissured cartilage, which may have led to the result that there was no difference in cartilage-related markers between the 2 groups, except for CS846. Third, the inflammatory markers studied were localized and common IL-1β, IL-6, and MMP-13, which are associated with cartilage degeneration, were not evaluated. The cartilage that can be harvested is the minimum amount of thin and small tissue necessary, taking into account postharvest effects. Further evaluation was difficult because of the dichotomy between histological evaluation and evaluation of gene protein expression. It is not ethically appropriate to collect an excessive amount of cartilage, or cartilage from areas that would not be routinely removed during cam resection. Fourth, although some FAIS patients have been shown to exhibit this cartilage lesion, this lesion has not been investigated to determine how FAIS affects this morphology of cartilage. Finally, the association of this cartilage lesion with clinical outcomes and progression of OA has not been evaluated. Further studies will include longer-term follow-up to assess this cartilage lesion and the incidence of OA by adding a control group that does not present this cartilage lesion.
Conclusion
The dimple pattern fissured cartilage of femoral head-neck junction caused by FAI, compared with normal cartilage, showed histologically significant cartilage degeneration and a significant increase in protein expression of CS846, a biomarker for early OA.
Footnotes
Final revision submitted June 19, 2023; accepted June 29, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: C.S.B. has received royalties from Iota Biosciences for a United States patent (041263). J.H. has received royalties from Cook Myosite. M.J.P. has received education payments from Smith & Nephew, Conmed, Linvatec, Ossur, Arthrex, Siemens Medical Solutions; consulting fees from Smith & Nephew, MIS, Olatec, and NICE Recovery Systems; nonconsulting fees from Smith & Nephew, MIS, Olatec, NICE Recovery Systems, and Synthes; royalties from Linvatec, Smith & Nephew, Arthrosurface, Bledsoe, Conmed, DJO, Slack, and Elsevier; and holds shares in Arthrosurface, MJP Innovations, Vail Valley Surgery Center, Vail MSO Holdings, MIS, EFFRx, Olatec, Arthrex, Manna Tree Partners, Stryker, Trimble, 3M, Bristol Myers, Squibb, Pfizer, AbbVie, and Johnson & Johnson. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Vail Health Hospital (ref No. v7 092321).
